Corrosion Performance of Nano-ZrO2 Modified Coatings in Hot Mixed Acid Solutions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Preparation of Nano-ZrO2 Concentrates
2.3. Preparation of Nano-ZrO2-Modified Coatings
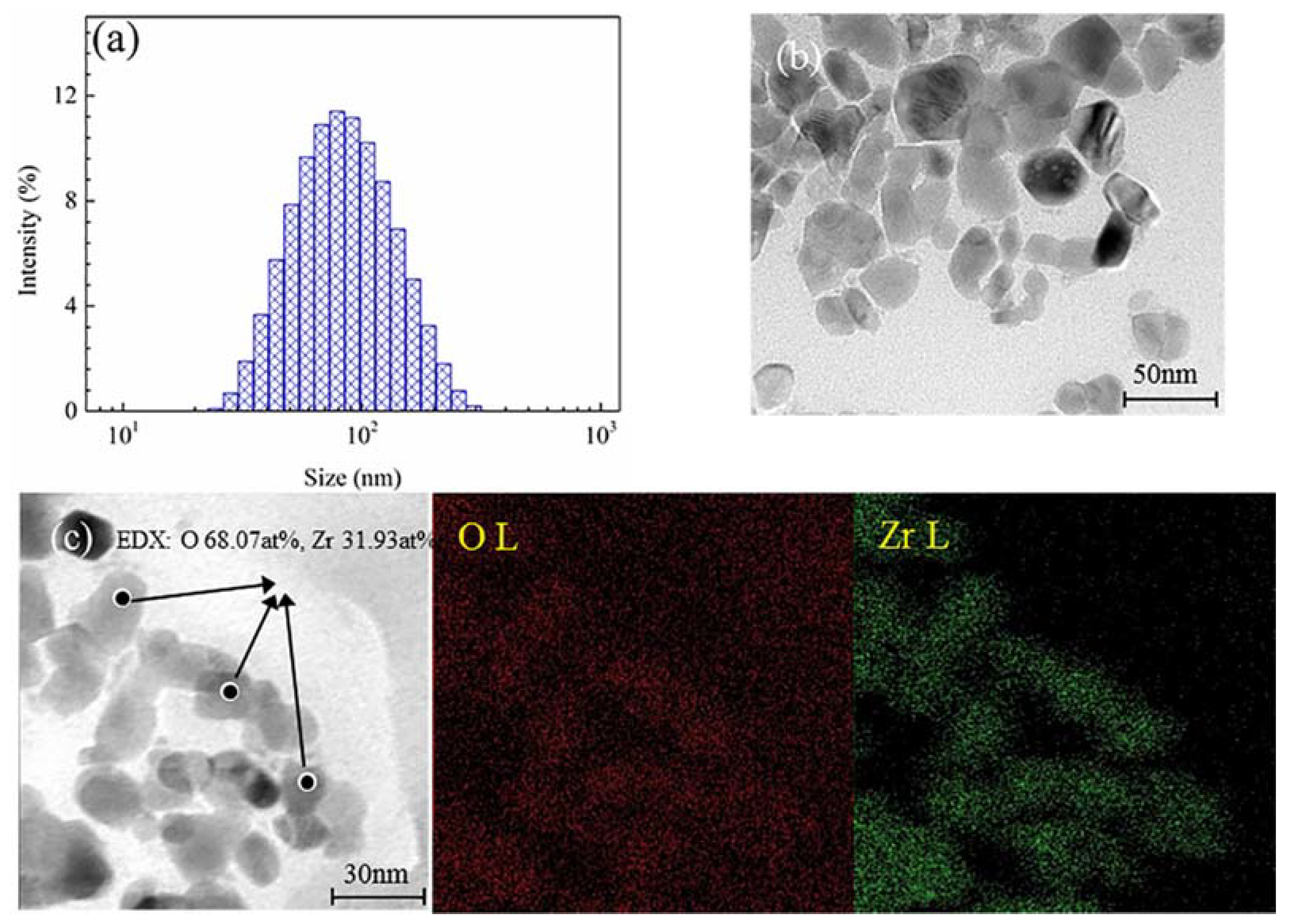
2.4. Particle Size Analysis of Nano-ZrO2 Concentrate
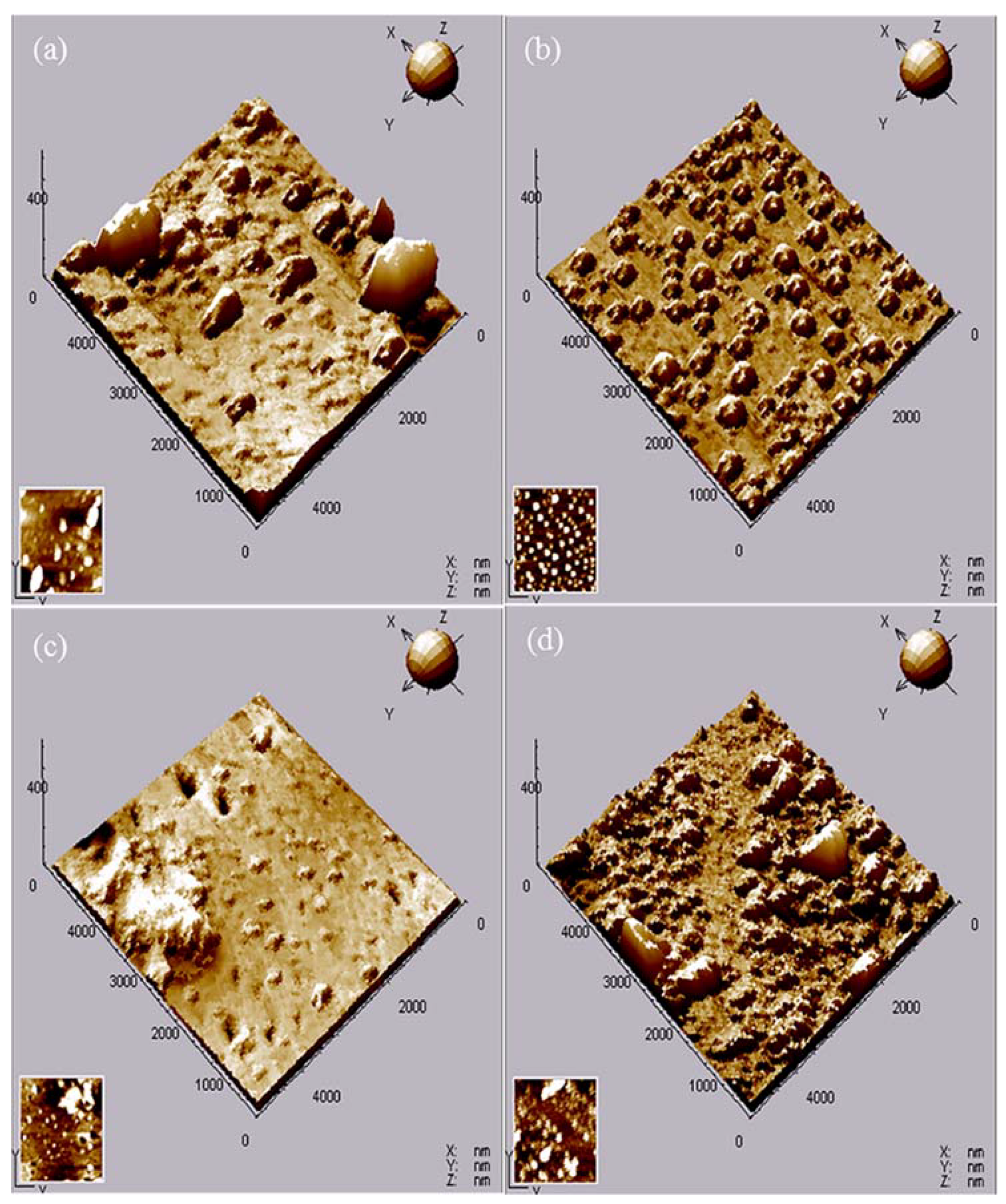
2.5. Surface Properties
2.6. Electrochemical Studies
3. Results and Discussion
3.1. Characterization of Nano-ZrO2 Concentrates
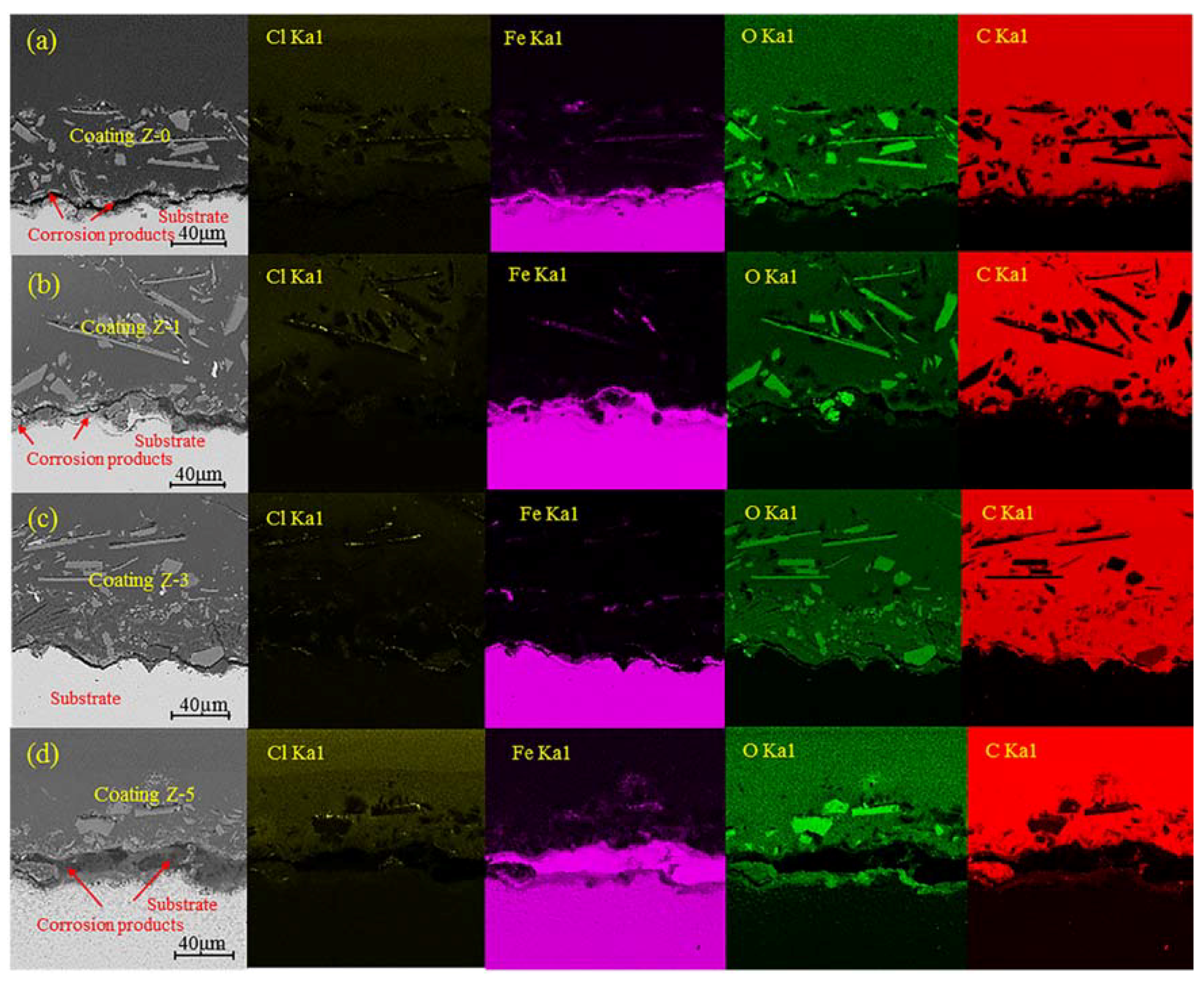
3.2. Acid Immersion Test and Corrosion Morphologies
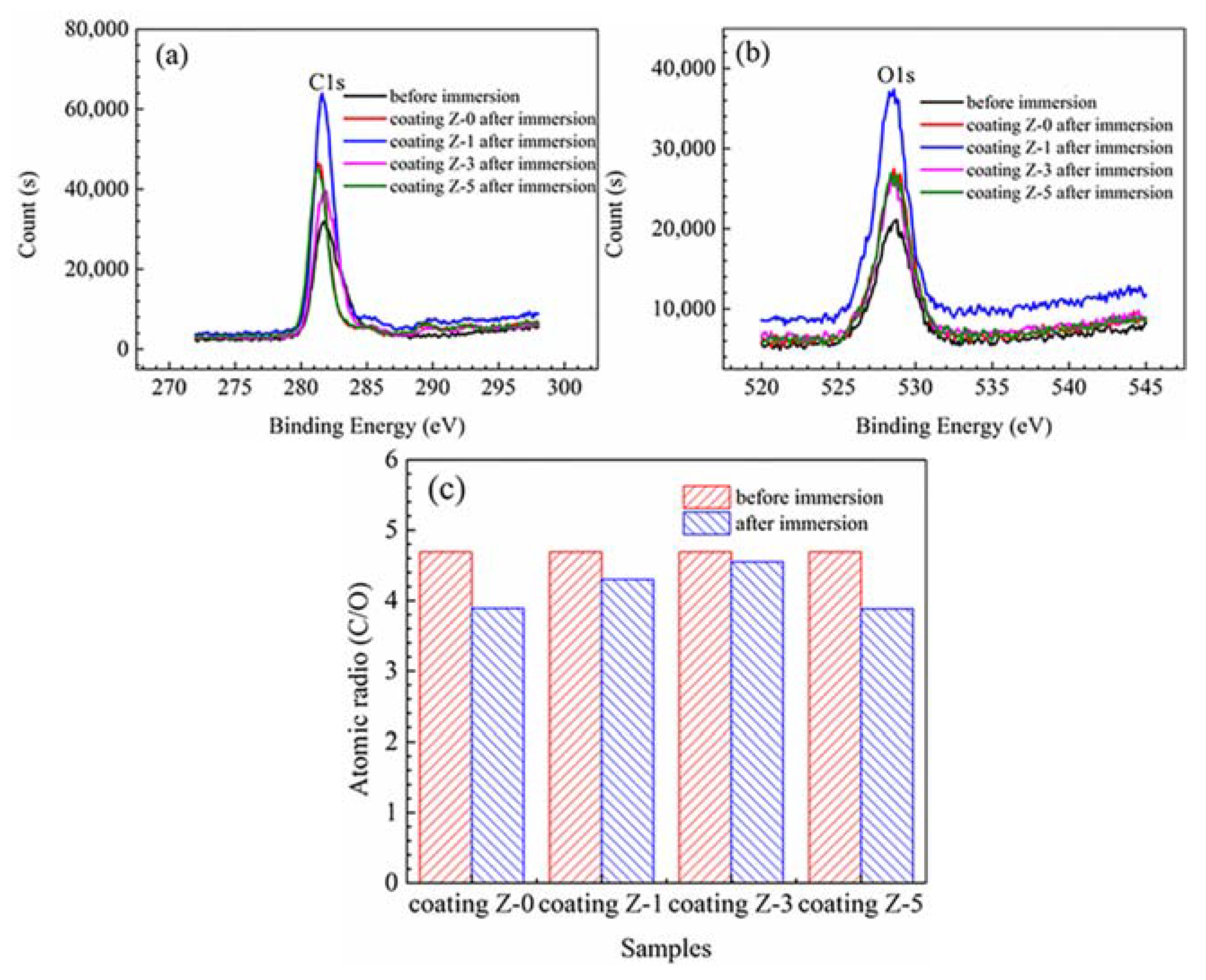
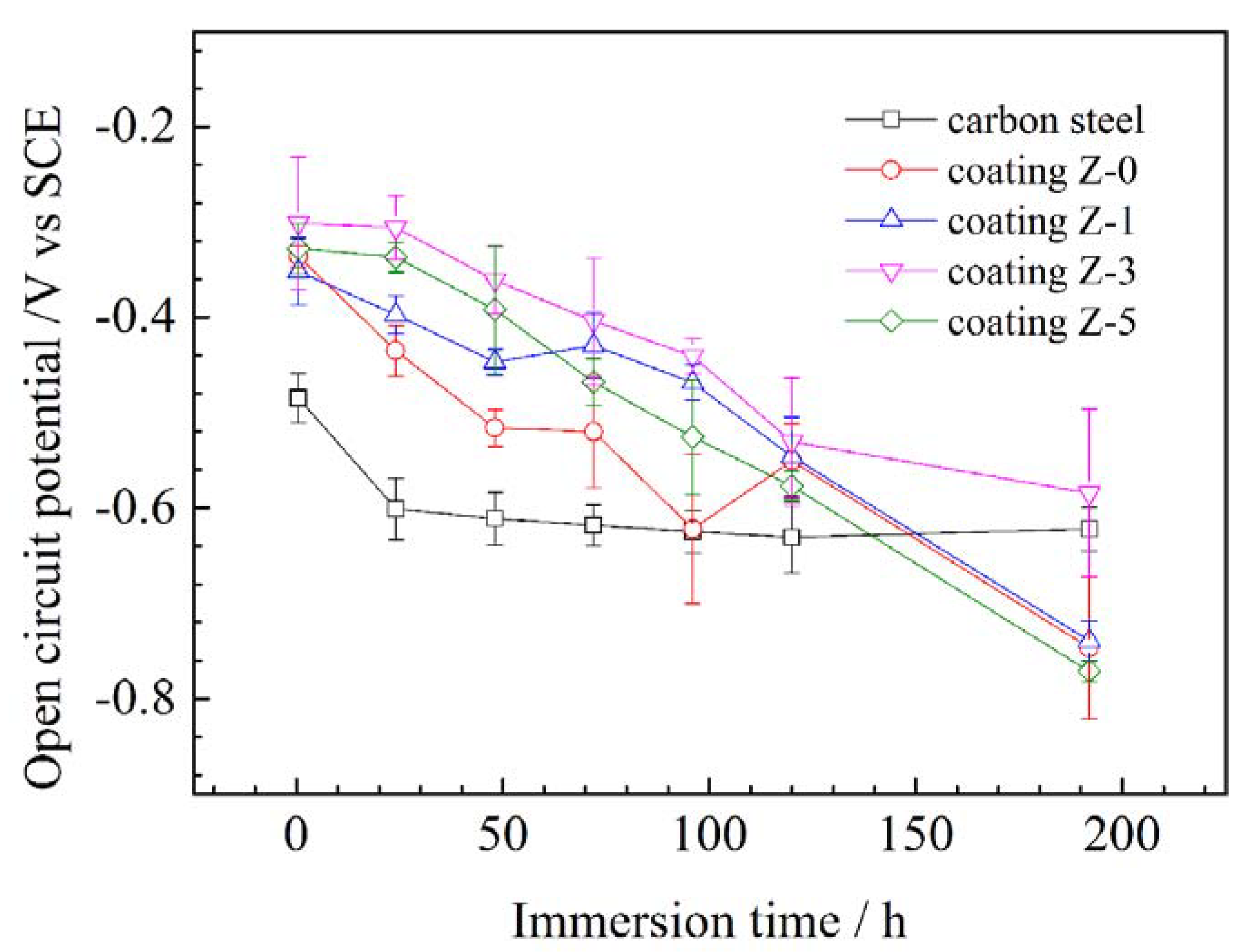
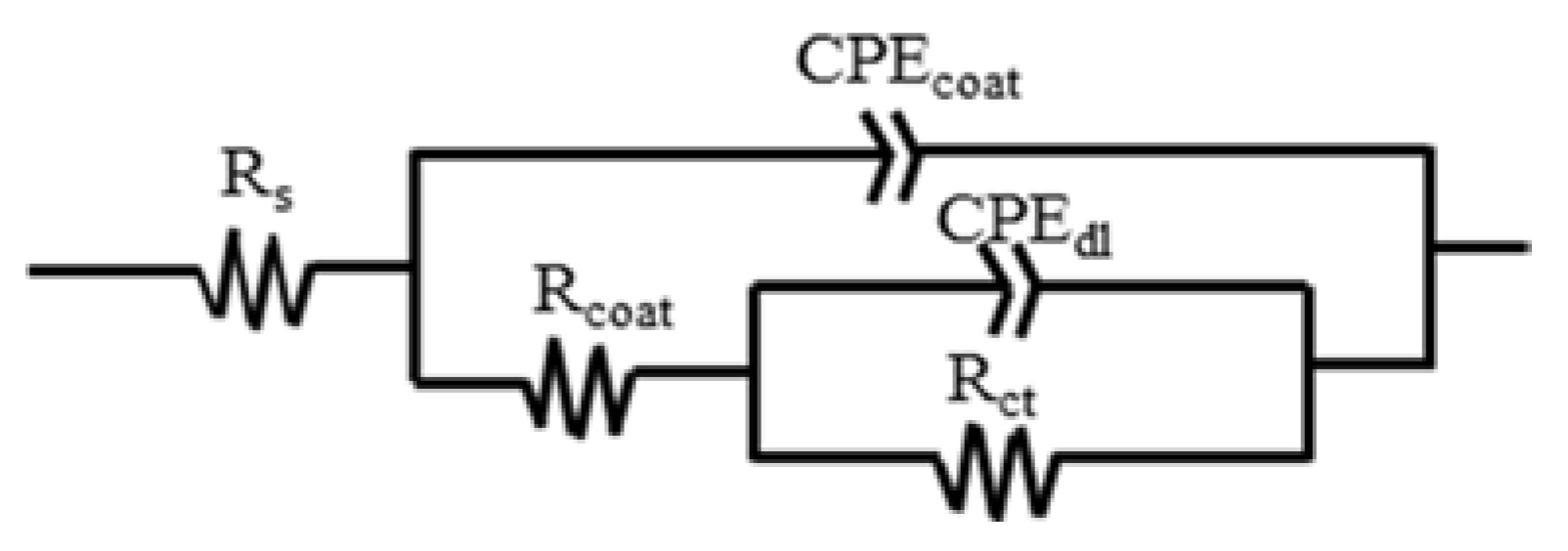
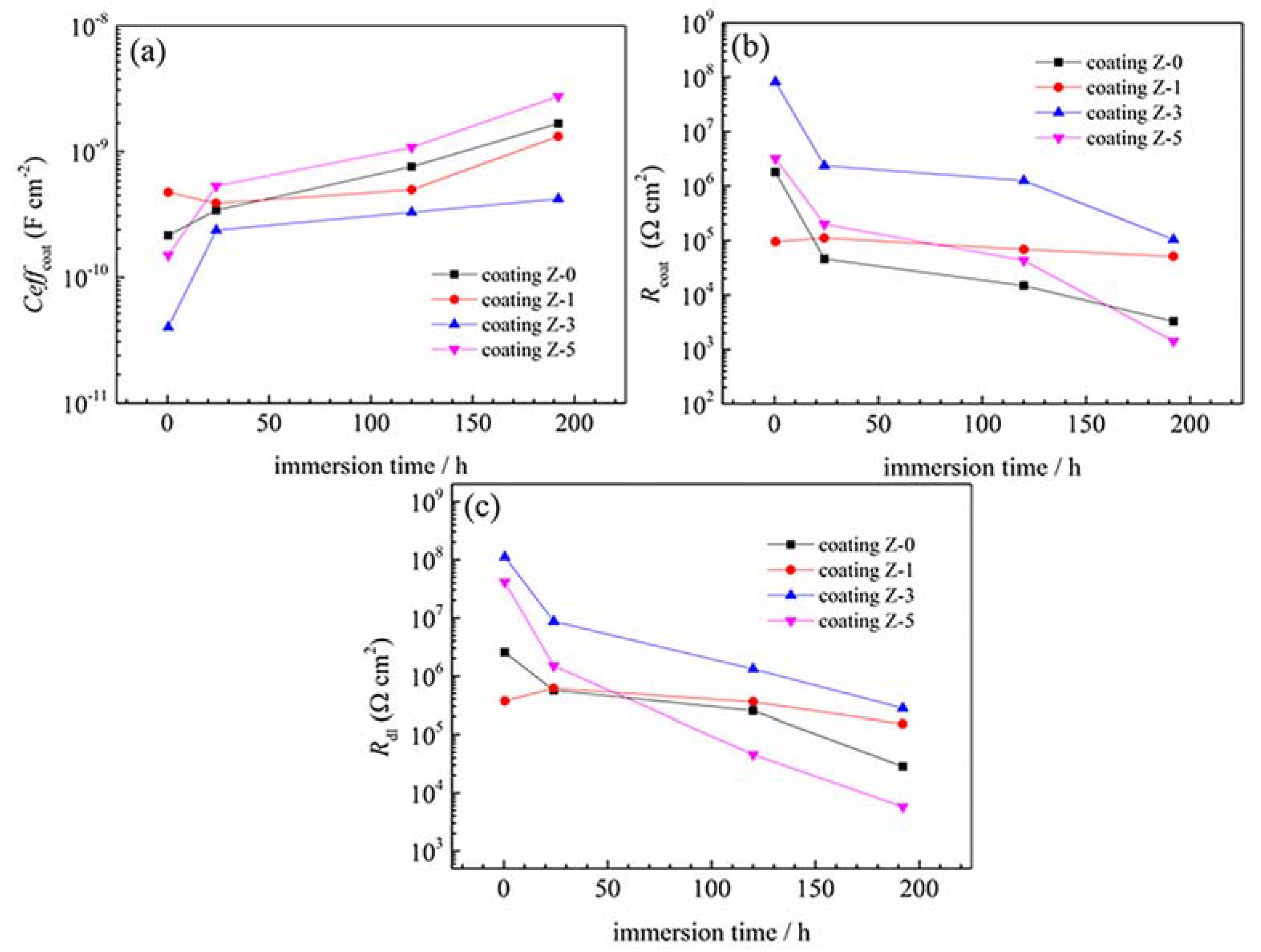
3.3. Electrochemically-Evaluated Corrosion Response
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stanger, R.; Wall, T. Sulphur impacts during pulverised coal combustion in oxy-fuel technology for carbon capture and storage. Prog. Energy Combust. Sci. 2011, 37, 69–88. [Google Scholar] [CrossRef]
- Shi, H.W.; Liu, F.C.; Yang, L.H.; Han, E.H. Characterization of protective performance of epoxy reinforced with nanometer-sized TiO2 and SiO2. Prog. Org. Coat. 2008, 62, 359–368. [Google Scholar] [CrossRef]
- Moller, V.B.; Dam-Johansen, K.; Frankaer, S.M.; Kiil, S. Acid-resistant organic coatings for the chemical industry: A review. J. Coat. Technol. Res. 2017, 14, 279–306. [Google Scholar] [CrossRef]
- Shi, X.M.; Nguyen, T.A.; Suo, Z.Y.; Liu, Y.J.; Avci, R. Effect of nanoparticles on the anticorrosion and mechanical properties of epoxy coating. Surf. Coat. Technol. 2009, 204, 237–245. [Google Scholar] [CrossRef]
- Mayavan, S.; Siva, T.; Sathiyanarayanan, S. Graphene ink as a corrosion inhibiting blanket for iron in an aggressive chloride environment. RSC Adv. 2013, 3, 24868–24871. [Google Scholar] [CrossRef]
- Cech, J.; Kretow, B. The effectiveness of toughening technologies on multifunctional epoxy resin systems. Surf. Coat. Aust. 2003, 40, 19–23. [Google Scholar]
- Wang, Z.; Han, E.; Liu, F.; Ke, W. Effect of Different Curing Agents on Cure Reaction and Exposure Resistance of Phenolic-Epoxy Resins in Hot Acid Solutions. Corrosion 2010, 66. [Google Scholar] [CrossRef]
- Sabzi, M.; Mirabedini, S.M.; Zohuriaan-Mehr, J.; Atai, M. Surface modification of TiO2 nano-particles with silane coupling agent and investigation of its effect on the properties of polyurethane composite coating. Prog. Org. Coat. 2009, 65, 222–228. [Google Scholar] [CrossRef]
- Alexandre, M.; Dubois, P. Polymer-layered silicate nanocomposites: Preparation, properties and uses of a new class of materials. Mater. Sci. Eng. R Rep. 2000, 28, 1–63. [Google Scholar] [CrossRef]
- Chaiko, D.J.; Leyva, A.A. Thermal transitions and barrier properties of olefinic nanocomposites. Chem. Mater. 2005, 17, 13–19. [Google Scholar] [CrossRef]
- Hang, T.T.X.; Truc, T.A.; Nam, T.H.; Oanh, V.K.; Jorcin, J.B.; Pebere, N. Corrosion protection of carbon steel by an epoxy resin containing organically modified clay. Surf. Coat. Technol. 2007, 201, 7408–7415. [Google Scholar] [CrossRef] [Green Version]
- Song, H.-J.; Zhang, Z.-Z. Investigation of the tribological properties of polyfluo wax/polyurethane composite coating filled with nano-SiC or nano-ZrO2. Mater. Sci. Eng. A 2006, 426, 59–65. [Google Scholar] [CrossRef]
- Behzadnasab, M.; Mirabedini, S.M.; Kabiri, K.; Jamali, S. Corrosion performance of epoxy coatings containing silane treated ZrO2 nanoparticles on mild steel in 3.5% NaCl solution. Corros. Sci. 2011, 53, 89–98. [Google Scholar] [CrossRef]
- Montesperelli, G.; Gusmano, G.; Rapone, M.; Padeletti, G.; Cusma, A.; Kaciulis, S.; Mezzi, A.; Di Maggio, R. Zirconia primers for corrosion resistant coatings. Surf. Coat. Technol. 2007, 201, 5822–5828. [Google Scholar]
- Ebrahim-Ghajari, M.; Allahkaram, S.R.; Mahdavi, S. Corrosion behaviour of electrodeposited nanocrystalline Co and Co/ZrO2nanocomposite coatings. Surf. Eng. 2014, 31, 251–257. [Google Scholar] [CrossRef]
- Hu, C.B.; Zheng, Y.S.; Qing, Y.Q.; Wang, F.L.; Mo, C.Y.; Mo, Q. Preparation of Poly(o-toluidine)/Nano Zirconium Dioxide (ZrO2)/Epoxy Composite Coating and Its Corrosion Resistance. J. Inorg. Organomet. Polym. Mater. 2015, 25, 583–592. [Google Scholar] [CrossRef]
- Zhou, C.L.; Lu, X.; Xin, Z.; Liu, J.; Zhang, Y.F. Polybenzoxazine/SiO2 nanocomposite coatings for corrosion protection of mild steel. Corros. Sci. 2014, 80, 269–275. [Google Scholar] [CrossRef]
- Sababi, M.; Pan, J.S.; Augustsson, P.E.; Sundell, P.E.; Claesson, P.M. Influence of polyaniline and ceria nanoparticle additives on corrosion protection of a UV-cure coating on carbon steel. Corros. Sci. 2014, 84, 189–197. [Google Scholar] [CrossRef]
- Liang, Y.; Wang, M.D.; Wang, C.; Feng, J.; Li, J.S.; Wang, L.J.; Fu, J.J. Facile Synthesis of Smart Nanocontainers as Key Components for Construction of Self-Healing Coating with Superhydrophobic Surfaces. Nanoscale Res. Lett. 2016, 11, 231. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.C.; Hao, Y.S.; Wang, Z.Y.; Shi, H.W.; Han, E.H.; Ke, W. Flaking and degradation of polyurethane coatings after 2 years of outdoor exposure in Lhasa. Chin. Sci. Bull. 2010, 55, 650–655. [Google Scholar] [CrossRef]
- Golczak, S.; Kanciurzewska, A.; Fahlman, M.; Langer, K.; Langer, J.J. Comparative XPS surface study of polyaniline thin films. Solid State Ion. 2008, 179, 2234–2239. [Google Scholar] [CrossRef]
- Radhakrishnan, S.; Siju, C.R.; Mahanta, D.; Patil, S.; Madras, G. Conducting polyaniline-nano-TiO2 composites for smart corrosion resistant coatings. Electrochim. Acta 2009, 54, 1249–1254. [Google Scholar] [CrossRef]
- Pour-Ali, S.; Dehghanian, C.; Kosari, A. In situ synthesis of polyaniline-camphorsulfonate particles in an epoxy matrix for corrosion protection of mild steel in NaCl solution. Corros. Sci. 2014, 85, 204–214. [Google Scholar] [CrossRef]
- Wang, Z.B.; Wang, Z.Y.; Hu, H.X.; Liu, C.B.; Zheng, Y.G. Corrosion Protection Performance of Nano-SiO2/Epoxy Composite Coatings in Acidic Desulfurized Flue Gas Condensates. J. Mater. Eng. Perform. 2016, 25, 3880–3889. [Google Scholar] [CrossRef]
- Sridhar, T.M.; Mudali, U.K.; Subbaiyan, M. Preparation and characterisation of electrophoretically deposited hydroxyapatite coatings on type 316L stainless steel. Corros. Sci. 2003, 45, 237–252. [Google Scholar] [CrossRef]
- Zhang, H.; Yao, G.C.; Wang, S.L.; Liu, Y.H.; Luo, H.J. A chrome-free conversion coating for magnesium-lithium alloy by a phosphate-permanganate solution. Surf. Coat. Technol. 2008, 202, 1825–1830. [Google Scholar] [CrossRef]
- Ruhi, G.; Bhandari, H.; Dhawan, S.K. Designing of corrosion resistant epoxy coatings embedded with polypyrrole/SiO2 composite. Prog. Org. Coat. 2014, 77, 1484–1498. [Google Scholar] [CrossRef]
- Shen, G.X.; Chen, Y.C.; Lin, L.; Lin, C.J.; Scantlebury, D. Study on a hydrophobic nano-TiO2 coating and its properties for corrosion protection of metals. Electrochim. Acta 2005, 50, 5083–5089. [Google Scholar] [CrossRef]
- Milosev, I.; Jovanovic, Z.; Bajat, J.B.; Jancic-Heinemann, R.; Miskovic-Stankovic, V.B. Surface Analysis and Electrochemical Behavior of Aluminum Pretreated by Vinyltriethoxysilane Films in Mild NaCl Solution. J. Electrochem. Soc. 2012, 159, C303–C311. [Google Scholar] [CrossRef]
- Liu, C.; Bi, Q.; Leyland, A.; Matthews, A. An electrochemical impedance spectroscopy study of the corrosion behaviour of PVD coated steels in 0.5 N NaCl aqueous solution: Part II. EIS interpretation of corrosion behaviour. Corros. Sci. 2003, 45, 1257–1273. [Google Scholar] [CrossRef]
- Liu, X.W.; Xiong, J.P.; Lv, Y.W.; Zuo, Y. Study on corrosion electrochemical behavior of several different coating systems by EIS. Prog. Org. Coat. 2009, 64, 497–503. [Google Scholar] [CrossRef]
- Yan, M.C.; Vetter, C.A.; Gelling, V.J. Corrosion inhibition performance of polypyrrole Al flake composite coatings for Al alloys. Corros. Sci. 2013, 70, 37–45. [Google Scholar] [CrossRef]
- Hirayama, R.; Haruyama, S. ELECTROCHEMICAL IMPEDANCE FOR DEGRADED COATED STEEL HAVING PORES. Corrosion 1991, 47, 952–958. [Google Scholar] [CrossRef]
- Kouisni, L.; Azzi, M.; Dalard, F.; Maximovitch, S. Phosphate coatings on magnesium alloy AM60 Part 2: Electrochemical behaviour in borate buffer solution. Surf. Coat. Technol. 2005, 192, 239–246. [Google Scholar]
- Cubides, Y.; Castaneda, H. Corrosion protection mechanisms of carbon nanotube and zinc-rich epoxy primers on carbon steel in simulated concrete pore solutions in the presence of chloride ions. Corros. Sci. 2016, 109, 145–161. [Google Scholar] [CrossRef]
- Singh, A.; Ahamad, I.; Singh, V.K.; Quraishi, M.A. Inhibition effect of environmentally benign Karanj (Pongamia pinnata) seed extract on corrosion of mild steel in hydrochloric acid solution. J. Solid State Electrochem. 2011, 15, 1087–1097. [Google Scholar] [CrossRef]
- Hirschorn, B.; Orazem, M.E.; Tribollet, B.; Vivier, V.; Frateur, I.; Musiani, M. Determination of effective capacitance and film thickness from constant-phase-element parameters. Electrochim. Acta 2010, 55, 6218–6227. [Google Scholar] [CrossRef]
- Su, X.M.; Zhou, Q.; Zhang, Q.Y.; Zhang, Y.; Zhang, H. Study on the deterioration process of bipolar coating using electrochemical impedance spectroscopy. Appl. Surf. Sci. 2011, 257, 6095–6101. [Google Scholar] [CrossRef]
- Zhou, C.L.; Lu, X.; Xin, Z.; Liu, J. Corrosion resistance of novel silane-functional polybenzoxazine coating on steel. Corros. Sci. 2013, 70, 145–151. [Google Scholar] [CrossRef]
- Castela, A.S.L.; Simoes, A.M.; Ferreira, M.G.S. EIS evaluation of attached and free polymer films. Prog. Org. Coat. 2000, 38, 1–7. [Google Scholar] [CrossRef]
- Yun, H.; Li, J.; Chen, H.B.; Lin, C.J. A study on the N-, S- and Cl-modified nano-TiO2 coatings for corrosion protection of stainless steel. Electrochim. Acta 2007, 52, 6679–6685. [Google Scholar] [CrossRef]
- Mansfeld, F. MODELS FOR THE IMPEDANCE BEHAVIOR OF PROTECTIVE COATINGS AND CASES OF LOCALIZED CORROSION. Electrochim. Acta 1993, 38, 1891–1897. [Google Scholar] [CrossRef]
- Hassan, H.H.; Abdelghani, E.; Amin, M.A. Inhibition of mild steel corrosion in hydrochloric acid solution by triazole derivatives—Part I. Polarization and EIS studies. Electrochim. Acta 2007, 52, 6359–6366. [Google Scholar] [CrossRef]
- Tang, N.; vanOoij, W.J.; Gorecki, G. Comparative EIS study of pretreatment performance in coated metals. Prog. Org. Coat. 1997, 30, 255–263. [Google Scholar] [CrossRef]
- Liu, J.G.; Gong, G.P.; Yan, C.W. EIS study of corrosion behaviour of organic coating/Dacromet composite systems. Electrochim. Acta 2005, 50, 3320–3332. [Google Scholar]



 : positive charge,
: positive charge,  : negative charge.
: negative charge.
 : positive charge,
: positive charge,  : negative charge.
: negative charge.
| Sample Coding 1 | Composition |
|---|---|
| Coating Z-0 | 65% phenolic-epoxy resin, 0.5% KH550 silane coupling agent, 34.1% 240 mesh glass flake, 0.4% BYK106 dispersant, 30% HN01 curing agent |
| Coating Z-1 | Coating Z-0 + 1.49% nano-ZrO2 concentrate (1% nano-ZrO2) |
| Coating Z-3 | Coating Z-0 + 4.48% nano-ZrO2 concentrate (3% nano-ZrO2) |
| Coating Z-5 | Coating Z-0 + 7.46% nano-ZrO2 concentrate (5% nano-ZrO2) |
| Samples | Ra (nm) | RMS (nm) |
|---|---|---|
| Z-0 Coating | 24.58 | 73.60 |
| Z-1 Coating | 12.79 | 27.44 |
| Z-3 Coating | 5.94 | 16.10 |
| Z-5 Coating | 20.42 | 52.03 |
| Samples | Element | At % | |
|---|---|---|---|
| Before immersion | Z-0, Z-1, Z-3 and Z-5 Coatings | C1s | 79.86 |
| O1s | 17.01 | ||
| N1s | 1.8 | ||
| After immersion | Z-0 Coating | C1s | 77.82 |
| O1s | 20.47 | ||
| N1s | 1.71 | ||
| Z-1 Coating | C1s | 80.37 | |
| O1s | 18.46 | ||
| N1s | 1.16 | ||
| Z-3 Coating | C1s | 80.77 | |
| O1s | 17.31 | ||
| N1s | 1.93 | ||
| Z-5 Coating | C1s | 76.99 | |
| O1s | 19.81 | ||
| N1s | 3.21 |
| Sample | Immersion Time/h | Rs/Ω·cm2 | Qcoat/sn·Ω−1·cm−2 | αcoat | Ceffcoat/F·cm−2 | Rcoat/Ω·cm−2 | Qdl/sn·Ω−1·cm−2 | αdl | Rct/Ω·cm2 | Chsq |
|---|---|---|---|---|---|---|---|---|---|---|
| Z-0 Coating | 30 min | 0.36 | 2.44 × 10−9 | 0.896 | 2.16 × 10−10 | 1.81 × 106 | 2.02 × 10−7 | 0.2517 | 2.54 × 106 | 1.30 × 10−3 |
| 24 h | 0.36 | 3.41 × 10−10 | 1 | 3.41 × 10−10 | 4.60 × 104 | 1.67 × 10−7 | 0.5321 | 5.63 × 105 | 2.21 × 10−3 | |
| 120 h | 393 | 2.88 × 10−9 | 0.9113 | 7.59 × 10−10 | 1.48× 104 | 1.99 × 10−4 | 0.1117 | 2.53 × 105 | 3.59 × 10−4 | |
| 192 h | 138 | 4.84 × 10−7 | 0.6298 | 1.66 × 10−9 | 3.26 × 103 | 5.58 × 10−4 | 0.4868 | 2.84 × 104 | 1.94 × 10−4 | |
| Z-1 Coating | 30 min | 0.36 | 2.10 × 10−9 | 0.9337 | 4.73 × 10−10 | 9.51 × 104 | 1.17 × 10−6 | 0.296 | 3.70 × 105 | 2.12 × 10−3 |
| 24 h | 0.37 | 3.88 × 10−10 | 1 | 3.88 × 10−10 | 1.11 × 105 | 4.76 × 10−7 | 0.3895 | 6.08 × 105 | 1.16 × 10−3 | |
| 120 h | 0.36 | 7.46 × 10−9 | 0.8793 | 4.97 × 10−10 | 6.89 × 104 | 2.24 × 10−6 | 0.1965 | 3.58 × 105 | 9.63 × 10−4 | |
| 192 h | 723 | 4.89 × 10−8 | 0.7395 | 1.32 × 10−9 | 5.10 × 104 | 2.47 × 10−5 | 0.4372 | 1.52 × 105 | 3.34 × 10−3 | |
| Z-3 Coating | 30 min | 0.36 | 2.16 × 10−9 | 0.8403 | 4.02 × 10−11 | 8.26 × 107 | 1.07 × 10−7 | 0.4962 | 1.11 × 108 | 1.68 × 10−3 |
| 24 h | 0.36 | 1.09 × 10−9 | 0.9344 | 2.37 × 10−10 | 2.38 × 106 | 1.76 × 10−8 | 0.3601 | 8.67 × 106 | 2.37 × 10−3 | |
| 120 h | 0.36 | 1.52 × 10−9 | 0.9331 | 3.28 × 10−10 | 1.26 × 106 | 3.28 × 10−7 | 0.4515 | 1.31 × 106 | 2.15 × 10−3 | |
| 192 h | 0.36 | 1.11 × 10−9 | 0.9572 | 4.21 × 10−10 | 1.04 × 105 | 8.61 × 10−6 | 0.2803 | 2.80 × 105 | 3.97 × 10−4 | |
| Z-5 Coating | 30 min | 0.36 | 2.47 × 10−9 | 0.8815 | 1.50 × 10−10 | 3.21 × 106 | 2.67 × 10−8 | 0.2855 | 4.09 × 107 | 2.55 × 10−3 |
| 24 h | 0.36 | 2.34 × 10−9 | 0.9338 | 5.31 × 10−10 | 2.01 × 105 | 2.61 × 10−7 | 0.3371 | 1.49 × 106 | 2.29 × 10−3 | |
| 120 h | 228 | 1.19 × 10−7 | 0.6911 | 1.08 × 10−9 | 4.31 × 104 | 4.41 × 10−5 | 0.3982 | 4.53 × 104 | 4.51 × 10−4 | |
| 192 h | 67.3 | 2.35 × 10−8 | 0.8619 | 2.74 × 10−9 | 1.41× 103 | 5.10 × 10−4 | 0.2736 | 5.76 × 103 | 4.13 × 10−4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, W.; Wang, Z.; Han, E.-H.; Wang, S.; Liu, Q. Corrosion Performance of Nano-ZrO2 Modified Coatings in Hot Mixed Acid Solutions. Materials 2018, 11, 934. https://doi.org/10.3390/ma11060934
Xu W, Wang Z, Han E-H, Wang S, Liu Q. Corrosion Performance of Nano-ZrO2 Modified Coatings in Hot Mixed Acid Solutions. Materials. 2018; 11(6):934. https://doi.org/10.3390/ma11060934
Chicago/Turabian StyleXu, Wenhua, Zhenyu Wang, En-Hou Han, Shuai Wang, and Qian Liu. 2018. "Corrosion Performance of Nano-ZrO2 Modified Coatings in Hot Mixed Acid Solutions" Materials 11, no. 6: 934. https://doi.org/10.3390/ma11060934





