Biohydrogen Fermentation from Sucrose and Piggery Waste with High Levels of Bicarbonate Alkalinity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Seed Sludge
2.2. Experimental Procedure
| Run | Bicarbonate Alkalinity Control | Initial Bicarbonate Alkalinity (mg CaCO3/L) | |||||
|---|---|---|---|---|---|---|---|
| pH 5 | pH 6 | pH 7 | pH 8 | pH 9 | pH 10 | ||
| I | No | 0 | 13 | 79 | 214 | 379 | 663 |
| II | Yes | 0 | 21 | 113 | 221 | 506 | 1,023 |
| III | Yes | 0 | 450 | 2,525 | 3,038 | 3,175 | 4,663 |
| IV | Yes | 0 | 978 | 3,129 | 4,201 | 4,488 | 6,551 |
| V | Yes | 0 | 575 | 5,050 | 6,113 | 6,363 | 13,100 |
2.3. Analysis
3. Results and Discussion
3.1. Hydrogen Yield under Various pHs Condition
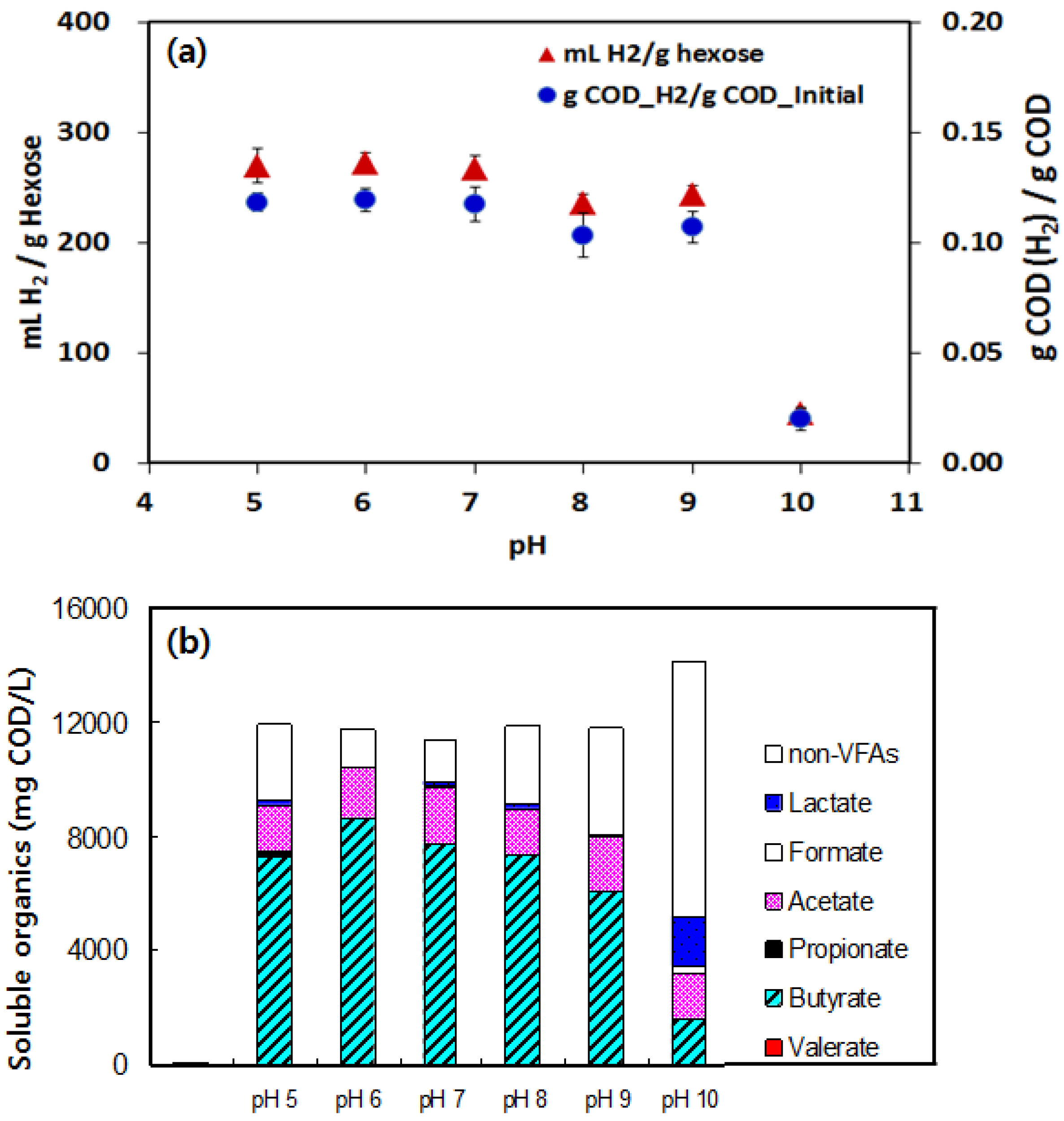
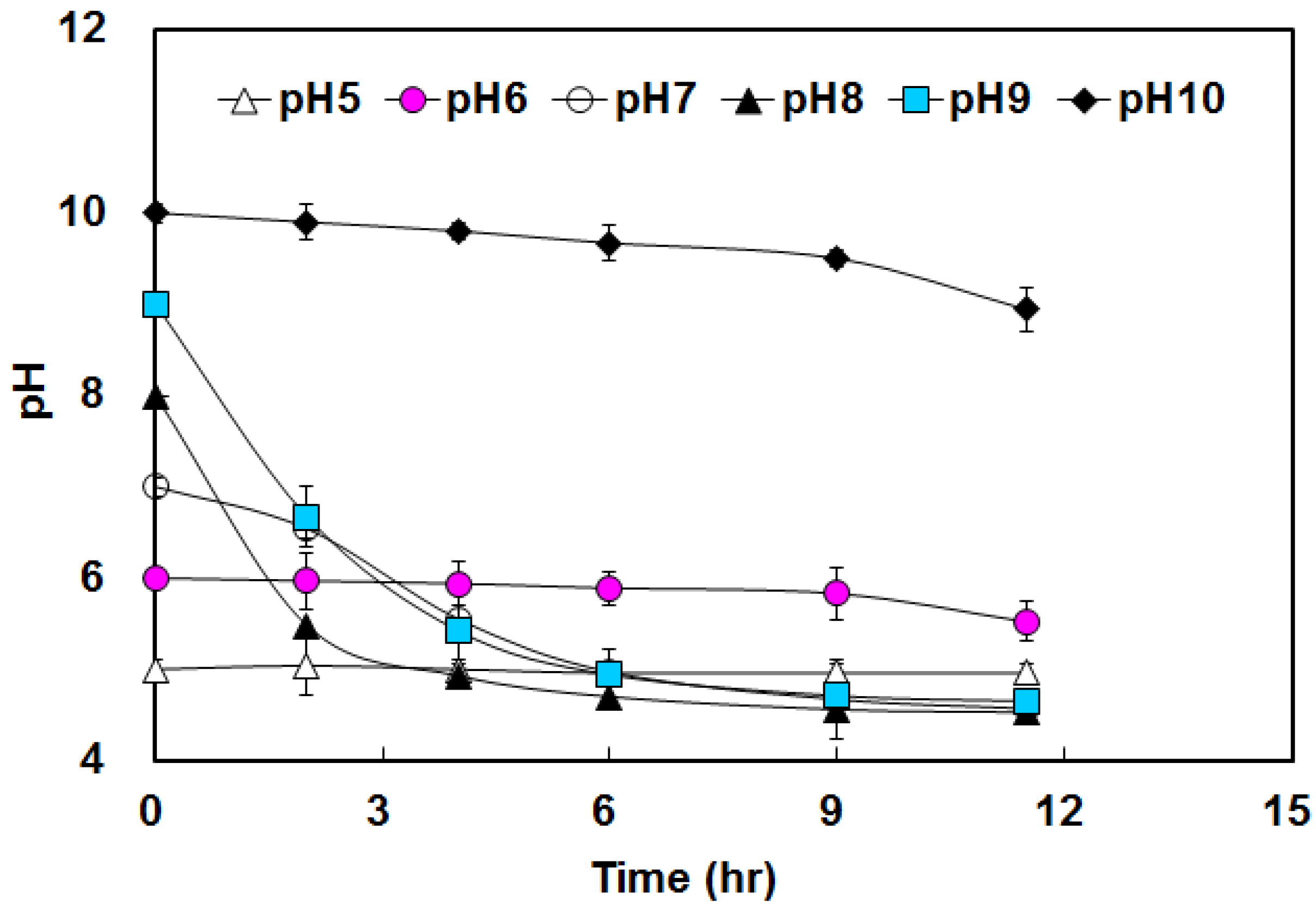
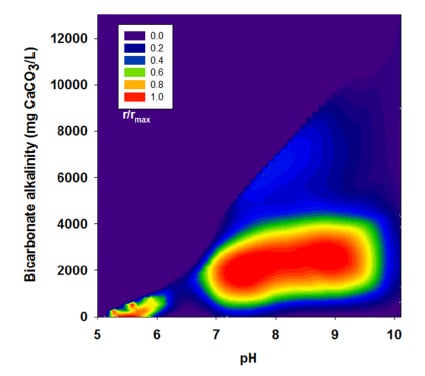
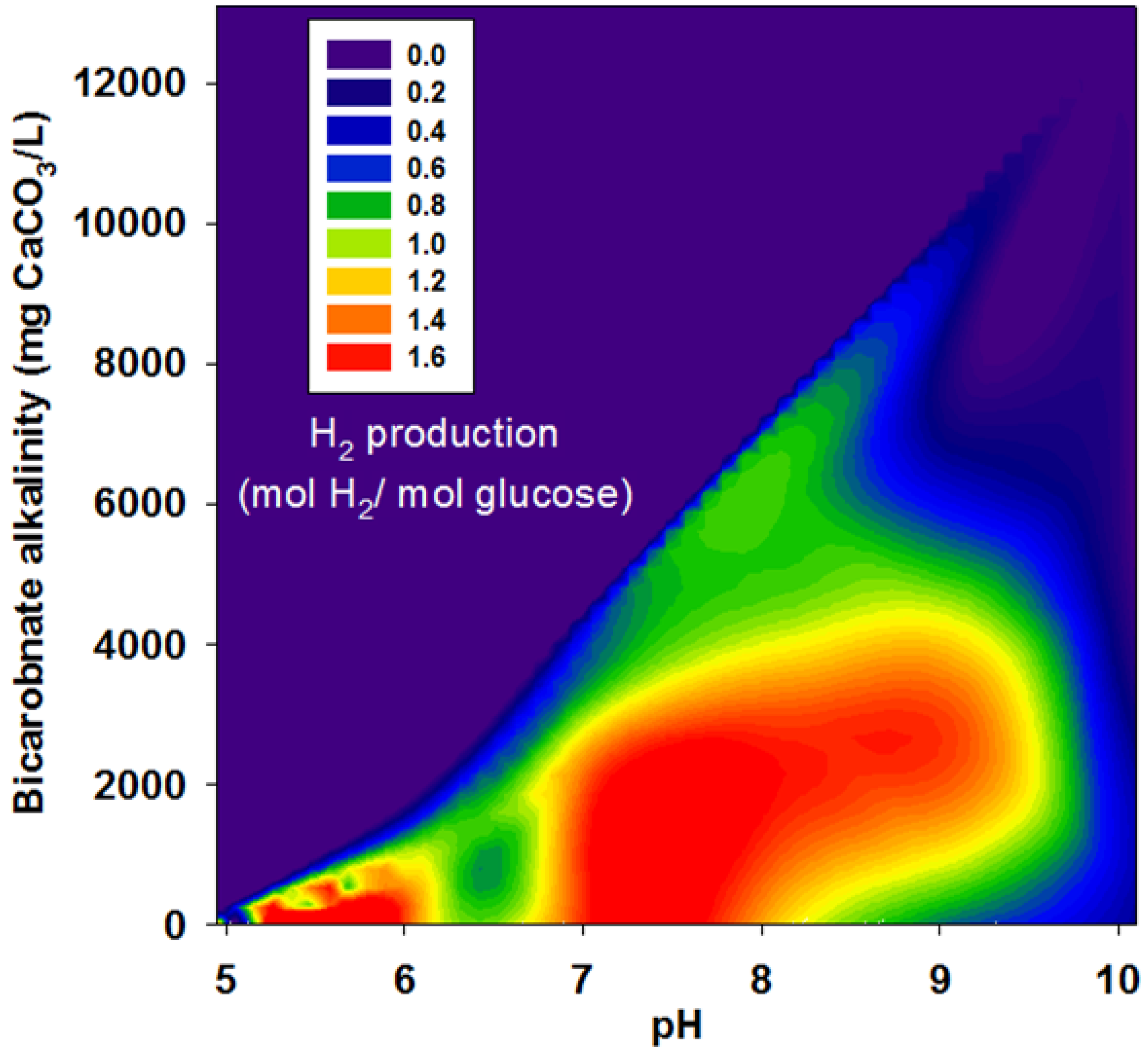
3.2. Effects of BA
| Run | pH | Bicarbonate Alkalinity | H2 Yield | R | r/rmax |
|---|---|---|---|---|---|
| (mg CaCO3/L) | (mol H2/mol glucose) | (mL H2/g VSS-d) | |||
| II | 4.95 | 0 | 1.38 | 63.9 | 0.2 |
| 6.00 | 21 | 1.51 | 71.8 | 0.23 | |
| 7.04 | 113 | 1.60 | 62.9 | 0.20 | |
| 8.05 | 221 | 1.28 | 49.3 | 0.15 | |
| 9.07 | 379 | 0.82 | 49.1 | 0.15 | |
| 9.98 | 663 | 0.43 | 18.1 | 0.06 | |
| III | 5.00 | 0 | 1.15 | 139.1 | 0.44 |
| 6.10 | 450 | 1.23 | 154.9 | 0.49 | |
| 7.10 | 2,525 | 1.36 | 258.3 | 0.81 | |
| 8.10 | 3,038 | 1.46 | 296.4 | 0.93 | |
| 8.95 | 3,175 | 1.48 | 318.5 | 1.00 | |
| 10.10 | 4,663 | 0.01 | 15.6 | 0.05 | |
| IV | 5.02 | 0 | 0.52 | 56.6 | 0.18 |
| 6.17 | 978 | 1.01 | 109.6 | 0.34 | |
| 7.13 | 3,129 | 1.00 | 143.9 | 0.45 | |
| 8.12 | 4,201 | 1.00 | 133.8 | 0.42 | |
| 9.01 | 4,488 | 1.02 | 121.5 | 0.38 | |
| 9.96 | 6,551 | 0.12 | 11.0 | 0.03 | |
| V | 5.14 | 0 | 0.52 | 40.3 | 0.13 |
| 6.27 | 575 | 0.88 | 65.4 | 0.21 | |
| 7.36 | 5,050 | 0.76 | 81.9 | 0.26 | |
| 8.10 | 6,113 | 0.85 | 101.6 | 0.32 | |
| 8.99 | 6,363 | 0.40 | 61.8 | 0.19 | |
| 10.10 | 13,100 | 0.00 | 0.00 | 0.00 |

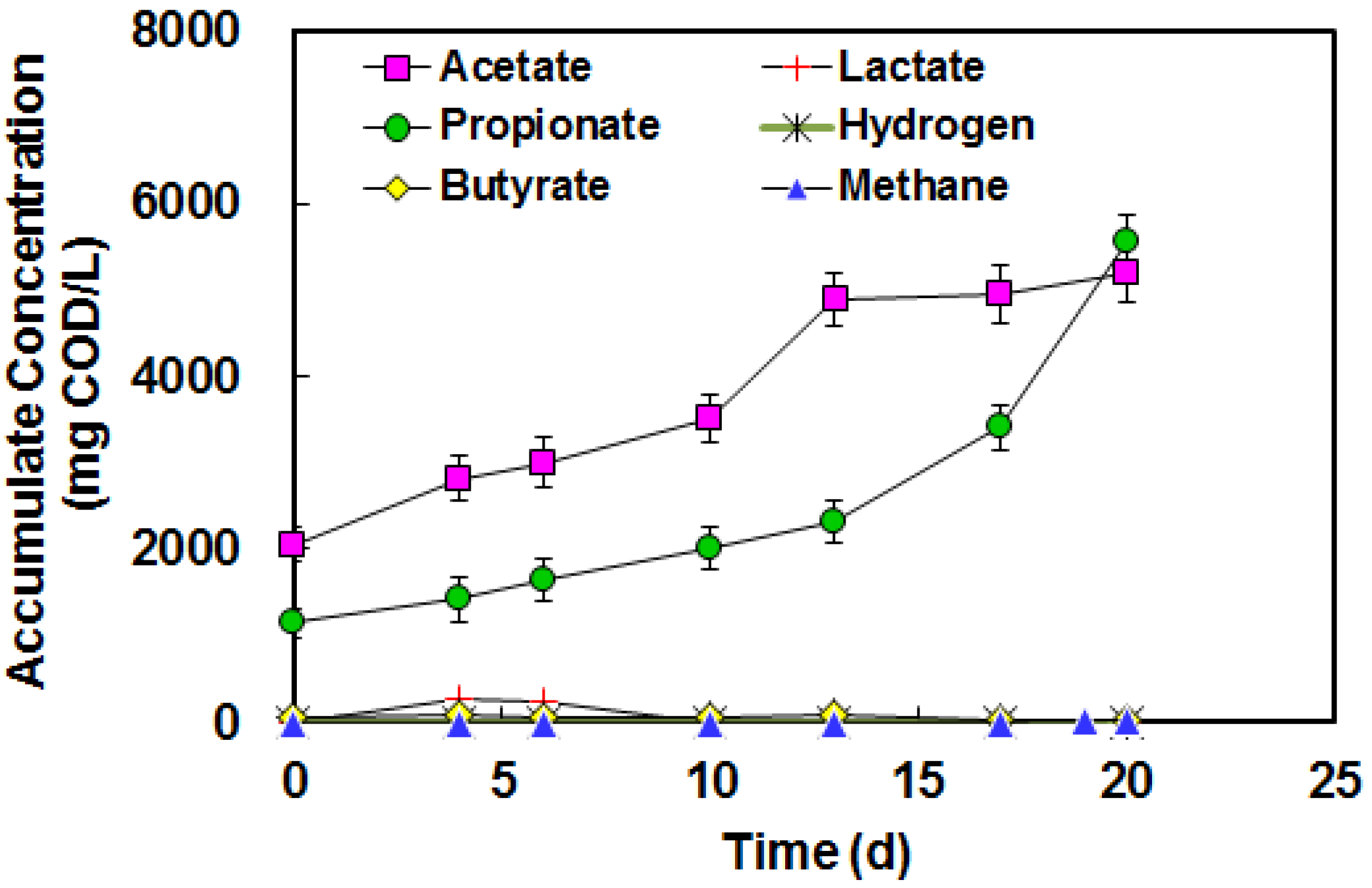
3.3. Acetate and Butyrate as the Final Byproducts
3.4. Lactate and Propionate as Final Byproducts

3.5. BA in High Strength Piggery Waste
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- A Multiyear Plan for the Hydrogen R & D Program, Rationale, Structure and Technology Roadmaps; U.S. Department of Energy, Energy Efficiency and Renewable Energy, Office of Power Delivery, Office of Power Technologies: Washington, DC, USA, 1999.
- Hawkes, F.R.; Dinsdale, R.; Hawkes, D.L.; Hussy, I. Sustainable fermentative hydrogen production: Challenges for process optimization. Int. J. Hydrog. Energy 2002, 27, 1339–1347. [Google Scholar] [CrossRef]
- Thauer, R.K.; Jungermann, K.; Decker, K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol. Rev. 1977, 41, 100–180. [Google Scholar] [PubMed]
- Van Ginkel, S.; Sung, S.; Lay, J.J. Biohydrogen production as a function of pH and substrate concentration. Environ. Sci. Technol 2001, 38, 4726–4730. [Google Scholar]
- Li, C.; Fang, H.H.P. Fermentative hydrogen production from wastewater and solid wastes by mixed cultures. Crit. Rev. Environ. Sci. Technol. 2007, 37, 1–39. [Google Scholar] [CrossRef]
- Khanal, S.K.; Chen, W.H.; Li, L.; Sung, S. Biological hydrogen production: Effect of pH and intermediate products. Int. J. Hydrog. Energy 2004, 29, 1123–1131. [Google Scholar]
- Lee, Y.; Miyahara, T.; Noike, T. Effect of pH on microbial hydrogen fermentation. J. Chem. Technol. Biotechnol. 2002, 77, 694–698. [Google Scholar] [CrossRef]
- Cai, M.; Liu, J.; Wei, Y. Enhanced biohydrogen production from sewage with alkaline pretreatment. Environ. Sci. Technol. 2004, 38, 3195–3202. [Google Scholar] [CrossRef] [PubMed]
- Ahn, Y.H.; Bae, J.; Park, S.; Min, K. Anaerobic digestion elutriated phased treatment of piggery waste. Water Sci. Technol. 2004, 49, 181–189. [Google Scholar] [PubMed]
- Ahn, Y.H.; Speece, R.E. Elutriated acid fermentation of municipal primary sludge. Water Res. 2006, 40, 2210–2220. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, P.; Ibrahim, S.; Mohamad Annuar, M.S. Effects of biomass, COD and bicarbonate concentrations on fermentative hydrogen production from POME by granulated sludge in a batch culture. Int. J. Hydrog. Energy 2012, 37, 17801–17808. [Google Scholar] [CrossRef]
- Valdez-Vazquez, I.; Poggi-Varaldo, H.M. Alkalinity and high total solids affecting H2 production from organic solid waste by anaerobic consortia. Int. J. Hydrog. Energy 2009, 34, 3639–3646. [Google Scholar] [CrossRef]
- Choi, J.; Ahn, Y. Characteristics of biohydrogen fermentation from various substrates. Int. J. Hydrog. Energy 2014, 39, 3152–3159. [Google Scholar] [CrossRef]
- Owen, W.F.; Stuckey, D.D.; Healy, J.B., Jr.; Young, L.Y.; McCarty, P.L. Bioassay for monitoring biochemical methane potential and anaerobic toxicity. Water Res. 1979, 13, 485–493. [Google Scholar] [CrossRef]
- Speece, R.E. Anaerobic Biotechnology for Industrial Wastewaters; Archae Press: Nashville, TN, USA, 1996. [Google Scholar]
- Standard Methods for the Examination of Water and Wastewater, 20th ed.; the American Public Health Association, the American Water Works Association and the Water Environment Federation: Washington, DC, USA, 1998.
- Buchauer, K. A comparison of two simple titration procedures to determine volatile fatty acids in influents to wastewater and sludge treatment processes. Water SA 1998, 24, 49–56. [Google Scholar]
- Lay, J.J.; Lee, Y.J.; Noike, T. Feasibility of biological hydrogen production from organic fraction of municipal solids waste. Water Res. 1999, 33, 2576–2586. [Google Scholar] [CrossRef]
- Zwietering, M.H.; Jongenburger, I.; Rombouts, F.M.; Van’s Riet, K. Modeling of the bacterial growth curve. Appl. Environ. Microbiol. 1990, 56, 1875–1881. [Google Scholar] [PubMed]
- Zhang, J.; Wang, Q. Buffering and nutrient effects of white mud from ammonia-soda process on thermophilic hydrogen fermentation from food waste. Int. J. Hydrog. Energy 2013, 38, 13564–13571. [Google Scholar] [CrossRef]
- Lin, C.Y.; Lay, C.H. Effects of carbonate and phosphate concentrations on hydrogen production using anaerobic sewage sludge microflora. Int. J. Hydrog. Energy 2004, 29, 275–281. [Google Scholar] [CrossRef]
- Chen, C.C.; Lin, C.Y.; Lin, M.C. Acid-base enrichment enhances on anaerobic hydrogen production process. Appl. Microbiol. Biotechnol. 2002, 57, 224–228. [Google Scholar]
- Hussy, I.; Hawkes, F.R.; Dinsdale, R.; Hawkes, D.L. Continuous fermentative hydrogen production from a wheat starch co-product by mixed microflora. Biotechnol. Bioeng. 2003, 84, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Parker, W.; Basnar, R.; Proracki, A.; Falletta, P.; Beland, M.; Seto, P. Biohydrogen production by anaerobic co-digestion of municipal food waste and sewage sludges. Int. J. Hydrog. Energy 2008, 33, 3561–3659. [Google Scholar] [CrossRef]
- Jones, D.T.; Woods, D.R. Solvent production. In Biotechnology Handbooks, Vol.3, Clostridia; Plenum Press: New York, NY, USA, 1989. [Google Scholar]
- Kaneuchi, C.; Benno, Y.; Mitsuoka, T. Clostridium coccoids, a new species from feces of mice. Int. J. Syst. Bacteriol. 1976, 26, 482–486. [Google Scholar] [CrossRef]
- Stadtman, E.R.; Stadtman, T.C.; Pastan, I.; Smith, L.D.S. Clostridium barkeri sp. n. J. Bacteriol. 1972, 110, 758–760. [Google Scholar] [PubMed]
- Min, B.; Kim, J.R.; Oh, S.E.; Regan, J.M.; Logan, B.E. Electricity generation from swine wastewater using microbial fuel cells. Water Res. 2005, 39, 4961–4968. [Google Scholar] [CrossRef] [PubMed]
- Kotsopoulos, T.; Fotidis, I.A.; Tsolakis, N.; Martzopoulos, G.G. Biohydrogen production from pig slurry in a CSTR reactor system with mixed cultures under hyper-thermophilic temperature (70 °C). Biomass Bioenergy 2009, 33, 1168–1174. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, Y.; Zhang, D.; Zhu, X. Waste activated sludge fermentation for hydrogen production enhanced by anaerobic process improvement and acetobacteria inhibition: The role of fermentation pH. Environ. Sci. Technol. 2010, 44, 3317–3323. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, J.; Ahn, Y. Biohydrogen Fermentation from Sucrose and Piggery Waste with High Levels of Bicarbonate Alkalinity. Energies 2015, 8, 1716-1729. https://doi.org/10.3390/en8031716
Choi J, Ahn Y. Biohydrogen Fermentation from Sucrose and Piggery Waste with High Levels of Bicarbonate Alkalinity. Energies. 2015; 8(3):1716-1729. https://doi.org/10.3390/en8031716
Chicago/Turabian StyleChoi, Jeongdong, and Youngho Ahn. 2015. "Biohydrogen Fermentation from Sucrose and Piggery Waste with High Levels of Bicarbonate Alkalinity" Energies 8, no. 3: 1716-1729. https://doi.org/10.3390/en8031716
APA StyleChoi, J., & Ahn, Y. (2015). Biohydrogen Fermentation from Sucrose and Piggery Waste with High Levels of Bicarbonate Alkalinity. Energies, 8(3), 1716-1729. https://doi.org/10.3390/en8031716