Enhancing the Ignition, Hardness and Compressive Response of Magnesium by Reinforcing with Hollow Glass Microballoons
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Processing
2.3. Methods
3. Results and Discussion
3.1. Microstructural Characterization
3.2. Density Measurement
3.3. Coefficient of Thermal Expansion (CTE)
3.4. Ignition Properties
3.5. Microhardness
3.6. Compression Properties and Fracture Behaviour
3.7. Elastic Modulus
4. Conclusions
- Mg matrix syntactic foams with hollow glass microballoons (GMB) as a reinforcement can be successfully synthesized using disintegrated melt deposition technique. Mg-25 foam exhibited an average density of 1.47 g/cc similar to polymers signifying its weight saving potential and elevated temperature usage.
- CTE values reduced with the incorporation of GMB in pure Mg with Mg-25 foam exhibiting the CTE value of 21.2 × 10−6/K indicating superior thermal and dimensional stability.
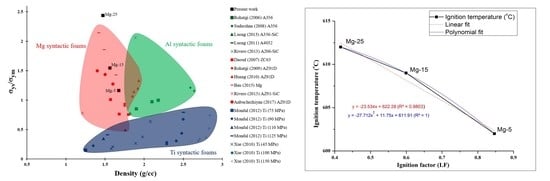
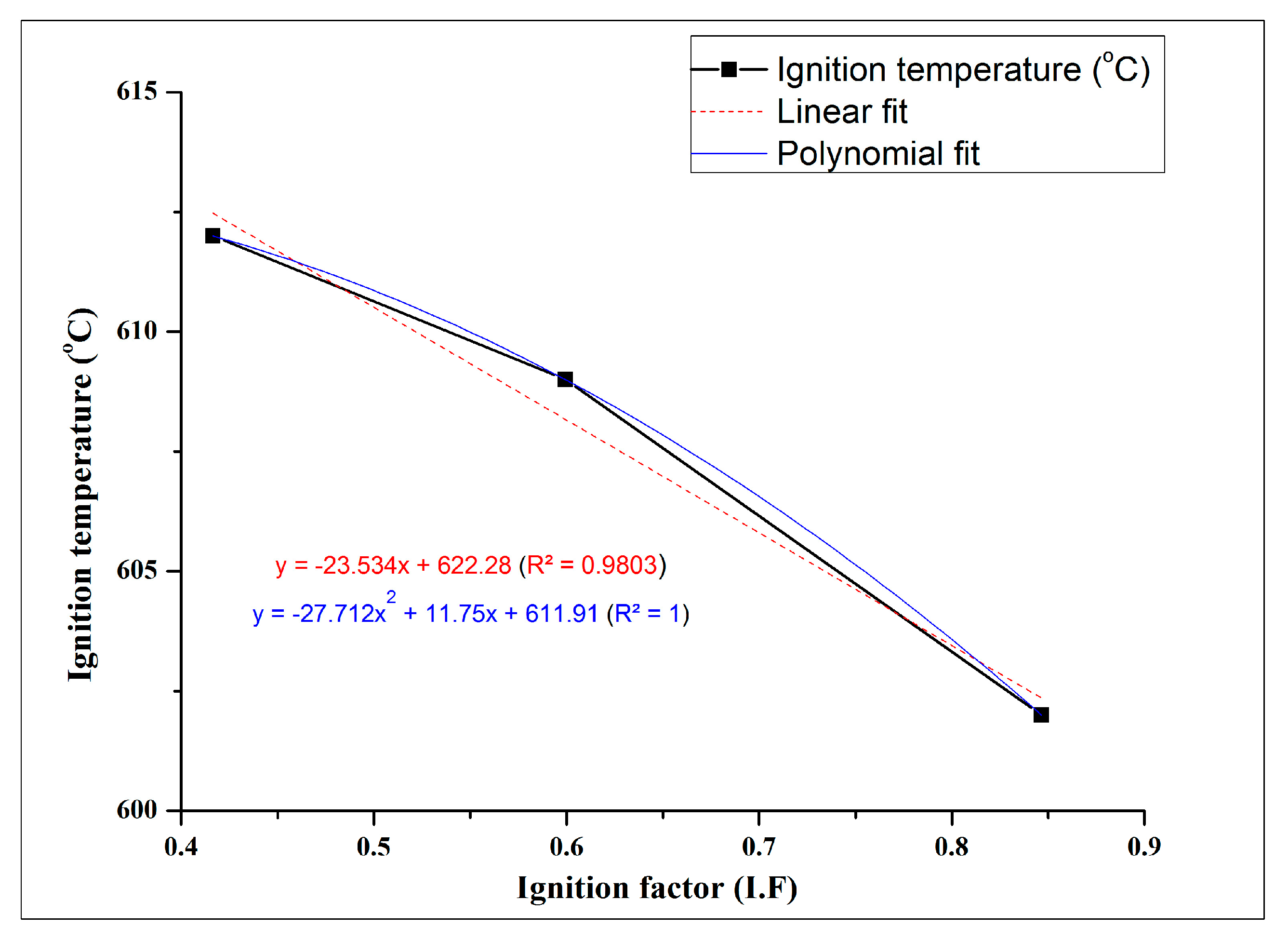
- The ignition temperature of Mg-25 showed the highest resistance to ignition, with an increase in ignition temperature of ~22 °C. Further, the ignition temperature of the developed foams exhibit a perfect first-degree polynomial with respect to ignition factor, a new parameter proposed by authors.
- The hardness of pure Mg increased with the increasing GMB content, with Mg-25 showing a maximum increase of ~127.7%.
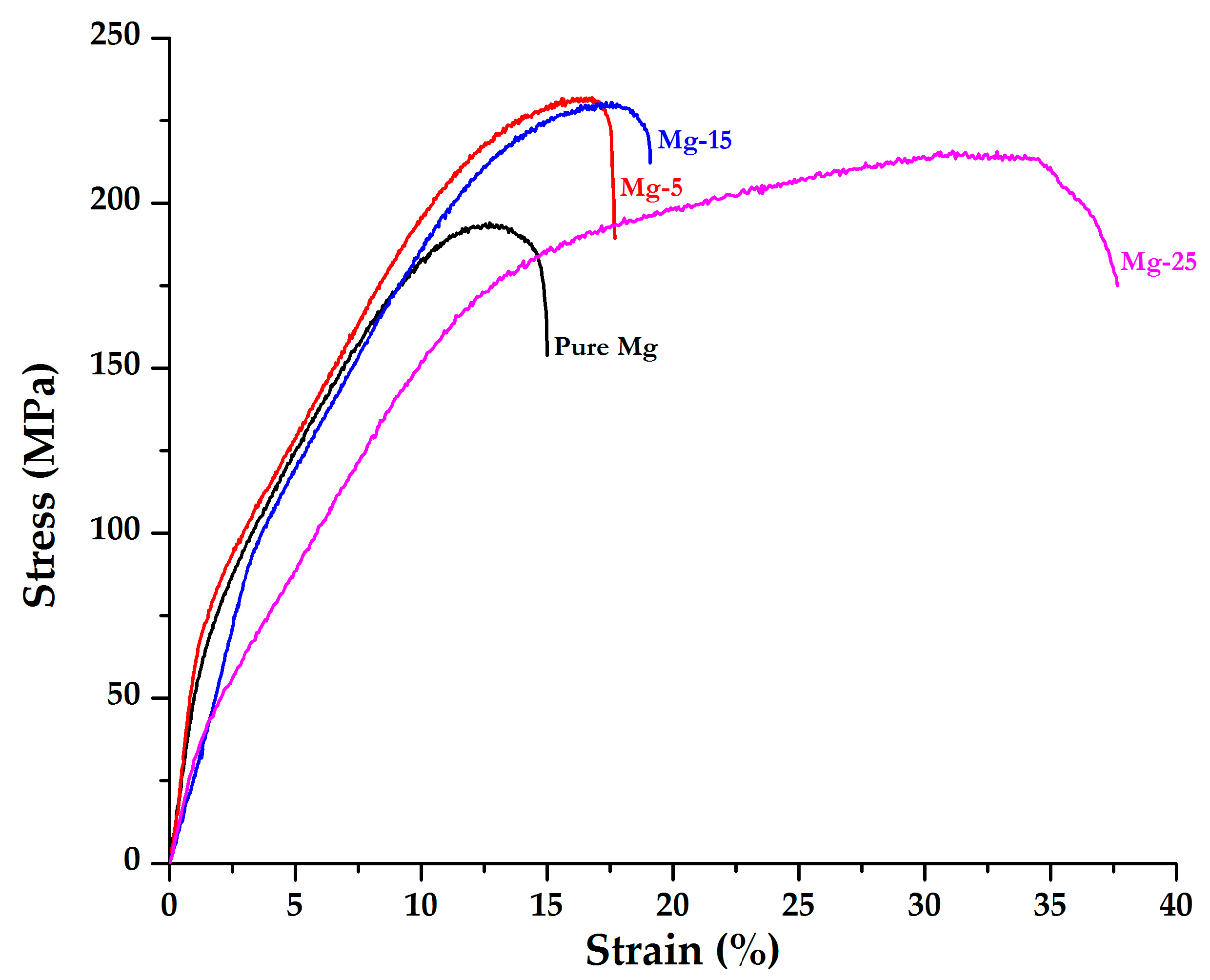
- Under compression loading, 0.2% CYS and compressive fracture strain of pure Mg increased with GMB addition. Mg-25 foam exhibited a 0.2% CYS and compressive fracture strain of ~161 MPa and ~37.7% respectively. However, the maximum UCS was observed in Mg-5 foam.
- Energy absorbed under compressive loading also increased with progressive addition of GMB, with Mg-25 foam showing a significant improvement of ~200% as compared to pure Mg.
- The superior compressive properties with elastic modulus closer to natural bone makes Mg/GMB syntactic foams a potential choice for implant materials.
Author Contributions
Conflicts of Interest
References
- Parande, G.; Manakari, V.; Meenashisundaram, G.K.; Gupta, M. Enhancing the hardness/compression/damping response of magnesium by reinforcing with biocompatible silica nanoparticulates. Int. J. Mater. Res. 2016, 107, 1091–1099. [Google Scholar] [CrossRef]
- Mordike, B.; Ebert, T. Magnesium: Properties—Applications—Potential. Mater. Sci. Eng. A 2001, 302, 37–45. [Google Scholar] [CrossRef]
- Chen, Y.; Tekumalla, S.; Guo, Y.B.; Gupta, M. Introducing mg-4zn-3gd-1ca/zno nanocomposite with compressive strengths matching/exceeding that of mild steel. Sci. Rep. 2016, 6, 32395. [Google Scholar] [CrossRef] [PubMed]
- Toda-Caraballo, I.; Galindo-Nava, E.I.; Rivera-Díaz-del-Castillo, P.E. Understanding the factors influencing yield strength on mg alloys. Acta Mater. 2014, 75, 287–296. [Google Scholar] [CrossRef]
- Johnston, S.; Shi, Z.; Atrens, A. The influence of ph on the corrosion rate of high-purity mg, az91 and ze41 in bicarbonate buffered hanks’ solution. Corros. Sci. 2015, 101, 182–192. [Google Scholar] [CrossRef]
- Omar, M.Y.; Xiang, C.; Gupta, N.; Strbik, O.M.; Cho, K. Syntactic foam core metal matrix sandwich composite under bending conditions. Mater. Des. 2015, 86, 536–544. [Google Scholar] [CrossRef]
- Rohatgi, P.K.; Gupta, N.; Schultz, B.F.; Luong, D.D. The synthesis, compressive properties, and applications of metal matrix syntactic foams. JOM J. Miner. Met. Mater. Soc. 2011, 63, 36–42. [Google Scholar]
- Manakari, V.; Parande, G.; Gupta, M. Effects of hollow fly-ash particles on the properties of magnesium matrix syntactic foams: A review. Mater. Perform. Charact. 2016, 5, 116–131. [Google Scholar] [CrossRef]
- Anantharaman, H.; Shunmugasamy, V.C.; Strbik, O.M.; Gupta, N.; Cho, K. Dynamic properties of silicon carbide hollow particle filled magnesium alloy (az91d) matrix syntactic foams. Int. J. Impact Eng. 2015, 82, 14–24. [Google Scholar] [CrossRef]
- Newsome, D.B.; Schultz, B.F.; Ferguson, J.; Rohatgi, P.K. Synthesis and quasi-static compressive properties of mg-az91d-al2o3 syntactic foams. Materials 2015, 8, 6085–6095. [Google Scholar] [CrossRef] [PubMed]
- Rohatgi, P.; Kim, J.; Gupta, N.; Alaraj, S.; Daoud, A. Compressive characteristics of a356/fly ash cenosphere composites synthesized by pressure infiltration technique. Compos. Part A Appl. Sci. Manuf. 2006, 37, 430–437. [Google Scholar] [CrossRef]
- Kumar, B.B.; Doddamani, M.; Zeltmann, S.E.; Gupta, N.; Ramesh, M.; Ramakrishna, S. Processing of cenosphere/hdpe syntactic foams using an industrial scale polymer injection molding machine. Mater. Des. 2016, 92, 414–423. [Google Scholar] [CrossRef]
- Manakari, V.; Parande, G.; Doddamani, M.; Gaitonde, V.; Siddhalingeshwar, I.; Shunmugasamy, V.C.; Gupta, N. Dry sliding wear of epoxy/cenosphere syntactic foams. Tribol. Int. 2015, 92, 425–438. [Google Scholar] [CrossRef]
- Zeltmann, S.E.; Kumar, B.B.; Doddamani, M.; Gupta, N. Prediction of strain rate sensitivity of high density polyethylene using integral transform of dynamic mechanical analysis data. Polymer 2016, 101, 1–6. [Google Scholar] [CrossRef]
- Bharath Kumar, B.; Zeltmann, S.E.; Doddamani, M.; Gupta, N.; Gurupadu, S.; Sailaja, R. Effect of cenosphere surface treatment and blending method on the tensile properties of thermoplastic matrix syntactic foams. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- Nguyen, Q.; Sharon Nai, M.; Nguyen, A.; Seetharaman, S.; Wai Leong, E.; Gupta, M. Synthesis and properties of light weight magnesium–cenosphere composite. Mater. Sci. Technol. 2016, 32, 1–7. [Google Scholar] [CrossRef]
- Sankaranarayanan, S.; Nguyen, Q.; Shabadi, R.; Almajid, A.; Gupta, M. Powder metallurgy hollow fly ash cenospheres’ particles reinforced magnesium composites. Powder Metall. 2016, 59, 188–196. [Google Scholar] [CrossRef]
- Rivero, G.A.R.; Schultz, B.F.; Ferguson, J.; Gupta, N.; Rohatgi, P.K. Compressive properties of al-a206/sic and mg-az91/sic syntactic foams. J. Mater. Res. 2013, 28, 2426–2435. [Google Scholar] [CrossRef]
- Anbuchezhiyan, G.; Mohan, B.; Sathianarayanan, D.; Muthuramalingam, T. Synthesis and characterization of hollow glass microspheres reinforced magnesium alloy matrix syntactic foam. J. Alloys Compd. 2017, 719, 125–132. [Google Scholar] [CrossRef]
- Zeltmann, S.E.; Chen, B.; Gupta, N. Thermal expansion and dynamic mechanical analysis of epoxy matrix–borosilicate glass hollow particle syntactic foams. J. Cell. Plast. 2017. [Google Scholar] [CrossRef]
- Poveda, R.L.; Dorogokupets, G.; Gupta, N. Carbon nanofiber reinforced syntactic foams: Degradation mechanism for long term moisture exposure and residual compressive properties. Polym. Degrad. Stab. 2013, 98, 2041–2053. [Google Scholar] [CrossRef]
- Gupta, M.; Wong, W. Magnesium-based nanocomposites: Lightweight materials of the future. Mater. Charact. 2015, 105, 30–46. [Google Scholar] [CrossRef]
- Gupta, M.; Lai, M.; Saravanaranganathan, D. Synthesis, microstructure and properties characterization of disintegrated melt deposited mg/sic composites. J. Mater. Sci. 2000, 35, 2155–2165. [Google Scholar] [CrossRef]
- ASTM E384-16, Standard Test Method for Microindentation Hardness of Materials; ASTM International: West Conshohocken, PA, USA, 2016.
- ASTM E9-09, Standard Test Methods of Compression Testing of Metallic Materials at Room Temperature; ASTM International: West Conshohocken, PA, USA, 2009.
- ASTM E1876-15, Standard Test Method for Dynamic Young’s Modulus, Shear Modulus, and Poisson’s Ratio by Impulse Excitation of Vibration; ASTM International: West Conshohocken, PA, USA, 2015.
- Güllü, A.; Özdemir, A.; Özdemir, E. Experimental investigation of the effect of glass fibres on the mechanical properties of polypropylene (pp) and polyamide 6 (pa6) plastics. Mater. Des. 2006, 27, 316–323. [Google Scholar] [CrossRef]
- Yung, K.; Zhu, B.; Yue, T.; Xie, C. Preparation and properties of hollow glass microsphere-filled epoxy-matrix composites. Compos. Sci. Technol. 2009, 69, 260–264. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, X.; Kuang, Y.; Liu, B.; Zhang, K.; Fang, D. Enhanced mechanical properties and degradation rate of mg-3zn-1y based alloy by cu addition for degradable fracturing ball applications. Mater. Lett. 2017, 195, 194–197. [Google Scholar] [CrossRef]
- Czerwinski, F. Overcoming barriers of magnesium ignition and flammability. Adv. Mater. Process. 2014, 172, 28–31. [Google Scholar]
- Tekumalla, S.; Gupta, M. An insight into ignition factors and mechanisms of magnesium based materials: A review. Mater. Des. 2017, 113, 84–98. [Google Scholar] [CrossRef]
- Jiao, C.; Wang, H.; Li, S.; Chen, X. Fire hazard reduction of hollow glass microspheres in thermoplastic polyurethane composites. J. Hazard. Mater. 2017, 332, 176–184. [Google Scholar] [CrossRef]
- Liu, L.; Hu, J.; Zhuo, J.; Jiao, C.; Chen, X.; Li, S. Synergistic flame retardant effects between hollow glass microspheres and magnesium hydroxide in ethylene-vinyl acetate composites. Polym. Degrad. Stab. 2014, 104, 87–94. [Google Scholar] [CrossRef]
- Kim, Y.M.; Yim, C.D.; Kim, H.S.; You, B.S. Key factor influencing the ignition resistance of magnesium alloys at elevated temperatures. Scr. Mater. 2011, 65, 958–961. [Google Scholar] [CrossRef]
- Parande, G.; Manakari, V.; Meenashisundaram, G.K.; Gupta, M. Enhancing the tensile and ignition response of monolithic magnesium by reinforcing with silica nanoparticulates. J. Mater. Res. 2017, 32, 2169–2178. [Google Scholar] [CrossRef]
- Kumar, B.B.; Singh, A.K.; Doddamani, M.; Luong, D.D.; Gupta, N. Quasi-static and high strain rate compressive response of injection-molded cenosphere/hdpe syntactic foam. JOM 2016, 68, 1861–1871. [Google Scholar] [CrossRef]
- Rohatgi, P.K.; Daoud, A.; Schultz, B.F.; Puri, T. Microstructure and mechanical behavior of die casting az91d-fly ash cenosphere composites. Compos. Part A Appl. Sci. Manuf. 2009, 40, 883–896. [Google Scholar] [CrossRef]
- Zhao, Y.; Tao, X. Behaviour of metal matrix syntactic foams in compression. Mater. Sci. Technol. 2009, 1785–1794. [Google Scholar]
- Daoud, A.; El-Khair, M.A.; Abdel-Aziz, M.; Rohatgi, P. Fabrication, microstructure and compressive behavior of zc63 mg–microballoon foam composites. Compos. Sci. Technol. 2007, 67, 1842–1853. [Google Scholar] [CrossRef]
- Luong, D.; Lehmhus, D.; Gupta, N.; Weise, J.; Bayoumi, M. Structure and compressive properties of invar-cenosphere syntactic foams. Materials 2016, 9, 115. [Google Scholar] [CrossRef] [PubMed]
- Trojanová, Z.; Gärtnerová, V.; Jäger, A.; Námešný, A.; Chalupová, M.; Palček, P.; Lukáč, P. Mechanical and fracture properties of an az91 magnesium alloy reinforced by si and sic particles. Compos. Sci. Technol. 2009, 69, 2256–2264. [Google Scholar] [CrossRef]
- Huang, Z.-Q.; Yu, S.-R.; Li, M.-Q. Microstructures and compressive properties of az91d/fly-ash cenospheres composites. Trans. Nonferrous Met. Soc. China 2010, 20, s458–s462. [Google Scholar] [CrossRef]
- Daoud, A. Effect of fly ash addition on the structure and compressive properties of 4032–fly ash particle composite foams. J. Alloys Compd. 2009, 487, 618–625. [Google Scholar] [CrossRef]
- Luong, D.D.; Gupta, N.; Daoud, A.; Rohatgi, P.K. High strain rate compressive characterization of aluminum alloy/fly ash cenosphere composites. JOM 2011, 63, 53–56. [Google Scholar]
- Luong, D.D.; Strbik, O.M.; Hammond, V.H.; Gupta, N.; Cho, K. Development of high performance lightweight aluminum alloy/sic hollow sphere syntactic foams and compressive characterization at quasi-static and high strain rates. J. Alloys Compd. 2013, 550, 412–422. [Google Scholar] [CrossRef]
- Mondal, D.; Das, S.; Ramakrishnan, N.; Bhasker, K.U. Cenosphere filled aluminum syntactic foam made through stir-casting technique. Compos. Part A Appl. Sci. Manuf. 2009, 40, 279–288. [Google Scholar] [CrossRef]
- Surappa, M. Synthesis of fly ash particle reinforced a356 al composites and their characterization. Mater. Sci. Eng. A 2008, 480, 117–124. [Google Scholar]
- Xue, X.; Kearns, V.; Williams, R.; Zhao, Y. Mechanical and biological properties of titanium syntactic foams. In Proceedings of the Minerals, Metals and Materials Society/AIME, 420 Commonwealth Dr., P. O. Box 430, Warrendale, PA, USA, 14–18 February 2010. [Google Scholar]
- Zeltmann, S.E.; Prakash, K.A.; Doddamani, M.; Gupta, N. Prediction of modulus at various strain rates from dynamic mechanical analysis data for polymer matrix composites. Compos. Part B Eng. 2017, 120, 27–34. [Google Scholar] [CrossRef]
- Gupta, N.; Shunmugasamy, V.C. Multifunctional Syntactic Foams. U.S. 9676916 B2, 13 June 2017. [Google Scholar]
- Manakari, V.; Parande, G.; Gupta, M. Selective laser melting of magnesium and magnesium alloy powders: A review. Metals 2016, 7, 2. [Google Scholar] [CrossRef]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Hossain, K.M.Z.; Patel, U.; Ahmed, I. Development of microspheres for biomedical applications: A review. Prog. Biomater. 2015, 4, 1–19. [Google Scholar] [CrossRef]
- Li, S.; Nguyen, L.; Xiong, H.; Wang, M.; Hu, T.C.-C.; She, J.-X.; Serkiz, S.M.; Wicks, G.G.; Dynan, W.S. Porous-wall hollow glass microspheres as novel potential nanocarriers for biomedical applications. Nanomedicine: Nanotechnol. Biol. Med. 2010, 6, 127–136. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.; Niinomi, M.; Nakai, M.; Liu, H.; Santos, P.F.; Itoh, Y.; Ikeda, M. Tensile and Compressive Properties of Low-Cost High-Strength β-Type Ti-mn Alloys Fabricated by Metal Injection Molding. In Proceedings of the 13th World Conference on Titanium; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2016. [Google Scholar] [CrossRef]



| Material | Theoretical Density (g/cc) | Measured Density (g/cc) | Matrix Porosity (vol %) | CTE (× 10−6/K) | Ignition Temperature (°C) |
|---|---|---|---|---|---|
| Mg | 1.738 | 1.701 ± 0.002 | 2.1 | 27.1 | 590 ± 1.2 |
| Mg-5 | 1.686 | 1.674 ± 0.015 (↓1.6%) | 0.72 | 24.2 (↓10.7%) | 602 ± 1 |
| Mg-15 | 1.588 | 1.559 ± 0.010 (↓8.3%) | 1.78 | 22.7 (↓16.2%) | 609 ± 0.8 |
| Mg-25 | 1.502 | 1.472 ± 0.018 (↓13.4%) | 1.98 | 21.2 (↓21.7%) | 612 ± 0.5 |
| Material | Hardness (Hv) | 0.2% CYS (MPa) | UCS (MPa) | Ultimate Compressive Strain (%) | Energy Absorbed (MJ/m3) |
|---|---|---|---|---|---|
| Pure Mg | 47 ± 2 | 66 ± 3.5 | 194 ± 8 | 15 ± 1 | 21.1 ± 1.2 |
| Mg-5 | 82 ± 4 (↑74.5%) | 77 ± 3 (↑16.7%) | 232 ± 7 (↑19.6%) | 17.2 ± 0.6 (↑14.7%) | 28.4 ± 1.3 (↑34.6%) |
| Mg-15 | 91 ± 5 (↑93.6%) | 102 ± 5 (↑54.5%) | 231 ± 6 (↑19.1%) | 19.1 ± 0.7 (↑27.3%) | 32.7 ± 1.6 (↑55%) |
| Mg-25 | 107 ± 6 (↑127.7%) | 161 ± 4 (↑144%) | 216 ± 6 (↑11.3%) | 37.7 ± 2 (↑151.3%) | 63.4 ± 3.2 (↑200%) |
| Material | Density (g/cc) | 0.2% CYS (MPa) | UCS (MPa) | Ultimate Compressive Strain (%) | Elastic Modulus (GPa) |
|---|---|---|---|---|---|
| Natural Bone | 1.8–2.1 | 130–180 | - | - | 3–20 |
| Cortical Bone | 1.3 ± 0.03 | - | 131–224 | 2–12 | 15–30 |
| Titanium alloy | 4.4–4.5 | 1040 | 1643–2324 | 29–49 | 110–117 |
| Stainless Steel | 7.9–8.1 | 170–310 | - | - | 189–205 |
| Pure Mg | 1.7014 | 66 | 194 | 15 | 43.3 |
| Mg-5 | 1.6739 | 77 | 232 | 17.2 | 42.56 (↓1.7%) |
| Mg-15 | 1.5597 | 102 | 231 | 19.1 | 41.10 (↓5.1%) |
| Mg-25 | 1.4723 | 161 | 216 | 37.7 | 39.85 (↓7.9%) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manakari, V.; Parande, G.; Doddamani, M.; Gupta, M. Enhancing the Ignition, Hardness and Compressive Response of Magnesium by Reinforcing with Hollow Glass Microballoons. Materials 2017, 10, 997. https://doi.org/10.3390/ma10090997
Manakari V, Parande G, Doddamani M, Gupta M. Enhancing the Ignition, Hardness and Compressive Response of Magnesium by Reinforcing with Hollow Glass Microballoons. Materials. 2017; 10(9):997. https://doi.org/10.3390/ma10090997
Chicago/Turabian StyleManakari, Vyasaraj, Gururaj Parande, Mrityunjay Doddamani, and Manoj Gupta. 2017. "Enhancing the Ignition, Hardness and Compressive Response of Magnesium by Reinforcing with Hollow Glass Microballoons" Materials 10, no. 9: 997. https://doi.org/10.3390/ma10090997