Investigating the Effects of Loading Factors on the In Vitro Pharmaceutical Performance of Mesoporous Materials as Drug Carriers for Ibuprofen
Abstract
:1. Introduction
2. Results
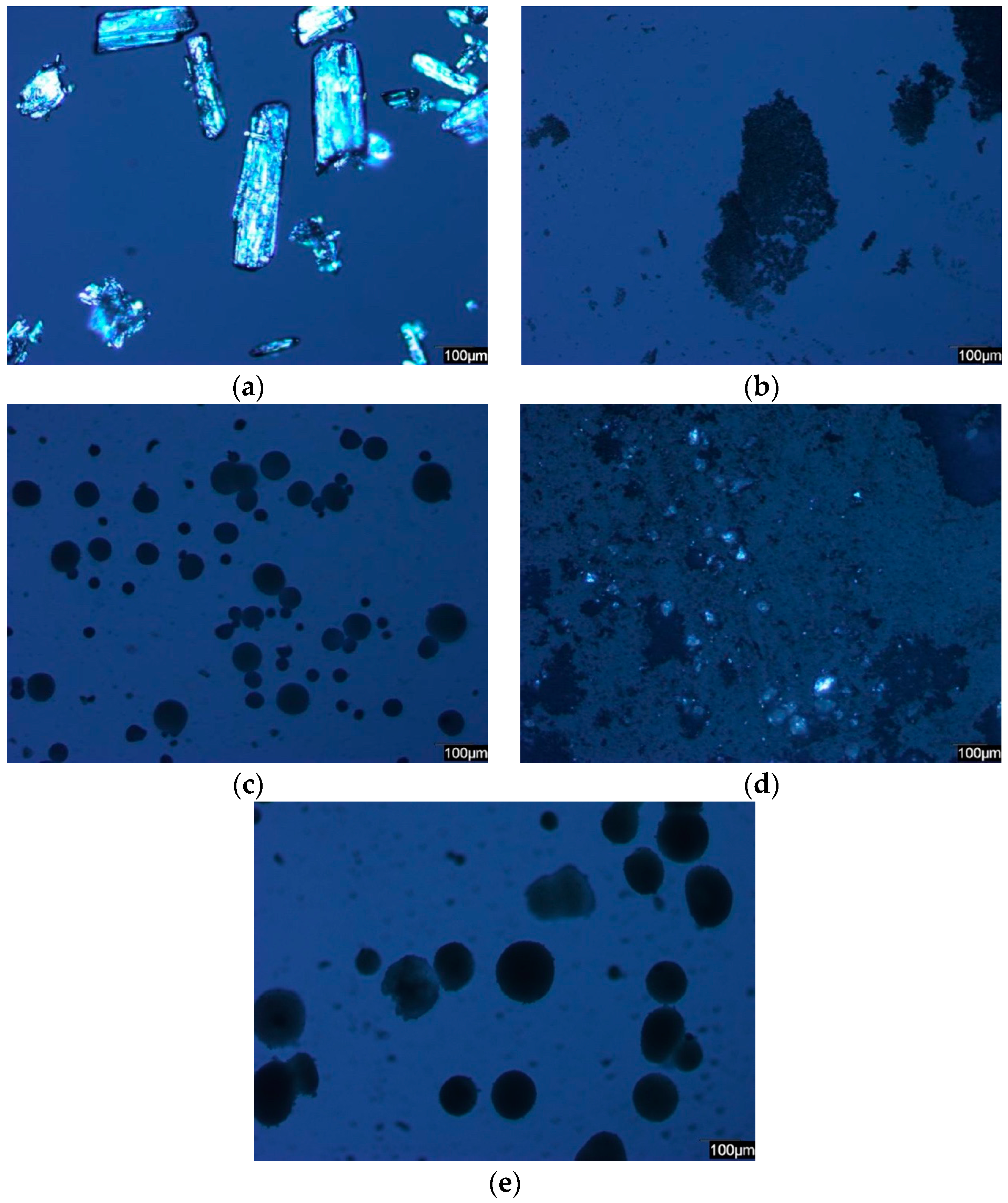
2.1. PLM
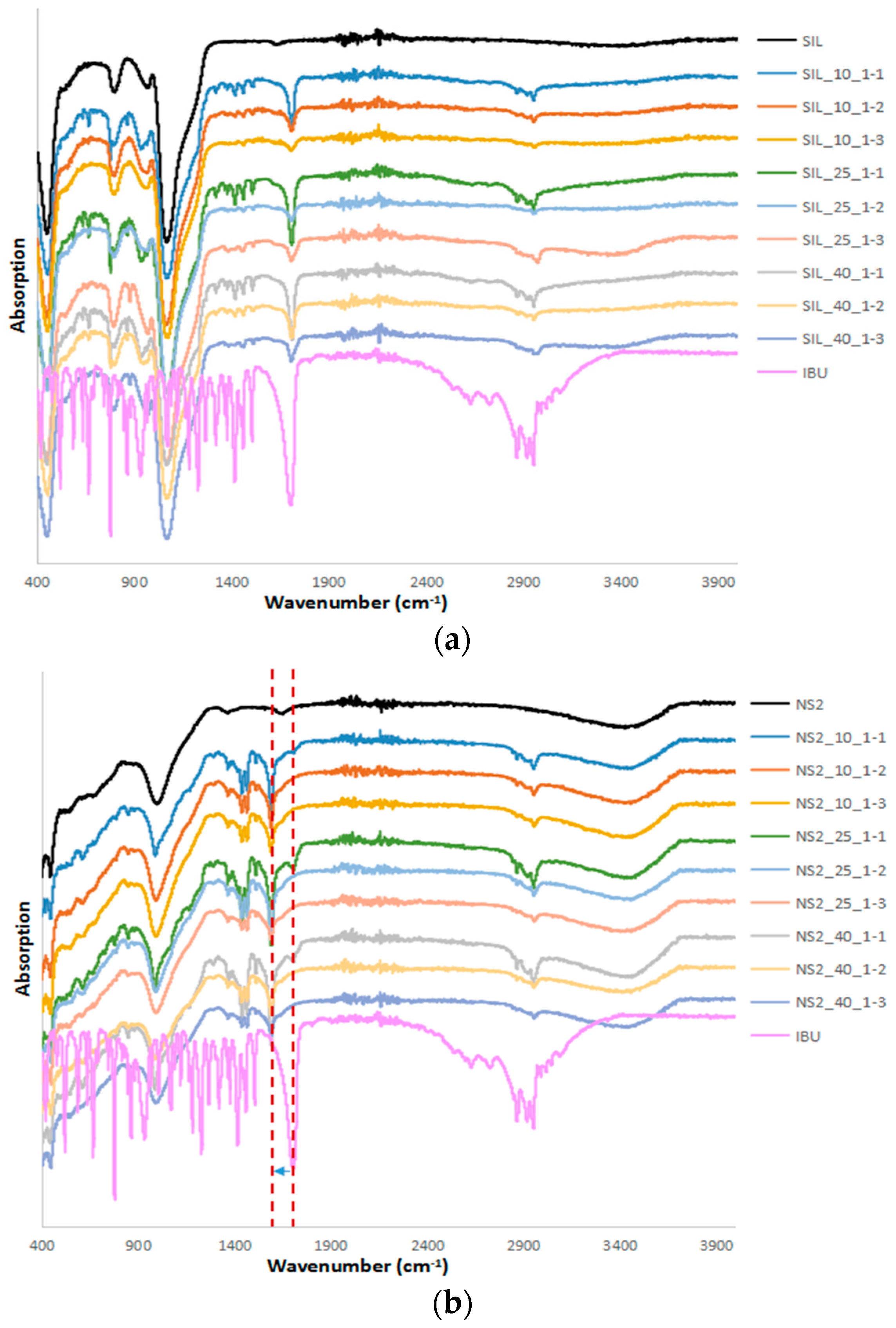
2.2. FTIR and Drug Desorption Recovery
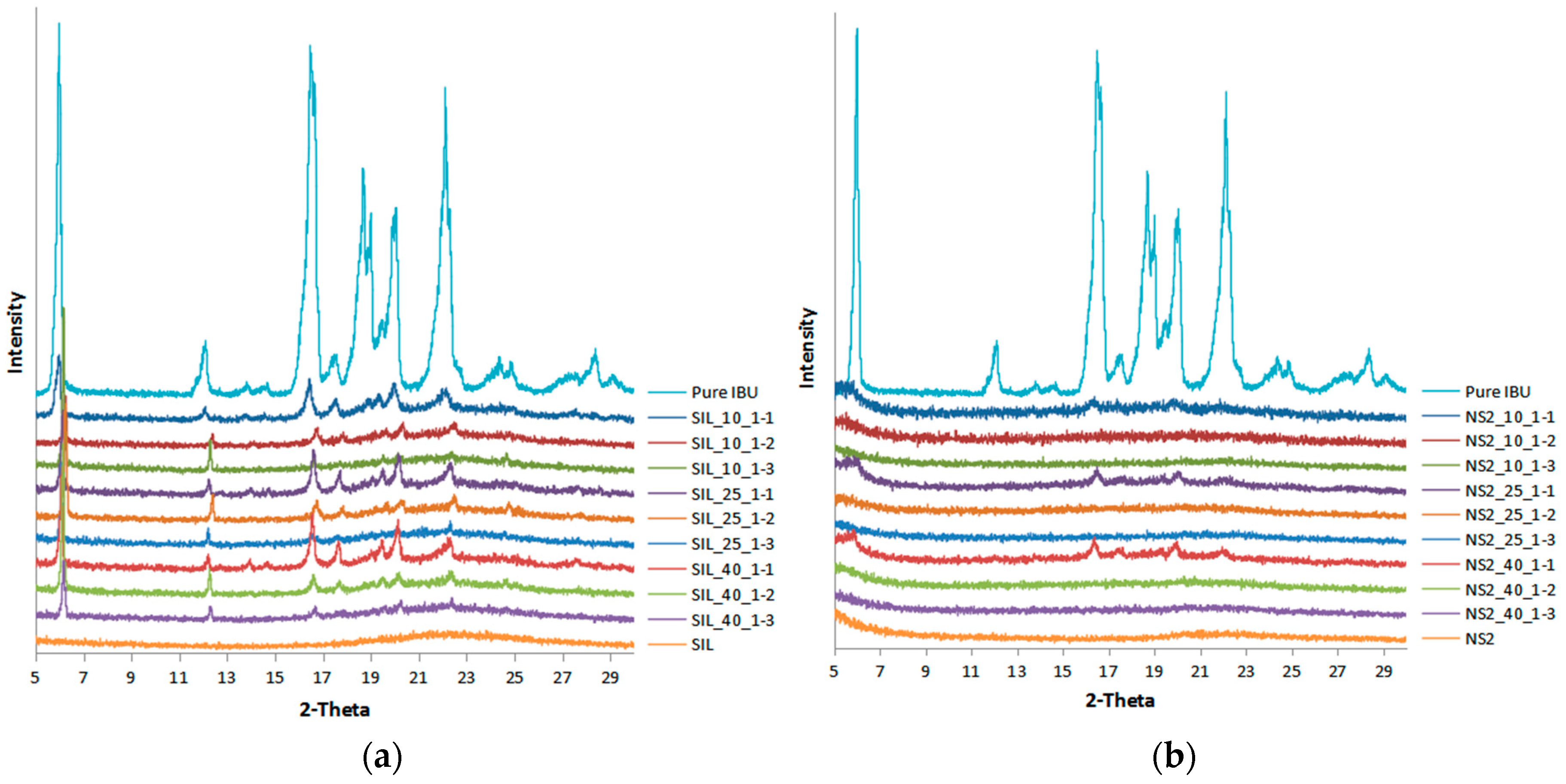
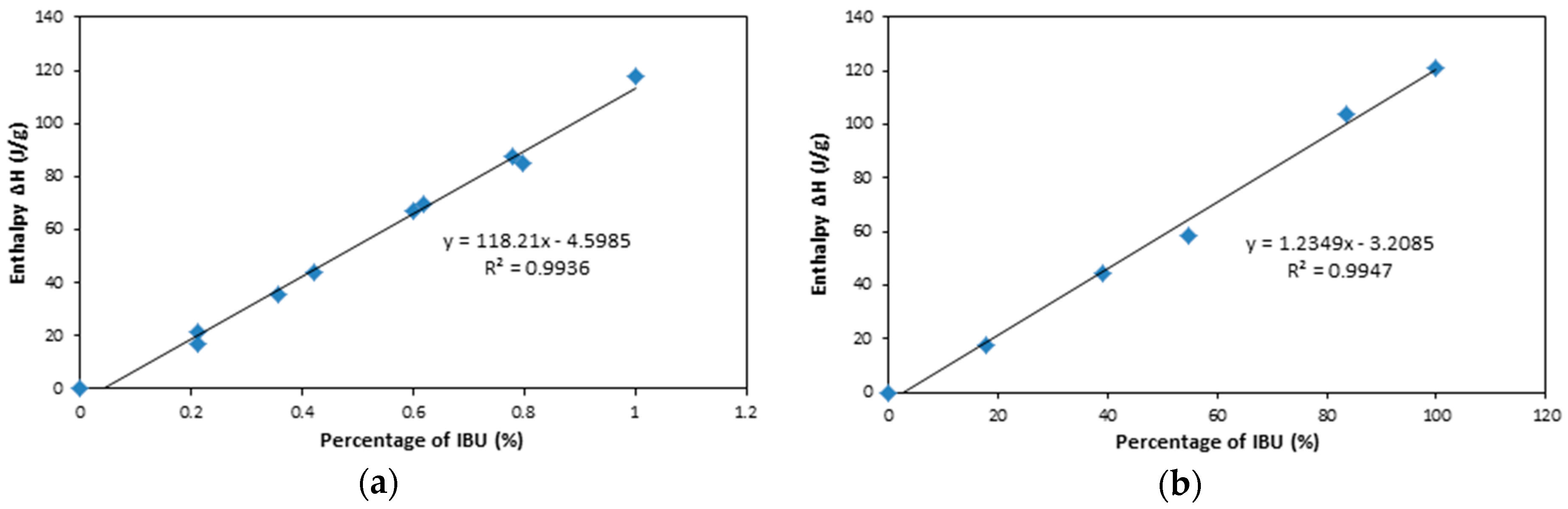
2.3. XRPD and DSC
2.4. Dissolution Efficiency
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Methods
4.2.1. Experimental Design and Drug Loading Procedure
4.2.2. Polarized Light Microscopy
4.2.3. Fourier Transform Infrared Spectrometer
4.2.4. Differential Scanning Calorimetry and Its Calibration Model to Determine the Percentage of the Crystalline IBU in the Drug-Loaded Samples
4.2.5. X-ray Powder Diffraction
4.2.6. Drug Desorption Recovery Rate Analysis
4.2.7. Dissolution Test
4.2.8. High Performance Liquid Chromatography Analysis
4.2.9. Dissolution Efficiency
4.2.10. Stability Test
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Singh, A.; Worku, Z.A.; Van den Mooter, G. Oral formulation strategies to improve solubility of poorly water-soluble drugs. Expert Opin. Drug Deliv. 2011, 8, 1361–1378. [Google Scholar] [CrossRef] [PubMed]
- Wang, S. Ordered mesoporous materials for drug delivery. Microporous Mesoporous Mater. 2009, 117, 1–9. [Google Scholar] [CrossRef]
- Xu, W.; Riikonen, J.; Lehto, V.-P. Mesoporous systems for poorly soluble drugs. Int. J. Pharm. 2013, 453, 181–197. [Google Scholar] [CrossRef] [PubMed]
- Salonen, J.; Laitinen, L.; Kaukonen, A.M.; Tuura, J.; Björkqvist, M.; Heikkilä, T.; Vähä-Heikkilä, K.; Hirvonen, J.; Lehto, V.P. Mesoporous silicon microparticles for oral drug delivery: Loading and release of five model drugs. J. Control. Release 2005, 108, 362–374. [Google Scholar] [CrossRef] [PubMed]
- Williams, H.D.; Van Speybroeck, M.; Augustijns, P.; Porter, C.J.H. Lipid-based formulations solidified via adsorption onto the mesoporous carrier Neusilin® US2: Effect of drug type and formulation composition on in vitro pharmaceutical performance. J. Pharm. Sci. 2014, 103, 1734–1746. [Google Scholar] [CrossRef] [PubMed]
- Limnell, T.; Riikonen, J.; Salonen, J.; Kaukonen, A.M.; Laitinen, L.; Hirvonen, J.; Lehto, V.P. Surface chemistry and pore size affect carrier properties of mesoporous silicon microparticles. Int. J. Pharm. 2007, 343, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Limnell, T.; Santos, H.A.; Mäkilä, E.; Heikkilä, T.; Salonen, J.; Murzin, D.Y.; Kumar, N.; Laaksonen, T.; Peltonen, L.; Hirvonen, J. Drug delivery formulations of ordered and nonordered mesoporous silica: Comparison of three drug loading methods. J. Pharm. Sci. 2011, 100, 3294–3306. [Google Scholar] [CrossRef] [PubMed]
- Kiekens, F.; Eelen, S.; Verheyden, L.; Daems, T.; Martens, J.; Den Mooter, G.V. Use of ordered mesoporous silica to enhance the oral bioavailability of ezetimibe in dogs. J. Pharm. Sci. 2012, 101, 1136–1144. [Google Scholar] [CrossRef] [PubMed]
- Kinnari, P.; Mäkilä, E.; Heikkilä, T.; Salonen, J.; Hirvonen, J.; Santos, H.A. Comparison of mesoporous silicon and non-ordered mesoporous silica materials as drug carriers for itraconazole. Int. J. Pharm. 2011, 414, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Van Speybroeck, M.; Mols, R.; Mellaerts, R.; Thi, T.D.; Martens, J.A.; Humbeeck, J.V.; Annaert, P.; Mooter, G.V.D.; Augustijns, P. Combined use of ordered mesoporous silica and precipitation inhibitors for improved oral absorption of the poorly soluble weak base itraconazole. Eur. J. Pharm. Biopharm. 2010, 75, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Qu, F.; Zhu, G.; Huang, S.; Li, S.; Sun, J.; Zhang, D.; Qiu, S. Controlled release of captopril by regulating the pore size and morphology of ordered mesoporous silica. Microporous Mesoporous Mater. 2006, 92, 1–9. [Google Scholar] [CrossRef]
- Cauda, V.; Mühlstein, L.; Onida, B.; Bein, T. Tuning drug uptake and release rates through different morphologies and pore diameters of confined mesoporous silica. Microporous Mesoporous Mater. 2009, 118, 435–442. [Google Scholar] [CrossRef]
- Kapoor, S.; Hegde, R.; Bhattacharyya, A.J. Influence of surface chemistry of mesoporous alumina with wide pore distribution on controlled drug release. J. Control. Release 2009, 140, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Goscianska, J.; Olejnik, A.; Nowak, I.; Marciniak, M.; Pietrzak, R. Ordered mesoporous silica modified with lanthanum for ibuprofen loading and release behaviour. Eur. J. Pharm. Biopharm. 2015, 94, 550–558. [Google Scholar] [CrossRef] [PubMed]
- Heikkilä, T.; Salonen, J.; Tuura, J.; Hamdy, M.S.; Mul, G.; Kumar, N.; Salmi, T.; Murzin, D.Y.; Laitinen, L.; Kaukonen, A.M.; et al. Mesoporous silica material TUD-1 as a drug delivery system. Int. J. Pharm. 2007, 331, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Forsgren, J.; Strømme, M. Stabilisation of amorphous ibuprofen in upsalite, a mesoporous magnesium carbonate, as an approach to increasing the aqueous solubility of poorly soluble drugs. Int. J. Pharm. 2014, 472, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Riikonen, J.; Mäkilä, E.; Salonen, J.; Lehto, V.-P. Determination of the physical state of drug molecules in mesoporous silicon with different surface chemistries. Langmuir 2009, 25, 6137–6142. [Google Scholar] [CrossRef] [PubMed]
- Mellaerts, R.; Jammaer, J.A.G.; Van Speybroeck, M.; Chen, H.; Humbeeck, J.V.; Augustijns, P.; Van den Mooter, G.; Martens, J.A. Physical state of poorly water soluble therapeutic molecules loaded into SBA-15 ordered mesoporous silica carriers: A case study with itraconazole and ibuprofen. Langmuir 2008, 24, 8651–8659. [Google Scholar] [CrossRef] [PubMed]
- W.R. Grace & Co. Company Literature on Syloid FP. Available online: www.discoverysciences.com (accessed on 6 Febuary 2017).
- Fuji Chemical Industries Co., Ltd. Company Literature on Neusilin. Available online: http://www.neusilin.com/product/ (accessed on 6 Febuary 2017).
- Álvarez, C.; Núñez, I.; Torrado, J.J.; Gordon, J.; Potthast, H.; García-Arieta, A. Investigation on the possibility of biowaivers for ibuprofen. J. Pharm. Sci. 2011, 100, 2343–2349. [Google Scholar] [CrossRef] [PubMed]
- British Pharmacopoeia Commission. British Pharmacopoeia; TSO: London, UK, 2010. [Google Scholar]
- Liu, L.; Gao, H. Molecular structure and vibrational spectra of ibuprofen using density function theory calculations. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2012, 89, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.I.; Dave, R.H. Evaluation of colloidal solid dispersions: Physiochemical considerations and in vitro release profile. AAPS PharmSciTech 2013, 14, 620–628. [Google Scholar] [CrossRef] [PubMed]
- Khanfar, M.; Fares, M.M.; Salem, M.t.S.; Qandil, A.M. Mesoporous silica based macromolecules for dissolution enhancement of irbesartan drug using pre-adjusted ph method. Microporous Mesoporous Mater. 2013, 173, 22–28. [Google Scholar] [CrossRef]
- Krupa, A.; Majda, D.; Jachowicz, R.; Mozgawa, W. Solid-state interaction of ibuprofen and Neusilin US2. Thermochim. Acta 2010, 509, 12–17. [Google Scholar] [CrossRef]
- Herzfeldt, C.D.; Kümmel, R. Dissociation constants, solubilities and dissolution rates of some selected nonsteroidal antiinflammatories. Drug Dev. Ind. Pharmacy 1983, 9, 767–793. [Google Scholar] [CrossRef]
- Lee, D.-J.; Lee, S.; Kim, I.W. Effects of humidity and surfaces on the melt crystallization of ibuprofen. Int. J. Mol. Sci. 2012, 13, 10296. [Google Scholar] [CrossRef] [PubMed]
- Hillerström, A.; Andersson, M.; Samuelsson, J.; van Stam, J. Solvent strategies for loading and release in mesoporous silica. Colloid Interface Sci. Commun. 2014, 3, 5–8. [Google Scholar] [CrossRef]
- Khan, K.A. The concept of dissolution efficiency. J. Pharmacy Pharmacol. 1975, 27, 48–49. [Google Scholar] [CrossRef]
- Anderson, N.H.; Bauer, M.; Boussac, N.; Khan-Malek, R.; Munden, P.; Sardaro, M. An evaluation of fit factors and dissolution efficiency for the comparison of in vitro dissolution profiles. J. Pharm. Biomed. Anal. 1998, 17, 811–822. [Google Scholar] [CrossRef]
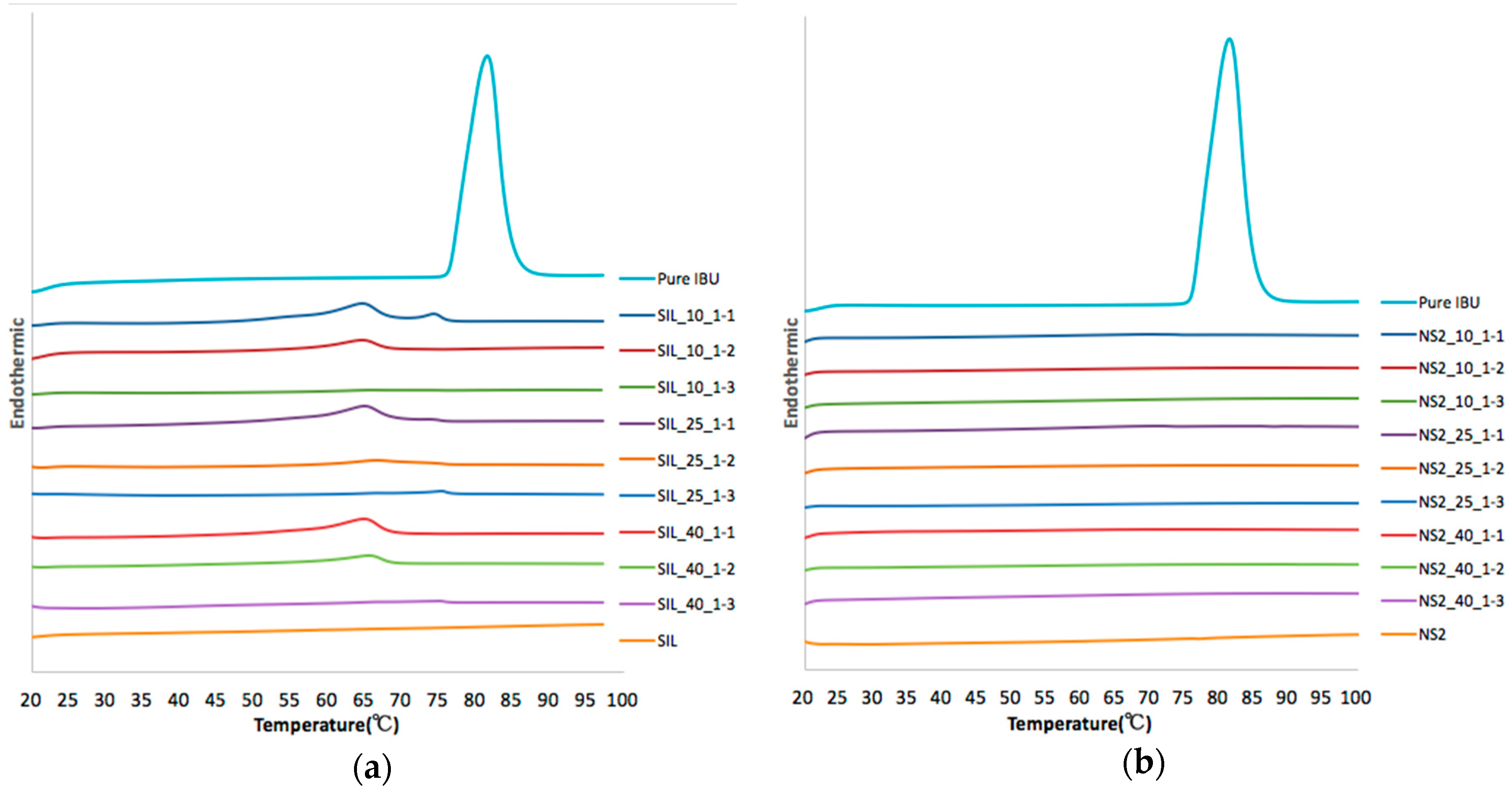

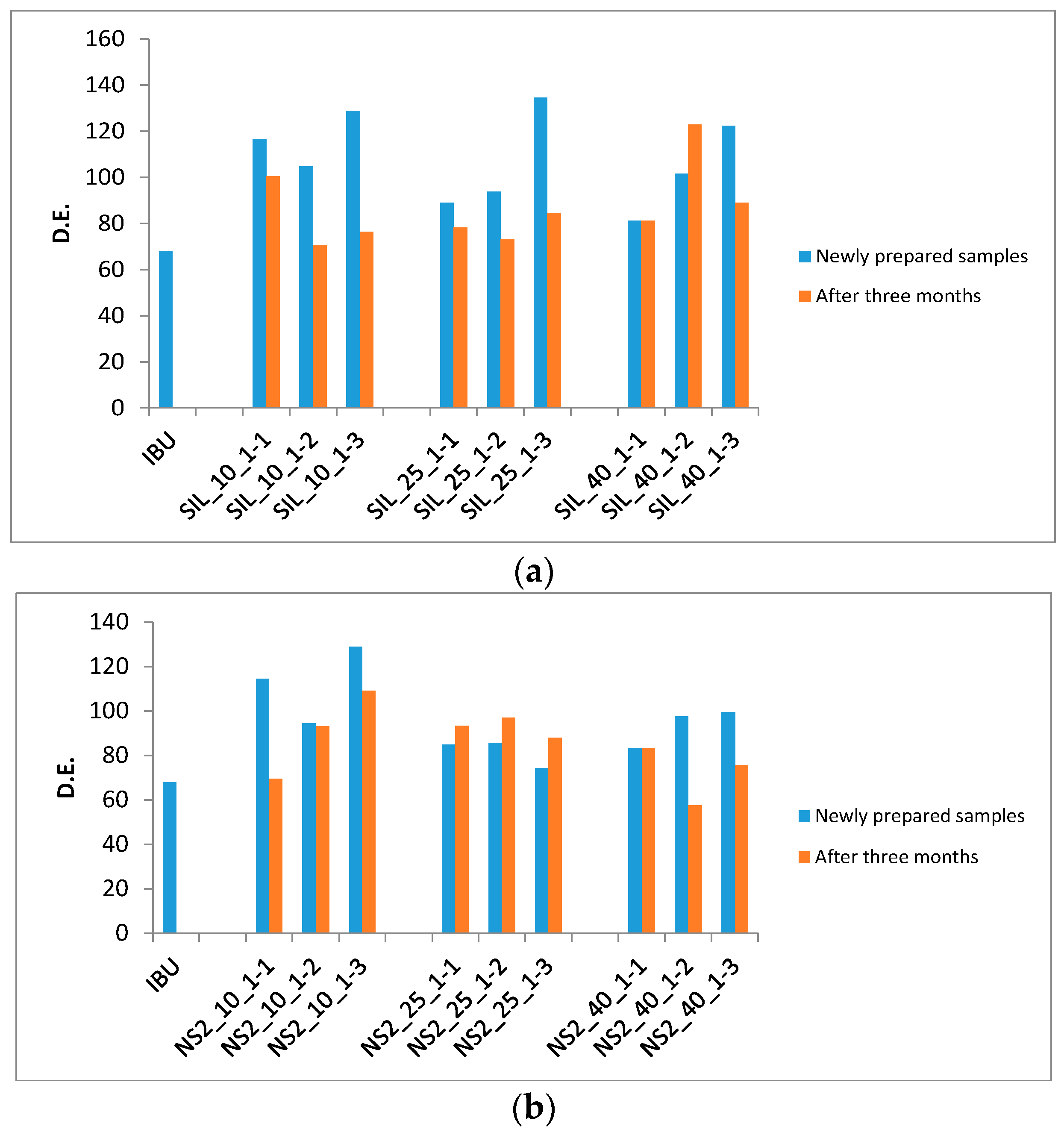
| Carrier | Number | Loading Factors | IBU Crystalline Percentage (%) | Drug Desorption Recovery (%) | Dissolution Efficiency (D.E. %) | |||
|---|---|---|---|---|---|---|---|---|
| IBU Concentration (mg/mL) | Drug to Carrier Ratio | Newly Prepared Samples | Samples after 3-Month Storage | Difference (%) | ||||
| S244FP | SIL_10_1-1 | 10 | 1:1 | 32.7 | 89.8 ± 3.3 | 116.5 | 100.3 | 16.1 |
| SIL_10_1-2 | 10 | 1:2 | 18.9 | 98.2 ± 1.4 | 104.6 | 70.4 | 34.2 | |
| SIL_10_1-3 | 10 | 1:3 | 9.1 | 78.8 ± 2.6 | 128.8 | 76.2 | 52.5 | |
| SIL_25_1-1 | 25 | 1:1 | 33.8 | 109.1 ± 1.1 | 88.9 | 78.1 | 10.7 | |
| SIL_25_1-2 | 25 | 1:2 | 14.1 | 88.2 ± 4.7 | 93.8 | 72.9 | 21.0 | |
| SIL_25_1-3 | 25 | 1:3 | 8.7 | 91.1 ± 1.2 | 134.6 | 84.4 | 50.2 | |
| SIL_40_1-1 | 40 | 1:1 | 30.5 | 107.5 ± 3.6 | 111.6 | 111.6 | 0.0 | |
| SIL_40_1-2 | 40 | 1:2 | 22.3 | 95.6 ± 2.2 | 101.4 | 122.9 | −21.5 | |
| SIL_40_1-3 | 40 | 1:3 | 6.8 | 100.1 ± 4.7 | 122.4 | 88.9 | 33.5 | |
| NS2 | NS2_10_1-1 | 10 | 1:1 | 6.4 | 53.7 ± 2.7 | 114.6 | 69.5 | 45.1 |
| NS2_10_1-2 | 10 | 1:2 | 0 | 46.4 ± 0.3 | 94.4 | 93.1 | 1.3 | |
| NS2_10_1-3 | 10 | 1:3 | 0 | 40.8 ± 1.3 | 128.9 | 109.2 | 19.7 | |
| NS2_25_1-1 | 25 | 1:1 | 4.1 | 52.2 ± 0.7 | 84.8 | 93.3 | −8.5 | |
| NS2_25_1-2 | 25 | 1:2 | 0 | 45.2 ± 0.7 | 85.6 | 97.0 | −11.4 | |
| NS2_25_1-3 | 25 | 1:3 | 0 | 48.4 ± 1.6 | 74.4 | 87.9 | −13.5 | |
| NS2_40_1-1 | 40 | 1:1 | 0 | 54.1 ± 2.2 | 83.3 | 83.3 | 0.0 | |
| NS2_40_1-2 | 40 | 1:2 | 0 | 53.5 ± 0.8 | 97.5 | 57.6 | 39.9 | |
| NS2_40_1-3 | 40 | 1:3 | 0 | 46.9 ± 0.5 | 99.5 | 75.7 | 23.8 | |
| Sample | Actual IBU Percentage (%) | Enthalpy ΔH (J/g) | Predicted IBU Percentage (%) | Prediction Error (%) |
|---|---|---|---|---|
| IBU and S244FP | 75.23 | 83.13 | 74.21 | 1.35 |
| 35.89 | 39.36 | 37.19 | 3.60 | |
| IBU and NS2 | 9.00 | 8.35 | 9.36 | 3.98 |
| 24.86 | 28.14 | 25.39 | 2.11 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lai, J.; Lin, W.; Scholes, P.; Li, M. Investigating the Effects of Loading Factors on the In Vitro Pharmaceutical Performance of Mesoporous Materials as Drug Carriers for Ibuprofen. Materials 2017, 10, 150. https://doi.org/10.3390/ma10020150
Lai J, Lin W, Scholes P, Li M. Investigating the Effects of Loading Factors on the In Vitro Pharmaceutical Performance of Mesoporous Materials as Drug Carriers for Ibuprofen. Materials. 2017; 10(2):150. https://doi.org/10.3390/ma10020150
Chicago/Turabian StyleLai, Junmin, Wu Lin, Peter Scholes, and Mingzhong Li. 2017. "Investigating the Effects of Loading Factors on the In Vitro Pharmaceutical Performance of Mesoporous Materials as Drug Carriers for Ibuprofen" Materials 10, no. 2: 150. https://doi.org/10.3390/ma10020150
APA StyleLai, J., Lin, W., Scholes, P., & Li, M. (2017). Investigating the Effects of Loading Factors on the In Vitro Pharmaceutical Performance of Mesoporous Materials as Drug Carriers for Ibuprofen. Materials, 10(2), 150. https://doi.org/10.3390/ma10020150