Hydrogen Storage for Mobility: A Review
Abstract
:1. Introduction
2. Hydrogen for Mobility
2.1. Overall Efficiency
2.2. Costs of Battery vs. Fuel Cell
2.3. Practical Advantages of Fuel Cells
3. Ideal Storage Method
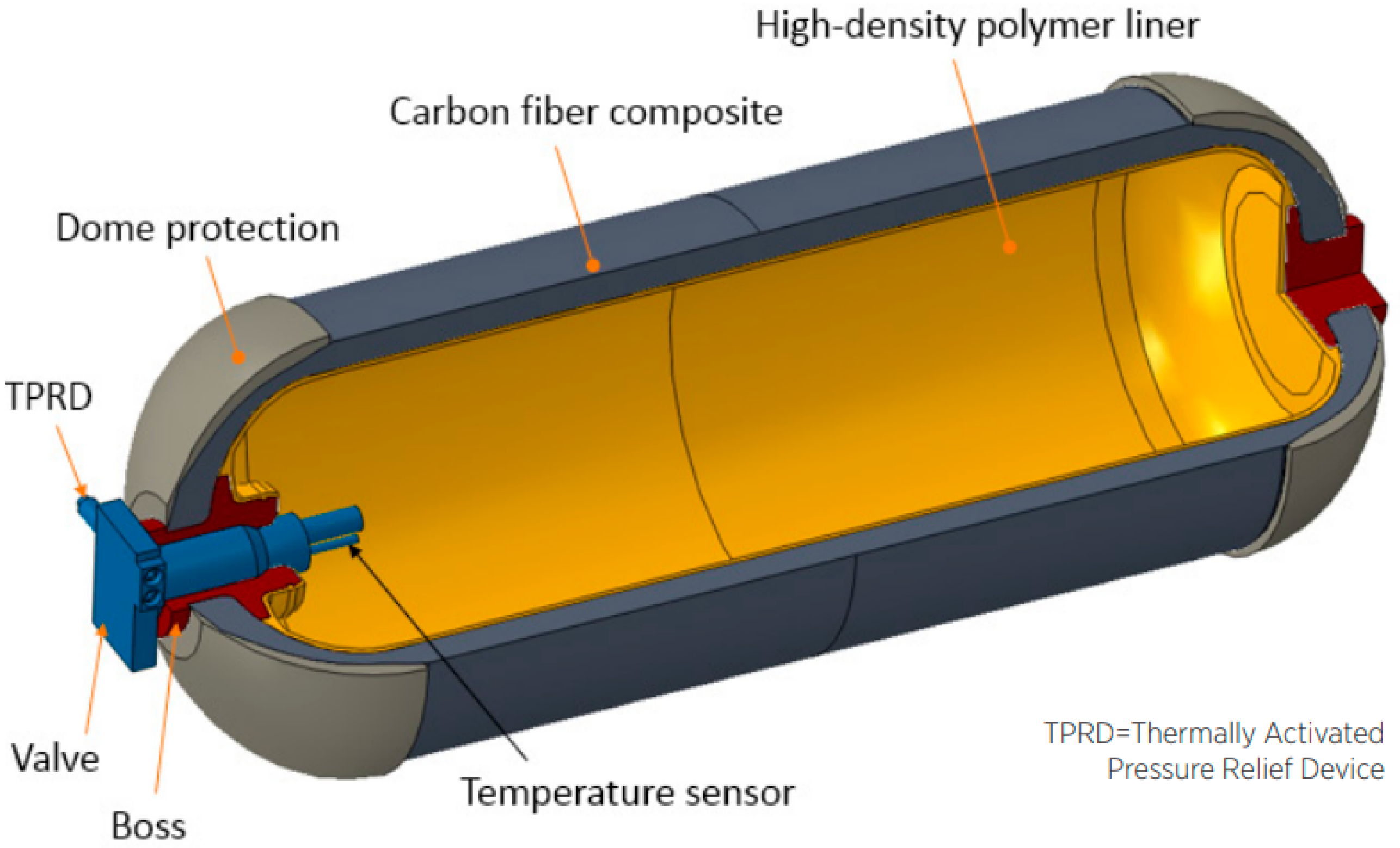
4. Present Industry Choice: Compressed Gas
5. Other Storage Methods
5.1. Liquid Hydrogen
5.2. Cold/Cryo Compression
5.3. Metal–Organic Framework
5.4. Carbon Nanostructures
5.5. Metal Hydrides
5.6. Metal Borohydrides
5.7. Kubas-Type Hydrogen
5.8. Liquid Organic Hydrogen Carriers
5.9. Chemical Hydrogen
6. Overview
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stocker, T.F.; Qin, D.; Plattner, G.-K.; Tignor, M.; Allen, S.K.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, V.; Midgley, P.M. IPCC, 2013: Summary for Policymakers. In Climate Change 2013: The Physical Science Basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013. [Google Scholar]
- Walsh, B.S.; Parratt, S.R.; Hoffmann, A.A.; Atkinson, D.; Snook, R.R.; Bretman, A.; Price, T.A.R. The Impact of Climate Change on Fertility. Trends Ecol. Evol. 2019, 34, 249–259. [Google Scholar] [CrossRef] [PubMed]
- CaraDonna, P.J.; Cunningham, J.L.; Iler, A.M. Experimental warming in the field delays phenology and reduces body mass, fat content and survival: Implications for the persistence of a pollinator under climate change. Funct. Ecol. 2018, 32, 2345–2356. [Google Scholar] [CrossRef]
- Hernandez-Ochoa, I.M.; Asseng, S.; Kassie, B.T.; Xiong, W.; Robertson, R.; Pequeno, D.N.L.; Sonder, K.; Reynolds, M.; Babar, M.A.; Milan, A.M.; et al. Climate change impact on Mexico wheat production. Agric. For. Meteorol. 2018, 263, 373–387. [Google Scholar] [CrossRef]
- Kontgis, C.; Schneider, A.; Ozdogan, M.; Kucharik, C.; Tri, V.P.D.; Duc, N.H.; Schatz, J. Climate change impacts on rice productivity in the Mekong River Delta. Appl. Geogr. 2019, 102, 71–83. [Google Scholar] [CrossRef]
- Raymundo, R.; Asseng, S.; Robertson, R.; Petsakos, A.; Hoogenboom, G.; Quiroz, R.; Hareau, G.; Wolf, J. Climate change impact on global potato production. Eur. J. Agron. 2018, 100, 87–98. [Google Scholar] [CrossRef]
- Mazdiyasni, O.; AghaKouchak, A. Substantial increase in concurrent droughts and heatwaves in the United States. Proc. Natl. Acad. Sci. USA 2015, 112, 11484–11489. [Google Scholar] [CrossRef] [Green Version]
- Ogburn, S.P. Indian Monsoons Are Becoming More Extreme. Scientific American. 2014. Available online: https://www.scientificamerican.com/article/indian-monsoons-are-becoming-more-extreme/ (accessed on 13 February 2019).
- Warner, K.; Ehrhart, C.; de Sherbinin, A.; Adamo, S.; Chai-Onn, T. In Search of Shelter: Mapping the Effects of Climate Change on Human Migration and Displacement; Climate Change CARE International: London, UK, 2009; 26p. [Google Scholar]
- Black, R.; Bennett, S.R.; Thomas, S.M.; Beddington, J.R. Climate change: Migration as adaptation. Nature 2011, 478, 447–449. [Google Scholar] [CrossRef] [PubMed]
- McLeman, R.; Smit, B. Migration as an Adaptation to Climate Change. Clim. Chang. 2006, 76, 31–53. [Google Scholar] [CrossRef]
- Reuveny, R. Climate change-induced migration and violent conflict. Political Geogr. 2007, 26, 656–673. [Google Scholar] [CrossRef]
- Nordås, R.; Gleditsch, N.P. Climate change and conflict. Political Geogr. 2007, 26, 627–638. [Google Scholar] [CrossRef]
- Barnett, J. Security and climate change. Glob. Environ. Chang. 2003, 13, 7–17. [Google Scholar] [CrossRef] [Green Version]
- AFHYPAC and FNCCR. Déployer les stations hydrogène dans votre territoire. 2018. Available online: http://www.afhypac.org/documents/divers/GUIDE-STATION-HYDROGENE-WEB.pdf (accessed on 13 February 2019).
- United States Environmental Protection Agency. Inventory of U.S. Greenhouse Gas Emissions and Sinks: 1990–2015. 2017. Available online: https://www.epa.gov/sites/production/files/2017-02/documents/2017_complete_report.pdf (accessed on 13 February 2019).
- BP Statistical Review of World Energy. 2018. Available online: https://www.bp.com/content/dam/bp/en/corporate/pdf/energy-economics/statistical-review/bp-stats-review-2018-full-report.pdf (accessed on 12 October 2018).
- Saito, S. Role of nuclear energy to a future society of shortage of energy resources and global warming. J. Nucl. Mater. 2010, 398, 1–9. [Google Scholar] [CrossRef]
- Salameh, M.G. Can renewable and unconventional energy sources bridge the global energy gap in the 21st century? Appl. Energy 2003, 75, 33–42. [Google Scholar] [CrossRef]
- Friedrichs, J. Global energy crunch: How different parts of the world would react to a peak oil scenario. Energy Policy 2010, 38, 4562–4569. [Google Scholar] [CrossRef]
- Lund, H.; Mathiesen, B.V. Energy system analysis of 100% renewable energy systems—The case of Denmark in years 2030 and 2050. Energy 2009, 34, 524–531. [Google Scholar] [CrossRef]
- Singer, S.; Denruyter, J.-P.; Yener, D. The Energy Report: 100% Renewable Energy by 2050. In Towards 100% Renewable Energy; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Züttel, A.; Borgschulte, A.; Schlapbach, L. Hydrogen as a Future Energy Carrier; John Wiley & Sons: Hoboken, NJ, USA, 2011; p. 441. [Google Scholar]
- Winter, C.-J.; Nitsch, J. Hydrogen as an Energy Carrier; Springer: Berlin/Heidelberg, Germany, 1988; p. 380. [Google Scholar]
- Mazloomi, K.; Gomes, C. Hydrogen as an energy carrier: Prospects and challenges. Renew. Sustain. Energy Rev. 2012, 16, 3024–3033. [Google Scholar] [CrossRef]
- Cipriani, G.; Di Dio, V.; Genduso, F.; La Cascia, D.; Liga, R.; Miceli, R.; Galluzzo, G.R. Perspective on hydrogen energy carrier and its automotive applications. Int. J. Hydrogen Energy 2014, 39, 8482–8494. [Google Scholar] [CrossRef]
- Züttel, A.; Remhof, A.; Borgschulte, A.; Friedrichs, O. Hydrogen: The future energy carrier. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010, 368, 3329–3342. [Google Scholar] [CrossRef]
- Nakicenovic, N.; Grübler, A.; McDonald, A. Global Energy Perspectives; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar]
- Arnold, R. Are Renewable Energy Cost Reductions Coming to an End? What Next? 2018. Available online: http://energypost.eu/are-renewable-energy-cost-reductions-coming-to-an-end-what-next/ (accessed on 13 February 2019).
- Kikuchi, Y.; Ichikawa, T.; Sugiyama, M.; Koyama, M. Battery-assisted low-cost hydrogen production from solar energy: Rational target setting for future technology systems. Int. J. Hydrogen Energy 2019, 44, 1451–1465. [Google Scholar] [CrossRef]
- Dincer, I.; Ratlamwala, T. Development of novel renewable energy based hydrogen production systems: A comparative study. Energy Convers. Manag. 2013, 72, 77–87. [Google Scholar] [CrossRef]
- Nafchi, F.M.; Baniasadi, E.; Afshari, E.; Javani, N. Performance assessment of a solar hydrogen and electricity production plant using high temperature PEM electrolyzer and energy storage. Int. J. Hydrogen Energy 2018, 43, 5820–5831. [Google Scholar] [CrossRef]
- Züttel, A. Materials for hydrogen storage. Mater. Today 2003, 6, 24–33. [Google Scholar] [CrossRef]
- Abdalla, A.M.; Hossain, S.; Nisfindy, O.B.; Azad, A.T.; Dawood, M.; Azad, A.K. Hydrogen production, storage, transportation and key challenges with applications: A review. Energy Convers. Manag. 2018, 165, 602–627. [Google Scholar] [CrossRef]
- Durbin, D.; Malardier-Jugroot, C. Review of hydrogen storage techniques for on board vehicle applications. Int. J. Hydrogen Energy 2013, 38, 14595–14617. [Google Scholar] [CrossRef]
- Wang, L.; Yang, R.T. New sorbents for hydrogen storage by hydrogen spillover–A review. Energy Environ. Sci. 2008, 1, 268–279. [Google Scholar] [CrossRef]
- Jain, I.; Jain, P.; Jain, A. Novel hydrogen storage materials: A review of lightweight complex hydrides. J. Alloy Compd. 2010, 503, 303–339. [Google Scholar] [CrossRef]
- Fakioğlu, E.; Yürüm, Y.; Nejat Veziroğlu, T. A review of hydrogen storage systems based on boron and its compounds. Int. J. Hydrogen Energy 2004, 29, 1371–1376. [Google Scholar] [CrossRef]
- Marbán, G.; Valdés-Solís, T. Towards the hydrogen economy? Int. J. Hydrogen Energy 2007, 32, 1625–1637. [Google Scholar] [CrossRef] [Green Version]
- Muradov, N.; Vezirolu, T. From hydrocarbon to hydrogen?carbon to hydrogen economy. Int. J. Hydrogen Energy 2005, 30, 225–237. [Google Scholar] [CrossRef]
- Barreto, L.; Makihira, A.; Riahi, K. The hydrogen economy in the 21st century: A sustainable development scenario. Int. J. Hydrogen Energy 2003, 28, 267–284. [Google Scholar] [CrossRef]
- Penner, S.S. Steps toward the hydrogen economy. Energy 2006, 31, 33–43. [Google Scholar] [CrossRef]
- Bossel, U. Does a Hydrogen Economy Make Sense? Proc. IEEE 2006, 94, 1826–1837. [Google Scholar] [CrossRef]
- ITM Power. Hydrogen Refuelling Infrastructure. 2017. Available online: http://www.level-network.com/wp-content/uploads/2017/02/ITM-Power.pdf (accessed on 18 March 2019).
- Gardiner, M.; Satyapal, S. Energy Requirements for Hydrogen Gas Compression and Liquefaction as Related to Vehicle Storage Needs. DOE Hydrogen and Fuel Cells Program Record; 2009. Available online: https://www.hydrogen.energy.gov/pdfs/9013_energy_requirements_for_hydrogen_gas_compression.pdf (accessed on 13 June 2019).
- Fuel Cell Technologies Office. Fuel Cells. 2015. Available online: https://www.energy.gov/sites/prod/files/2015/11/f27/fcto_fuel_cells_fact_sheet.pdf (accessed on 18 March 2019).
- Office of Energy Efficiency and Renewable Energy. All-Electric Vehicles. Available online: https://www.fueleconomy.gov/feg/evtech.shtml (accessed on 18 March 2019).
- Curry, C. Lithium-ion Costs and Market. 2017. Available online: https://data.bloomberglp.com/bnef/sites/14/2017/07/BNEF-Lithium-ion-battery-costs-and-market.pdf (accessed on 13 June 2019).
- Tajitsu, N.; Shiraki, M. Toyota Plans to Expand Production, Shrink Cost of Hydrogen Fuel Cell Vehicles, in Business News; Reuters: London, UK, 2018. [Google Scholar]
- Brown, E.G. Joint Agency Staff Report on Assembly Bill 8: Assessment of Time and Cost Needed to Attain 100 Hydrogen Refueling Stations in California; California Energy Commission: Sacramento, CA, USA, 2015.
- Thomas, C. Fuel cell and battery electric vehicles compared. Int. J. Hydrogen Energy 2009, 34, 6005–6020. [Google Scholar] [CrossRef] [Green Version]
- Heraeus, Heraeus Precious Appraisal. 2018. Available online: https://www.heraeus.com/media/media/hpm/doc_hpm/precious_metal_update/en_6/Appraisal_20190429.pdf (accessed on 13 June 2019).
- ToolBox, T.E. Fossil and Alternative Fuels-Energy Content. Available online: https://www.engineeringtoolbox.com/fossil-fuels-energy-content-d_1298.html (accessed on 13 February 2019).
- AFHYPAC. Les données de base physico-chimiques sur l’hydrogène. Mémento de l’Hydrogène. 2013. Available online: http://www.afhypac.org/documents/tout-savoir/fiche_1.2_donnees_physicochimiques_rev.mars_2013.pdf (accessed on 13 February 2019).
- International Group of Liquefied Natural Gas Importers. Basic Properties of LNG. LNG Information Papers. Available online: http://www.kosancrisplant.com/media/5648/1-lng_basics_82809_final_hq.pdf (accessed on 13 February 2019).
- Mori, D.; Hirose, K. Recent challenges of hydrogen storage technologies for fuel cell vehicles. Int. J. Hydrogen Energy 2009, 34, 4569–4574. [Google Scholar] [CrossRef]
- Crabtree, R.H. Hydrogen storage in liquid organic heterocycles. Energy Environ. Sci. 2008, 1, 134. [Google Scholar] [CrossRef]
- Eberle, U.; Arnold, G.; Von Helmolt, R. Hydrogen storage in metal–hydrogen systems and their derivatives. J. Power Sources 2006, 154, 456–460. [Google Scholar] [CrossRef]
- Aardahl, C.; Rassat, S. Overview of systems considerations for on-board chemical hydrogen storage. Int. J. Hydrogen Energy 2009, 34, 6676–6683. [Google Scholar] [CrossRef]
- Zhang, J.; Fisher, T.S.; Ramachandran, P.V.; Gore, J.P.; Mudawar, I. A Review of Heat Transfer Issues in Hydrogen Storage Technologies. J. Heat Transf. 2005, 127, 1391–1399. [Google Scholar] [CrossRef]
- U.S. Department of Energy. Fuel Cell Technologies Office Multi-Year Research, Development, and Demonstration Plan. 2012. Available online: https://www.energy.gov/eere/fuelcells/downloads/fuel-cell-technologies-office-multi-year-research-development-and-22 (accessed on 6 February 2019).
- SAE International. SAE J2600 Compressed Hydrogen Surface Vehicle Fueling Connection Devices. 2015. Available online: https://www.sae.org/standards/content/j2600_201211/ (accessed on 13 February 2019).
- Yamashita, A.; Kondo, M.; Goto, S.; Ogami, N. Development of High-Pressure Hydrogen Storage System for the Toyota “Mirai”. SAE Tech. Pap. Ser. 2015. [Google Scholar] [CrossRef]
- Legault, M. Pressure Vessel Tank Types. 2012. Available online: https://www.compositesworld.com/articles/pressure-vessel-tank-types (accessed on 13 February 2019).
- Law, K.; Rosenfeld, J. Cost Analyses of Hydrogen Storage Materials and On-Board Systems. 2011. Available online: https://www.hydrogen.energy.gov/pdfs/review11/st002_law_2011_o.pdf (accessed on 18 March 2019).
- Hua, T.; Ahluwalia, R.; Peng, J.-K.; Kromer, M.; Lasher, S.; McKenney, K.; Law, K.; Sinha, J. Technical Assessment of Compressed Hydrogen Storage Tank Systems for Automotive Applications; Office of Scientific and Technical Information (OSTI): Oak Ridge, TN, USA, 2010.
- Fuel Cell Technologies Office, Hydrogen Storage. 2017. Available online: https://www.energy.gov/sites/prod/files/2017/03/f34/fcto-h2-storage-fact-sheet.pdf (accessed on 13 June 2019).
- Toyota. 2017 Mirai Product Information. 2017. Available online: https://ssl.toyota.com/mirai/assets/core/Docs/Mirai%20Specs.pdf (accessed on 13 February 2019).
- Gye, H.-R.; Seo, S.-K.; Bach, Q.-V.; Ha, D.; Lee, C.-J. Quantitative risk assessment of an urban hydrogen refueling station. Int. J. Hydrogen Energy 2019, 44, 1288–1298. [Google Scholar] [CrossRef]
- Alperowicz, N. Ube Industries’ New Nylon Resin to be Used in Toyota Fuel-Cell Vehicles. 2014. Available online: chemweek.com (accessed on 13 June 2019).
- Wyckaert, P.; Nadeau, S.; Bouzid, H.A. Analysis of risks of pressure vessels. In Proceedings of the Kongress der Gesellschaft für Arbeitswissenschaft, Zurich, Switzerland, 15–17 February 2017. [Google Scholar]
- Rossini, F.D. Report on International Practical Temperature Scale of 1968. J. Chem. Thermodyn. 1970, 2, 447–459. [Google Scholar] [CrossRef]
- Xu, W.; Li, Q.; Huang, M. Design and analysis of liquid hydrogen storage tank for high-altitude long-endurance remotely-operated aircraft. Int. J. Hydrogen Energy 2015, 40, 16578–16586. [Google Scholar] [CrossRef]
- Babac, G.; Şişman, A.; Çimen, T. Two-dimensional thermal analysis of liquid hydrogen tank insulation. Int. J. Hydrogen Energy 2009, 34, 6357–6363. [Google Scholar] [CrossRef]
- Petitpas, G. Simulation of boil-off losses during transfer at a LH2 based hydrogen refueling station. Int. J. Hydrogen Energy 2018, 43, 21451–21463. [Google Scholar] [CrossRef]
- Gürsu, S.; Lordgooei, M.; Sherif, S.; Veziroglu, T. An optimization study of liquid hydrogen boil-off losses. Int. J. Hydrogen Energy 1992, 17, 227–236. [Google Scholar] [CrossRef]
- Zhou, L. Progress and problems in hydrogen storage methods. Renew. Sustain. Energy Rev. 2005, 9, 395–408. [Google Scholar] [CrossRef]
- Amos, W.A. Costs of Storing and Transporting Hydrogen; National Technical Information Service (NTIS): Springfield, VA, USA, 1998. [Google Scholar]
- Colozza, A.J. Hydrogen Storage for Aircraft Applications Overview; NASA: Washington, DC, USA, 2002.
- Sirosh, N. Hydrogen Composite Tank Program. In Proceedings of the 2002 U.S. DOE Hydrogen Program Review, Golden, CO, USA, 6–10 May 2002. [Google Scholar]
- Hydrogen Delivery Technical Team Roadmap. 2017. Available online: energy.gov (accessed on 13 June 2019).
- Krasae-In, S.; Stang, J.H.; Neksa, P. Development of large-scale hydrogen liquefaction processes from 1898 to 2009. Int. J. Hydrogen Energy 2010, 35, 4524–4533. [Google Scholar] [CrossRef]
- Petitpas, G.; Simon, A.J. Liquid Hydrogen Infrastructure Analysis; Lawrence Livermore National Laboratory: Livermore, CA, USA, 2017.
- Meneghelli, B.; Tamburello, D.; Fesmire, J.; Swanger, A. Integrated Insulation System for Automotive Cryogenic; U.S. DOE Hydrogen and Fuel Cells Program, 2017. Available online: https://www.hydrogen.energy.gov/pdfs/progress17/iv_d_4_meneghelli_2017.pdf (accessed on 13 June 2019).
- D1655-18a; ASTM International: West Conshohocken, PA, USA, 2018.
- Crabtree, G.W.; Dresselhaus, M.S.; Buchanan, M.V. The Hydrogen Economy. Phys. Today 2004, 57, 39–44. [Google Scholar] [CrossRef]
- Moreno-Blanco, J.; Petitpas, G.; Espinosa-Loza, F.; Elizalde-Blancas, F.; Martinez-Frias, J.; Aceves, S.M. The fill density of automotive cryo-compressed hydrogen vessels. Int. J. Hydrogen Energy 2019, 44, 1010–1020. [Google Scholar] [CrossRef]
- Kunze, K.; Oliver, K. Cryo-Compressed Hydrogen Storage; BMW Group: Munich, Germany, 2012. [Google Scholar]
- Ahluwalia, R.K.; Peng, J.-K.; Hua, T.Q. Cryo-compressed hydrogen storage. In Compendium of Hydrogen Energy; Woodhead Publishing: Cambridge, UK, 2016; pp. 119–145. [Google Scholar]
- Argonne National Laboratory. Technical Assessment of Cryo-Compressed Hydrogen Storage Tank Systems for Automotive Applications; U.S. Department of Energy: Oak Ridge, TN, USA, 2009.
- Ahluwalia, R.; Peng, J.; Roh, H.; Hua, T.; Houchins, C.; James, B. Supercritical cryo-compressed hydrogen storage for fuel cell electric buses. Int. J. Hydrogen Energy 2018, 43, 10215–10231. [Google Scholar] [CrossRef]
- Reed, R.; Golda, M. Cryogenic properties of unidirectional composites. Cryogenics 1994, 34, 909–928. [Google Scholar] [CrossRef]
- Ahluwalia, R.K.; Hua, T.Q.; Peng, J.-K.; Lasher, S.; McKenney, K.; Sinha, J.; Llc, T. Nuclear Engineering Division Technical assessment of cryo-compressed hydrogen storage tank systems for automotive applications. Int. J. Hydrogen Energy 2010, 35, 4171–4184. [Google Scholar] [CrossRef]
- Aceves, S.M.; Petitpas, G.; Espinosa-Loza, F.; Matthews, M.J.; Ledesma-Orozco, E. Safe, long range, inexpensive and rapidly refuelable hydrogen vehicles with cryogenic pressure vessels. Int. J. Hydrogen Energy 2013, 38, 2480–2489. [Google Scholar] [CrossRef]
- He, Y.; Chen, F.; Li, B.; Qian, G.; Zhou, W.; Chen, B. Porous metal–organic frameworks for fuel storage. Coord. Chem. Rev. 2018, 373, 167–198. [Google Scholar] [CrossRef]
- Zhao, R.; Liang, Z.; Zou, R.; Xu, Q. Metal-Organic Frameworks for Batteries. Joule 2018, 2, 2235–2259. [Google Scholar] [CrossRef] [Green Version]
- Mehtab, T.; Yasin, G.; Arif, M.; Shakeel, M.; Korai, R.M.; Nadeem, M.; Muhammad, N.; Lu, X. Metal-organic frameworks for energy storage devices: Batteries and supercapacitors. J. Energy Storage 2019, 21, 632–646. [Google Scholar] [CrossRef]
- Qiu, J.; Zhang, X.; Feng, Y.; Zhang, X.; Wang, H.; Yao, J. Modified metal-organic frameworks as photocatalysts. Appl. Catal. B Environ. 2018, 231, 317–342. [Google Scholar] [CrossRef]
- Lan, G.; Ni, K.; Lin, W. Nanoscale metal–organic frameworks for phototherapy of cancer. Coord. Chem. Rev. 2019, 379, 65–81. [Google Scholar] [CrossRef]
- Rosi, N.L.; Eckert, J.; Eddaoudi, M.; Vodak, D.T.; Kim, J.; O’Keeffe, M.; Yaghi, O.M. Hydrogen Storage in Microporous Metal-Organic Frameworks. Science 2003, 300, 1127–1129. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, A.; Liu, Y.; Purewal, J.; Tran, L.D.; Wong-Foy, A.G.; Veenstra, M.; Matzger, A.J.; Siegel, D.J. Balancing gravimetric and volumetric hydrogen density in MOFs. Energy Environ. Sci. 2017, 10, 2459–2471. [Google Scholar] [CrossRef]
- Choi, J.-S.; Son, W.-J.; Kim, J.; Ahn, W.-S. Metal–organic framework MOF-5 prepared by microwave heating: Factors to be considered. Microporous Mesoporous Mater. 2008, 116, 727–731. [Google Scholar] [CrossRef]
- Koizumi, K.; Nobusada, K.; Boero, M. Hydrogen storage mechanism and diffusion in metal–organic frameworks. Phys. Chem. Chem. Phys. 2019, 21, 7756–7764. [Google Scholar] [CrossRef] [PubMed]
- Saha, D.; Wei, Z.; Deng, S. Equilibrium, kinetics and enthalpy of hydrogen adsorption in MOF-177. Int. J. Hydrogen Energy 2008, 33, 7479–7488. [Google Scholar] [CrossRef]
- Yuan, S.; Feng, L.; Wang, K.; Pang, J.; Bosch, M.; Lollar, C.; Sun, Y.; Qin, J.; Yang, X.; Zhang, P.; et al. Stable Metal-Organic Frameworks: Design, Synthesis, and Applications. Adv. Mater. 2018, 30, e1704303. [Google Scholar] [CrossRef] [PubMed]
- Wikipedia Contributors. MOF-5. Wikipedia, The Free Encyclopedia. Available online: https://de.wikipedia.org/wiki/MOF-5 (accessed on 13 February 2019).
- Chakraborty, A.; Kumar, S. Thermal management and desorption modeling of a cryo-adsorbent hydrogen storage system. Int. J. Hydrogen Energy 2013, 38, 3973–3986. [Google Scholar] [CrossRef]
- Richard, M.-A.; Benard, P.; Chahine, R. Gas adsorption process in activated carbon over a wide temperature range above the critical point. Part 1: Modified Dubinin-Astakhov model. Adsorpt 2009, 15, 43–51. [Google Scholar] [CrossRef]
- Richard, M.-A.; Bénard, P.; Chahine, R. Gas adsorption process in activated carbon over a wide temperature range above the critical point. Part 2: Conservation of mass and energy. Adsorption 2009, 15, 53–63. [Google Scholar] [CrossRef]
- Kumar, V.S.; Raghunathan, K.; Kumar, S. A lumped-parameter model for cryo-adsorber hydrogen storage tank. Int. J. Hydrogen Energy 2009, 34, 5466–5475. [Google Scholar] [CrossRef]
- Kumar, V.S.; Kumar, S. Generalized model development for a cryo-adsorber and 1-D results for the isobaric refueling period. Int. J. Hydrogen Energy 2010, 35, 3598–3609. [Google Scholar] [CrossRef]
- Ghosh, I.; Naskar, S.; Bandyopadhyay, S.S. Cryosorption storage of gaseous hydrogen for vehicular application–A conceptual design. Int. J. Hydrogen Energy 2010, 35, 161–168. [Google Scholar] [CrossRef]
- Hermosilla-Lara, G.; Momen, G.; Marty, P.; Le Neindre, B.; Hassouni, K. Hydrogen storage by adsorption on activated carbon: Investigation of the thermal effects during the charging process. Int. J. Hydrogen Energy 2007, 32, 1542–1553. [Google Scholar] [CrossRef] [Green Version]
- Momen, G.; Hermosilla, G.; Michau, A.; Pons, M.; Firdaous, M.; Marty, P.; Hassouni, K. Experimental and numerical investigation of the thermal effects during hydrogen charging in packed bed storage tank. Int. J. Heat Mass Transf. 2009, 52, 1495–1503. [Google Scholar] [CrossRef]
- Zhan, L.; Li, K.; Zhang, R.; Liu, Q.; Lü, C.; Ling, L. Improvements of the DA equation for application in hydrogen adsorption at supercritical conditions. J. Supercrit. Fluids 2004, 28, 37–45. [Google Scholar] [CrossRef]
- Paggiaro, R.; Michl, F.; Benard, P.; Polifke, W. Cryo-adsorptive hydrogen storage on activated carbon. II: Investigation of the thermal effects during filling at cryogenic temperatures. Int. J. Hydrogen Energy 2010, 35, 648–659. [Google Scholar] [CrossRef]
- Schütz, W.; Michl, F.; Polifke, W.; Paggiaro, R. Storage System for Storing a Medium and Method for Loading a Storage System with a Storage Medium and Emptying the Same Therefrom. U.S. Patent Application No. 10/578,172, 24 January 2008. [Google Scholar]
- Xiao, J.; Tong, L.; Deng, C.; Benard, P.; Chahine, R. Simulation of heat and mass transfer in activated carbon tank for hydrogen storage. Int. J. Hydrogen Energy 2010, 35, 8106–8116. [Google Scholar] [CrossRef]
- Vasiliev, L.L.; Kanonchik, L.E. Activated carbon fibres and composites on its base for high performance hydrogen storage system. Chem. Eng. Sci. 2010, 65, 2586–2595. [Google Scholar] [CrossRef]
- Huang, B.; Ni, Z.; Millward, A.; McGaughey, A.; Uher, C.; Kaviany, M.; Yaghi, O. Thermal conductivity of a metal-organic framework (MOF-5): Part II. Measurement. Int. J. Heat Mass Transf. 2007, 50, 405–411. [Google Scholar] [CrossRef]
- Hardy, B.; Tamburello, D.; Corgnale, C. Hydrogen storage adsorbent systems acceptability envelope. Int. J. Hydrogen Energy 2018, 43, 19528–19539. [Google Scholar] [CrossRef]
- Proch, S.; Herrmannsdörfer, J.; Kempe, R.; Kern, C.; Jess, A.; Seyfarth, L.; Senker, J. Pt@MOF-177: Synthesis, Room-Temperature Hydrogen Storage and Oxidation Catalysis. Chem. A Eur. J. 2019, 14, 8204–8212. [Google Scholar] [CrossRef]
- Rubio-Martinez, M.; Avci-Camur, C.; Thornton, A.W.; Imaz, I.; Maspoch, D.; Hill, M.R. New synthetic routes towards MOF production at scale. Chem. Soc. Rev. 2017, 46, 3453–3480. [Google Scholar] [CrossRef] [Green Version]
- DeSantis, D.; Mason, J.A.; James, B.D.; Houchins, C.; Long, J.R.; Veenstra, M. Techno-economic Analysis of Metal–Organic Frameworks for Hydrogen and Natural Gas Storage. Energy Fuels 2017, 31, 2024–2032. [Google Scholar] [CrossRef]
- Cheng, J.; Yuan, X.; Zhao, L.; Huang, D.; Zhao, M.; Dai, L.; Ding, R. GCMC simulation of hydrogen physisorption on carbon nanotubes and nanotube arrays. Carbon 2004, 42, 2019–2024. [Google Scholar] [CrossRef]
- Ariharan, A.; Viswanathan, B.; Nandhakumar, V. Nitrogen-incorporated carbon nanotube derived from polystyrene and polypyrrole as hydrogen storage material. Int. J. Hydrogen Energy 2018, 43, 5077–5088. [Google Scholar] [CrossRef]
- Kaskun, S.; Kayfeci, M. The synthesized nickel-doped multi-walled carbon nanotubes for hydrogen storage under moderate pressures. Int. J. Hydrogen Energy 2018, 43, 10773–10778. [Google Scholar] [CrossRef]
- Silambarasan, D.; Surya, V.; Iyakutti, K.; Asokan, K.; Vasu, V.; Kawazoe, Y.; Kandasami, A. Gamma (γ)-ray irradiated multi-walled carbon nanotubes (MWCNTs) for hydrogen storage. Appl. Surf. Sci. 2017, 418, 49–55. [Google Scholar] [CrossRef]
- Masika, E.; Mokaya, R. Preparation of ultrahigh surface area porous carbons templated using zeolite 13X for enhanced hydrogen storage. Prog. Nat. Sci. 2013, 23, 308–316. [Google Scholar] [CrossRef] [Green Version]
- Ahn, C. Enhanced Hydrogen Dipole Physisorption. 2006. Available online: https://www.hydrogen.energy.gov/pdfs/review06/stp_16_ahn.pdf (accessed on 25 March 2019).
- Broom, D.; Webb, C.; Fanourgakis, G.; Froudakis, G.; Trikalitis, P.; Hirscher, M. Concepts for improving hydrogen storage in nanoporous materials. Int. J. Hydrogen Energy 2019, 44, 7768–7779. [Google Scholar] [CrossRef]
- Shriniwasan, S.; Gor, N.; Tatiparti, S.S.V. Hydrogen Sorption Mechanism of Magnesium (Hydride). Mater. Today Proc. 2018, 5, 23235–23241. [Google Scholar] [CrossRef]
- Zuliani, D.; Reeson, D. Making Magnesium a More Cost and Environmentally Competitive Option. In Proceedings of the 9th International Conference on Magnesium Alloys and Their Applications, Vancouver, BC, Canada, 8–12 July 2012. [Google Scholar]
- Wu, G.; Zhang, J.; Li, Q.; Chou, K. A new model to describe absorption kinetics of Mg-based hydrogen storage alloys. Int. J. Hydrogen Energy 2011, 36, 12923–12931. [Google Scholar] [CrossRef]
- Li, J.; Zhou, C.; Fang, Z.Z.; Bowman, R.C., Jr.; Lu, J.; Ren, C. Isothermal hydrogenation kinetics of ball-milled nano-catalyzed magnesium hydride. Materials 2019, 5, 100227. [Google Scholar] [CrossRef]
- Vigelholm, B.; Kjøller, J.; Larsen, B.; Pedersen, A.S. Formation and decomposition of magnesium hydride. J. Less Common Met. 1983, 89, 135–144. [Google Scholar] [CrossRef]
- Paskevicius, M.; Sheppard, D.A.; Buckley, C.E. Thermodynamic Changes in Mechanochemically Synthesized Magnesium Hydride Nanoparticles. J. Am. Chem. Soc. 2010, 132, 5077–5083. [Google Scholar] [CrossRef] [PubMed]
- Zaluska, A.; Zaluski, L.; Ström–Olsen, J. Nanocrystalline magnesium for hydrogen storage. J. Alloy. Compd. 1999, 288, 217–225. [Google Scholar] [CrossRef]
- Yahya, M.; Ismail, M. Synergistic catalytic effect of SrTiO3 and Ni on the hydrogen storage properties of MgH2. Int. J. Hydrogen Energy 2018, 43, 6244–6255. [Google Scholar] [CrossRef]
- Bogdanović, B.; Felderhoff, M.; Pommerin, A.; Schüth, F.; Spielkamp, N. Advanced Hydrogen-Storage Materials Based on Sc-, Ce-, and Pr-Doped NaAlH4. Adv. Mater. 2006, 18, 1198–1201. [Google Scholar] [CrossRef]
- Rusman, N.; Dahari, M. A review on the current progress of metal hydrides material for solid-state hydrogen storage applications. Int. J. Hydrogen Energy 2016, 41, 12108–12126. [Google Scholar] [CrossRef]
- Garrier, S.; Chaise, A.; De Rango, P.; Marty, P.; Delhomme, B.; Fruchart, D.; Miraglia, S. MgH2 intermediate scale tank tests under various experimental conditions. Int. J. Hydrogen Energy 2011, 36, 9719–9726. [Google Scholar] [CrossRef]
- National Center for Biotechnology Information. Magnesium Hydride (CID=5486771). 2019. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/5486771 (accessed on 13 February 2019).
- Sakintuna, B.; Lamaridarkrim, F.; Hirscher, M. Metal hydride materials for solid hydrogen storage: A review. Int. J. Hydrogen Energy 2007, 32, 1121–1140. [Google Scholar] [CrossRef]
- Ley, M.B.; Jepsen, L.H.; Lee, Y.-S.; Cho, Y.W.; Von Colbe, J.M.B.; Dornheim, M.; Rokni, M.; Jensen, J.O.; Sloth, M.; Filinchuk, Y.; et al. Complex hydrides for hydrogen storage–new perspectives. Mater. Today 2014, 17, 122–128. [Google Scholar] [CrossRef]
- Li, C.; Peng, P.; Zhou, D.; Wan, L. Research progress in LiBH4 for hydrogen storage: A review. Int. J. Hydrogen Energy 2011, 36, 14512–14526. [Google Scholar] [CrossRef]
- Li, H.-W.; Akiba, E.; Orimo, S.-I. Comparative study on the reversibility of pure metal borohydrides. J. Alloys Compd. 2013, 580, S292–S295. [Google Scholar] [CrossRef]
- Sun, T.; Xiao, F.; Tang, R.; Wang, Y.; Dong, H.; Li, Z.; Wang, H.; Liuzhang, O.; Zhu, M. Hydrogen storage performance of nano Ni decorated LiBH4 on activated carbon prepared through organic solvent. J. Alloys Compd. 2014, 612, 287–292. [Google Scholar] [CrossRef]
- Lombardo, L.; Yang, H.; Züttel, A. Study of borohydride ionic liquids as hydrogen storage materials. J. Energy Chem. 2019, 33, 17–21. [Google Scholar] [CrossRef]
- Salmi, T.; Russo, V. Reaction engineering approach to the synthesis of sodium borohydride. Chem. Eng. Sci. 2019, 199, 79–87. [Google Scholar] [CrossRef]
- Lee, J.; Shin, H.; Choi, K.S.; Lee, J.; Choi, J.-Y.; Yu, H.K. Carbon layer supported nickel catalyst for sodium borohydride (NaBH4) dehydrogenation. Int. J. Hydrogen Energy 2019, 44, 2943–2950. [Google Scholar] [CrossRef]
- Au, M.; Walters, R.T. Reversibility aspect of lithium borohydrides. Int. J. Hydrogen Energy 2010, 35, 10311–10316. [Google Scholar] [CrossRef]
- Skipper, C.V.J.; Hamaed, A.; Antonelli, D.M.; Kaltsoyannis, N. The Kubas interaction in M(II) (M = Ti, V, Cr) hydrazine-based hydrogen storage materials: A DFT study. Dalton Trans. 2012, 41, 8515–8523. [Google Scholar] [CrossRef]
- Kubas, G.J. Hydrogen activation on organometallic complexes and H2 production, utilization, and storage for future energy. J. Organomet. Chem. 2009, 694, 2648–2653. [Google Scholar] [CrossRef]
- Kubas, G.J. Metal–dihydrogen and σ-bond coordination: The consummate extension of the Dewar–Chatt–Duncanson model for metal–olefin π bonding. J. Organomet. Chem. 2001, 635, 37–68. [Google Scholar] [CrossRef]
- Morris, L.; Hales, J.J.; Trudeau, M.L.; Georgiev, P.; Embs, J.P.; Eckert, J.; Kaltsoyannis, N.; Antonelli, D.M.; Embs, J.P.P. A manganese hydride molecular sieve for practical hydrogen storage under ambient conditions. Energy Environ. Sci. 2019, 12, 1580–1591. [Google Scholar] [CrossRef]
- Hales, J.J.; Trudeau, M.L.; Antonelli, D.M.; Kaltsoyannis, N. Computational study of H2 binding to MH3 (M = Ti, V, or Cr). Dalton Trans. 2019, 48, 4921–4930. [Google Scholar] [CrossRef]
- Preuster, P.; Papp, C.; Wasserscheid, P. Liquid Organic Hydrogen Carriers (LOHCs): Toward a Hydrogen-free Hydrogen Economy. Acc. Chem. Res. 2017, 50, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Hu, P.; Fogler, E.; Diskin-Posner, Y.; Iron, M.A.; Milstein, D. A novel liquid organic hydrogen carrier system based on catalytic peptide formation and hydrogenation. Nat. Commun. 2015, 6, 6859. [Google Scholar] [CrossRef] [PubMed]
- Taube, M.; Taube, P. A Liquid Organic Carrier of Hydrogen as a Fuel for Automobiles; Institut für Reaktorforschung: Würenlingen, Switzerland, 1979. [Google Scholar]
- Eblagon, K.M.; Rentsch, D.; Friedrichs, O.; Remhof, A.; Zuettel, A.; Ramirez-Cuesta, A.; Tsang, S.C.; Ramirez-Cuesta, A. Hydrogenation of 9-ethylcarbazole as a prototype of a liquid hydrogen carrier. Int. J. Hydrogen Energy 2010, 35, 11609–11621. [Google Scholar] [CrossRef]
- Geburtig, D.; Preuster, P.; Bösmann, A.; Müller, K.; Wasserscheid, P. Chemical utilization of hydrogen from fluctuating energy sources–Catalytic transfer hydrogenation from charged Liquid Organic Hydrogen Carrier systems. Int. J. Hydrogen Energy 2016, 41, 1010–1017. [Google Scholar] [CrossRef]
- He, T.; Pei, Q.; Chen, P. Liquid organic hydrogen carriers. J. Energy Chem. 2015, 24, 587–594. [Google Scholar] [CrossRef] [Green Version]
- Teichmann, D.; Arlt, W.; Wasserscheid, P. Liquid Organic Hydrogen Carriers as an efficient vector for the transport and storage of renewable energy. Int. J. Hydrogen Energy 2012, 37, 18118–18132. [Google Scholar] [CrossRef]
- Teichmann, D.; Arlt, W.; Wasserscheid, P.; Freymann, R. A future energy supply based on Liquid Organic Hydrogen Carriers (LOHC). Energy Environ. Sci. 2011, 4, 2767–2773. [Google Scholar] [CrossRef]
- Sotoodeh, F.; Smith, K.J. Structure sensitivity of dodecahydro-N-ethylcarbazole dehydrogenation over Pd catalysts. J. Catal. 2011, 279, 36–47. [Google Scholar] [CrossRef]
- Sotoodeh, F.; Zhao, L.; Smith, K.J. Kinetics of H2 recovery from dodecahydro-N-ethylcarbazole over a supported Pd catalyst. Appl. Catal. A Gen. 2009, 362, 155–162. [Google Scholar] [CrossRef]
- Gleichweit, C.; Schernich, S.; Lorenz, M.P.A.; Höfert, O.; Brückner, N.; Steinrück, H.-P.; Amende, M.; Zhao, W.; Wasserscheid, P.; Libuda, J.; et al. Dehydrogenation of Dodecahydro-N-ethylcarbazole on Pt(111). ChemSusChem 2013, 6, 974–977. [Google Scholar] [CrossRef] [PubMed]
- Sobota, M.; Nikiforidis, I.; Amende, M.; Zanón, B.S.; Staudt, T.; Höfert, O.; Lykhach, Y.; Papp, C.; Hieringer, W.; Laurin, M.; et al. Dehydrogenation of Dodecahydro-N-ethylcarbazole on Pd/Al2O3 Model Catalysts. Chem. A Eur. J. 2011, 17, 11542–11552. [Google Scholar] [CrossRef] [PubMed]
- Amende, M.; Schernich, S.; Sobota, M.; Nikiforidis, I.; Hieringer, W.; Assenbaum, D.; Gleichweit, C.; Drescher, H.-J.; Papp, C.; Steinrück, H.-P.; et al. Dehydrogenation Mechanism of Liquid Organic Hydrogen Carriers: Dodecahydro- N -ethylcarbazole on Pd(111). Chem. A Eur. J. 2013, 19, 10854–10865. [Google Scholar] [CrossRef] [PubMed]
- Amende, M.; Gleichweit, C.; Werner, K.; Schernich, S.; Zhao, W.; Lorenz, M.P.A.; Höfert, O.; Papp, C.; Koch, M.; Wasserscheid, P.; et al. Model Catalytic Studies of Liquid Organic Hydrogen Carriers: Dehydrogenation and Decomposition Mechanisms of Dodecahydro-N-ethylcarbazole on Pt(111). ACS Catal. 2014, 4, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, C.W.; Baker, R.T.; Staubitz, A.; Manners, I. B–N compounds for chemical hydrogen storage. Chem. Soc. Rev. 2009, 38, 279–293. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Gong, X.; Wang, B.; Wu, Z.; Fang, T. A experimental study on the dehydrogenation performance of dodecahydro-N-ethylcarbazole on M/TiO2 catalysts. Int. J. Hydrogen Energy 2019, 44, 2951–2959. [Google Scholar] [CrossRef]
- Glaister, B.J.; Mudd, G.M. The environmental costs of platinum–PGM mining and sustainability: Is the glass half-full or half-empty? Miner. Eng. 2010, 23, 438–450. [Google Scholar] [CrossRef]
- He, Y.; Song, Y.; Cullen, D.A.; Laursen, S. Selective and Stable Non-Noble-Metal Intermetallic Compound Catalyst for the Direct Dehydrogenation of Propane to Propylene. J. Am. Chem. Soc. 2018, 140, 14010–14014. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information. 9-Ethylcarbazole (CID=6836). PubChem Compound Database; 2019. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/6836 (accessed on 13 February 2019).
- Sultan, O.; Shaw, H. Study of Automotive Storage of Hydrogen using Recyclable Liquid Chemical Carriers. 1975. Available online: http://adsabs.harvard.edu/abs/1975STIN. 7633642S (accessed on 13 February 2019).
- Chiyoda Corporation. What Is “SPERA HYDROGEN” System? 2017. Available online: https://www.chiyodacorp.com/en/service/spera-hydrogen/innovations/ (accessed on 1 February 2019).
- Chiyoda Corporation. Performance of 10,000 Hours of Operation in Chiyoda’s Demo Plant. 2017. Available online: https://www.chiyodacorp.com/en/service/spera-hydrogen/demo-plant/ (accessed on 1 February 2019).
- Kim, H.-Y.; Kang, M.-G.; Kim, T.-G.; Kang, C.-W. Toxicity of Methylcyclohexane and Its Effect on the Reproductive System in SD Rats. Saf. Health Work 2011, 2, 290–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, J.W.; Neff, J.M.; Cox, B.A.; Tatem, H.E.; Hightower, G.M. Characteristics of dispersions and water-soluble extracts of crude and refined oils and their toxicity to estuarine crustaceans and fish. Mar. Boil. 1974, 27, 75–88. [Google Scholar] [CrossRef]
- Campo, P.; Lataye, R.; Cossec, B.; Placidi, V. Toluene-induced hearing loss: A mid-frequency location of the cochlear lesions. Neurotoxicol. Teratol. 1997, 19, 129–140. [Google Scholar] [CrossRef]
- Hydrogeit. Dibenzyltoluene: The Future of Hydrogen Storage. 2018. Available online: https://www.h2-international.com/2018/09/03/dibenzyltoluene-the-future-of-hydrogen-storage/ (accessed on 1 February 2019).
- Leinweber, A.; Müller, K. Hydrogenation of the Liquid Organic Hydrogen Carrier Compound Monobenzyl Toluene: Reaction Pathway and Kinetic Effects. Energy Technol. 2018, 6, 513–520. [Google Scholar] [CrossRef]
- Modisha, P.M.; Jordaan, J.H.; Bösmann, A.; Wasserscheid, P.; Bessarabov, D. Analysis of reaction mixtures of perhydro-dibenzyltoluene using two-dimensional gas chromatography and single quadrupole gas chromatography. Int. J. Hydrogen Energy 2018, 43, 5620–5636. [Google Scholar] [CrossRef]
- Heller, A.; Rausch, M.H.; Schulz, P.S.; Wasserscheid, P.; Fröba, A.P. Binary Diffusion Coefficients of the Liquid Organic Hydrogen Carrier System Dibenzyltoluene/Perhydrodibenzyltoluene. J. Chem. Eng. Data 2016, 61, 504–511. [Google Scholar] [CrossRef]
- Müller, K.; Stark, K.; Emel’Yanenko, V.N.; Varfolomeev, M.A.; Zaitsau, D.H.; Shoifet, E.; Schick, C.; Verevkin, S.P.; Arlt, W. Liquid Organic Hydrogen Carriers: Thermophysical and Thermochemical Studies of Benzyl- and Dibenzyl-toluene Derivatives. Ind. Eng. Chem. Res. 2015, 54, 7967–7976. [Google Scholar] [CrossRef]
- Müller, K.; Aslam, R.; Fischer, A.; Stark, K.; Wasserscheid, P.; Arlt, W. Experimental assessment of the degree of hydrogen loading for the dibenzyl toluene based LOHC system. Int. J. Hydrogen Energy 2016, 41, 22097–22103. [Google Scholar] [CrossRef]
- Brückner, N.; Obesser, K.; Bösmann, A.; Teichmann, D.; Arlt, W.; Dungs, J.; Wasserscheid, P. Evaluation of Industrially Applied Heat-Transfer Fluids as Liquid Organic Hydrogen Carrier Systems. ChemSusChem 2014, 7, 229–235. [Google Scholar]
- Arkema. GPS Safety Summary: Dibenzyltoluene. 2013. Available online: https://www.arkema.com/export/shared/.content/media/downloads/socialresponsability/safety-summuries/Hydrogen-Peroxide-Dibenzyltoluene-GPS-2013-02-10-V0.pdf (accessed on 1 February 2019).
- Shi, L.; Qi, S.; Qu, J.; Che, T.; Yi, C.; Yang, B. Integration of hydrogenation and dehydrogenation based on dibenzyltoluene as liquid organic hydrogen energy carrier. Int. J. Hydrogen Energy 2019, 44, 5345–5354. [Google Scholar] [CrossRef]
- Rönsch, S.; Schneider, J.; Matthischke, S.; Schluter, M.; Götz, M.; Lefebvre, J.; Prabhakaran, P.; Bajohr, S. Review on methanation–From fundamentals to current projects. Fuel 2016, 166, 276–296. [Google Scholar] [CrossRef]
- Götz, M.; Lefebvre, J.; Mörs, F.; Koch, A.M.; Graf, F.; Bajohr, S.; Reimert, R.; Kolb, T. Renewable Power-to-Gas: A technological and economic review. Renew. Energy 2016, 85, 1371–1390. [Google Scholar] [CrossRef] [Green Version]
- Thauer, R.K.; Kaster, A.-K.; Seedorf, H.; Buckel, W.; Hedderich, R. Methanogenic archaea: Ecologically relevant differences in energy conservation. Nat. Rev. Genet. 2008, 6, 579–591. [Google Scholar] [CrossRef] [PubMed]
- Seifert, A.; Rittmann, S.K.-M.R.; Herwig, C. Analysis of process related factors to increase volumetric productivity and quality of biomethane with Methanothermobacter marburgensis. Appl. Energy 2014, 132, 155–162. [Google Scholar] [CrossRef]
- Hashimoto, K.; Yamasaki, M.; Fujimura, K.; Matsui, T.; Izumiya, K.; Komori, M.; El-Moneim, A.; Akiyama, E.; Habazaki, H.; Kumagai, N.; et al. Global CO2 recycling—novel materials and prospect for prevention of global warming and abundant energy supply. Mater. Sci. Eng. A 1999, 267, 200–206. [Google Scholar] [CrossRef]
- Götz, M.; Bajohr, S.; Buchholz, D. Speicherung elektrischer Energie aus regenerativen Quellen im Erdgasnetz. Energ. Wasser Prax. 2011, 62, 72–76. [Google Scholar]
- Lefebvre, J.; Götz, M.; Bajohr, S.; Reimert, R.; Kolb, T. Improvement of three-phase methanation reactor performance for steady-state and transient operation. Fuel Process. Technol. 2015, 132, 83–90. [Google Scholar] [CrossRef]
- de Vasconcelos, B.R.; Minh, D.P.; Lyczko, N.; Phan, T.S.; Sharrock, P.; Nzihou, A. Upgrading greenhouse gases (methane and carbon dioxide) into syngas using nickel-based catalysts. Fuel 2018, 226, 195–203. [Google Scholar] [CrossRef] [Green Version]
- Weger, L.; Abánades, A.; Butler, T. Methane cracking as a bridge technology to the hydrogen economy. Int. J. Hydrogen Energy 2017, 42, 720–731. [Google Scholar] [CrossRef] [Green Version]
- Joglekar, M.; Nguyen, V.; Pylypenko, S.; Ngo, C.; Li, Q.; O’Reilly, M.E.; Gray, T.S.; Hubbard, W.A.; Gunnoe, T.B.; Herring, A.M.; et al. Organometallic Complexes Anchored to Conductive Carbon for Electrocatalytic Oxidation of Methane at Low Temperature. J. Am. Chem. Soc. 2016, 138, 116–125. [Google Scholar] [CrossRef] [PubMed]
- Douvartzides, S.; Coutelieris, F.; Tsiakaras, P. Exergy analysis of a solid oxide fuel cell power plant fed by either ethanol or methane. J. Power Sources 2004, 131, 224–230. [Google Scholar] [CrossRef]
- Amiri, A.; Tang, S.; Steinberger-Wilckens, R.; Tadé, M.O. Evaluation of fuel diversity in Solid Oxide Fuel Cell system. Int. J. Hydrogen Energy 2018, 43, 23475–23487. [Google Scholar] [CrossRef] [Green Version]
- Valera-Medina, A.; Xiao, H.; Owen-Jones, M.; David, W.I.F.; Bowen, P.J. Ammonia for power. Prog. Energy Combust. Sci. 2018, 69, 63–102. [Google Scholar] [CrossRef]
- Lamb, K.E.; Dolan, M.D.; Kennedy, D.F. Ammonia for hydrogen storage; A review of catalytic ammonia decomposition and hydrogen separation and purification. Int. J. Hydrogen Energy 2019, 44, 3580–3593. [Google Scholar] [CrossRef]
- Christensen, C.H.; Johannessen, T.; Sørensen, R.Z.; Nørskov, J.K. Towards an ammonia-mediated hydrogen economy? Catal. Today 2006, 111, 140–144. [Google Scholar] [CrossRef]
- Perna, A.; Minutillo, M.; Jannelli, E.; Cigolotti, V.; Nam, S.; Han, J. Design and performance assessment of a combined heat, hydrogen and power (CHHP) system based on ammonia-fueled SOFC. Appl. Energy 2018, 231, 1216–1229. [Google Scholar] [CrossRef]
- Aziz, M.; Oda, T.; Morihara, A.; Kashiwagi, T. Combined nitrogen production, ammonia synthesis, and power generation for efficient hydrogen storage. Energy Procedia 2017, 143, 674–679. [Google Scholar] [CrossRef]
- Yapicioglu, A.; Dincer, I. A review on clean ammonia as a potential fuel for power generators. Renew. Sustain. Energy Rev. 2019, 103, 96–108. [Google Scholar] [CrossRef]
- Cinti, G.; Frattini, D.; Desideri, U.; Bidini, G.; Jannelli, E. Coupling Solid Oxide Electrolyser (SOE) and ammonia production plant. Appl. Energy 2017, 192, 466–476. [Google Scholar] [CrossRef] [Green Version]
- Lange, N.A.; Dean, J.A. Lange’s Handbook of Chemistry; McGraw-Hill: New York, NY, USA, 1967. [Google Scholar]
- Siddiqui, O.; Dincer, I. A review and comparative assessment of direct ammonia fuel cells. Therm. Sci. Eng. Prog. 2018, 5, 568–578. [Google Scholar] [CrossRef]
- Afif, A.; Radenahmad, N.; Cheok, Q.; Shams, S.; Kim, J.H.; Azad, A.K. Ammonia-fed fuel cells: A comprehensive review. Renew. Sustain. Energy Rev. 2016, 60, 822–835. [Google Scholar] [CrossRef]
- Cooper, S.J.; Brandon, N.P. Chapter 1 - An Introduction to Solid Oxide Fuel Cell Materials, Technology and Applications. In Solid Oxide Fuel Cell Lifetime and Reliability; Academic Press: Cambridge, MA, USA, 2017; pp. 1–18. [Google Scholar]
- Schmidt, O.; Gambhir, A.; Staffell, I.; Hawkes, A.; Nelson, J.; Few, S. Future cost and performance of water electrolysis: An expert elicitation study. Int. J. Hydrogen Energy 2017, 42, 30470–30492. [Google Scholar] [CrossRef]
- Kobayashi, H.; Hayakawa, A.; Somarathne, K.D.K.A.; Okafor, E.C. Science and technology of ammonia combustion. Proc. Combust. Inst. 2018, 37, 109–133. [Google Scholar] [CrossRef]
- Berg, J.M.; Tymoczko, J.L.; Stryer, L. Biochemistry; W.H. Freeman: New York, NY, USA, 2002. [Google Scholar]
- Wikipedia Contributors. Haber Process. Wikipedia, The Free Encyclopedia. Available online: https://en.wikipedia.org/wiki/Haber_process (accessed on 19 February 2019).





| Property | Hydrogen | Natural Gas |
|---|---|---|
| Lower heating value (LHV, MJ/kg) | 120 [53] | 52 [53] |
| Higher heating value (HHV, MJ/kg) | 142 [53] | 47 [53] |
| Density at 273 K (kg/m3) | 0.09 [54] | 0.65 [54] |
| Boiling point at atmospheric pressure(K) | 20.3 [54] | 111.2 [55] |
| Liquid density (kg/m3) | 70.8 [54] | 450.0 [55] |
| Flammability concentration limits in air (vol %) | 4–75 [54] | 5–15 [54] |
| Diffusion coefficient in air (cm2/s) | 0.61 [54] | 0.16 [54] |
| Storage System Targets | Gravimetric Density System (wt %) | Volumetric Density System (MJ/L) | Cost ($/kWh) |
|---|---|---|---|
| Current status (700 bar compressed) | 4.2 | 2.9 | 15 |
| 2020 | 4.5 | 3.6 | 10 |
| Ultimate | 6.5 | 6.1 | 8 |
| Type | Materials | Typical Pressure (bar) | Cost ($/kg) | Gravimetric Density (wt %) |
|---|---|---|---|---|
| I | All-metal construction | 300 | 83 | 1.7 |
| II | Mostly metal, composite overwrap in the hoop direction | 200 | 86 | 2.1 |
| III | Metal liner, full composite overwrap | 700 | 700 [65] | 4.2 [66] |
| IV | All-composite construction | 700 | 633 [65] | 5.7 (Toyota Mirai) |
| Method | Gravimetric Energy Density (wt %) | Volumetric Energy Density (MJ/L) | Temperature (K) | Pressure (barg) | Remarks |
|---|---|---|---|---|---|
| Compressed | 5.7 | 4.9 | 293 | 700 | Current industry standard |
| Liquid | 7.5 | 6.4 | 20 | 0 | Boil-off constitues major disadvantage |
| Cold/cryo compressed | 5.4 | 4.0 | 40–80 | 300 | Boil-off constitues major disadvantage |
| MOF | 4.5 | 7.2 | 78 | 20–100 | Attractive densities only at very low temperatures. |
| Carbon nanostructures | 2.0 | 5.0 | 298 | 100 | Volumetric density based on powder density of 2.1 g/mL and 2.0 wt % storage capacity. |
| Metal hydrides | 7.6 | 13.2 | 260–425 | 20 | Requires thermal management system. |
| Metal borohydrides | 14.9–18.5 | 9.8–17.6 | 130 | 105 | Low temperature, high pressure thermal management required |
| Kubas-type | 10.5 | 23.6 | 293 | 120 | |
| LOHC | 8.5 | 7 | 293 | 0 | Highly endo/exothermal requires processing plant and catalyst. Not suitable for mobility |
| Chemical | 15.5 | 11.5 | 298 | 10 | Requires SOFC fuel cell. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivard, E.; Trudeau, M.; Zaghib, K. Hydrogen Storage for Mobility: A Review. Materials 2019, 12, 1973. https://doi.org/10.3390/ma12121973
Rivard E, Trudeau M, Zaghib K. Hydrogen Storage for Mobility: A Review. Materials. 2019; 12(12):1973. https://doi.org/10.3390/ma12121973
Chicago/Turabian StyleRivard, Etienne, Michel Trudeau, and Karim Zaghib. 2019. "Hydrogen Storage for Mobility: A Review" Materials 12, no. 12: 1973. https://doi.org/10.3390/ma12121973







