A Biofuel Similar to Biodiesel Obtained by Using a Lipase from Rhizopus oryzae, Optimized by Response Surface Methodology †
Abstract
: A new biodiesel-like biofuel is obtained by the enzymatic ethanolysis reaction of sunflower oil with ethanol, in free solvent media, by using BIOLIPASE-R, a multipurpose alimentary additive from Biocon®-Spain that is a low cost lipase from a strain of Rhizopus oryzae. This biofuel is composed by two parts of fatty acid ethyl esters (FAEE) and one of monoglyceride (MG), which in this form integrates glycerol, through the application of the 1,3-selective lipases. Thus, this process minimizes waste generation and maximizes the efficiency of the process because no residual glycerol is produced. Response surface methodology (RSM) is employed to evaluate the main reaction parameters (reaction temperature, oil/ethanol ratio and pH) on the sunflower oil conversion. Water content and amount of lipase were also previously investigated. Regarding the results, we found that it operates optimally with a water content of the reaction medium of 0.15%, 0.05%–0.1% lipase by weight relative to the weight of oil used, 20 °C, volume ratio (mL/mL) oil/ethanol 12/3.5 and pH 12 (by addition of 50 μL of 10 N NaOH solution). These results have proven a very good efficiency of the biocatalyst in the studied selective process.1. Introduction
The production of biodiesel has become very important in recent years as a potential alternative to partially satisfy the future energetic demands in the transport sector [1–3] since the availability of fossil fuels, from the last century until nowadays the main primary source of energy, is becoming increasingly more limited. In this respect, among the different existing methods to produce biofuels, transesterification with short chain alcohols is currently the most attractive and widely accepted methodology for biodiesel production [4]. This usually involves the use of homogeneous base catalysts operating under mild conditions. In order to shift the equilibrium to the production of fatty acid methyl esters (FAME), an excess of methanol is normally utilized in the process to produce biodiesel and glycerol is always obtained as the main by-product through the stepwise process. Thus, besides the alkaline impurities that need to be removed in the conventional method, the accumulation of glycerol is the main drawback of this method, not only because it supposes a lowering in the atomic yield of the process, but also because this residual glycerol must be removed from the obtained biodiesel to avoid problems of polymerization and of course of performance in direct injection (DI) motors. In this way, several consecutive water washing steps are generally applied, where a lot of water it is actually spent to achieve the complete elimination of the glycerol [5].
To avoid the problems associated with the generation of glycerol in the conventional process, a series of alternative methods are under investigation. They all are based on preparing various glycerol derivatives in the same transesterification process. These novel methodologies are able to prepare methyl esters of fatty acids from lipids, using different acyl acceptors, instead of methanol, in the transesterification process, which directly affords alternative glycerol derivative co-products [6]. Thus, the transesterification reaction of triglycerides with dimethyl carbonate (DMC) [7], ethyl acetate [8] or methyl acetate [9] can generate a mixture of three molecules of FAME or fatty acid ethyl esters (FAEE) and one of glycerol carbonate (GC) or glycerol triacetate (triacetin) [10].
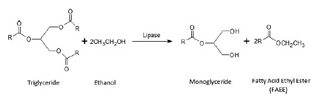
In this way, our research group have recently developed a protocol for the preparation of a new biodiesel-like biofuel, that integrates glycerol into its composition via 1,3-regiospecific enzymatic transesterification of sunflower oil using free [11–14] and immobilized [11,14,15] porcine pancreatic lipase (PPL). It was found that compared to the conventional biodiesel preparation method, the operating conditions of such an enzymatic process were much smoother and did not generate any acidic or alkaline impurities. Thus, the so-called Ecodiesel biofuel [11–15], synthesized through the partial ethanolysis of triglycerides with 1,3-selective lipases, is constituted by a mixture of two parts of FAEE and one of monoacylglyceride (MG). These MGs integrate the glycerol as a derivative product that is soluble in the FAEE mixture, thus working as a biodiesel-like biofuel. In this case, ethanol is used as a cheap reagent, instead of the more expensive ones such as dimethyl carbonate or methyl acetate. This procedure takes advantage of the 1,3-selective nature of the most known lipases, which allows stopping the process in the second step of the alcoholysis, thereby obtaining the previously commented mixture of two moles of FAEE and one of MG as products (Scheme 1), reducing in this way the environmental impact of the process.
In summary, the enzymatic process to obtain this new biofuel operates under much smoother conditions, besides, impurities are no produced and the biofuel produced exhibits similar physicochemical properties to those of conventional biodiesel. Last, but not least, monoacylglycerides (MG) enhance biodiesel lubricity, as demonstrated by recent studies [16–18]. Moreover, the ethanol that is not spent in the enzymatic process also remains in the reaction mixture in such a way that, the product blends obtained after the reaction can be directly used as a fuel. In this respect, current studies [19–21] have proven that blends of diesel fuel and ethanol with biodiesel produce a little less maximum power output than regular diesel. No significant difference in the emissions of CO2, CO, and NOx between regular diesel and biodiesel, ethanol and diesel blends was observed, but the use of these blends resulted in a reduction of particulate matter. Thus, the term Ecodiesel is being currently ascribed to blends of fatty acid alkyl ester with ethanol, alone or with any proportion of diesel fuel [21,22].
The current existing limitations to applying industrial lipases have been mainly associated with their high production costs, which can be overcome through the application of molecular technologies to achieve the production of purified enzymes in sufficiently high quantities [23,24]. In this way, to achieve economic viability, the crucial factors affecting productivity of enzymatic biodiesel synthesis are suitable raw materials and the selected lipase. The latter can be properly modified to improve stability and catalytic efficiency, by optimization of parameters like the molar substrate ratio, temperature, water activity and pH of the enzyme's microenvironment [25]. In this respect, although Ecodiesel was initially produced using porcine pancreatic lipases (PPL), remarkable results have been also obtained with a low cost purified microbial lipase, Lipopan 50 BG (Novozymes AS, Bagsværd, Denmark) [12], from the microorganism Thermomyces lanuginosus, usually used as bread emulsifier (bread improver) [26]. The application of an available lipase on an industrial scale is a significant advance to achieve an economically feasible biofuel production by enzymatic methods.
In this context of research targeted at improving the viability and competitiveness of the enzymatic process, the present study aims to evaluate the BIOLIPASE-R a low cost powdered enzyme preparation containing lipases from a strain of Rhizopus oryzae (ROL). This is a multipurpose additive from Biocon®-Spain, used in the food industry. Although the use of Rhizopus oryzae lipase in the synthesis of conventional biodiesel as well as in other transesterification processes has been described [27], under our best knowledge, BIOLIPASE-R never has been used in any green chemical process, including oil transesterification. Thus, we have tried to evaluate the 1,3 selective behaviour of this low cost, industrial commercial lipase, to make feasible the profitable production of alternative biofuels, using an enzymatic approach. In this respect, in order to evaluate the influence of several crucial reaction parameters in the transesterification reaction, optimum values of water content and lipase amount were firstly determined. After that, using these optimum values a multi-factorial design of experiments and response surface methodology was applied for other reaction parameters such as temperature, oil/ethanol volumetric ratio and pH, controlled by adding different quantities of aqueous solutions of NaOH 10 N, to optimize the catalytic behaviour of this 1,3-selective BIOLIPASE-R. In the current partial ethanolysis of sunflower oil, a biofuel that integrates glycerol as MG, together to the different FAEEs obtained in the enzymatic ethanolysis process is obtained, as well as an excess of unreacted ethanol. This biofuel mixture currently named Ecodiesel is able to directly operate in diesel engines, alone or in whichever mixture with diesel fuel, without any further separation or purification.
2. Results and Discussion
2.1. Comparative Chromatograms of Standardized Reaction Products
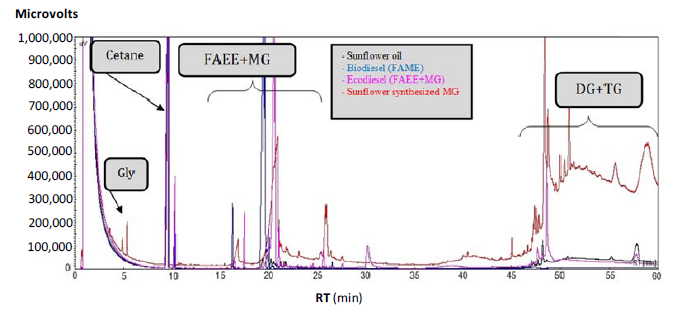
To identify the most characteristic components of biofuels obtained by enzymatic alcoholysis, as well as to compare their rheological properties, several commercial FAME reference standards of FAEE, MG and TG (triacylglycerides) were used, as shown in Figure 1. Here a representative sample of sunflower oil monoglycerides is also included, that was easily produced by the substitution of methanol or ethanol by glycerol, in a conventional alcoholysis process with KOH as homogeneous catalyst following standard experimental conditions.
In Figure 1 we can see that the different fatty acids esters (FAEs), that comprise the lipid profile of the sunflower oil, display retention times (RT) slightly higher than that of cetane (n-hexadecane), used as internal standard. Thus, whereas the RT of cetane is around 10 min, all RT of FAEs appear in the range of 16 to 26 min. These are composed of methyl, ethyl and glycerol esters (the later including MGs) of palmitic, stearic, linoleic and oleic acids. Thus, palmitic acid (C16:0) derivatives are grouped in a narrow range of RT, 16 to 17 min. Derivatives of oleic (C18:1) and linoleic acid (C18:2) are grouped in RT of 19 to 21 min, with the exception of the glycerol ester of oleic acid, or what is the same, the MG of oleic acid, has a different behaviour, with a RT = 26 min. The RT of glycerol is 5 min, before cetane. The absence of this compound in the obtained chromatograms clearly demonstrates the 1,3 regioselective nature of the studied enzymatic transesterification reaction.
In Figure 1 the presence of DGs (diacylglycerides) with higher retention times, 40–60 min, that do not allow their integration in the GC chromatogram can also be seen, so that it is necessary to determine DG together and TG, by using an internal standard such as the cetane here employed. It should be noted that the differences in RT values between MG and DG are much higher than those existing between MG and FAME or FAEE, as expected from the differences between their corresponding molecular weights. At the same time, it is clear that the FAMEs, FAEEs and MGs display somewhat higher RT values than cetane, but within the same molecular weight range, which allows us to anticipate that the FAE should have similar chemical-physical properties to the hydrocarbons that constitute diesel.
Since the retention times of different fatty acid derivatives are considered very closely related to their chemical-physical properties, the great similarity of the obtained RT values is a clear demonstration of the similarity between the rheological properties of the different MGs with their corresponding FAMEs or FAEEs, which is crucial to allow its use as a fuel capable of substituting for petroleum products. Consequently, conversion (as wt %) is a reaction parameter where all molecules (FAEE, MG and DG) obtained in the ethanolysis of TG are included, and it will be considered as a very different parameter, respect to the selectivity (as wt %), where only FAEEs and MGs are included, all with RT values lower than 26 min. These molecules exhibit RT values similar to those of the hydrocarbons present in conventional diesel, so they could exhibit similar physicochemical and rheological properties. However a high conversion could indicate a high proportion of DG molecules, with high molecular weight and high viscosity values. Consequently, a very high selectivity, indicating a very high percentage of FAEEs and MGs, could result in a viscosity value close to that of petroleum diesel, so that the highest conversion value is not a sufficient guarantee of lower viscosity values. Thus, both parameters will be provided as GC analysis results of the reaction products. Taking into account that retention times of the complex mixture of hydrocarbons constituting fossil diesel fuel range from 1 to 25 min, as a reference value for different biofuels (FAME, FAEE, MG) as selectivity value, all those FAEs that present RT values coincident with the hydrocarbons constituting diesel, or those with RT lower than 25 min is used, as it is expected they also present similar physicochemical and rheological properties as conventional diesel.
2.2. Variables Effect on Enzyme Activity
To carry out an evaluation of enzyme activity and optimize reaction conditions for this enzyme, a multivariable experimental design in which the influence of the temperature, pH environment and the oil/ethanol volume ratio has been analyzed, as well as the magnitude of these influences, has been performed. According to previous results [12], before carrying out the multivariable experimental design for Analysis of Variance (ANOVA), a more detailed study about the influence of enzyme and water amounts was developed to operate under optimum conditions with respect to these two strongly influential variables. Thus, both variables have been studied separately, obtaining in this way further information about their influence and simplifying the subsequent multivariable experimental design (ANOVA). The current study has also followed in this case the One Variable at a Time (OVAT) methodology, for which initial conditions of pH, temperature and oil/alcohol volume ratio have been set and variables were modified one by one.
2.2.1. Effect of Water Content
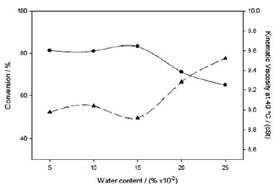
The water content is a very important parameter in enzymatic transesterification, through the water activity (aw), that accounts for the intensity with which water associates with lipases to obtain the best enzymatic activity, especially in solvent-free systems. A series of experiments under optimal conditions for temperature, pH and oil/ethanol ratio, obtained from previous RSM studies [12], were carried out to evaluate the effect of this parameter. Figure 2 shows the effect of water content in the reaction yield achieved in the transesterification reaction of sunflower oil. A minimum kinematic viscosity, which corresponds to a maximum conversion, was achieved at a concentration of 0.15% of added water in the reaction medium. Consequently, the water content is a very important parameter that must be controlled in the ethanolysis processes, so this parameter has been set to the optimum value (0.15 wt % water) to carry out the multivariable experimental design to analyze the influence of the temperature, pH environment and the oil/ethanol volume ratio as well as the magnitude of these influences.
2.2.2. Effect of the Quantity of Lipase
Figure 3 depicts the effect of the quantity of enzyme utilized on conversion and kinematic viscosity. Twenty mg of lipase was selected as the optimum value in all reactions, as this quantity was shown to be sufficient to provide a combined good yield. It can be also seen that there is a subsequent yield decrease as the amount of lipase added is increased. The is probably due to the effects of enzymatic agglomeration already described for other lipases in free form [12]. Therefore, for subsequent experiments, the optimum amount of catalyst to be used was fixed at 20 mg.
2.2.3. Analysis of Variance (ANOVA) and Optimization of the Reaction Parameters by RSM
The analysis of variance methods has become very attractive in reaction parameter optimization and in the evaluation of the effects of the parameters in the TG transesterification reaction [9,12,28] due to its effectiveness in the analysis of variables. Thus, results are obtained in 36 runs, each one with different experimental conditions, selected by a multifactorial design of experiments with three factors, using the software STATGRAPHICS CENTURION version XV.I. (Sigma-plus, Levallois-Perret France), where two of them are developed at three levels, and the other at two levels, as indicated in Table 1.
Results achieved following this methodology are shown in Table 2. The quantity of biocatalyst (BIOLIPASE-R) in all these experiments was fixed at 20 mg. All experiments were duplicated and run in a random way in order to avoid experimental errors.
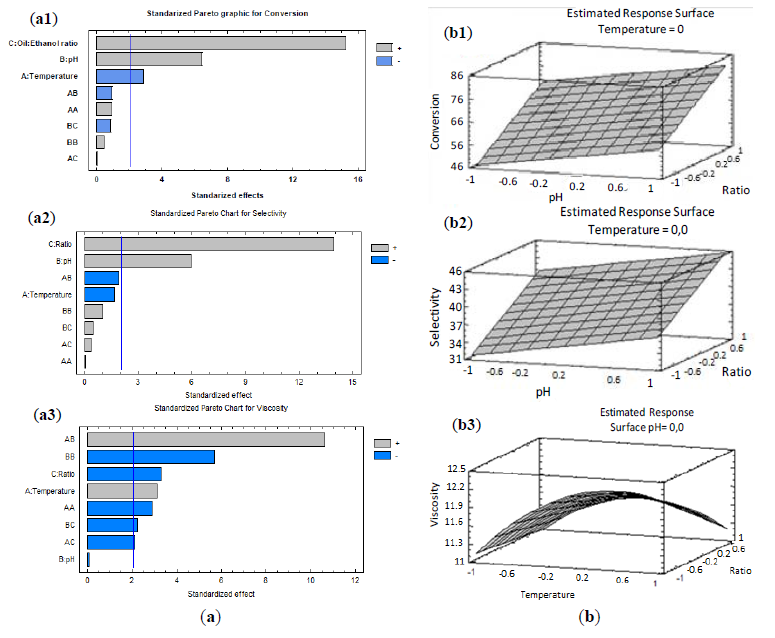
From these data and using the Statgraphics software, a multivariate statistical analysis (ANOVA) has been performed to determine the correlation or “effects” of the experimental studied variables with output variables (conversion, selectivity and kinematic viscosity). The software gives us different data outputs that allow us to analyze the influence of the independent variables in the dependent variables. The quadratic polynomial model was highly significant and sufficient to explain the relationship between conversion/selectivity/kinematic viscosity and important experimental variables, as summarized in Tables 3, 4 and 5. Thus, the results of the factorial design suggested that the major factors affecting the transesterification, for the production of biofuels integrating glycerol as monoacylglycerols, were pH and oil/ethanol ratio (v/v) in conversion and selectivity, however in kinematic viscosity, temperature and oil/ethanol volumetric ratio were the most influential reaction parameters.
The correlation coefficient values R2 were 0.916 for Conversion, 0.901 for Selectivity and 0.877 for Kinematic viscosity, respectively, which imply a good fit between models and experimental data in Pareto graphics, respect to Conversion, Selectivity and Viscosity, as indicated in Figure 4a. The adjusted correlation coefficients R2 were 0.891, 0.872 and 0.84 for Conversion, Selectivity and Kinematic viscosity, respectively. The obtained results pointed out that the temperature, pH and oil/ethanol ratios were also important parameters influencing the conversion, selectivity and viscosity in the systems (p < 0.05).
The software also allows obtaining equations, after the elimination of non-influential parameters in the model for conversion, selectivity and kinematic viscosity, and the R2 values for these dependent variables were 0.871, 0.873 and 0.876, respectively, and the equations obtained (Equations (1)–(3)) were remarkably simpler as compared to the initial ones. These equations describe the model created and gives solutions for the dependent variable based on the independent variable combinations, whether they are or not significant in the response. Thus, taking into account that R is the Oil/Ethanol ratio (v/v), pH is the obtained by the addition of different μL of NaOH 10 N and T the reaction temperature:
The surface plots in Figure 4b, described by the regression model were drawn to display the effects of the independent variables on Conversion, Selectivity and Kinematic viscosity. Here the influence of the different variables in the reaction performance of the systems can be clearly seen. This model showed that the optimum values for the parameters to maximize transesterification yield (Conversion, Selectivity, Kinematic Viscosity) were lower temperatures (20 °C), maximum amount of aqueous NaOH 10 N added (50 μL) and the maximum oil/ethanol (mL/mL) ratio = 12/3.5 (1/6 molar ratio) studied. Conversions up to 80%, Selectivities as high as 70% and Kinematic viscosity values of about 10 mm2·s−1 could be achieved under these conditions, which in theory will render feasible the utilization of the obtained biofuel in blends with diesel. For example, by the addition of only 35% of fossil fuel diesel to this biofuel, a viscosity reduction at 4.8 mm2·s−1 is obtained, a value within the acceptance limits of the EN 14214 standard [3].
2.2.4. Experimental Validation of Proposed Model
To validate the proposed models, a series of three experiments has been carried out, whose reaction conditions have been selected among the range of variables. In this way, reaction conditions and their yields (conversion, selectivity) for each reaction performed are shown in Table 6. Thus, achieved experimental values are similar to theoretical calculated data provided by the proposed models.
2.2.5. Influence of Temperature
To get more accurate information about the influence of temperature on the biocatalytic behavior of BIOLIPASE-R, a series of reactions at different temperatures are performed, operating under the optimum experimental conditions determined here. In this way, it is confirmed that the biocatalyst really shows an optimum of efficiency in the conditions estimated by the mltivariate statistical analysis (ANOVA). Data from these experiments are shown in Figure 5.
3. Experimental Section
3.1. Materials
Commercial sunflower oil was locally obtained. The chromatographically pure ethyl esters of palmitic acid, stearic acid, oleic acid, linoleic acid and linolenic acid were commercially obtained from Accustandard (New Haven, CT, USA), and the hexadecane (cetane) was from Sigma-Aldrich (St Louis, MO, USA). Other chemicals like absolute ethanol and sodium hydroxide were pure analytical compounds (99.5%) obtained commercially from Panreac (Barcelona, Spain). BIOLIPASE-R, a low cost powdered enzyme preparation containing obtained lipases from a strain of Rhizopus oryzae, was kindly provided by Biocon®-Spain (Barcelona, Spain).
3.2. Ethanolysis Reactions
These reactions were performed according to the previously described experimental procedure [11–15] to determine the optimal conditions for obtaining the selective ethanolysis reaction, such as amount of lipase, water content, pH (controlled by addition of different volumes of 10 N NaOH solution), the oil/ethanol molar ratio (v/v) and temperature. Thus, enzymatic assays are carried out with commercial sunflower oil (9.4 g, 12 mL, 0.01 mol) at controlled temperatures (20–40 °C) in a 25 mL round bottom flask. Reaction mixtures were stirred with a conventional magnetic stirrer at a higher stirring speed than 300 rpm, to avoid mass transfer limitations, for a reaction time of 2 h. Variable oil/alcohol volume ratios at different pH, several water contents and different quantities of lipase are studied. The different oil/ethanol ratios (v/v) are obtained by introducing absolute ethanol volumes in the 1.75–3.5 mL range, the influence of different amounts of lipase are studied in the 0.05–0.5 g range, the water content is varied from 0.05 to 0.25 wt % by adding deionized water, the sample is left under stirring with the oil for about 15 min to homogenize and stabilize the system. The influence of pH values was examined by adding different volumes (12.5–50 μL) of 10 N aqueous NaOH solution. In this regard, a blank reaction in the presence of the highest quantity of solution of NaOH was performed to rule out a potential contribution from the homogeneous NaOH catalysed reaction. Less than 10% conversion of the starting material was obtained, so the contribution of the homogenous base catalysis can be considered as negligible under the investigated conditions. All variables were studied and optimized according to a factorial experimental design and a response surface methodology.
3.3. Analytical Methods
Reaction products were monitored by capillary column gas chromatography, using a Varian 430-GC gas chromatograph (Walnut Creek, CA, USA), connected to a HT5 capillary column (25 m × 0.32 mm ID × 0.1 μm, SGE, Supelco) with a flame ionization detector (FID) (Walnut Creek, CA, USA) at 450 °C and splitless injection at 350 °C. Helium is used as carrier gas, with a flow of 1.5 mL/min. It has been applied a heating ramp from 90 to 200 °C at a rate of 7 °C/min, followed by another ramp from 200 to 360 °C at a rate of 15 °C/min, maintaining the temperature of the oven at 360 °C for 10 min using as internal standard n-hexadecane (cetane) to quantify the content of ethyl esters and the different glycerides (-mono, di and triglycerides) with the help of several commercial standard fatty acid esters. This method allows us to completely analyse the sample in a single injection and in a time no longer than 60 min, which simplifies the process and increases the speed of analysis [11–15].
Considering that sunflower oil is constituted by a mixture of fatty acids (mainly linoleic, oleic, palmitic and stearic acids) in variable proportions, the reactions results are expressed as the relative amounts of the corresponding ethyl esters (Fatty Acid Ethyl Esters–FAEE,), monoglycerides (MG) and diglycerides (DG) that are integrated in the chromatogram. The amount of triglycerides (TG) which has not reacted is calculated from the difference to the internal standard (cetane). Thus, the conversion includes the total amount of triglyceride transformed (FAEE + MG + DG) in the ethanolysis process and selectivity makes reference to the relative amount of FAEE + MG obtained. The latter are those ones having retention times close to the cetane standard, which is the reference hydrocarbon for diesel fuel.
3.4. Viscosity Measurements
Transesterification reactions of oils and fats are basically carried out to obtain an important reduction in the viscosity of these materials, as they share similar values of other significant chemical-physical parameters with fossil diesel, except for the viscosity. In this respect, most of oils exhibit viscosities in the 30–45 mm2/s cSt range, while fossil diesel is in the 2.5–6 cSt range. Thus, due to the importance of viscosity for the correct running of diesel engines, this parameter becomes in the critical factor to change in the chemical-physical properties of vegetable oils before their use as biofuel. The transesterification process of oils and fats is actually developed in order to obtain a noticeable lowering in viscosity of the oils so the resulting product can be employed as biofuel in current existing diesel engines. Thus, accurate viscosity measurements are critical to assess the quality of the produced biofuels, since unsuitable viscosity values can decisively affect the correct working conditions of the diesel engine. Therefore, the characterization of this parameter is essential to evaluate the results obtained in the ethanolysis process.
Viscosities were determined in a Oswald Proton Cannon-Fenske Routine Viscometer 33,200 (Sigma-Aldrich, St. Louis, MO, USA), size 150 capillary viscometer. This is based on determining the time needed for a given volume of fluid to pass between two points marked on the instrument. The kinematic viscosity is given by the ratio between the dynamic viscosity (h, in Poise, g/cm s) and the density (r, in g/cm3); υ = h/r, in cm2/s or centistokes, cSt, mm2/s. Samples, previously centrifuged at 3500 rpm for 10 min and filtered at 50 °C, are immersed in a thermostatic bath at 40 °C for 15 min, making sure that the temperature remains stable. Then, samples are introduced into the viscometer and this, in turn, in the water bath, ensuring that it is rigorously positioned vertically, with the bottom end at a minimum distance of 2 cm from the floor of the bath [11–15].
3.5. Experimental Design
The effect of process parameters in the enzymatic transesterification reaction to obtain the optimum conditions for the viscosity, selectivity and conversion were studied using a multifactorial design of experiments with three factors run by the software Statgraphics version XV.I. Two of them were developed at three levels, and the last one at two levels, so that it gives 36 runs. The experiments were performed in random order. The experimental parameters selected for this study were reaction temperature, oil/ethanol ratio (v/v) and different pH values obtained by the addition of variable volumes, in μL, of 10 N NaOH. Table 1 shows the coded and actual values of the process parameters used in the design matrix.
3.6. Statistical Analysis
The experimental data obtained from experimental design were analyzed by response surface methodology (RSM) [9,12,28]. A mathematical model, following a second-order polynomial equation, was developed to describe the relationships between the predicted response variable (viscosity, conversion and selectivity) and the independent variables of reaction conditions, as it is shown in the Equation (4), where Y, is the predicted response variable; β0, βi, βii, βij the intercept, linear, quadratic and interaction constant coefficients of the model, respectively; Xi, Xj (i = 1, 3; j = 1, 3; i ≠ j) represent the coded independent variables:
Response surface plots were developed using the fitted quadratic polynomial equation obtained from regression analysis, holding one of the independent variables at constant values corresponding to the stationary point and changing the order two variables. The quality of the fit of the polynomial model equation was evaluated by the coefficient of determination R2, and its regression coefficient significance was checked with an F-test. Confirmatory experiments were carried out in order to validate the model, using combinations of independent variables which were not part of the original experimental design, but within the experimental region.
4. Conclusions
In order to improve a new methodology that integrates glycerol in different monoacylglycerol molecules, the commercial BIOLIPASE-R, lipase from a strain of Rhizopus oryzae, a multipurpose alimentary additive from Biocon®-Spain, was evaluated as biocatalyst in the 1,3 selective ethanolysis of sunflower oil. Results show that ethanol/sunflower oil substrate molar ratio, pH and reaction temperature, as well as the water content and biocatalyst amount, have a significant effect on the percentage of reaction yield (conversion, selectivity and cinematic viscosity). On the basis of RSM analysis, we found that the optimum conditions are a reaction at 20 °C, with a volume ratio (mL/mL) oil/ethanol 12/3.5 and at pH 12 (obtained by addition of 50 μL of 10 N NaOH dissolution). Previously it was determined that optimal water content of the reaction medium was of 0.15%, and the best lipase amount by weight relative to the weight of oil used was 0.05%–0.1%.
Thus, the present results indicate that the studied low cost commercial lipase is specially efficient in catalyzing 1,3 selective ethanolysis processes, where glycerol is kept as MG in the biofuel mixture, with the different obtained FAEEs. The obtained mixture of MGs and FAEEs with the excess of unreacted ethanol, constitutes the Ecodiesel biofuel that can be used directly, without any kind of subsequent cleaning, separation or purification process. In this way, this biofuel can be economically viable and environmentally sustainable with the help of BIOLIPASE-R since by using a low cost and industrially available lipase, an important cost reduction is produced of the process, with high yield in very short reaction times (less than 1 h) and under mild reaction conditions. Besides, not only is a higher atomic yield achieved, respect to the conventional biodiesel reaction, (because no glycerol byproduct is generated), but also no purification step of the residual glycerol is necessary, so it can be used directly after its production, thus avoiding an important part of the production cost currently existing in the fabrication of conventional biodiesel.
Accordingly, the 1,3 selective behaviour of these free lipase, could open a new technically feasible and economically viable way to deal with the production of alternative biodiesel using an enzymatic approach.
Acknowledgments
Grants from the Spanish Ministry of Economy and Competitiveness (Project ENE 2011-27017), Spanish Ministry of Education and Science (Projects CTQ2010-18126 and CTQ2011-28954-C02-02), FEDER funds (Regional Development European Funds) and Junta de Andalucía FQM 0191, PO8-RMN-03515 and P11-TEP-7723 are gratefully acknowledged by the authors. We are also grateful to Biocon®-Spain, for kindly supplying the BIOLIPASE-R.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Demirbas, A. Political, economic and environmental impacts of biofuels: A review. Appl. Energy 2009, 86, S108–S117. [Google Scholar]
- Luque, R.; Herrero-Davila, L.; Campelo, J.M.; Clark, J.H.; Hidalgo, J.M.; Luna, D.; Marinas, J.M.; Romero, A.A. Biofuels: A technological perspective. Energy Environ. Sci. 2008, 1, 542–564. [Google Scholar]
- Luna, D.; Calero, J.; Sancho, E.D.; Luna, C.; Posadillo, A.; Bautista, F.M.; Romero, A.A.; Berbel, J.; Verdugo, C. Technological challenges for the production of biodiesel in arid lands. J. Arid Environ. 2014, 102, 127–138. [Google Scholar]
- Oh, P.P.; Lau, H.L.N.; Chen, J.H.; Chong, M.F.; Choo, Y.M. A review on conventional technologies and emerging process intensification (PI) methods for biodiesel production. Renew. Sustain. Energy Rev. 2012, 16, 5131–5145. [Google Scholar]
- Saleh, J.; Dube, M.A.; Tremblay, A.Y. Separation of glycerol from fame using ceramic membranes. Fuel Process. Technol. 2011, 92, 1305–1310. [Google Scholar]
- Ganesan, D.; Rajendran, A.; Thangavelu, V. An overview on the recent advances in the transesterification of vegetable oils for biodiesel production using chemical and biocatalysts. Rev. Environ. Sci. Biotechnol. 2009, 8, 367–394. [Google Scholar]
- Ilham, Z.; Saka, S. Two-step supercritical dimethyl carbonate method for biodiesel production from jatropha curcas oil. Bioresour. Technol. 2010, 101, 2735–2740. [Google Scholar]
- Kim, S.J.; Jung, S.M.; Park, Y.C.; Park, K. Lipase catalyzed transesterification of soybean oil using ethyl acetate, an alternative acyl acceptor. Biotechnol. Bioprocess Eng. 2007, 12, 441–445. [Google Scholar]
- Tan, K.T.; Lee, K.T.; Mohamed, A.R. Response to “Comment on a glycerol-free process to produce biodiesel by supercritical methyl acetate technology: An optimization study via response surface methodology”. Bioresour. Technol. 2011, 102, 3990–3991. [Google Scholar]
- Casas, A.; Ruiz, J.R.; Ramos, M.J.; Perez, A. Effects of triacetin on biodiesel quality. Energy Fuels 2010, 24, 4481–4489. [Google Scholar]
- Caballero, V.; Bautista, F.M.; Campelo, J.M.; Luna, D.; Marinas, J.M.; Romero, A.A.; Hidalgo, J.M.; Luque, R.; Macario, A.; Giordano, G. Sustainable preparation of a novel glycerol-free biofuel by using pig pancreatic lipase: Partial 1,3-regiospecific alcoholysis of sunflower oil. Process Biochem. 2009, 44, 334–342. [Google Scholar]
- Verdugo, C.; Luna, D.; Posadillo, A.; Sancho, E.D.; Rodriguez, S.; Bautista, F.; Luque, R.; Marinas, J.M.; Romero, A.A. Production of a new second generation biodiesel with a low cost lipase derived from thermomyces lanuginosus: Optimization by response surface methodology. Catal. Today 2011, 167, 107–112. [Google Scholar]
- Luna, D.; Bautista, F.M.; Caballero, V.; Campelo, J.M.; Marinas, J.M. Method for Producing Biodiesel Using Porcine Pancreatic Lipase as an Enzymatic Catalyst. European Patent EP2050823 A1, 22 April 2009. [Google Scholar]
- Luna, D.; Posadillo, A.; Caballero, V.; Verdugo, C.; Bautista, F.M.; Romero, A.A.; Sancho, E.D.; Luna, C.; Calero, J. New biofuel integrating glycerol into its composition through the use of covalent immobilized pig pancreatic lipase. Int. J. Mol. Sci. 2012, 13, 10091–10112. [Google Scholar]
- Luna, C.; Sancho, E.; Luna, D.; Caballero, V.; Calero, J.; Posadillo, A.; Verdugo, C.; Bautista, F.M.; Romero, A.A. Biofuel that keeps glycerol as monoglyceride by 1, 3-selective ethanolysis with pig pancreatic lipase covalently immobilized on alpo4 support. Energies 2013, 6, 3879–3900. [Google Scholar]
- Wadumesthrige, K.; Ara, M.; Salley, S.O.; Ng, K.Y.S. Investigation of lubricity characteristics of biodiesel in petroleum and synthetic fuel. Energy Fuels 2009, 23, 2229–2234. [Google Scholar]
- Xu, Y.F.; Wang, Q.J.; Hu, X.G.; Li, C.; Zhu, X.F. Characterization of the lubricity of bio-oil/diesel fuel blends by high frequency reciprocating test rig. Energy 2010, 35, 283–287. [Google Scholar]
- Haseeb, A.; Sia, S.Y.; Fazal, M.A.; Masjuki, H.H. Effect of temperature on tribological properties of palm biodiesel. Energy 2010, 35, 1460–1464. [Google Scholar]
- Çelikten, I. The effect of biodiesel, ethanol and diesel fuel blends on the performance and exhaust emissions in a di diesel engine. Gazi Univ. J. Sci. 2011, 24, 341–346. [Google Scholar]
- Cheenkachorn, K.; Fungtammasan, B. Biodiesel as an additive for diesohol. Int. J. Green Energy 2009, 6, 57–72. [Google Scholar]
- Jaganjac, M.; Prah, I.O.; Cipak, A.; Cindric, M.; Mrakovcic, L.; Tatzber, F.; Ilincic, P.; Rukavina, V.; Spehar, B.; Vukovic, J.P.; et al. Effects of bioreactive acrolein from automotive exhaust gases on human cells in vitro. Environ. Toxicol. 2012, 27, 644–652. [Google Scholar]
- Pang, X.B.; Mu, Y.J.; Yuan, J.; He, H. Carbonyls emission from ethanol-blended gasoline and biodiesel-ethanol-diesel used in engines. Atmos. Environ. 2008, 42, 1349–1358. [Google Scholar]
- Macario, A.; Verri, F.; Diaz, U.; Corma, A.; Giordano, G. Pure silica nanoparticles for liposome/lipase system encapsulation: Application in biodiesel production. Catal. Today 2013, 204, 148–155. [Google Scholar]
- Macario, A.; Giordano, G. Catalytic conversion of renewable sources for biodiesel production: A comparison between biocatalysts and inorganic catalysts. Catal. Lett. 2013, 143, 159–168. [Google Scholar]
- Szczesna-Antczak, M.; Kubiak, A.; Antczak, T.; Bielecki, S. Enzymatic biodiesel synthesis—Key factors affecting efficiency of the process. Renew. Energy 2009, 34, 1185–1194. [Google Scholar]
- Moayedallaie, S.; Mirzaei, M.; Paterson, J. Bread improvers: Comparison of a range of lipases with a traditional emulsifier. Food Chem. 2010, 122, 495–499. [Google Scholar]
- Yara-Varon, E.; Joli, J.E.; Torres, M.; Sala, N.; Villorbina, G.; Mendez, J.J.; Canela-Garayoa, R. Solvent-free biocatalytic interesterification of acrylate derivatives. Catal. Today 2012, 196, 86–90. [Google Scholar]
- Chang, C.; Chen, J.H.; Chang, C.M.J.; Wu, T.T.; Shieh, C.J. Optimization of lipase-catalyzed biodiesel by isopropanolysis in a continuous packed-bed reactor using response surface methodology. New Biotechnol. 2009, 26, 187–192. [Google Scholar]


| Variables | Unit | Levels | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| Temperature | °C | 20 | 30 | 40 |
| Oil/Ethanol ratio (v/v) | mL/mL | 12/1.75 | – | 12/3.5 |
| pH | (μL NaOH 10 N) | 8 (12.5) | 10 (5) | 12 (50) |
| Run | Studied variables | Output variables | ||||
|---|---|---|---|---|---|---|
| Temperature | pH | Oil/Ethanol ratio | Conversion (%) | FAEE (%) | Cinematic viscosity (mm2·s−1) | |
| 1 | −1 | 0 | 1 | 81.2 | 43.5 | 9.8 |
| 2 | 1 | −1 | −1 | 44.7 | 31.9 | 9.7 |
| 3 | 0 | 0 | 1 | 70 | 43.5 | 12.2 |
| 4 | 1 | 1 | 1 | 75.8 | 42.1 | 11.8 |
| 5 | −1 | 1 | 1 | 83.6 | 47.6 | 8.1 |
| 6 | 0 | 1 | −1 | 67.1 | 39.6 | 11.4 |
| 7 | −1 | −1 | 1 | 74.8 | 40.6 | 12.0 |
| 8 | 1 | 1 | −1 | 55.5 | 34.8 | 12.5 |
| 9 | 1 | 0 | −1 | 47.2 | 32.4 | 12.3 |
| 10 | −1 | 1 | −1 | 60 | 36.2 | 8.8 |
| 11 | 1 | 0 | 1 | 75.9 | 43.8 | 11.2 |
| 12 | 0 | −1 | −1 | 46.8 | 32.6 | 11.3 |
| 13 | 0 | 0 | −1 | 51.6 | 32.4 | 12.7 |
| 14 | 0 | −1 | 1 | 60.8 | 38.5 | 11.2 |
| 15 | 0 | 1 | 1 | 81.1 | 49.7 | 10.6 |
| 16 | −1 | −1 | −1 | 47.9 | 32.6 | 11.6 |
| 17 | 1 | −1 | 1 | 73.4 | 43.7 | 9.1 |
| 18 | −1 | 0 | −1 | 55.0 | 34.9 | 11.2 |
| Run | Repetition of experiments | Conversion (%) | Selectivity (%) | Cinematic viscosity (mm2·s−1) | ||
| Temperature | pH | Oil/Ethanol ratio | ||||
| 19 | 1 | 0 | −1 | 46.2 | 31.5 | 12.7 |
| 20 | 0 | 0 | 1 | 73.2 | 40.7 | 11.8 |
| 21 | 0 | −1 | −1 | 47.2 | 31.9 | 10.7 |
| 22 | −1 | 0 | −1 | 56.2 | 35.3 | 10.9 |
| 23 | 1 | 1 | −1 | 54.3 | 34.5 | 12.5 |
| 24 | −1 | 1 | −1 | 58.1 | 35.7 | 9.4 |
| 25 | 1 | −1 | 1 | 74.7 | 43.1 | 9.3 |
| 26 | −1 | −1 | −1 | 48.2 | 32.6 | 10.9 |
| 27 | 1 | 0 | 1 | 75 | 43.8 | 11.1 |
| 28 | 0 | 1 | 1 | 83.3 | 46.9 | 9.9 |
| 29 | −1 | −1 | 1 | 76.3 | 42.2 | 11.7 |
| 30 | −1 | 1 | 1 | 83.2 | 47.3 | 9.2 |
| 31 | 0 | 0 | −1 | 50.3 | 32.8 | 11.4 |
| 32 | 1 | 1 | 1 | 74.3 | 42.9 | 10.9 |
| 33 | 1 | −1 | −1 | 45.9 | 32.2 | 9.4 |
| 34 | 0 | 1 | −1 | 65.8 | 39.9 | 11.6 |
| 35 | 0 | −1 | 1 | 59.7 | 36.9 | 10.0 |
| 36 | −1 | 0 | 1 | 80.8 | 44.6 | 11.4 |
| Source | Sum of squares | Degrees of freedom | Mean square | F-value | p-value |
|---|---|---|---|---|---|
| A: Temperature | 162.24 | 1 | 162.24 | 8.06 | 0.0087 |
| B: pH | 836.62 | 1 | 836.62 | 41.55 | 0.0000 |
| C: Oil/ethanol ratio | 4648.97 | 1 | 4648.97 | 230.89 | 0.0000 |
| AA | 16.4356 | 1 | 16.4356 | 0.82 | 0.3746 |
| AB | 17.0156 | 1 | 17.0156 | 0.85 | 0.3664 |
| AC | 0.0266667 | 1 | 0.0266667 | 0.00 | 0.9712 |
| BB | 4.15681 | 1 | 4.15681 | 0.21 | 0.6533 |
| BC | 14.2604 | 1 | 14.2604 | 0.71 | 0.4077 |
| Blocks | 0.0025 | 1 | 0.0025 | 0.00 | 0.9912 |
| Total error | 523.511 | 26 | 20.1351 | – | – |
| Total (corrected) | 6223.24 | 35 | – | – | – |
R2 = 91.59%; R2 (adjusted) = 89.10%.
| Source | Sum of squares | Degrees of freedom | Mean square | F-value | p-value |
|---|---|---|---|---|---|
| A: Temperature | 11.2067 | 1 | 11.2067 | 2.78 | 0.1072 |
| B: pH | 142.107 | 1 | 142.107 | 35.31 | 0.0000 |
| C: Oil/ethanol ratio | 780.271 | 1 | 780.271 | 193.89 | 0.0000 |
| AA | 0.0138889 | 1 | 0.0138889 | 0.00 | 0.9536 |
| AB | 14.8225 | 1 | 14.8225 | 3.68 | 0.0660 |
| AC | 0.54 | 1 | 0.54 | 0.13 | 0.7171 |
| BB | 4.30222 | 1 | 4.30222 | 1.07 | 0.3107 |
| BC | 0.881667 | 1 | 0.881667 | 0.22 | 0.6436 |
| Blocks | 0.871111 | 1 | 0.871111 | 0.22 | 0.6456 |
| Total error | 104.633 | 26 | 4.02435 | – | – |
| Total (corrected) | 1059.65 | 35 | – | – | – |
R2 = 90.13%; R2 (adjusted) = 87.20%.
| Source | Sum of squares | Degrees of freedom | Mean square | F-value | p-value |
|---|---|---|---|---|---|
| A: Temperature | 2.34375 | 1 | 2.34375 | 9.69 | 0.0045 |
| B: pH | 0.00166667 | 1 | 0.00166667 | 0.01 | 0.9345 |
| C: Oil/ethanol ratio | 2.61361 | 1 | 2.61361 | 10.81 | 0.0029 |
| AA | 2.03347 | 1 | 2.03347 | 8.41 | 0.0075 |
| AB | 27.3006 | 1 | 27.3006 | 112.88 | 0.0000 |
| AC | 1.08375 | 1 | 1.08375 | 4.48 | 0.0440 |
| BB | 7.86722 | 1 | 7.86722 | 32.53 | 0.0000 |
| BC | 1.215 | 1 | 1.215 | 5.02 | 0.0338 |
| Blocks | 0.2025 | 1 | 0.2025 | 0.84 | 0.3686 |
| Total error | 6.28813 | 26 | 0.241851 | – | – |
| Total (corrected) | 50.9497 | 35 | – | – | – |
R2 = 87.66%; R2 (adjusted) = 84.00%.
| Temperature °C | pH | Oil/ethanol ratio | Conversion (%) | Selectivity (%) | ||
|---|---|---|---|---|---|---|
| Exp. | Adj. | Exp. | Adj. | |||
| 25 | 11.5 | 12/2.9 | 68.8 | 72.7 | 39.5 | 41.5 |
| 30 | 10.5 | 12/2.75 | 63.7 | 66.8 | 42.3 | 39.4 |
| 50 | 9 | 12/3.3 | 70.9 | 68.1 | 44.7 | 40.9 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Luna, C.; Verdugo, C.; Sancho, E.D.; Luna, D.; Calero, J.; Posadillo, A.; Bautista, F.M.; Romero, A.A. A Biofuel Similar to Biodiesel Obtained by Using a Lipase from Rhizopus oryzae, Optimized by Response Surface Methodology. Energies 2014, 7, 3383-3399. https://doi.org/10.3390/en7053383
Luna C, Verdugo C, Sancho ED, Luna D, Calero J, Posadillo A, Bautista FM, Romero AA. A Biofuel Similar to Biodiesel Obtained by Using a Lipase from Rhizopus oryzae, Optimized by Response Surface Methodology. Energies. 2014; 7(5):3383-3399. https://doi.org/10.3390/en7053383
Chicago/Turabian StyleLuna, Carlos, Cristobal Verdugo, Enrique D. Sancho, Diego Luna, Juan Calero, Alejandro Posadillo, Felipa M. Bautista, and Antonio A. Romero. 2014. "A Biofuel Similar to Biodiesel Obtained by Using a Lipase from Rhizopus oryzae, Optimized by Response Surface Methodology" Energies 7, no. 5: 3383-3399. https://doi.org/10.3390/en7053383








