3.1. Ion-Exchange Method for Nickel(II) Perrhenate Production
The study consisted of 10 process cycles before the ion-exchange resin regeneration, and eight cycles after regeneration. It was found that the application of the previously-described conditions (see
Section 2.2) resulted in high sorption efficiency, exceeding 99%. Additionally, the saturation degree of the resin with nickel was also high, and amounted to 5.8%. About 10 dm
3 of the solution was treated in a single cycle. It was established that the ion-exchanger, after the regeneration, also guaranteed a stable course for the sorption process—for each cycle, the sorption efficiency exceeded 99.7%. The results of nickel ion sorption tests, using the ion-exchange resin, before and after the regeneration, are shown in
Table 1 and
Table 2, respectively.
The elution efficiencies of nickel ions (
Table 3 and
Table 4) were in the range of 53.4%–99.7%, and gradually declined in subsequent cycles. A very large drop in the elution efficiency in the 9th cycle was observed. Elution test results, after the resin regeneration, indicated that this operation is necessary for stable operation of the ion-exchanger. Elution efficiencies after the regeneration were higher, more stable, and equal to at least 99.0%.
The produced nickel(II) perrhenate tetrahydrate had elevated concentrations of sodium, magnesium, and calcium, harmful contaminants, as well as cobalt and iron, components often present in sintered heavy alloys; other contaminant levels were as expected. The content of zinc, bismuth, copper, and arsenic did not exceed 0.0002% in each studied sample. The lead, chromium, aluminum, molybdenum, and potassium levels in the separated compound were below 0.0005%, and carbon was <0.002%. The contents of impurities in the selected nickel(II) perrhenate tetrahydrate samples are shown in
Table 5.
Significant increases in calcium, magnesium, sodium, cobalt, and iron levels in the separated Ni(ReO4)2 were observed for the subsequent cycles of the process. Other element concentrations, such as carbon, molybdenum, manganese, zinc, potassium, copper, bismuth, aluminum, chromium, and arsenic, remained unchanged.
The first purification stage allowed the removal of all the calcium, iron, cobalt, and magnesium, as well as ca. 90% of the sodium. In the next purification step, the remaining sodium content was removed. The studies resulted in the development of an anhydrous nickel(II) perrhenate production method with the following composition: 10.5% Ni, 66.5% Re and <0.0005% Ca, <0.0005% K, <0.0005% Mg, <0.0005% Cu, <0.0005% Al, <0.0005% Cr, <0.0005% Mn, <0.0005% Na, <0.0005% Mo, <0.0005% Co, <0.0005% Pb, <0.0005% Fe, <0.002% C, <0.0002% Zn, <0.0002% As, and <0.0002% Bi. About 50 kg of the substance, with a standardized composition, was produced using this method. The manufactured anhydrous nickel(II) perrhenate was used in the investigation of tungsten-based, sintered heavy alloy production.
3.2. Reduction of Anhydrous Nickel(II) Perrhenate
The produced Re-Ni powder was examined using X-ray phase analysis, mass loss determination from the thermal reduction process, hydrogen loss measurements, metallographic observations of the produced powders, bulk and tapped density measurements, powder granulometry, and the determination of Re, Ni, and impurity content using chemical analysis.
It was found that complete material reduction can be achieved for nickel(II) perrhenate; thermal reduction was conducted at 800 °C for one hour.
Figure 2 and
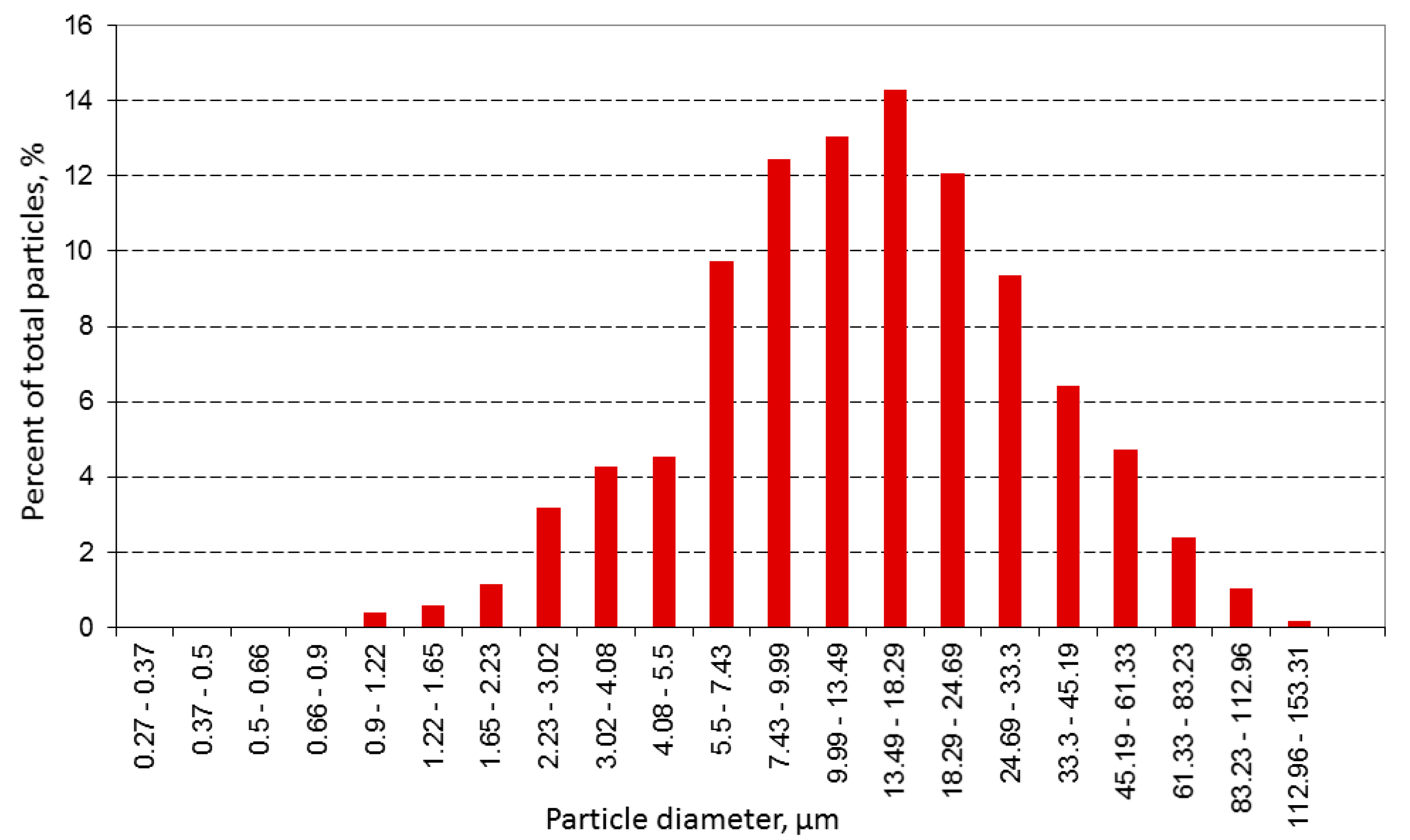
Figure 3 show the microstructure and size distribution of the reduced powder, respectively.
It was determined that powder particles formed agglomerates after reduction. Therefore, the powder should have been comminuted, e.g., using a ball mill, before its application in sintered alloy manufacturing.
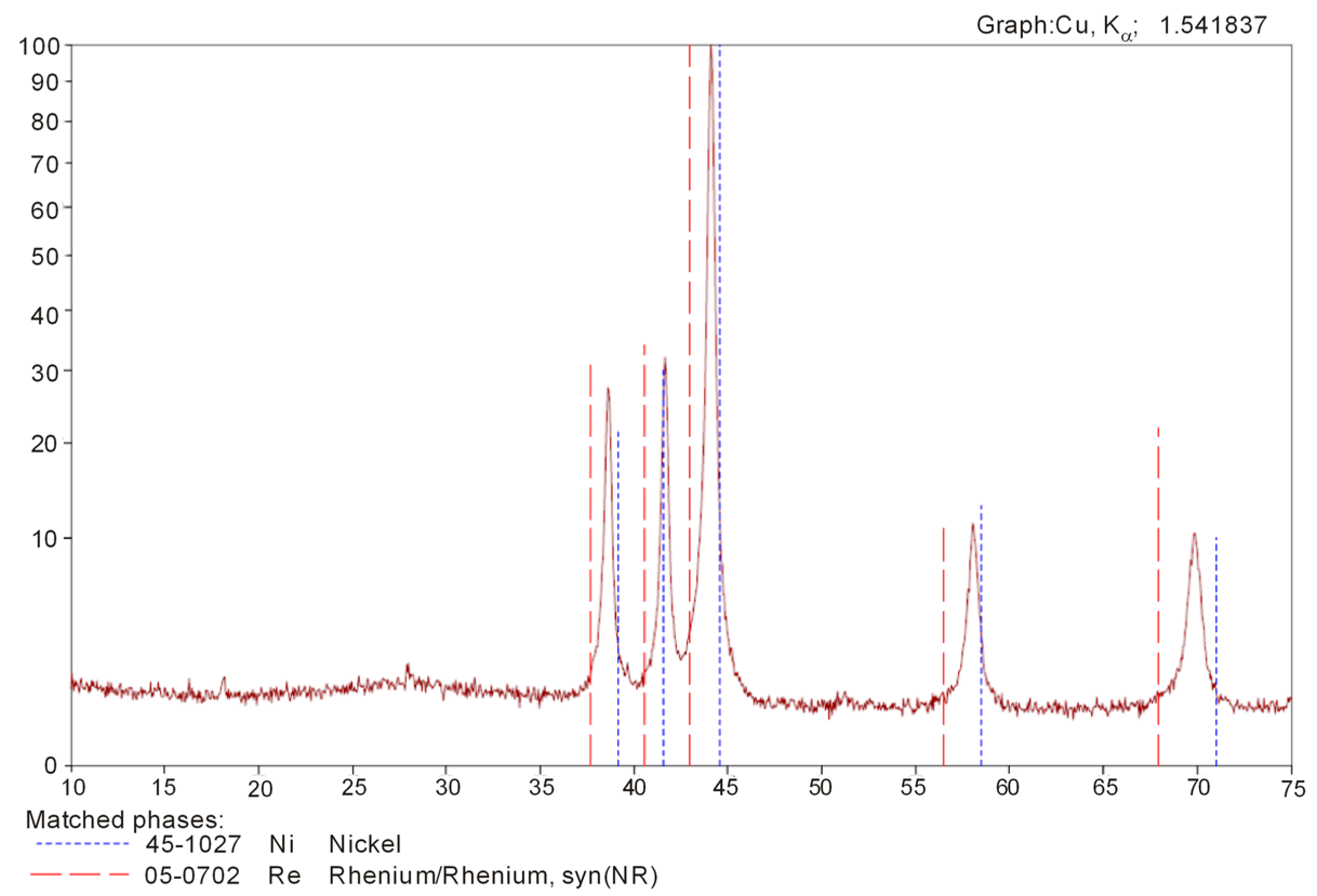
Figure 4 presents the diffraction pattern of the Re-Ni powder phase analysis. The thermal reduction process resulted in a completely alloyed powder, which contained: 13.6% Ni, 86.4% Re, <0.0005% Ca, <0.0005% K, <0.0005% Mg, <0.0005% Cu, <0.0005% Al, <0.0005% Cr, <0.0005% Mn, <0.0005% Na, <0.0005% Mo, <0.0005% Co, <0.0005% Pb, <0.0005% Fe, <0.002% C, <0.0002% Zn, <0.0002% As, and <0.0002% Bi.
3.3. Examination of 77W-20Re-3Ni Sintered Alloys
Selected examination results of 77W-20Re-3Ni sintered alloys, obtained according to the previously described procedure (see
Section 2.4) are presented below.
Figure 5 shows the microstructure of the produced sintered alloy. The particles of the high-melting phase and a small area of the bonding phase on the particle edges can be observed.
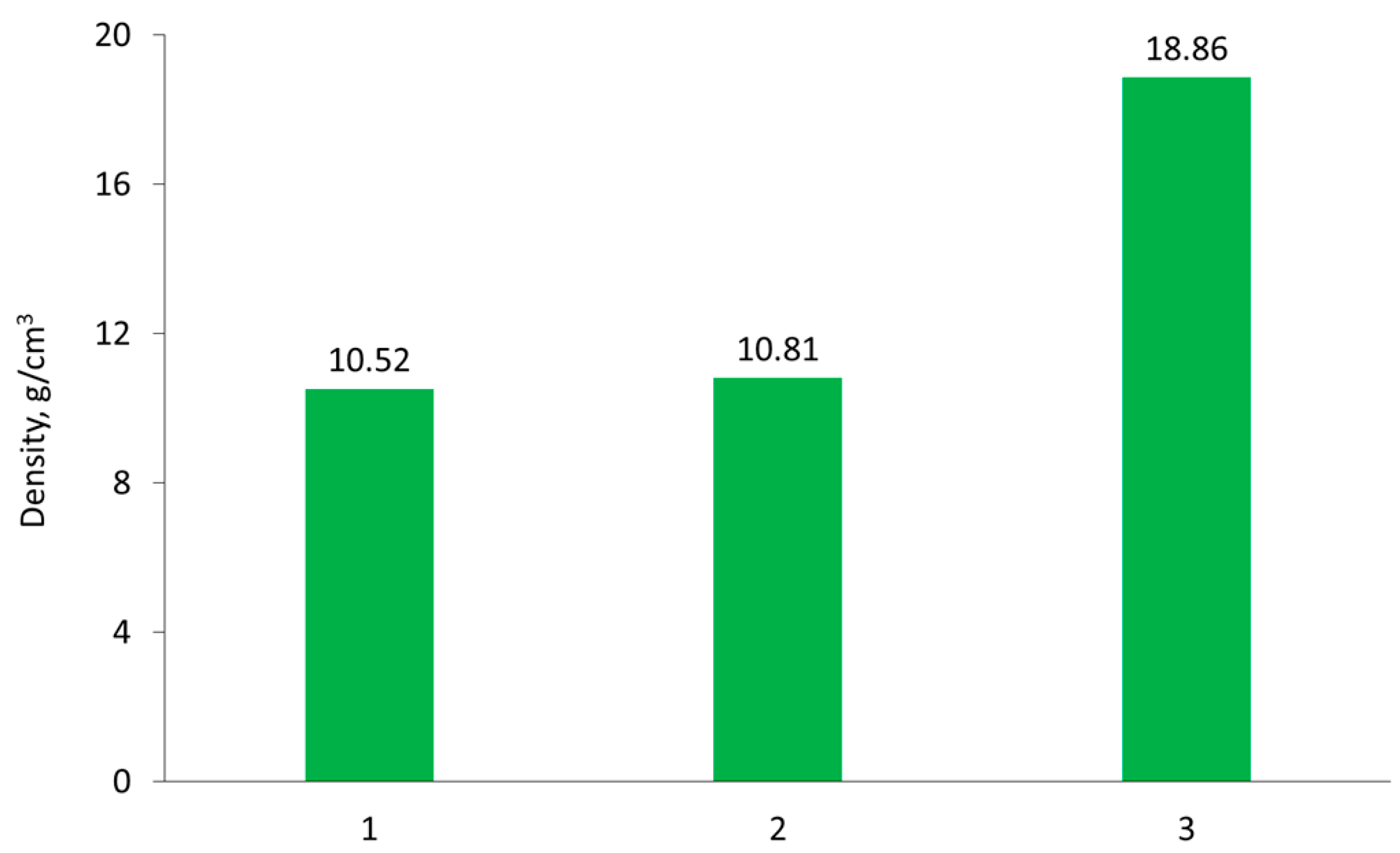
Figure 6 illustrates the density changes of the tested samples after various stages of the process.
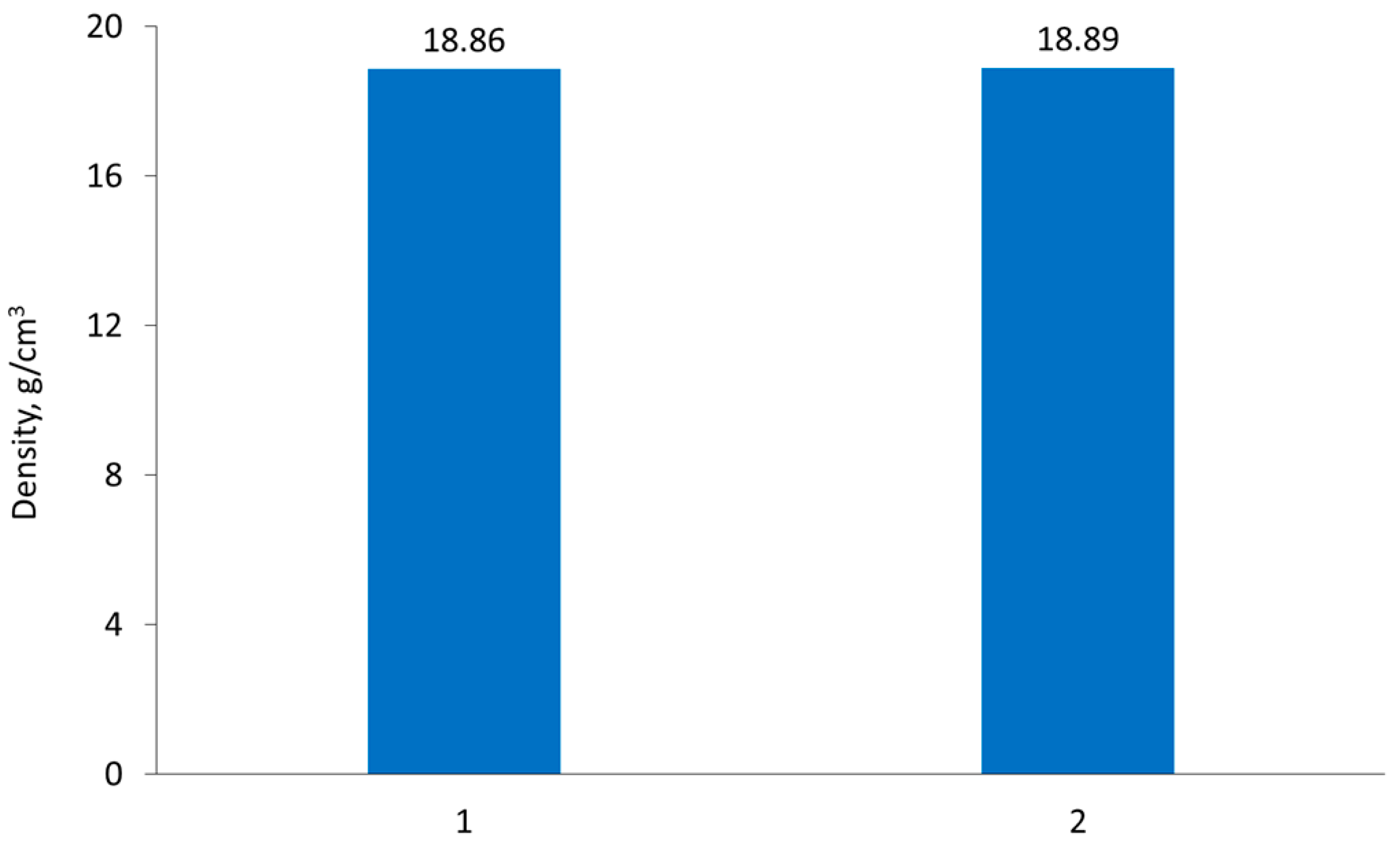
The influence of vacuum-sintering temperature on final product density was also determined.
Figure 7 shows that the temperature increase, from 1520 °C to 1650 °C, had no influence on density. Therefore, it may be concluded that a sintering temperature of 1520 °C provides sufficient process conditions for 77W-20Re-3Ni sintered alloy.
Table 6 presents selected properties of the sintered alloys, produced using Re-Ni alloy powder, compared to an alloy made from elemental powders (metallic rhenium obtained using ammonium perrhenate thermal reduction, as was used in comparative studies). The results presented in
Table 6 confirm that the application of Re-Ni alloy powder results in a sintered alloy with lower porosity, higher strength, and increased ductility, compared to that produced with elemental powders.