Can More Nanoparticles Induce Larger Viscosities of Nanoparticle-Enhanced Wormlike Micellar System (NEWMS)?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. Rheological Measurements
3. Results and Discussion
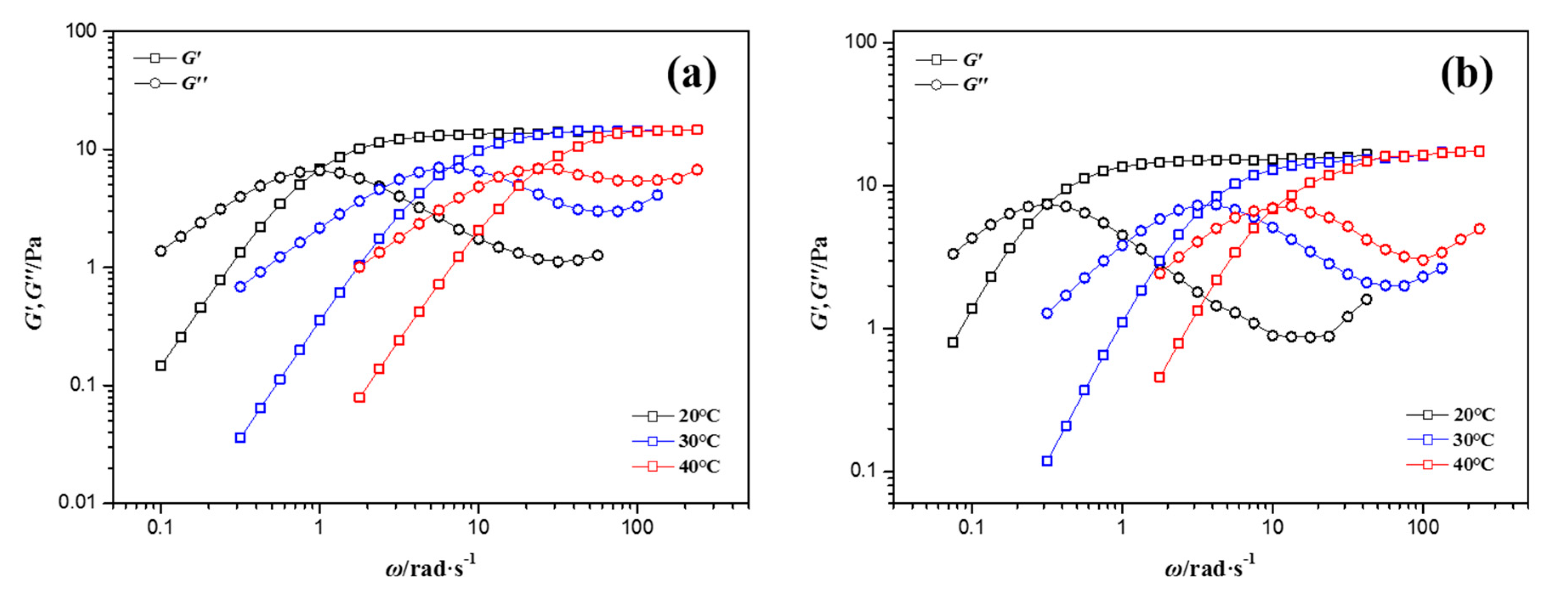
3.1. Rheological Properties of NEWMS
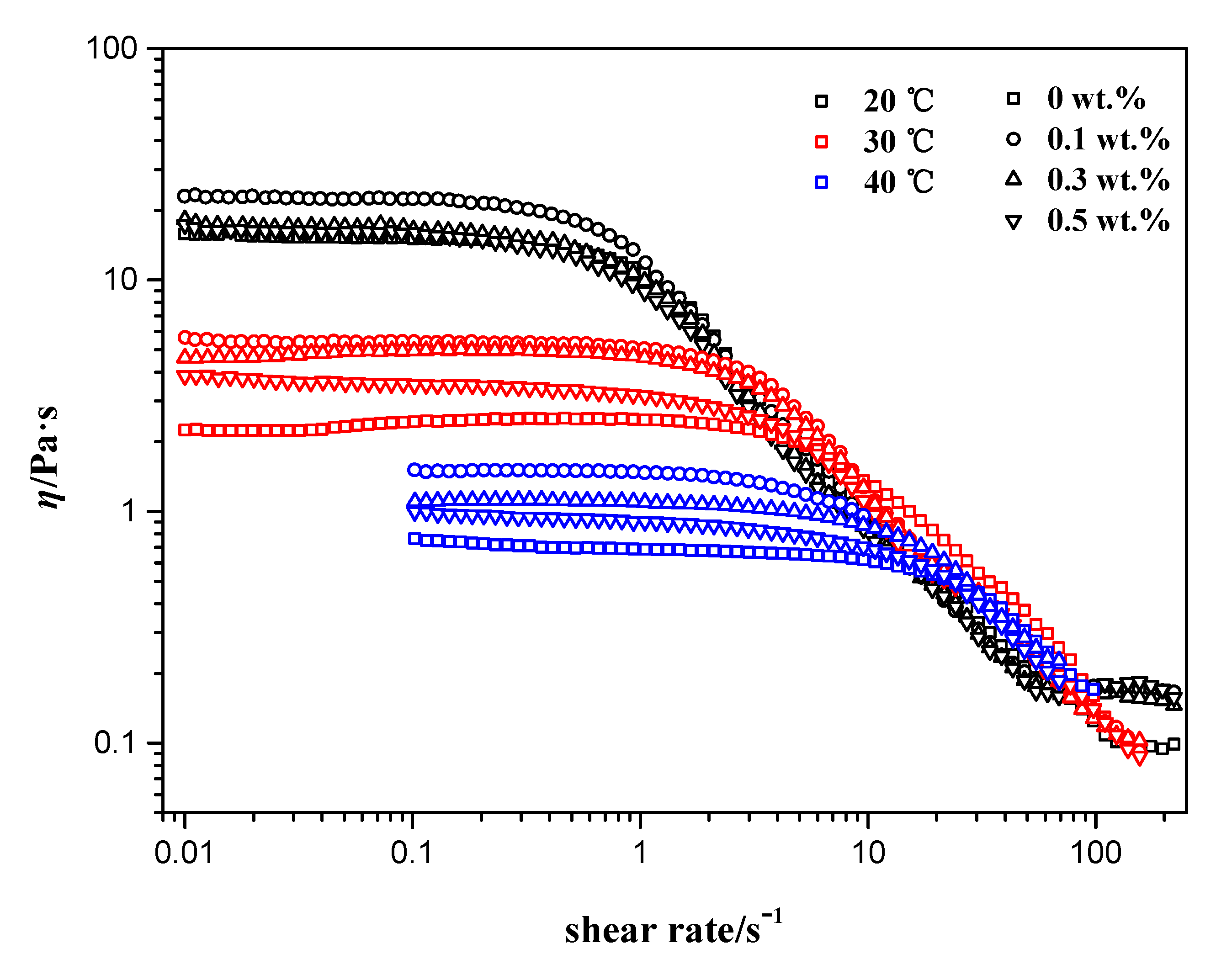
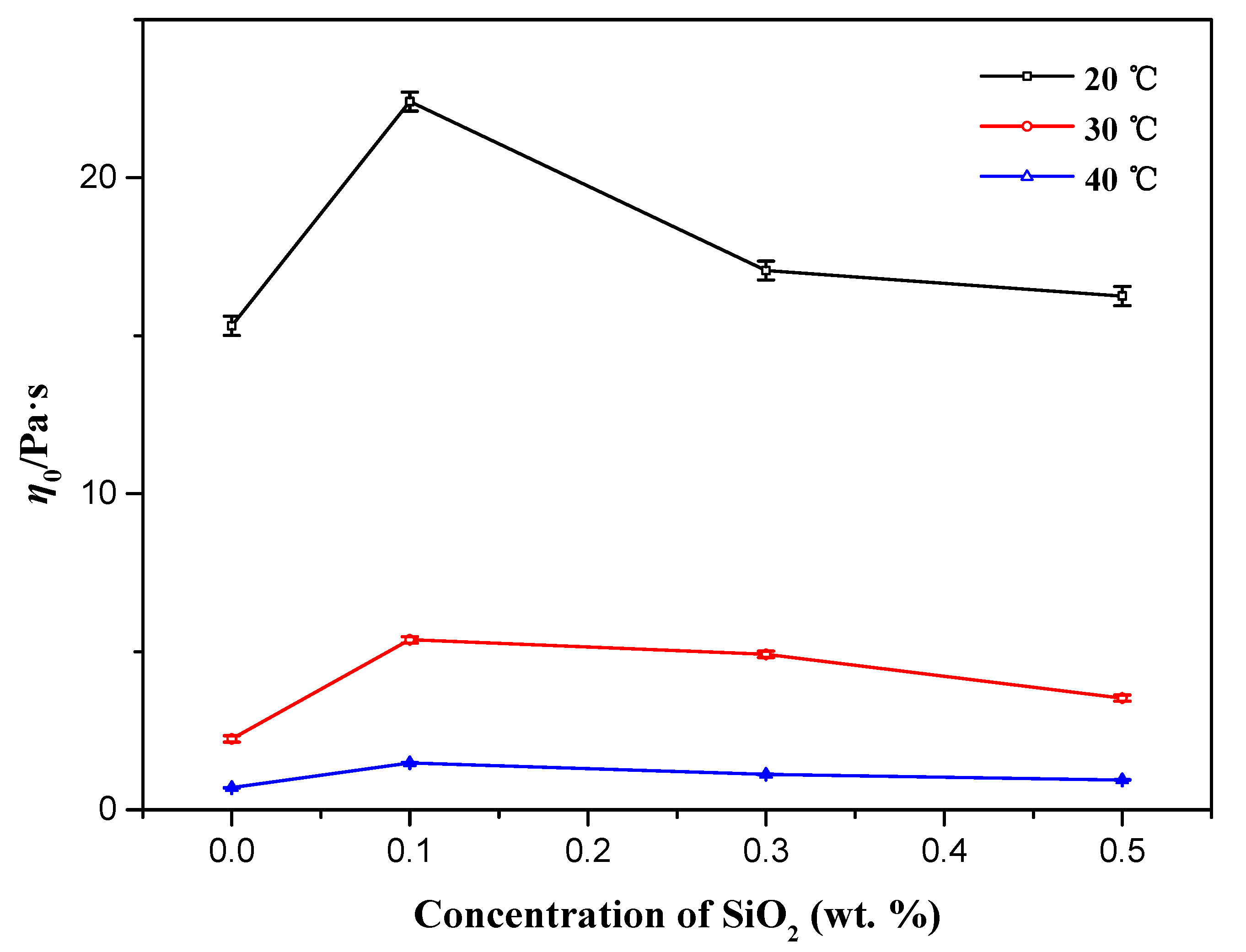
3.2. Effects of Silica Nanoparticle Concentration
3.3. Effects of Temperature
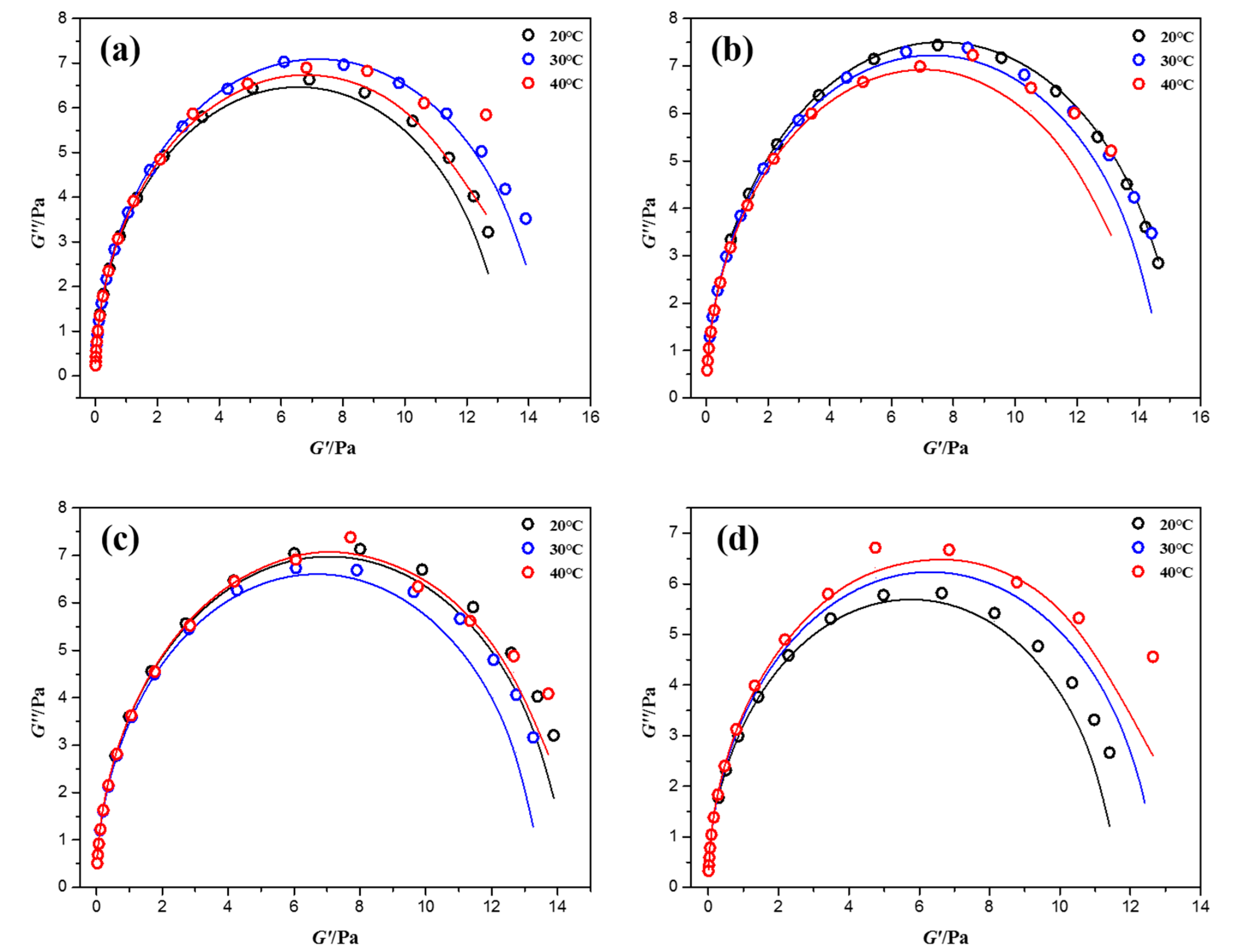
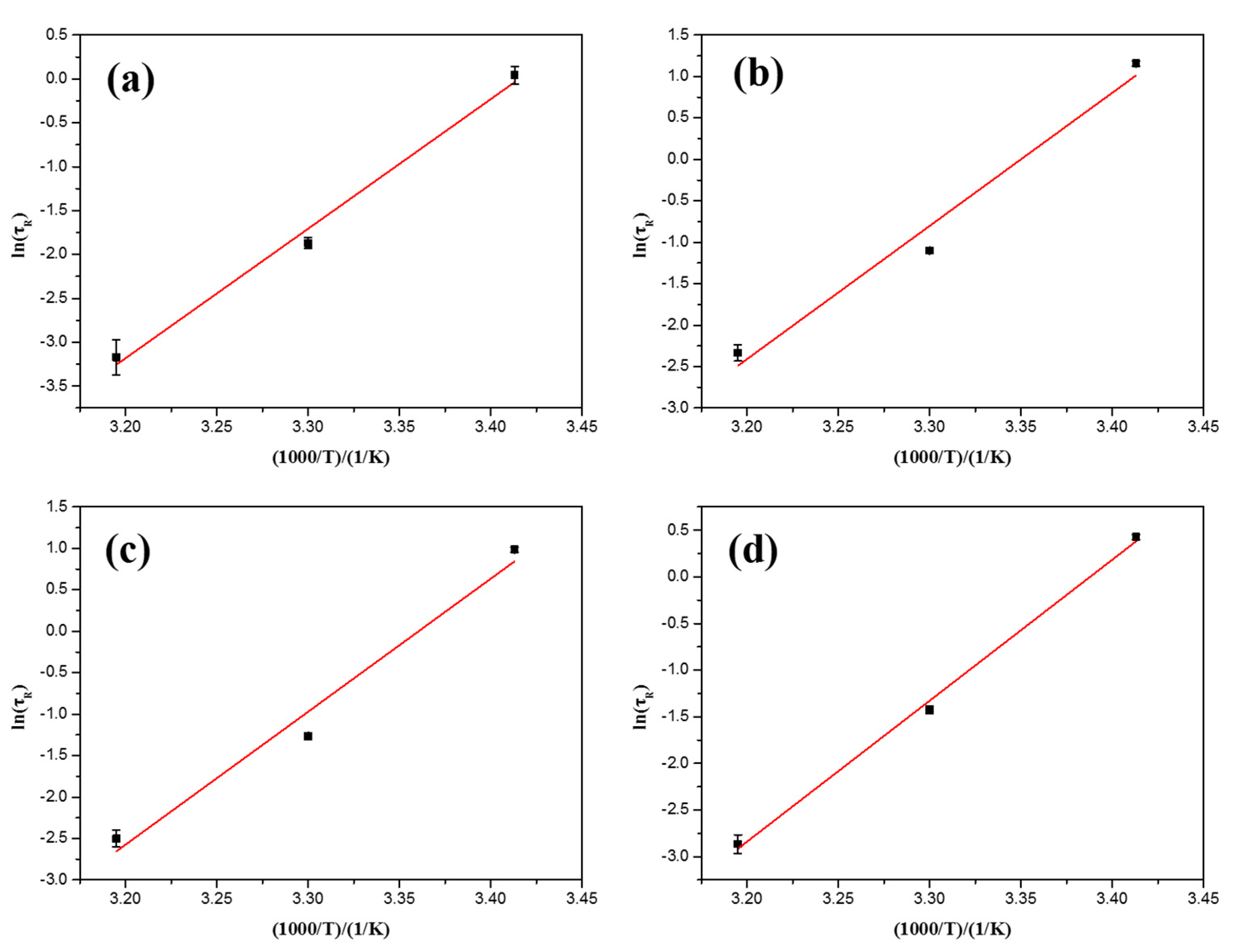
3.4. Mechanism Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Paul, D.R.; Robeson, L.M. Polymer nanotechnology: Nanocomposites. Polymer 2008, 49, 3187–3204. [Google Scholar] [CrossRef]
- Ansari, S.A.; Husain, Q. Potential applications of enzymes immobilized on/in nano materials: A review. Biotechnol. Adv. 2012, 30, 512–523. [Google Scholar] [CrossRef] [PubMed]
- Guerfi, A.; Sévigny, S.; Lagacé, M.; Hovington, P.; Kinoshita, K.; Zaghib, K. Nano-particle Li4Ti5O12 spinel as electrode for electrochemical generators. J. Power Sources 2003, 119, 88–94. [Google Scholar] [CrossRef]
- Bahiraei, M. Particle migration in nanofluids: A critical review. Int. J. Therm. Sci. 2016, 109, 90–113. [Google Scholar] [CrossRef]
- Bahiraei, M.; Alighardashi, M. Investigating non-newtonian nanofluid flow in a narrow annulus based on second law of thermodynamics. J. Mol. Liquids 2016, 219, 117–127. [Google Scholar] [CrossRef]
- Bahiraei, M.; Gharagozloo, K.; Alighardashi, M.; Mazaheri, N. CFD simulation of irreversibilities for laminar flow of a power-law nanofluid within a minichannel with chaotic perturbations: An innovative energy-efficient approach. Energy Convers. Manag. 2017, 144, 374–387. [Google Scholar] [CrossRef]
- Bahiraei, M.; Khosravi, R.; Heshmatian, S. Assessment and optimization of hydrothermal characteristics for a non-newtonian nanofluid flow within miniaturized concentric-tube heat exchanger considering designer’s viewpoint. Appl. Therm. Eng. 2017, 123, 266–276. [Google Scholar] [CrossRef]
- Bahiraei, M.; Mazaheri, N.; Alighardashi, M. Development of chaotic advection in laminar flow of a non-newtonian nanofluid: A novel application for efficient use of energy. Appl. Therm. Eng. 2017, 124, 1213–1223. [Google Scholar] [CrossRef]
- Yang, Z.Z.; Zhou, G.C.; Yong-Jun, L.U.; Zhu, C.Y. The mechanism of viscoelastic clean fracturing fluid and its application in oilfield. Drill. Fluid Complet. Fluld 2005, 22, 48–54. [Google Scholar]
- Acharya, D.P.; Kunieda, H. Wormlike micelles in mixed surfactant solutions. Adv. Colloid Interface Sci. 2006, 123–126, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Chu, Z.; Dreiss, C.A.; Feng, Y. Smart wormlike micelles. Chem. Soc. Rev. 2013, 42, 7174–7203. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Yuan, J.; Zheng, L. Spontaneous formation of vesicles by N-dodecyl-N-methylpyrrolidinium bromide (C 12 MPB) ionic liquid and sodium dodecyl sulfate (SDS) in aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2012, 407, 116–120. [Google Scholar] [CrossRef]
- Shrestha, R.G.; Shrestha, L.K.; Aramaki, K. Wormlike micelles in mixed amino acid-based anionic/nonionic surfactant systems. J. Colloid Interface Sci. 2008, 322, 596. [Google Scholar] [CrossRef] [PubMed]
- Spenley, N.A.; Cates, M.E.; Mcleish, T.C. Nonlinear rheology of wormlike micelles. Phys. Rev. Lett. 1993, 71, 939–942. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, R.; Sood, A.K. Effect of silica colloids on the rheology of viscoelastic gels formed by the surfactant cetyl trimethylammonium tosylate. J. Colloid Interface Sci. 2004, 283, 585. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Jia, Z.; Sun, H.; Liao, L.; Wen, Q. Rheological behavior and microstructure of an anionic surfactant micelle solution with pyroelectric nanoparticle. Colloids Surf. A Physicochem. Eng. Asp. 2012, 395, 267–275. [Google Scholar] [CrossRef]
- Wu, Y.; Luo, Z.; Yin, H.; Wang, T. Rheological characteristics of viscoelastic surfactant fluid mixed with silica nanoparticles. In Proceedings of the ASME 2013 International Conference on Micro/Nanoscale Heat and MASS Transfer, Hong Kong, China, 11–14 December 2013; p. V001T002A006. [Google Scholar]
- Narayanan, R.; El-Sayed, M.A. Catalysis with transition metal nanoparticles in colloidal solution: Nanoparticle shape dependence and stability. J. Phys Chem. B 2005, 36, 12663–12676. [Google Scholar] [CrossRef] [PubMed]
- Fanzatovich, I.I.; Aleksandrovich, K.D.; Rinatovich, I.A.; Evna, B.N.Y.; Yarullovna, Z.L.; Rashidovna, A.M.; Evgenevna, K.N. Supramolecular system based on cylindrical micelles of anionic surfactant and silica nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2016, 507, 255–260. [Google Scholar] [CrossRef]
- Nettesheim, F.; Liberatore, M.W.; Hodgdon, T.K.; Wagner, N.J.; Kaler, E.W.; Vethamuthu, M. Influence of nanoparticle addition on the properties of wormlike micellar solutions. Langmuir ACS J. Surf. Colloids 2008, 24, 7718. [Google Scholar] [CrossRef] [PubMed]
- Helgeson, M.E.; Hodgdon, T.K.; Kaler, E.W.; Wagner, N.J.; Vethamuthu, M.; Ananthapadmanabhan, K.P. Formation and rheology of viscoelastic “double networks” in wormlike micelle-nanoparticle mixtures. Langmuir ACS J. Surf. Colloids 2010, 26, 8049. [Google Scholar] [CrossRef] [PubMed]
- Cates, M.E.; Candau, S.J. Statics and dynamics of worm-like surfactant micelles. J. Phys. Condens. Matter 1999, 2, 9790–9797. [Google Scholar] [CrossRef]
- Zhao, M.; Zheng, L. Micelle formation by N-alkyl-N-methylpyrrolidinium bromide in aqueous solution. Phys. Chem. Chem. Phys. PCCP 2010, 13, 1332. [Google Scholar] [CrossRef] [PubMed]
- Walker, L.M. Rheology and structure of worm-like micelles. Curr. Opin. Colloid Interf. Sci. 2001, 6, 451–456. [Google Scholar] [CrossRef]
- Zhao, M.; Yan, Z.; Dai, C.; Du, M.; Li, H.; Zhao, Y.; Wang, K.; Ding, Q. Formation and rheological properties of wormlike micelles by N-hexadecyl-N-methylpiperidinium bromide and sodium salicylate. Colloid Polym. Sci. 2015, 293, 1–10. [Google Scholar] [CrossRef]
- Ezrahi, S.; Tuval, E.; Aserin, A. Properties, main applications and perspectives of worm micelles. Adv. Colloid Interface Sci. 2006, 128–130, 77–102. [Google Scholar] [CrossRef] [PubMed]
- Won, Y.Y.; Davis, H.T.; Bates, F.S. Giant wormlike rubber micelles. Science 1999, 283, 960–963. [Google Scholar] [CrossRef] [PubMed]
- Cates, M.E.; Fielding, S.M. Rheology of giant micelles. Adv. Phys. 2006, 55, 799–879. [Google Scholar] [CrossRef]
- Granek, R.; Cates, M.E. Stress relaxation in living polymers: Results from a poisson renewal model. J. Chem. Phys. 1992, 96, 4758–4767. [Google Scholar] [CrossRef]
- Klur, G.C.; Raghavan, S.R. Anionic wormlike micellar fluids that display cloud points: Rheology and phase behavior. J. Phys. Chem. B 2005, 109, 8599. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhao, M.; Zheng, L. Salt-induced wormlike micelles formed by N-alkyl-N-methylpyrrolidinium bromide in aqueous solution. Colloids Surf. A Physicochem. Eng. Asp. 2012, 396, 16–21. [Google Scholar] [CrossRef]

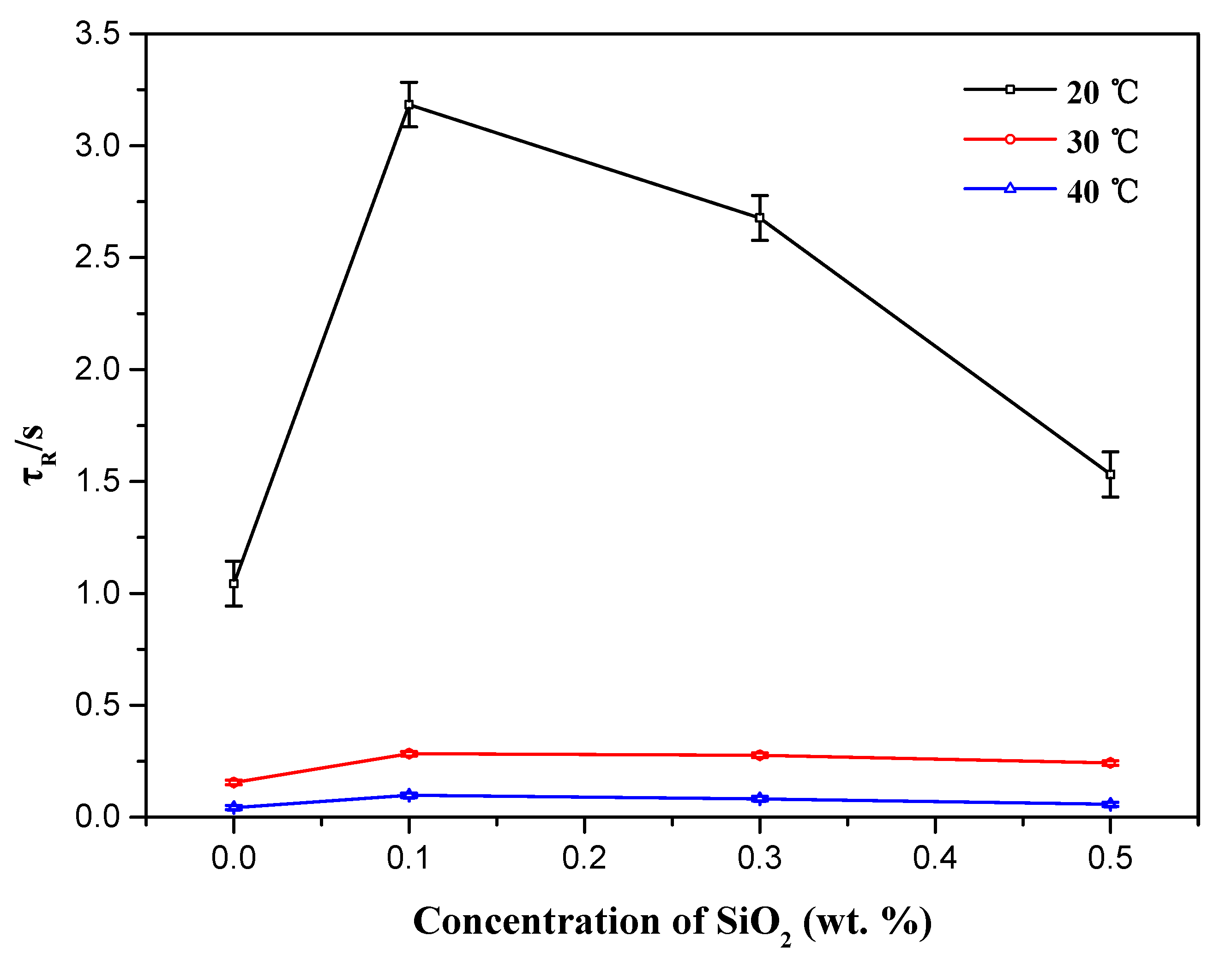

| Csilica (wt %) | T (°C) | η0 (Pa·s) | G′∞ (Pa) | G″min (Pa) | τR (s) | ξM (nm) | le (nm) | L (nm) |
|---|---|---|---|---|---|---|---|---|
| 0 | 20 | 15.311 ± 0.30 | 13.106 ± 0.02 | 1.118 ± 0.01 | 1.043 ± 0.10 | 67.62 ± 0.03 | 131–184 | 1536–2157 |
| 30 | 2.242 ± 0.10 | 13.954 ± 0.02 | 2.978 ± 0.01 | 0.154 ± 0.01 | 66.91 ± 0.03 | 129–181 | 604–848 | |
| 40 | 0.694 ± 0.01 | 13.664 ± 0.02 | 5.415 ± 0.01 | 0.042 ± 0.01 | 68.13 ± 0.03 | 133–187 | 336–472 | |
| 0.1 | 20 | 22.411 ± 0.30 | 14.691 ± 0.02 | 0.872 ± 0.01 | 3.184 ± 0.10 | 65.12 ± 0.03 | 123–173 | 2072–2914 |
| 30 | 5.375 ± 0.10 | 14.634 ± 0.02 | 1.996 ± 0.01 | 0.276 ± 0.01 | 65.91 ± 0.03 | 126–177 | 924–1298 | |
| 40 | 1.484 ± 0.01 | 14.001 ± 0.02 | 3.021 ± 0.01 | 0.097 ± 0.01 | 67.64 ± 0.03 | 131–184 | 607–853 | |
| 0.3 | 20 | 17.063 ± 0.30 | 14.139 ± 0.02 | 0.932 ± 0.01 | 2.677 ± 0.10 | 65.92 ± 0.03 | 126–177 | 1911–2685 |
| 30 | 4.917 ± 0.10 | 13.382 ± 0.02 | 2.072 ± 0.01 | 0.283 ± 0.01 | 67.93 ± 0.03 | 132–186 | 853–1201 | |
| 40 | 1.113 ± 0.01 | 14.292 ± 0.02 | 3.289 ± 0.01 | 0.082 ± 0.01 | 67.11 ± 0.03 | 130–182 | 564–791 | |
| 0.5 | 20 | 16.253 ± 0.30 | 11.537 ± 0.02 | 1.064 ± 0.01 | 1.532 ± 0.10 | 70.52 ± 0.03 | 140–197 | 1518–2136 |
| 30 | 3.531 ± 0.10 | 12.633 ± 0.02 | 2.204 ± 0.01 | 0.241 ± 0.01 | 69.23 ± 0.03 | 136–192 | 779–1100 | |
| 40 | 0.943 ± 0.01 | 13.178 ± 0.02 | 4.101 ± 0.01 | 0.057 ± 0.01 | 68.92 ± 0.03 | 135–190 | 434–611 |
| Csilica (wt %) | Ea (kJ/mol) |
|---|---|
| 0 | 122 |
| 0.1 | 134 |
| 0.3 | 131 |
| 0.5 | 125 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, M.; Zhang, Y.; Zou, C.; Dai, C.; Gao, M.; Li, Y.; Lv, W.; Jiang, J.; Wu, Y. Can More Nanoparticles Induce Larger Viscosities of Nanoparticle-Enhanced Wormlike Micellar System (NEWMS)? Materials 2017, 10, 1096. https://doi.org/10.3390/ma10091096
Zhao M, Zhang Y, Zou C, Dai C, Gao M, Li Y, Lv W, Jiang J, Wu Y. Can More Nanoparticles Induce Larger Viscosities of Nanoparticle-Enhanced Wormlike Micellar System (NEWMS)? Materials. 2017; 10(9):1096. https://doi.org/10.3390/ma10091096
Chicago/Turabian StyleZhao, Mingwei, Yue Zhang, Chenwei Zou, Caili Dai, Mingwei Gao, Yuyang Li, Wenjiao Lv, Jianfeng Jiang, and Yining Wu. 2017. "Can More Nanoparticles Induce Larger Viscosities of Nanoparticle-Enhanced Wormlike Micellar System (NEWMS)?" Materials 10, no. 9: 1096. https://doi.org/10.3390/ma10091096






