3.1. Characteristics of Thin Stillage and Rhizopus sp. Cultivation in Pentose Sugars
The thin stillage from corn-based ethanol production has previously been shown to be a good growth medium for production of nutritionally rich biomass using filamentous fungi [
5]. However, research performed on thin stillage derived from ethanol production using other cereals such as wheat is scarce in literature. In this study wheat-based thin stillage from a Swedish ethanol facility was mostly used and some of its characteristics are presented in
Table 1. Altogether, organic acids, glycerol, ethanol and pentose sugars represented around 13% of the
ca. 92 g·L
−1 of total solids. The thin stillage also contained around 5 g·L
−1 of nitrogen reinforcing its high potential to be used as the sole cultivation medium. The remaining fraction of the thin stillage most likely consists of fiber, oil, other cereal-unfermented components, salts and dead yeast cells, which are also potential nutrient sources.
Reasonably, microorganisms able to consume pentose sugars would be preferable for cultivation in thin stillage. Contrary to
Saccharomyces cerevisiae,
Zygomycetes fungi are well known for their capability to consume pentose sugars. However, their ability to assimilate xylose is far better investigated than for arabinose [
8].
Rhizopus sp. was first examined for assimilation of arabinose and xylose in semi-synthetic medium. It consumed 83% ± 1% of the initial arabinose after 12 days of cultivation. The fastest consumption rate of (2.0 ± 0.1) g·L
−1·d
−1 was reached after six days. The produced biomass was (4.1 ± 1.0) g·L
−1, corresponding to a yield of (285 ± 78) mg·biomass·g
−1 consumed arabinose. No metabolites were produced upon arabinose consumption. The
Rhizopus sp. consumed all xylose within 60 h of cultivation. The highest xylose consumption rate of about (570 ± 90) mg·L
−1·h
−1 was reached after 36 h. The produced biomass was 5.1 ± 0.0 g·L
−1, corresponding to a yield of (306 ± 2) mg·biomass·g
−1 consumed xylose. Other than biomass,
Rhizopus sp. produced ethanol, glycerol, lactic acid and xylitol, with yields of (128 ± 12) mg·g
−1, (88 ± 1) mg·g
−1, (74 ± 11) mg·g
−1 and (47 ± 15) mg·g
−1 consumed xylose, respectively. The production of the intermediate xylitol indicates that xylose conversion occurs through the general fungal pathway [
24]. The
Rhizopus strain used in this study is evidently a potential candidate to be used as a catalyst in fermentations of pentose-containing substrates such as thin stillage.
Table 1.
Characterisation of the industrial thin stillage derived from wheat-based ethanol production process used in the present work.
Table 1.
Characterisation of the industrial thin stillage derived from wheat-based ethanol production process used in the present work.
| Parameter | Value | Parameter | Value |
|---|
| pH | 3.5 | Arabinose (g·L−1) | 1.5 ± 0.1 |
| Total nitrogen (g·L−1) | 5.0 ± 0.4 | Glycerol (g·L−1) | 7.0 ± 0.1 |
| Soluble total nitrogen (g·L−1) | 2.1 ± 0.4 | Lactic acid (g·L−1) | 1.8 ± 0.1 |
| Total solids (%, w/v) | 9.2 ± 0.9 | Acetic acid (g·L−1) | 0.21 ± 0.01 |
| Suspended solids (%, w/v) | 3.8 ± 0.3 | Ethanol (g·L−1) | 1.2 ± 0.2 |
| Xylose (g·L−1) | 0.8 ± 0.1 | | |
3.2. Cultivation in Thin Stillage with a Rhizopus sp.
The growth performance of filamentous fungi has been studied in corn-based thin stillage. Mitra
et al. [
6] and Liang
et al. [
15] have reported growth of
Mucor circinelloides and
Pythium irregulare in 6% total solids thin stillage, respectively. The latter was further shown to perform better in 50% diluted thin stillage. The need for the dilution of thin stillage containing 8% or more total solids has been reported when using
Rhizopus oligosporus [
5]. In this work,
Rhizopus sp. was successfully grown in mostly wheat-based thin stillage containing
ca. 9% total solids, and its growth profiles within the range 25–45 °C are depicted in
Figure 2. Similar produced biomass values (around 6.5 g·L
−1) were achieved at the beginning of cultivation within the range 30–40 °C; a lower value was achieved at 45 °C (
ca. 4 g·L
−1); and a longer lag phase was observed at 25 °C. The highest maximum produced biomass value (
ca. 15 g·L
−1) was obtained at 30 °C, while the lowest one was obtained at 45 °C (<10 g·L
−1) (Table 2). Significant statistical differences were found on biomass production within examined temperatures (
p = 0.000); 45 °C was found to be statistically different from the other tested temperatures. Maximum biomass productivities of (366 ± 26) mg·L
−1·h
−1, (364 ± 52) mg·L
−1·h
−1, (358 ± 19) mg·L
−1·h
−1 and (234 ± 40) mg·L
−1·h
−1 were obtained after 18 h of cultivation at 30 °C, 35 °C, 40 °C and 45 °C, respectively; a maximum of (282 ± 2) mg·L
−1·h
−1 was obtained after 24 h cultivation at 25 °C. The
Rhizopus sp. also exhibited different growth morphologies during cultivation at different temperatures. It grew as small mycelial clumps at 30 °C, 35 °C and 40 °C, while it grew as small mycelial pellets at 25 °C and 45 °C. The capacity of
Rhizopus sp. to grow well up to 40° C is very relevant, since this could potentially lead to energy savings during cooling of the thin stillage after distillation.
When evaluating the potential to use fungal biomass as a nutrient source for feed applications, a few compositional aspects are of special interest such as protein contents. The final crude protein of the
Rhizopus sp. biomass was found to be within the range 49%–55% of biomass dry weight (Table 2), which was found to be similar to that at the beginning of cultivation (52%–54%).
Zygomycetes fungi are also well known for the presence of chitosan in their cell walls. This polymer has been gathering increasing interest due to its wide applications in e.g., biomedical and environmental fields [
25]. Its acetylated form chitin can also be found in the
Zygomycetes cell walls. Chitosan and chitin are polymers of glucosamine (GlcN) and
N-acetylglucosamine (GlcNAc) in which GlcN is dominant (60%–100%) in chitosan, while GlcNAc is dominant (60%–100%) in chitin [
26]. The cell wall fraction was measured as alkali-insoluble material (AIM), which presented an increasing trend during cultivation at all tested temperatures. Overall, the cell wall fraction of
Rhizopus sp. biomass was in the range 10%–15% of biomass dry weight. Maximum concentration of (2.2 ± 0.4) g·L
−1 and (1.5 ± 0.0) g·L
−1 of the AIM were obtained at the end of cultivation at both 30 °C and 40 °C and (1.6 ± 0.1) g·L
−1 after 72 h when cultivating
Rhizopus sp. at 35 °C. Both glucosamine and
N-acetylglucosamine contents were found to increase by 45%–50% of the AIM dry weight during cultivation in the thin stillage. Final glucosamine and
N-acetylglucosamine contents were not found to be statistically different at the temperatures examined (
p = 0.953 and
p = 0.199, respectively). These monomers were present at about the same fractions (10%–25% each), making up to 40%–50% of the
Rhizopus sp. cell wall (Table 2). Together, glucosamine and
N-acetylglucosamine made up to 6% of the
Rhizopus sp. biomass; up to (960 ± 77) mg·L
−1, (657 ± 127) mg·L
−1 and (778 ± 92) mg·L
−1 of glucosamine and
N-acetylglucosamine together could be produced during cultivation at 30 °C, 35 °C and 40 °C, respectively. Similar glucosamine trends were found by Ferreira
et al. [
23] when cultivating another
Rhizopus strain in spent sulphite liquor. Higher production of glucosamine was observed either when changing from semi-synthetic medium to spent sulphite liquor or during cultivation under more unfavourable temperatures.
N-acetylglucosamine content was found to increase at all tested conditions.
Figure 2.
Rhizopus sp. biomass concentration during cultivation in thin stillage at different temperatures.
Figure 2.
Rhizopus sp. biomass concentration during cultivation in thin stillage at different temperatures.
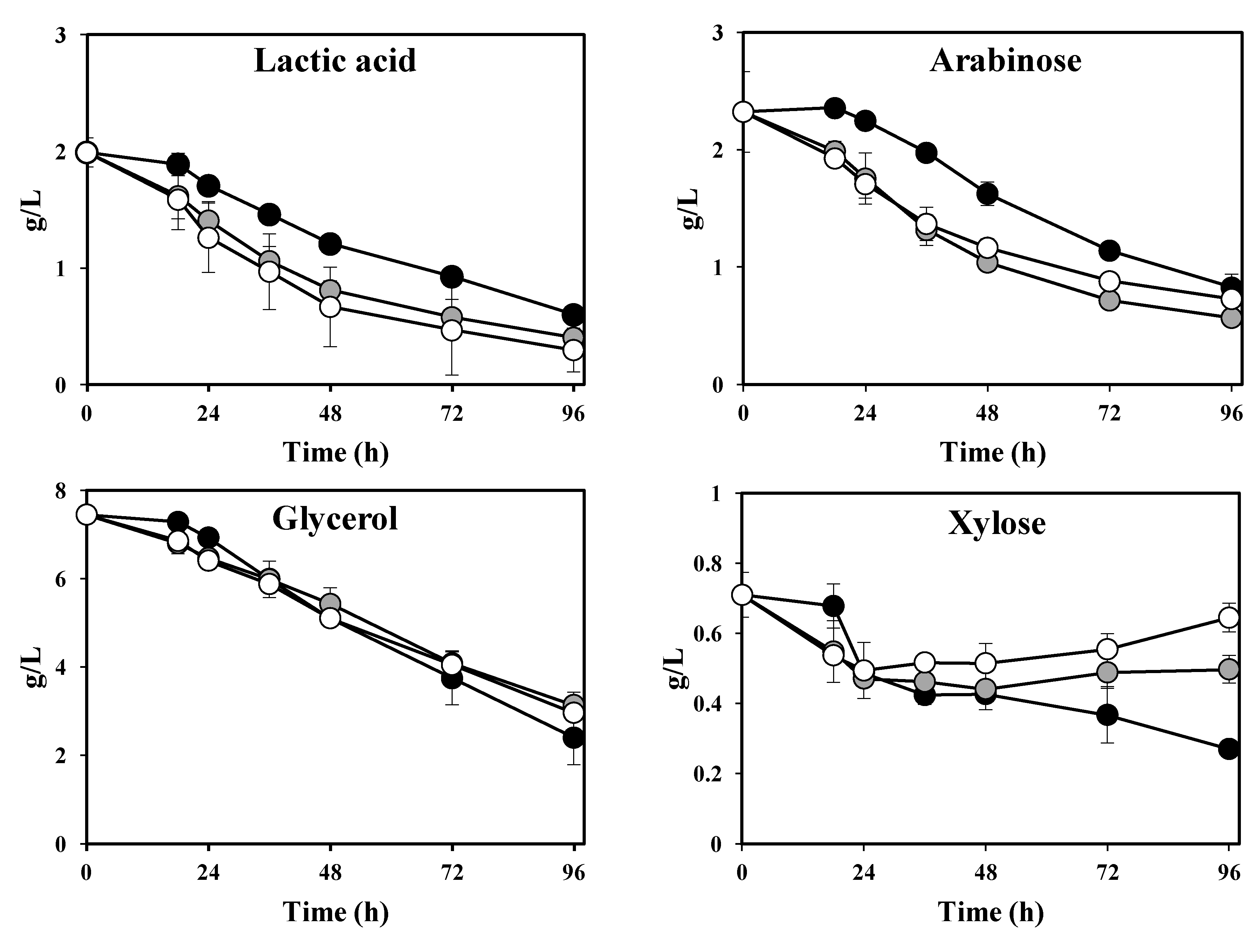
In addition to the production of high-quality fungal biomass, a post-cultivation thin stillage with a lower level of organic load is of interest. The
Rhizopus sp. was able to consume 70%–85% of the initial lactic acid and 58%–68% of the initial glycerol within the applied temperature ranges (
Figure 3; Table 2). Final lactic acid and glycerol reduction values were not found to be statistically different between temperatures (
p = 0.065 and
p = 0.068, respectively). Acetic acid was completely consumed after 18 h of cultivation. In addition, a net output of 1 g·L
−1 of ethanol was produced during cultivation at all tested temperatures (
Figure 3; Table 2). The concentration of the main sugars in the thin stillage (arabinose and xylose) showed different patterns during cultivation (
Figure 3). Arabinose decreased continuously during cultivation; reduction maxima of 64% ± 0%, 76% ± 0% and 69% ± 0% were achieved during cultivation at 30 °C, 35 °C and 40 °C, respectively. On the other hand, xylose presented a decreasing trend at 30 °C (maximum reduction of 61.9% ± 0.4%), whereas it presented a constant or slightly increasing trend after 24 h of cultivation at 35 °C and 40 °C (
Figure 3). Overall, total solids and suspended solids in the thin stillage were comparably reduced by 16%–21% and 37%–54%, respectively, after cultivation with
Rhizopus sp. No statistical differences were found between the tested temperatures (
p = 0.188 and
p = 0.072, respectively). However, if the sum of the consumed glycerol, lactic acid, arabinose and xylose is considered (around 8 g·L
−1 at all tested temperatures), it is considerably lower than the reduction in total solids and suspended solids (15–20 g·L
−1 and 14–20 g·L
−1, respectively). Clearly, other components of the thin stillage were assimilated by the
Rhizopus strain. This might explain the constant high level of protein during cultivation. It is well known that
Zygomycetes fungi can produce a wide range of enzymes depending upon the substrate they grow on being able to assimilate different carbon and nitrogen sources [
8]. In absolute terms, the biomass production might be overestimated due to entanglement with solids in the fungal mycelium.
Figure 3.
Concentration of lactic acid, glycerol, ethanol, arabinose and xylose during cultivation of Rhizopus sp. in thin stillage at 30 °C (black), 35 °C (grey) and 40 °C (white).
Figure 3.
Concentration of lactic acid, glycerol, ethanol, arabinose and xylose during cultivation of Rhizopus sp. in thin stillage at 30 °C (black), 35 °C (grey) and 40 °C (white).
Altogether, cultivation in the thin stillage with Rhizopus sp. resulted in the production of biomass containing 49%–55% protein and 6% glucosamine-based polymers as well as a spent medium with a reduced amount of total solids (up to 21%). No addition of nutrients was required; pH adjustment with 25% NH3 instead of NaOH did not lead to any further improvement in the biomass production (data not shown).
3.3. Cultivation in Thin Stillage Using Ascomycetes Fungi
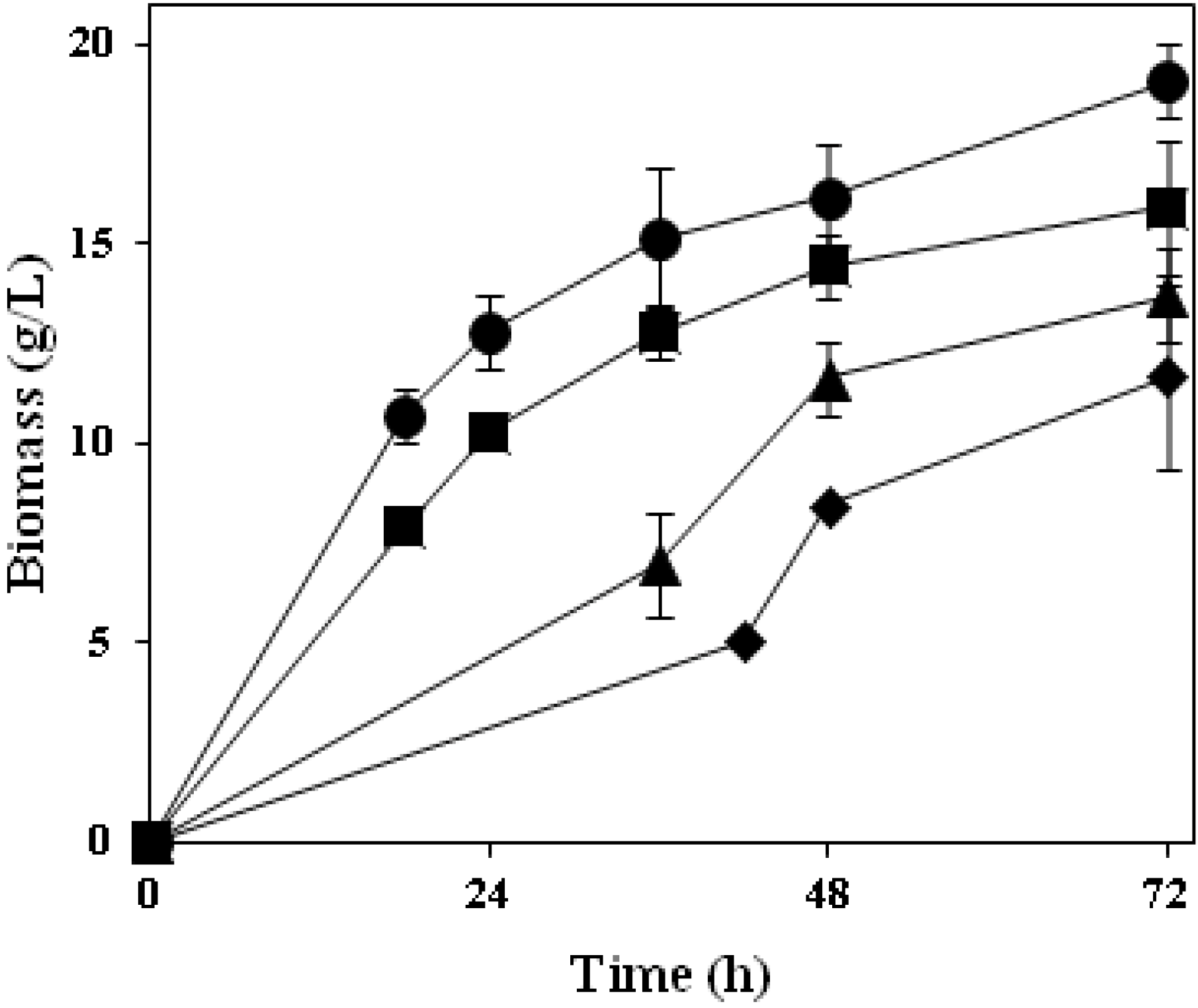
A preliminary study on thin stillage valorisation was performed using four
Ascomycetes fungi; their biomass production profiles are depicted in
Figure 4. Common aspects for all of them are either their long tradition in the production of food products or their extensive use at industrial scale that make them well-known/studied microorganisms. Similar to the
Rhizopus strain used, all four strains examined were able to grow extensively in the thin stillage containing 9% total solids. Cultivation with
A. oryzae resulted in the highest amount of produced fungal biomass (19 g·L
−1);
N. intermedia gave rise up to 16 g·L
−1 of biomass while
F. venenatum and
M. purpureus presented a longer lag phase and
ca. 14 g·L
−1 and 12 g·L
−1 of biomass were reached at the end of cultivation, respectively (
Figure 4; Table 2). The final biomass concentration produced was found to be statistically different among tested strains (
p = 0.003); final produced biomass with
A. oryzae was statistically different from that of
M. purpureus and
F. venenatum but not from that of
N. intermedia. Maximum biomass productivities of (595 ± 36) mg·L
−1·h
−1 and (439 ± 13) mg·L
−1·h
−1 were achieved after 18 h of cultivation with
A. oryzae and
N. intermedia, respectively, while (242 ± 19) mg·L
−1·h
−1 and (176 ± 2) mg·L
−1·h
−1 were achieved after 48 h of cultivation with
F. venenatum and
M. purpureus, respectively.
A. oryzae together with
F. venenatum grew in a well dispersed mycelial form, whereas
N. intermedia and
M. purpureus grew as dense mycelial suspensions. All fungi examined were also found to contain high crude protein content since ranges of 44%–56% of biomass dry weight were obtained (Table 2).
A. oryzae and
M. purpureus exhibited a slight decrease in their crude protein during cultivation (52%–48% and 49%–44%, respectively). In contrast, the crude protein of
F. venenatum increased slightly during cultivation (53%–56%), while that of
N. intermedia remained somewhat constant. Final crude protein levels were statistically different (
p = 0.001) among tested strains. No statistically significant differences were found between
A. oryzae and
M. purpureus and between
F. venenatum and
N. intermedia.
Figure 4.
Produced biomass profiles during cultivation of Ascomycetes filamentous fungi in thin stillage at 30 °C. A. oryzae (circles), N. intermedia (squares), F. venenatum (triangles), M. purpureus (diamonds).
Figure 4.
Produced biomass profiles during cultivation of Ascomycetes filamentous fungi in thin stillage at 30 °C. A. oryzae (circles), N. intermedia (squares), F. venenatum (triangles), M. purpureus (diamonds).
Interesting differences were found in the thin stillage after cultivation with different
Ascomycetes.
M. purpureus,
F. venenatum and
N. intermedia reduced the glycerol concentration by 7%–14%, while cultivation with
A. oryzae resulted in a 54% reduction (
Figure 5; Table 2).
Final glycerol reduction percentages were found to be statistically different (
p = 0.000); total glycerol reductions by
A. oryzae and
F. venenatum were statistically different among each other as well as statistically different from those obtained by the remaining strains, while no statistical difference was found between glycerol reduction values by
M. purpureus and
N. intermedia. The initial acetic acid present in the stillage had been completely consumed after 18 h, while the concentration of lactic acid remained constant during cultivation with all applied
Ascomycetes fungi (Table 2). The main sugars present in the thin stillage (arabinose and xylose) were also followed during cultivation with
Ascomycetes (
Figure 5). Both
A. oryzae and
M. purpureus assumed a consumption trend of arabinose reaching a maximum reduction of 45% ± 5% and 69% ± 3%, respectively. On the other hand, arabinose concentration remained constant during cultivation with
F. venenatum, and increased during cultivation with
N. intermedia; a maximum of (3.6 ± 0.0) g·L
−1 was recorded at 24 h of cultivation (
Figure 5). In contrast, an increase in xylose concentration was observed for all strains examined. The highest concentration of (1.9 ± 0.0) g·L
−1 was recorded during cultivation with
M. purpureus. Cultivation with
N. intermedia resulted in the lowest final amount of xylose (Table 2).
Figure 5.
Concentration profiles of glycerol, ethanol, arabinose and xylose during cultivation of Ascomycetes in thin stillage at 30 °C. A. oryzae (circles), N. intermedia (squares), F. venenatum (triangles), M. purpureus (diamonds).
Figure 5.
Concentration profiles of glycerol, ethanol, arabinose and xylose during cultivation of Ascomycetes in thin stillage at 30 °C. A. oryzae (circles), N. intermedia (squares), F. venenatum (triangles), M. purpureus (diamonds).
Additionally,
ca. 5 g·L
−1 extra ethanol were produced by
N. intermedia. Cultivation with
A. oryzae and
F. venenatum gave rise to
ca. 2 g·L
−1 additional ethanol, while
M. purpureus produced
ca. 0.5 g·L
−1 of extra ethanol (
Figure 5). Maximum amounts of ethanol were found to be statistically different among used strains (
p = 0.000); the pair-wise comparison between the extra ethanol amount produced by
A. oryzae and
F. venenatum was the only one found to be not statistically different. Reports on ethanol production from thin stillage are scarce in literature; one exception relates to the work performed with a metabolically engineered
Escherichia coli strain performed by Gonzalez
et al. [
17]. An ethanol yield of 0.42 g·g
−1 based on consumed glycerol, maltose and glucose present in thin stillage was reported. Altogether, the reduction of total solids was around 32%, 21%, 16% and 34%, and for suspended solids, the reduction was around 55%, 40%, 58% and 69% during cultivation with
A. oryzae,
F. venenatum,
M. purpureus and
N. intermedia, respectively. The reduction of total solids was found to be statistically different among strains (
p = 0.007). No statistically significant differences were found between
A. oryzae and
N. intermedia and between
F. venenatum and
M. purpureus. On the other hand, no statistical differences were found for the reduction of suspended solids (
p = 0.079).
The results support the degradation of arabinan by
F. venenatum and
N. intermedia and xylan by all examined
Ascomycetes strains and xylan degradation by
Rhizopus sp. when cultivated at 35 °C and 40 °C. These polymers are traditionally found in thin stillage [
27]. In the present conditions,
Ascomycetes did not consume lactic acid during cultivation. Reasons for such difference in comparison to the
Rhizopus sp. might include the presence of other more suitable substrates for the
Ascomycetes, the initial pH or nitrogen-limitation. During cultivation with
A. oryzae, the highest amount of biomass (19 g·L
−1) containing 48% crude protein as well as the highest glycerol reduction (54%) were obtained. Cultivation of
N. intermedia led to the production of 16 g·L
−1 biomass containing 56% crude protein and a reduction of around 34% of total solids. Additionally,
N. intermedia produced the highest amount of ethanol (
ca. 5 g·L
−1). Considering an ethanol facility producing 200 m
3 thin stillage per hour (corresponding to
ca. 200,000 m
3 ethanol per year), the inclusion of
N. intermedia could potentially lead to the production of 11,000 m
3 extra ethanol per year. Furthermore, the recovery of this extra ethanol would not need additional steps since it could be sent back into the process after the series of evaporations as is carried out in the present established industrial processes [
18].