Thermophysical Characterization of MgCl2·6H2O, Xylitol and Erythritol as Phase Change Materials (PCM) for Latent Heat Thermal Energy Storage (LHTES)
Abstract
:1. Introduction
2. Material and Methods
2.1. Investigated PCM
2.2. Heat Capacity, Melting Temperature and Latent Heat
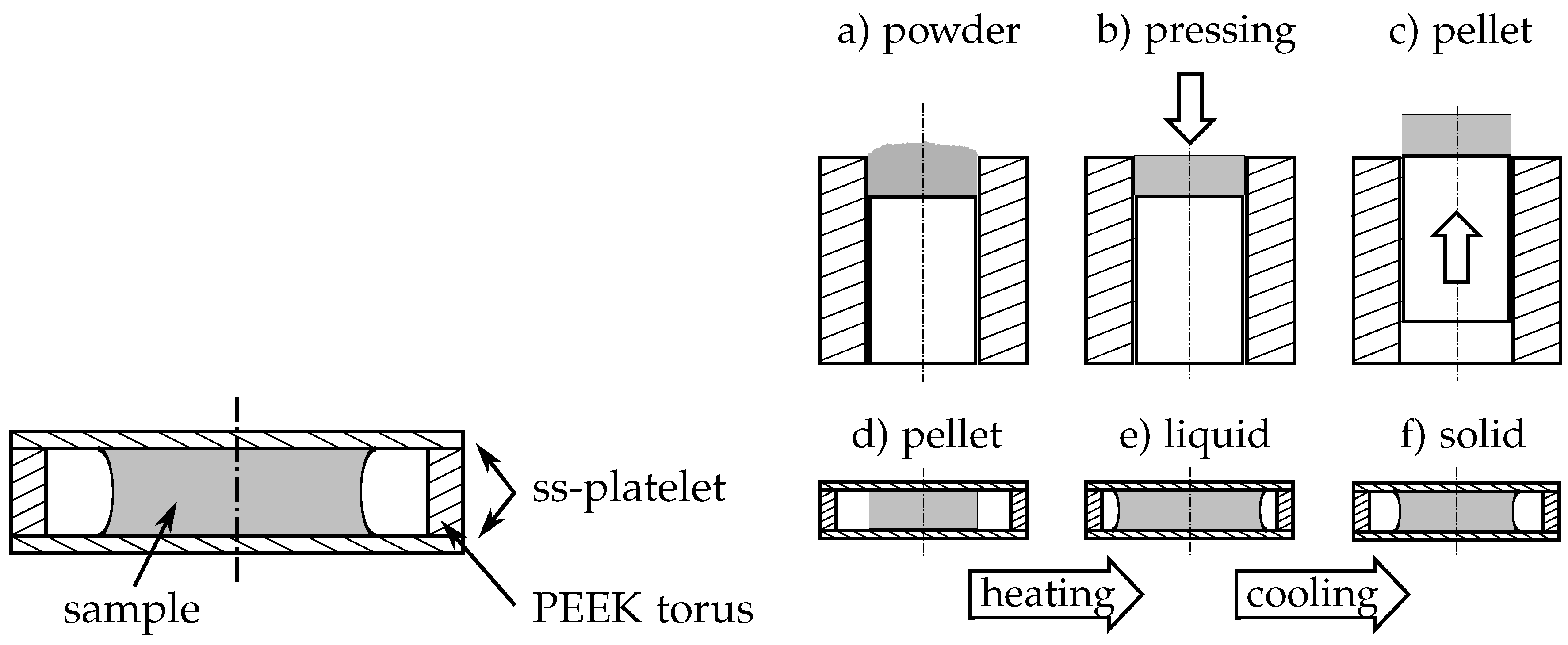
2.2.1. DSC
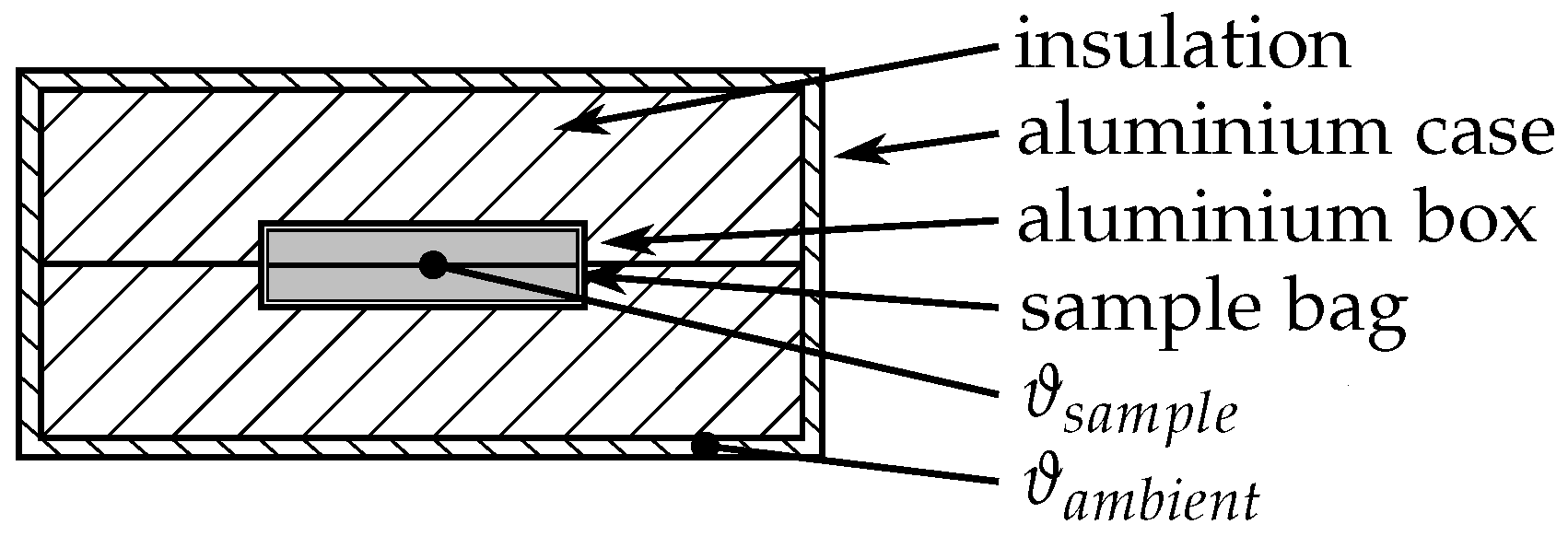
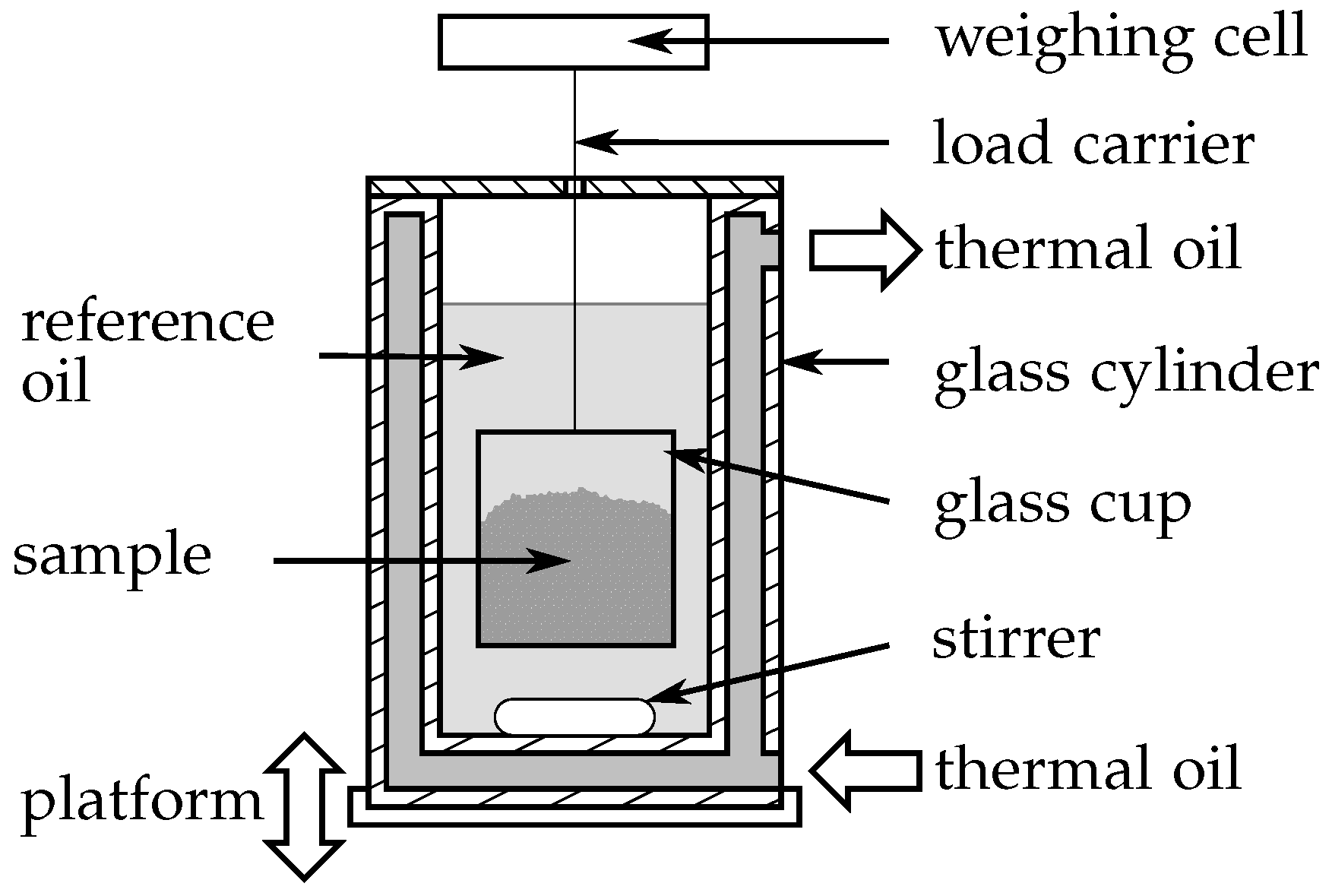
2.2.2. Three-Layer-Calorimeter
2.3. Density
2.4. Thermal Diffusivity and Conductivity
2.5. Cycling
3. Results and Discussion
3.1. Heat Capacity, Melting Temperature and Latent Heat
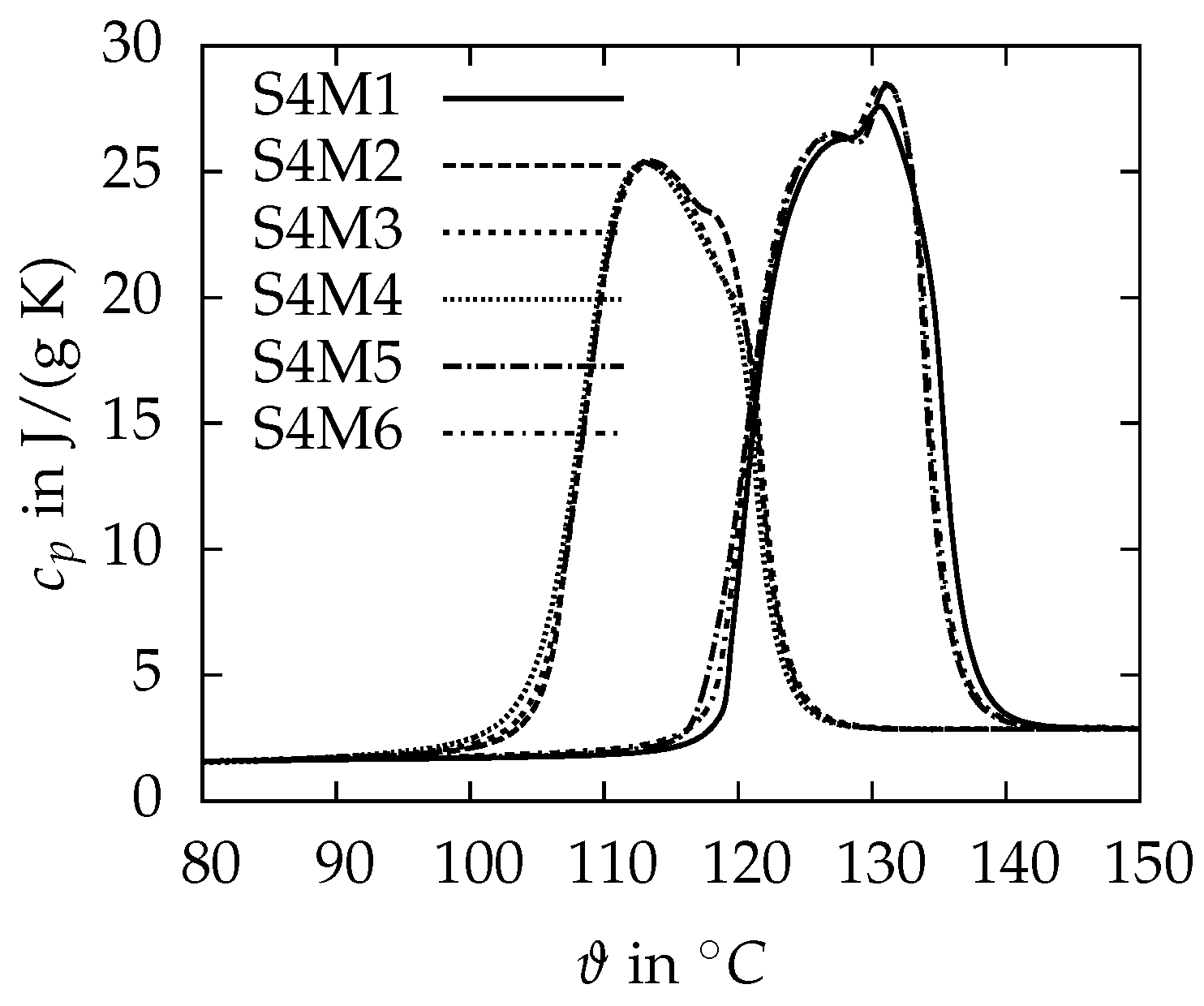
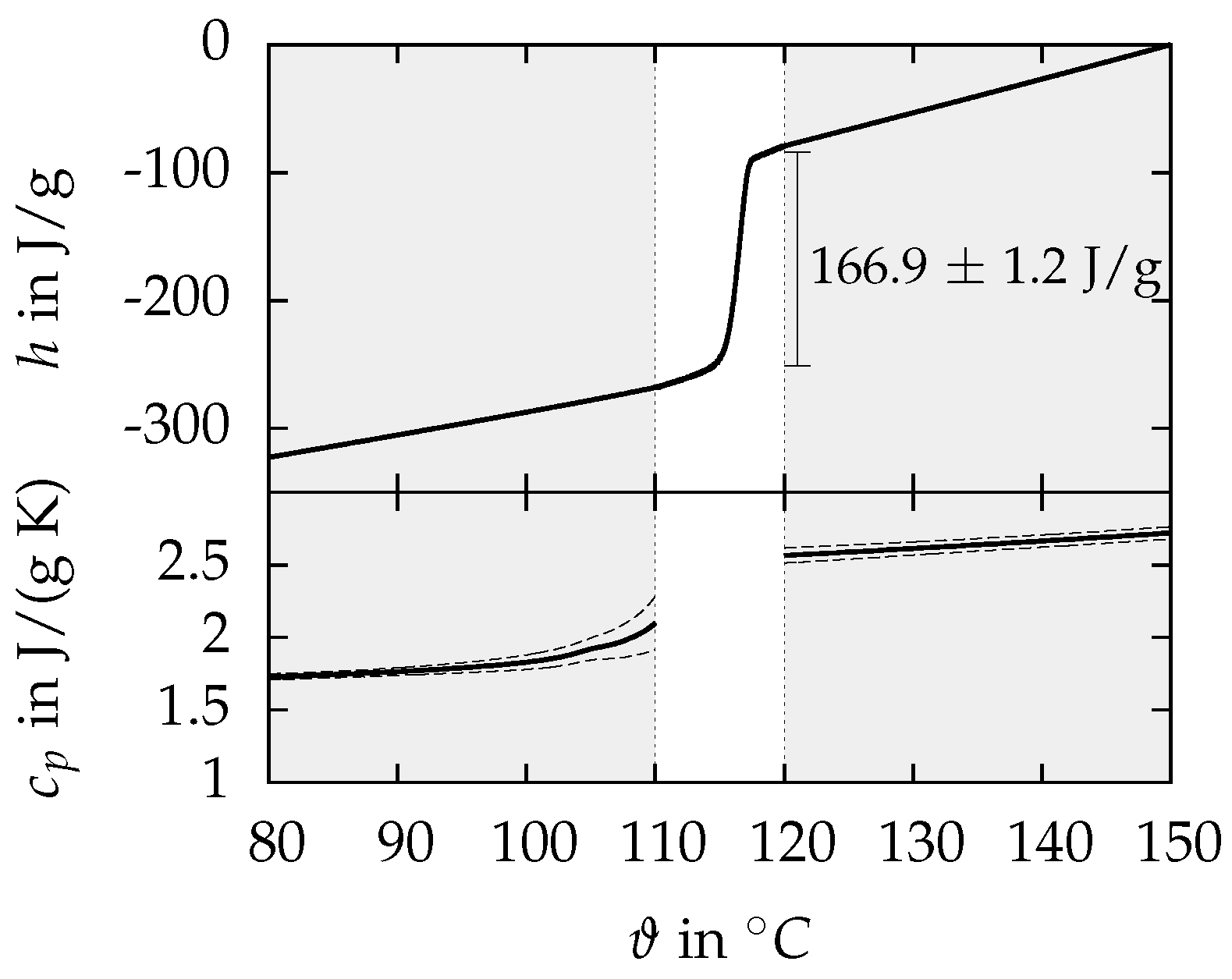
3.1.1. DSC
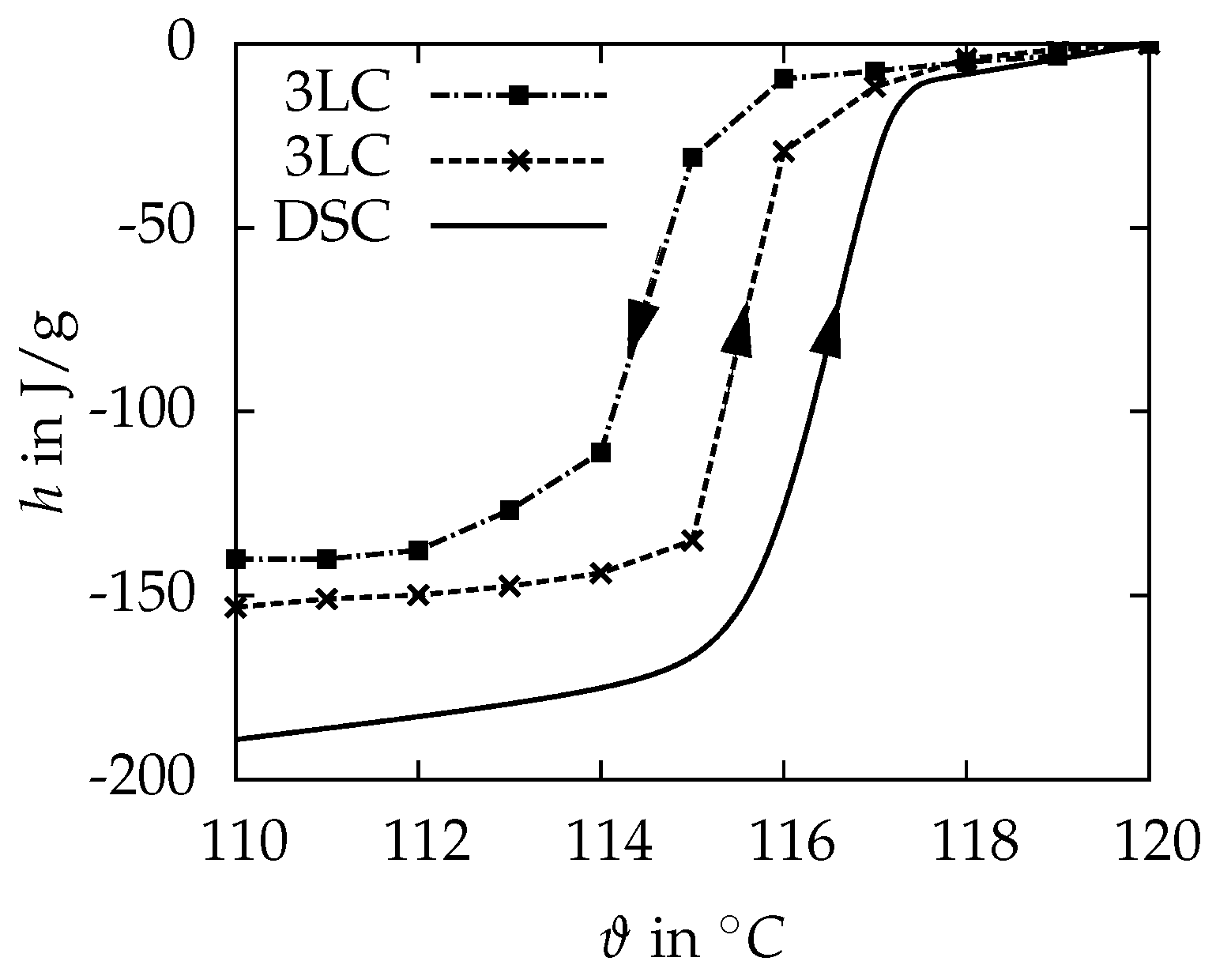
3.1.2. Three-Layer-Calorimeter
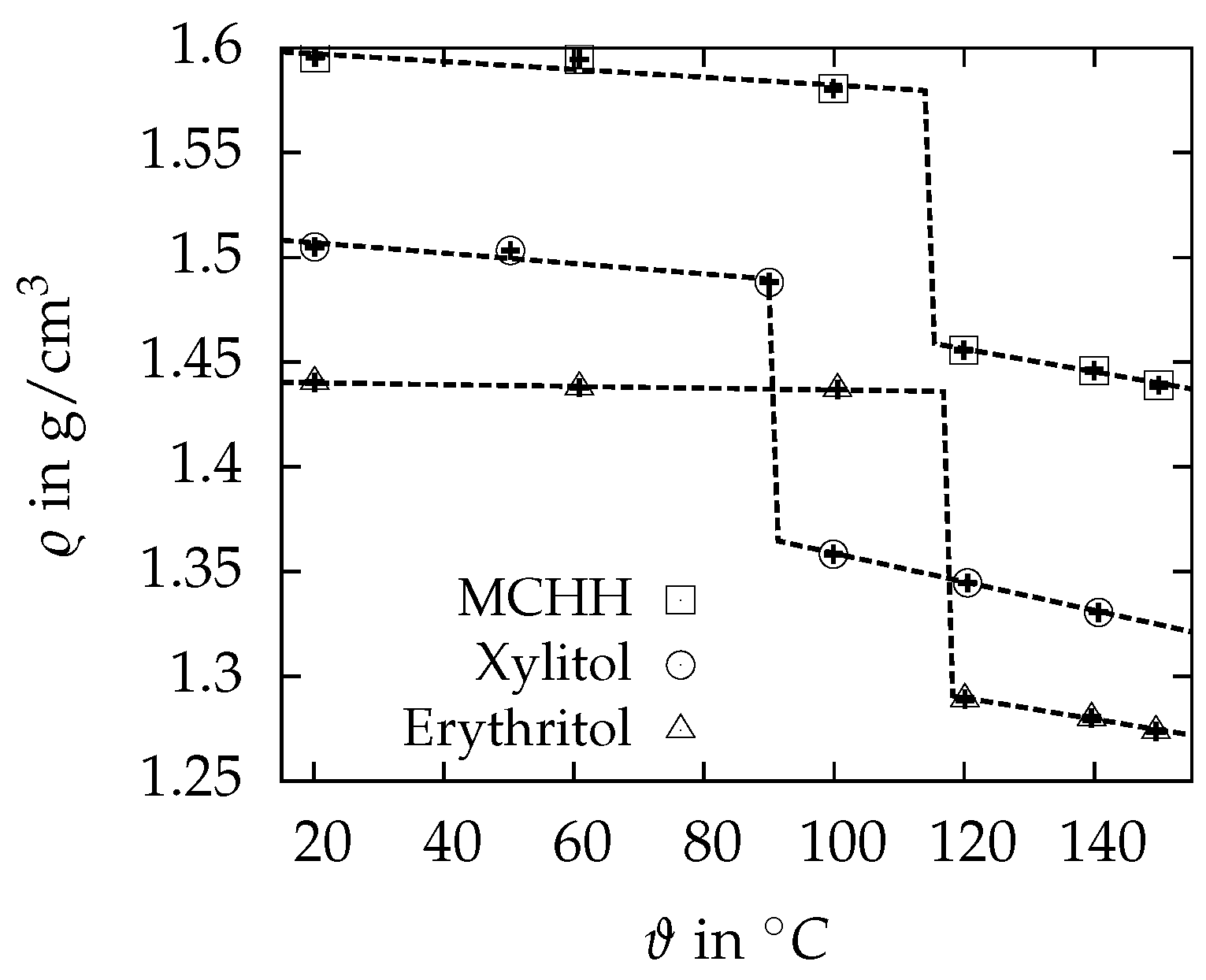
3.2. Density
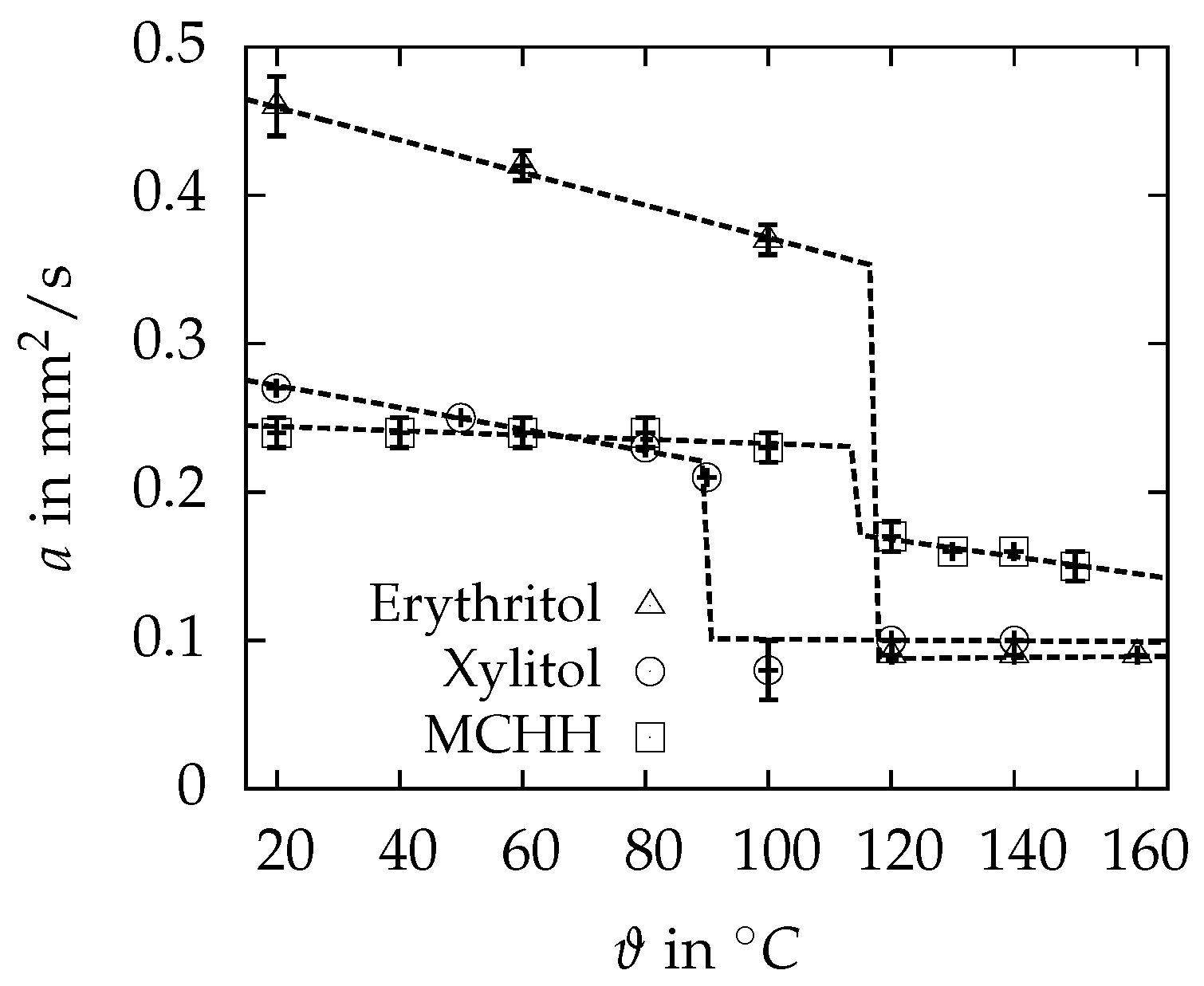
3.3. Thermal Diffusivity and Conductivity
3.4. Cycling
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 3LC | 3-layer-calorimeter | |
| DSC | Differential scanning calorimeter | |
| FEP | Fluorinated ethylene propylene | |
| LFA | Light flash apparatus | |
| LHTES | Latent heat thermal energy storage | |
| M | Measurement | |
| MCHH | Magnesiumchloride hexahydrate | |
| PEEK | Polyether ether ketone | |
| PCM | Phase change material | |
| ss | Stainless steel | |
| S | Sample | |
| a | Thermal diffusivity | |
| Heat capacity | ||
| , | Factors for linear equations | |
| , | Factors for linear equations | |
| h | Enthalpy | |
| k | Coverage factor | |
| W | Weighing value | |
| Volumetric coefficient of thermal expansion | ||
| % | Change of density from solid to liquid state | |
| Temperature | ||
| Supercooling | ||
| Thermal conductivity | ||
| Density | ||
| Reference state | ||
| Liquid (state) | ||
| Melting | ||
| Solid (state) |
References
- Storch, G.; Hauer, A. Cost-effectiveness of a heat energy distribution system based on mobile storage units: Two case studies. In Proceedings of the ECOSTOCK Conference, Stockton, NJ, USA, 31 May–2 June 2006. [Google Scholar]
- KTG Group. Available online: http://www.ktg-agrar.de/geschaeftsfelder/energieproduktion-biogas/latherm.html (accessed on 21 April 2017).
- Deckert, M.; Scholz, R.; Binder, S.; Hornung, A. Economic efficiency of mobile latent heat storages. Energy Procedia 2014, 46, 171–177. [Google Scholar] [CrossRef]
- Miró, L.; Gasia, J.; Cabeza, L.F. Thermal energy storage (TES) for industrial waste heat (IWH) recovery: A review. Appl. Energy 2016, 179, 284–301. [Google Scholar] [CrossRef]
- Mehling, H.; Cabeza, L.F. Heat and Cold Storage with PCM; Heat and Mass Transfer; Springer: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- Cohen, S.; Marcus, Y.; Migron, Y.; Dikstein, S.; Shafran, A. Water sorption, binding and solubility of polyols. J. Chem. Soc. Faraday Trans. 1993, 89, 3271. [Google Scholar] [CrossRef]
- Kaizawa, A.; Maruoka, N.; Kawai, A.; Kamano, H.; Jozuka, T.; Senda, T.; Akiyama, T. Thermophysical and heat transfer properties of phase change material candidate for waste heat transportation system. Heat Mass Transf. 2007, 44, 763–769. [Google Scholar] [CrossRef]
- Lopes Jesus, A.J.; Nunes, S.C.C.; Ramos Silva, M.; Matos Beja, A.; Redinha, J.S. Erythritol: Crystal growth from the melt. Int. J. Pharm. 2010, 388, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Shukla, A.; Buddhi, D.; Sawhney, R.L. Thermal cycling test of few selected inorganic and organic phase change materials. Renew. Energy 2008, 33, 2606–2614. [Google Scholar] [CrossRef]
- Sari, A.; Eroglu, R.; Biçer, A.; Karaipekli, A. Synthesis and Thermal Energy Storage Properties of Erythritol Tetrastearate and Erythritol Tetrapalmitate. Chem. Eng. Technol. 2011, 34, 87–92. [Google Scholar] [CrossRef]
- Ona, E.P.; Kojima, Y.; Matsuda, H.; Hidaka, H.; Kakiuchi, H. Latent Heat Storage Characteristics of Erythritol-Magnesium Chloride Hexahydrate Mixtures. Asian Pac. Confed. Chem. Eng. Congr. Program Abstr. 2004, 2004, 536. [Google Scholar]
- Rathgeber, C.; Miró, L.; Cabeza, L.F.; Hiebler, S. Measurement of enthalpy curves of phase change materials via DSC and T-History: When are both methods needed to estimate the behaviour of the bulk material in applications? Thermochim. Acta 2014, 596, 79–88. [Google Scholar] [CrossRef]
- Wei, L.; Ohsasa, K. Supercooling and Solidification Behavior of Phase Change Material. ISIJ Int. 2010, 50, 1265–1269. [Google Scholar] [CrossRef]
- Agyenim, F.; Eames, P.; Smyth, M. Experimental study on the melting and solidification behaviour of a medium temperature phase change storage material (Erythritol) system augmented with fins to power a LiBr/H2O absorption cooling system. Renew. Energy 2011, 36, 108–117. [Google Scholar] [CrossRef]
- Sharma, S.D.; Iwata, T.; Kitano, H.; Sagara, K. Thermal performance of a solar cooker based on an evacuated tube solar collector with a PCM storage unit. Sol. Energy 2005, 78, 416–426. [Google Scholar] [CrossRef]
- Pawar, U.C.; Shankargouda, S.J.; Honguntiker, P. Solar Energy for Cooking Food in Urban Buildings. Int. J. Eng. Technol. Manag. Appl. Sci. 2015, 3, 190–194. [Google Scholar]
- Kaizawa, A.; Kamano, H.; Kawai, A.; Jozuka, T.; Senda, T.; Maruoka, N.; Okinaka, N.; Akiyama, T. Technical Feasibility Study of Waste Heat Transportation System Using Phase Change Material from Industry to City. ISIJ Int. 2008, 48, 540–548. [Google Scholar] [CrossRef]
- Wang, W. Mobilized Thermal Energy Storage for Heat Recovery for Distributed Heating; Number 92; Mälardalen University: Västerås, Sweden, 2010. [Google Scholar]
- Seppälä, A.; Meriläinen, A.; Wikström, L.; Kauranen, P. The effect of additives on the speed of the crystallization front of xylitol with various degrees of supercooling. Exp. Therm. Fluid Sci. 2010, 34, 523–527. [Google Scholar] [CrossRef]
- Lane, G.A. Solar Heat Storage: Latent Heat Material Volume 2; CRC Press: Boca Raton, FL, USA, 1983. [Google Scholar]
- Cantor, S. DSC study of melting and solidification of salt hydrates. Thermochim. Acta 1979, 33, 69–86. [Google Scholar] [CrossRef]
- Choi, J.C.; Kim, S.D. Heat-transfer characteristics of a latent heat storage system using MgCl2·6H2O. Energy 1992, 17, 1153–1164. [Google Scholar] [CrossRef]
- Gonçalves, L.C.C.; Probert, S.D. Thermal-energy storage: Dynamic performance characteristics of cans each containing a phase-change material, assembled as a packed-bed. Appl. Energy 1993, 45, 117–155. [Google Scholar] [CrossRef]
- Pilar, R.; Svoboda, L.; Honcova, P.; Oravova, L. Study of magnesium chloride hexahydrate as heat storage material. Thermochim. Acta 2012, 546, 81–86. [Google Scholar] [CrossRef]
- El-Sebaii, A.A.; Al-Amir, S.; Al-Marzouki, F.M.; Faidah, A.S.; Al-Ghamdi, A.A.; Al-Heniti, S. Fast thermal cycling of acetanilide and magnesium chloride hexahydrate for indoor solar cooking. Energy Convers. Manag. 2009, 50, 3104–3111. [Google Scholar] [CrossRef]
- El-Sebaii, A.A.; Al-Heniti, S.; Al-Agel, F.; Al-Ghamdi, A.A.; Al-Marzouki, F. One thousand thermal cycles of magnesium chloride hexahydrate as a promising PCM for indoor solar cooking. Energy Convers. Manag. 2011, 52, 1771–1777. [Google Scholar] [CrossRef]
- Hasnain, S.M. Review on sustainable thermal energy storage technologies, Part I: Heat storage materials and techniques. Energy Convers. Manag. 1998, 39, 1127–1138. [Google Scholar] [CrossRef]
- Talja, R.A.; Roos, Y.H. Phase and state transition effects on dielectric, mechanical, and thermal properties of polyols. Thermochim. Acta 2001, 380, 109–121. [Google Scholar] [CrossRef]
- Diarce, G.; Gandarias, I.; Campos-Celador, A.; García-Romero, A.; Griesser, U. Eutectic mixtures of sugar alcohols for thermal energy storage in the 50–90 °C temperature range. Sol. Energy Mater. Sol. Cells 2015, 134, 215–226. [Google Scholar] [CrossRef]
- Barone, G.; Gatta, G.D.; Ferro, D.; Piacente, V. Enthalpies and entropies of sublimation, vaporization and fusion of nine polyhydric alcohols. J. Chem. Soc. Faraday Trans. 1990, 86, 75. [Google Scholar] [CrossRef]
- Kakiuchi, H.; Yamazaki, M.; Yabe, M.; Chihara, S.; Terunuma, Y.; Sakata, Y.; Usami, T. A Study of Erythritol as Phase Change Material. In Proceedings of the 2nd workshop IEA annex 10, Phase Change Materials and Chemical Reactions for Thermal Energy Storage, Sofia, Bulgaria, 11–13 April 1998. [Google Scholar]
- Ushak, S.; Gutierrez, A.; Galleguillos, H.; Fernandez, A.G.; Cabeza, L.F.; Grágeda, M. Thermophysical characterization of a by-product from the non-metallic industry as inorganic PCM. Sol. Energy Mater. Sol. Cells 2015, 132, 385–391. [Google Scholar] [CrossRef]
- Gschwander, S.; Haussmann, T.; Hagelstein, G.; Sole, A.; Hohenauer, W.; Lager, D.; Rathgeber, C.; Lazaro, A.; Mehling, H. Standard to Determine the Heat Storage Capacity of PCM Using hf-DSC with Constant Heating/Cooling Rate (Dynamic Mode)—DSC 4229 PCM. Standard—A Technical Report of Subtask A2.1 of IEA-SHC 42/ECES Annex 29. 31 January 2015. Available online: http://task42.iea-shc.org/publications (accessed on 21 April 2017).
- NETZSCH. Bestimmung der Spezifischen Wärme Mit DSC—Schwerpunktkurs DSC. Available online: https://www.netzsch-thermal-analysis.com/en/products-solutions/dilatometer/dil-402-expedis-select-supreme/ (accessed on 21 April 2017).
- Höhne, G.W.H.; Hemminger, W.F.; Flammersheim, H.J. Differential Scanning Calorimetry; Springer: Berlin/Heidelberg, Germany, 2003. [Google Scholar]
- Laube, A. W&A—Waerme—Und Anwendungstechnische Pruefungen. Available online: http://www.waermepruefung.de (accessed on 21 April 2017).
- Solé, A.; Miró, L.; Barreneche, C.; Martorell, I.; Cabeza, L.F. Review of the T-history method to determine thermophysical properties of phase change materials (PCM). Renew. Sustain. Energy Rev. 2013, 26, 425–436. [Google Scholar] [CrossRef]
- Breitwieser, M. IMETER—MSB Breitwieser. Available online: http://www.imeter.de (accessed on 21 April 2017).
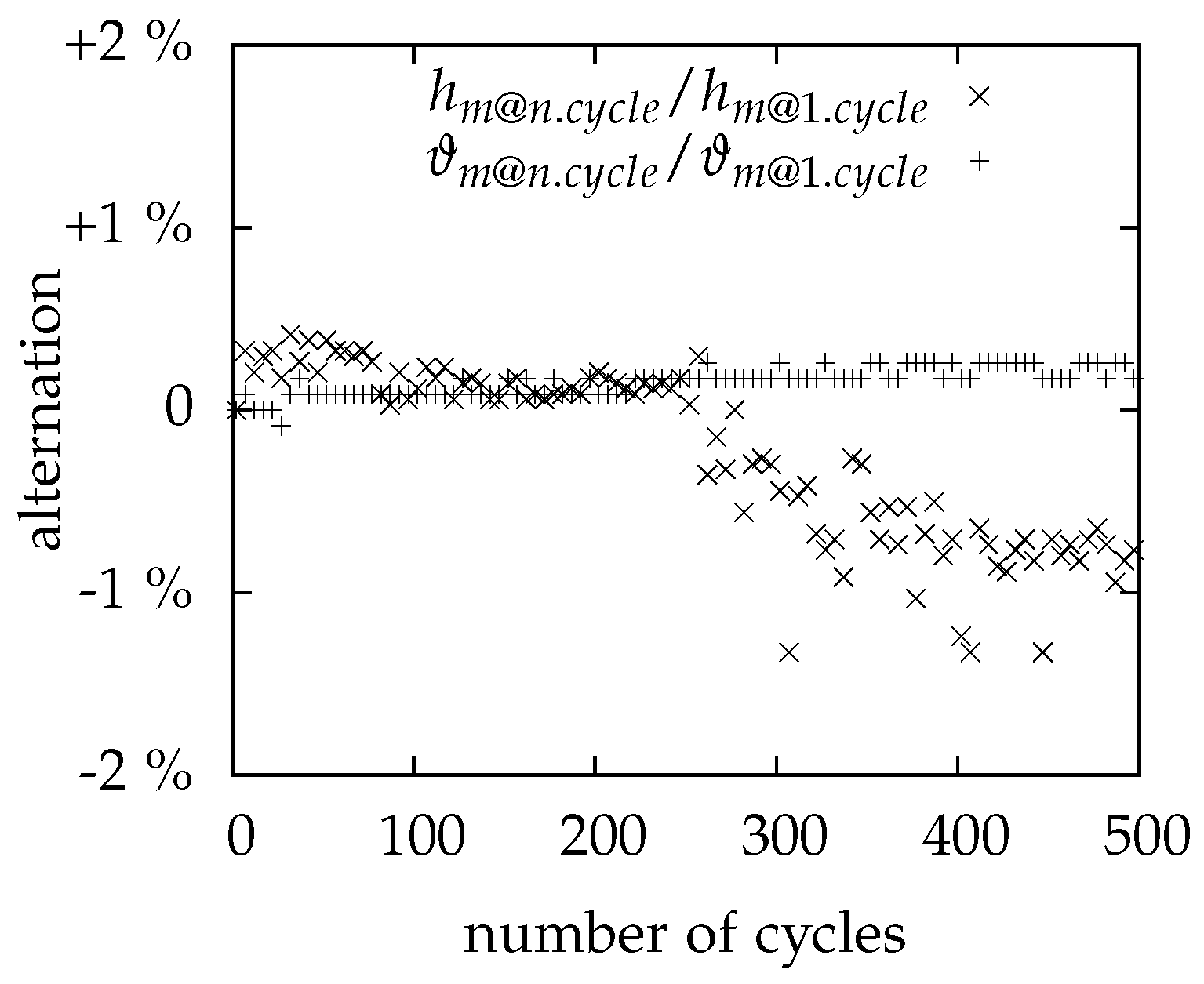
| Property | Erythritol | Source | Xylitol | Source | MCHH | Source |
|---|---|---|---|---|---|---|
| in | 117–120 | [5,7,9,10,28,29] | 92–94 | [5,7,19,28,29,30] | 110.8–117.5 | [5,20,21,24,25,26] |
| in | 315–379.57 | [5,7,9,10,12,28,29] | 232–280 | [5,7,19,28,29,30] | 133.9–200 | [5,12,20,21,24,25,26] |
| in | 1.38 (20 ) | [31] | 1.33 | [30] | 2.25 (100 ) | [20] |
| 2.1 (25 ) | [32] | |||||
| in | 2.76 (140 ) | [31] | 2.36 | [30] | 2.61 (120 ) | [20] |
| in | 0.733 (20 ) | [5] | - | - | 0.704 (110 ) | [5,20] |
| in | 0.326 (140 ) | [5] | - | - | 0.570 (120 ) | [5,20] |
| in | 1.480 (20 ) | [5] | 1.500 (20 ) | [5] | 1.569 (20 ) | [5,20] |
| in | 1.300 (140 ) | [5] | - | - | 1.450 (120 ) | [5,20] |
| 1.422 (128 ) | [32] |
| Range | ||
|---|---|---|
| solid (80–110 ) | 1.155 | |
| liquid (120–150 ) | 1.945 |
| Range | ||
|---|---|---|
| Erythritol | ||
| s. (20–118 ) | ||
| l. (118–150 ) | ||
| Xylitol | ||
| s. (20–90 ) | ||
| l. (90–140 ) | ||
| MgCl6HO | ||
| s. (20–115 ) | ||
| l. (115–150 ) | ||
| Property | Erythritol | Xylitol | MCHH |
|---|---|---|---|
| in | 105.1 ± 0.1 | 90 ± 1 | 115.1 ± 0.1 |
| 118.1 ± 0.6 | |||
| in | 118.2 | - | 115.8 |
| in | 60 | >90 | 30 |
| in | 47 | - | 2.8 |
| in | 316 ± 1 (90–135 ) | 237.6 ± 1.3 (70–116 ) | 166.9 ± 1.2 (114–118 ) |
| 352.9 ± 0.7 (110–145 ) | |||
| in | 337 (110–125 ) | - | 143.4 (114–118 ) |
| in | 1.34 ± 0.09 (20 ) | 1.27 ± 0.05 (20 ) | 1.83 ± 0.06 (100 ) |
| in | 2.87 ± 0.03 (150 ) | 2.73 ± 0.08 (120 ) | 2.57 ± 0.06 (120 ) |
| in | 0.456 ± 0.018 | 0.270 ± 0.002 | 0.244 ± 0.011 |
| in | 0.088 ± 0.001 | 0.100 ± 0.001 | 0.173 ± 0.008 |
| in | 1.4404 ± 0.0005 | 1.5050 ± 0.0004 | 1.5955 ± 0.0002 |
| in | 2.94 · 10−5 | 1.64 · 10−4 | 1.17 · 10−4 |
| in | 1.2891 ± 0.0008 | 1.3446 ± 0.0003 | 1.4557 ± 0.0004 |
| in | 3.95 · 10−4 | 5.02 · 10−4 | 3.76 · 10−4 |
| in % | 10.1 | 8.4 | 7.7 |
| in | 0.89 ± 0.06 (20 ) | 0.52 ± 0.04 (20 ) | 0.70 ± 0.05 (110 ) |
| in | 0.33 ± 0.02 (140 ) | 0.36 ± 0.03 (140 ) | 0.63 ± 0.04 (120 ) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Höhlein, S.; König-Haagen, A.; Brüggemann, D. Thermophysical Characterization of MgCl2·6H2O, Xylitol and Erythritol as Phase Change Materials (PCM) for Latent Heat Thermal Energy Storage (LHTES). Materials 2017, 10, 444. https://doi.org/10.3390/ma10040444
Höhlein S, König-Haagen A, Brüggemann D. Thermophysical Characterization of MgCl2·6H2O, Xylitol and Erythritol as Phase Change Materials (PCM) for Latent Heat Thermal Energy Storage (LHTES). Materials. 2017; 10(4):444. https://doi.org/10.3390/ma10040444
Chicago/Turabian StyleHöhlein, Stephan, Andreas König-Haagen, and Dieter Brüggemann. 2017. "Thermophysical Characterization of MgCl2·6H2O, Xylitol and Erythritol as Phase Change Materials (PCM) for Latent Heat Thermal Energy Storage (LHTES)" Materials 10, no. 4: 444. https://doi.org/10.3390/ma10040444






