Comparative Analysis of Milled Wood Lignins (MWLs) Isolated from Sugar Maple (SM) and Hot-Water Extracted Sugar Maple (ESM)
Abstract
: To further elucidate the advantageous effects of hot-water extraction (HWE) on delignification, milled wood lignin (MWL) was isolated from sugar maple (SM) and from hot-water extracted sugar maple (ESM). Ball-milled wood was analyzed for particle size distribution (PSD) before and after dioxane:water (DW) extraction. The MWL samples were analyzed by analytical and spectral methods. The results indicated that the MWL isolated from SM and ESM was mainly released from the middle lamella (ML) and the secondary wall (SW), respectively. The cleavage of dibenzodioxocin (DB) and spirodienone (SD) lignin substructures during HWE is suggested. The removal of lignin during acetone:water (AW) extraction of hot-water extracted wood indicates that including an additional operation in a hardwood HWE-based biorefinery would be beneficial for processing of wood.1. Introduction
Hot-water extraction (HWE) has been recommended as the first step in a hardwood-based biorefinery (model species: sugar maple (SM), Acer saccharum; HWE at 160 °C for 2 h). This method of pretreatment of hardwood chips results in the removal of a significant amount of hemicelluloses/xylan (∼80% of total hemicelluloses), a small amount of easily accessible lignin (up to 15% of the total lignin), some water-soluble inorganics and hydrophilic/phenolic extractives. It has been shown that characteristic mild acidic conditions of this process (final pH ∼3.5, P-factor = 540 (the P-factor as described by Sixta [1]) contributes to high retention of cellulose and minimal degradation of released carbohydrates [2]. After HWE the hydrolyzate can be processed using several operations, for example membrane filtration, and further purified to generate a stream of valuable chemicals (e.g., acetic acid) and hydrolyzed sugars (monosaccharides/xyloses). These monosaccharides can be further used for the production of chemicals, biofuels or bioplastics by biochemical processes [3,4]. Membrane separation (ultrafiltration) of the hydrolyzate has been shown to furnish retentates which vary in lignin content, as high as 90%, depending on the pH and molecular weight limits of the membrane [2]. The lignin present in the retentates account for >60% of the easily accessible fraction dissolved during HWE. Analysis of the retentates by 2D nuclear magnetic resonance spectroscopy (NMR) experiments, namely heteronuclear single quantum coherence spectroscopy (HSQC) NMR have revealed typical lignin substructures such as β-O-4, β-β, and β-5, and benzaldehyde- and cinnamaldehyde-end units. A relatively high syringyl:guaiacyl (S/G) ratio of 2.6 was determined for this lignin by nitrobenzene oxidation (NBO) [2].
Our previous studies have indicated that delignification of hot-water extracted sugar maple (ESM) chips is more efficient than delignification of unextracted (native) SM chips. In kraft pulping, lower H-factors are required to reach approximately the same kappa number of pulp [2]. In organosolv pulping of ESM [for example, in oxygen-pressurized acetone:water (AW) ], delignification is characterized by a higher selectivity, i.e., the pulp produced under the same pulping conditions has a lower amount of residual lignin and a higher amount of retained carbohydrates [5]. ESM is more porous and has a significantly higher content of free phenolic hydroxyl (PhOH) groups [5,6]. An increase in the amount of PhOH groups indicates that HWE results in the cleavage of aryl ether bonds, including β-aryl ether bonds in lignin [7]. This cleavage is also reflected in a reduced presence of the fractions of higher molecular weight lignin in milled wood lignin (MWL) isolated from Eucalyptus hydrothermally treated at P-factor 600 than in MWL of native Eucalyptus [7]. These effects of HWE along with the expected cleavage of lignin-carbohydrate bonds, such as benzyl-aryl ether bonds [8,9] contribute to the documented delignification of ESM. In addition, HWE leads to a higher solubility of extracted wood (ground wood, 30 mesh) in the AW mixture (9:1) [5]. The AW mixture is a solvent characterized by the Hildebrand solubility parameter δ of 11.14 cal1/2 cm−3/2, which is very close to the reference value of the parameter for good lignin solvents (11 cal1/2 cm−3/2) [10]. Extraction of ESM with AW in the standard Soxhlet extraction removes 11.4% of ground wood and results in delignification. In our previous studies it has been shown that after sequential HWE and AW extraction, the delignification level of SM reached ∼50% (based on the original lignin in wood), with >35% of the total lignin in wood being removed during AW step. This may be attributed to lignin with a higher content of free PhOH groups and a relatively lower molecular weight present in ESM than in SM [5,7]. In contrast to these results, AW extraction of unextracted (native) SM leads to the removal of only 3.2% of ground wood, containing low-molecular weight extractives and presumably not lignin [11,12] (Table 1; [5]).
Although the benefits of HWE on the accessibility and reactivity of lignin in hardwoods have been recognized, the modifications of lignin during this process are not fully understood. Recently some important studies in this area have been reported [7,9,13]. Our attempts in this direction include isolation of MWL (in accordance with the Björkman procedure [14] with minor modifications) from AW pre-extracted SM (SM_AW) and ESM [pre-extracted prior to ball-milling with AW, ESM_AW or dichloromethane (DCM), ESM_DCM]. In contrast to AW, the DCM extraction does not lead to lignin removal from ESM (Table 1) in accordance with the Hildebrand solubility parameter of 9.93 cal1/2 cm−3/2 (versus 11 cal1/2 cm−3/2 for lignin) [15]. The ball-milling results were assessed by determination of the particle size distribution (PSD) of ball-milled wood samples. 2D NMR analysis was subsequently performed on isolated fractions of lignin, i.e., lignin-rich AW extract of hot-water extracted wood (AW_ESM) and MWL of native and hot-water extracted wood. The ratio of main lignin units, S/G ratio of wood and MWL samples, and the PhOH group content of MWL were also determined. The S/G ratio was determined by the traditional NBO method, as well as 2D HSQC NMR.
2. Results and Discussion
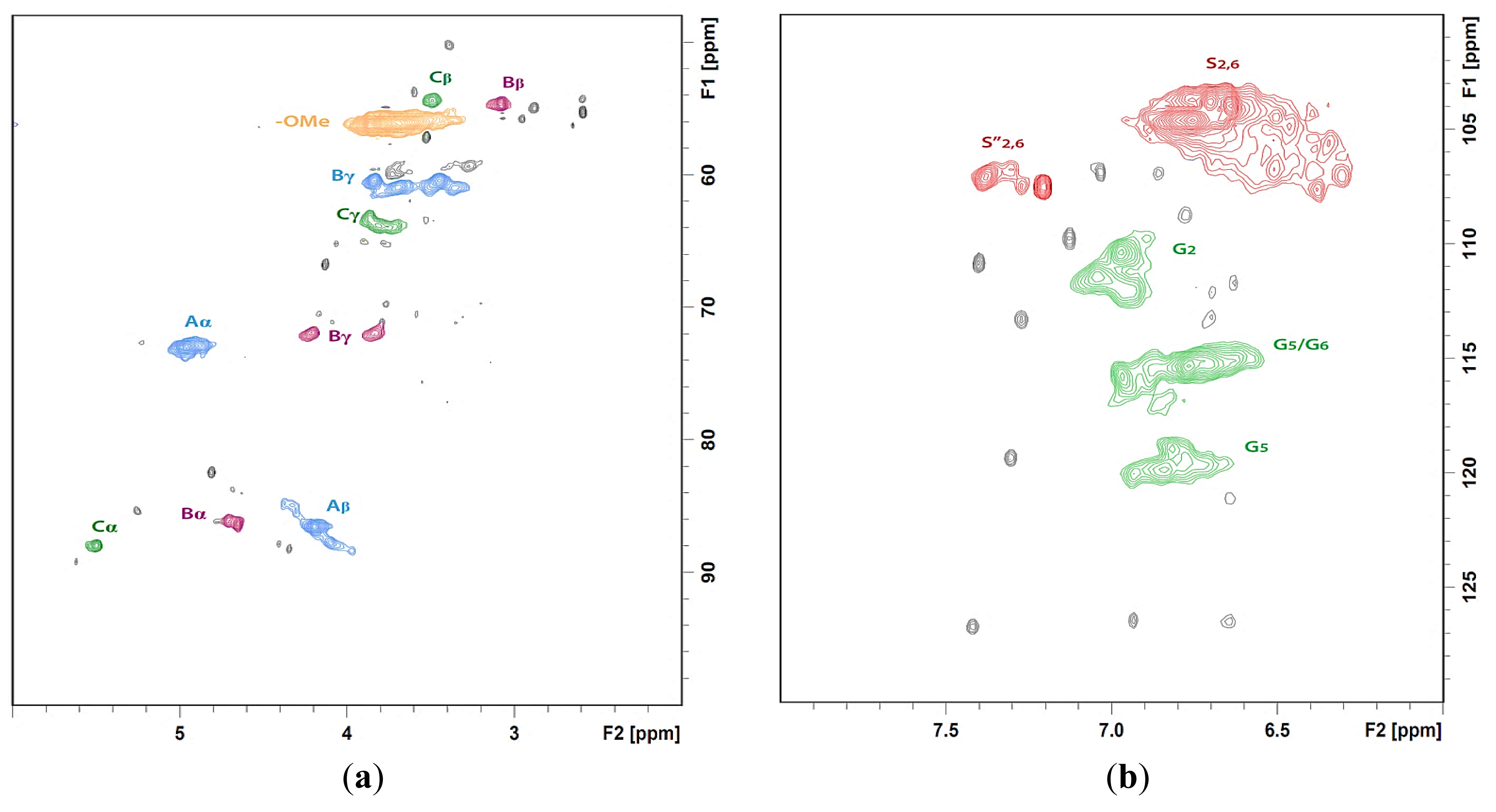
The S/G ratio of SM determined by NBO was found to decrease during HWE (S/G: SM 1.92 versus ESM 1.78). This result corroborates the high S/G ratio (2.6) [2] of lignin dissolved during HWE, which is a fraction of naturally easily accessible lignin. HWE also creates a new vulnerable fraction of lignin, which has been found to be soluble during AW extraction of ESM (AW_ESM) [5]. In this study, the AW_ESM extracts were analyzed by HSQC spectroscopy (Figure 1). Typical lignin side chain correlations (β-O-4, β-β and β-5 substructures as shown in Figure 2) were observed in the sample (Figure 1a). Like the lignin dissolved from SM during HWE, the AW-soluble lignin of ESM is also rich in S-units; with a relatively high S/G ratio of 3.1 which was determined by HSQC (refer to Section 3: Materials and Methods). Therefore, it appears that the two fractions of lignin recovered from two mild consecutive treatments of SM (HWE followed by AW extraction) mostly originate from fibers since in comparison to vessels, fibers contain a higher amount of S-units [16]. These findings demonstrate that the addition of an AW step (e.g., AW washing) to operations in a HWE-based biorefinery would be beneficial to further processing of ESM (or hot-water extracted hardwoods in general) by releasing easily accessible lignin. For the purpose of MWL isolation, based on the aforementioned results, hot-water ESM was extracted in AW (ESM_AW) but also in DCM (ESM_DCM) to retain most of the lignin in wood [5].
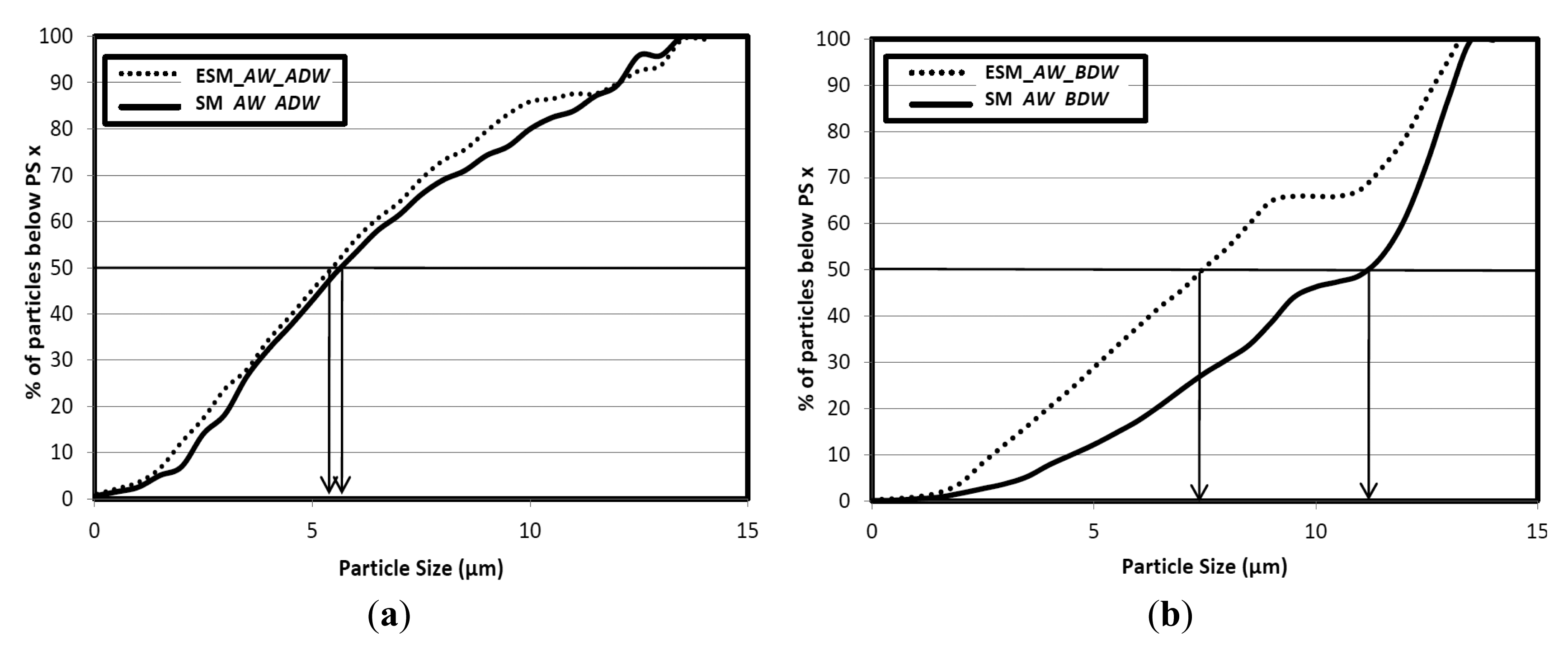
Ball-milling is the most critical operation in the isolation of MWL because the yield of MWL depends strongly on the size of the ball-milled wood particles [14,17–19]. In this study, ball-milling performed under the same conditions produced different results on native SM and on hot-water ESM (Table 2 and Figure 5a). For example, ESM_AW was affected more than SM_AW since ESM_AW particles were smaller than SM_AW particles throughout the whole range of particle sizes as observed by PSD measurements. This result may be attributed to the removal of hemicelluloses (mostly xylans) and an increased porosity of wood after HWE [3,6]; Hot-water extracted wood seems to collapse earlier and results in more efficient milling. For comparative purposes, we observed that ball-milled aspen produced after 2 h of milling at the Forest Products Laboratory (FPL) had lower average particle diameter (Dav) than ball-milled SM and of greater Dav than ball-milled ESM (Table 2).
A relatively low yield of MWL isolated from SM_AW (Table 3) is in accordance with the relatively large particles obtained during ball milling. It may be proposed that this early-released lignin originates mainly from the middle lamella (ML) [18]. The yield of MWLcrude isolated from ESM extracted in DCM (ESM_DCM) increased by more than four times indicating that a significant portion of lignin was released from the secondary wall (SW). This result is not surprising since HWE removes hemicelluloses from the SW, liberating lignin that was bound in lignin carbohydrate complex (LCC) [2,8,9]). In addition, HWE results in lignin of lower molecular weight [7] which contributes to higher extractability of lignin during MWL isolation. Also, based on the results of Klason lignin determination and purification yields, MWLcrude of ESM_DCM was found to be less contaminated by carbohydrates than MWLcrude of SM_AW, corroborating the cleavage of LCC bonds during HWE [8,9]. The yield of MWLcrude of ESM_AW was less than that of MWLcrude of ESM_DCM demonstrating that the AW extraction performed prior to the MWL isolation that provided a removal of easily accessible lignin was not instrumental in increasing accessibility of the remaining lignin. The MWLcrude of ESM_AW was also of a higher content of remaining carbohydrates than the MWLcrude of ESM_DCM. For this reason, a final purification of the MWLcrude was performed only for the ESM_DCM to provide a less contaminated lignin sample for spectral (HSQC) and chemical (S/G, PhOH) analyses.
The results of PSD of native and hot-water extracted ball-milled SM before and after dioxane:water (DW) extraction may be used in considerations of the morphological origin of MWL (Table 2 and Figure 5). It can be seen that even though the mass removal was lower for native SM based on the lower yield of MWL (Table 3), the diameter of the particles decreased more after DW extraction in the case of native SM (SM_AW: D50 = 11.3 μm and Dav = 9.898 μm before DW; D50 = 5.7 μm and Dav = 6.601 μm after DW) than in the case of hot-water ESM (for example, ESM_AW: D50 = 7.5 μm and Dav = 8.4 μm before DW; D50 = 5.4 μm and Dav = 6.2 μm after DW). In accordance with the previously suggested morphological origin of MWL based on the MWL yields, these results also imply that in the case of SM, lignin was extracted mainly from the particle surface (i.e., ML), while in the case of ESM, lignin was extracted from the surface (i.e., ML) and from the inner layers of the particles (i.e., SW).
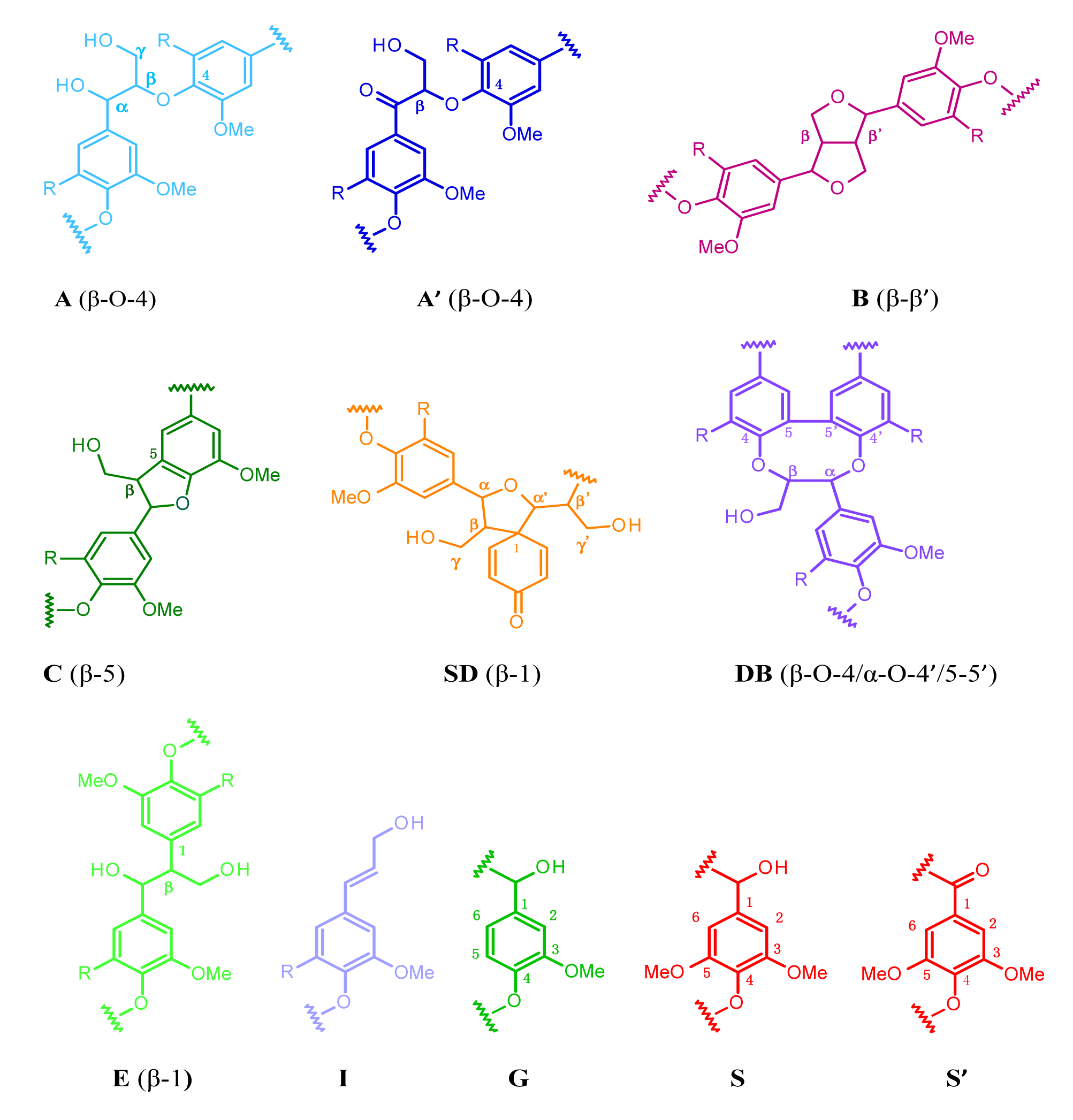
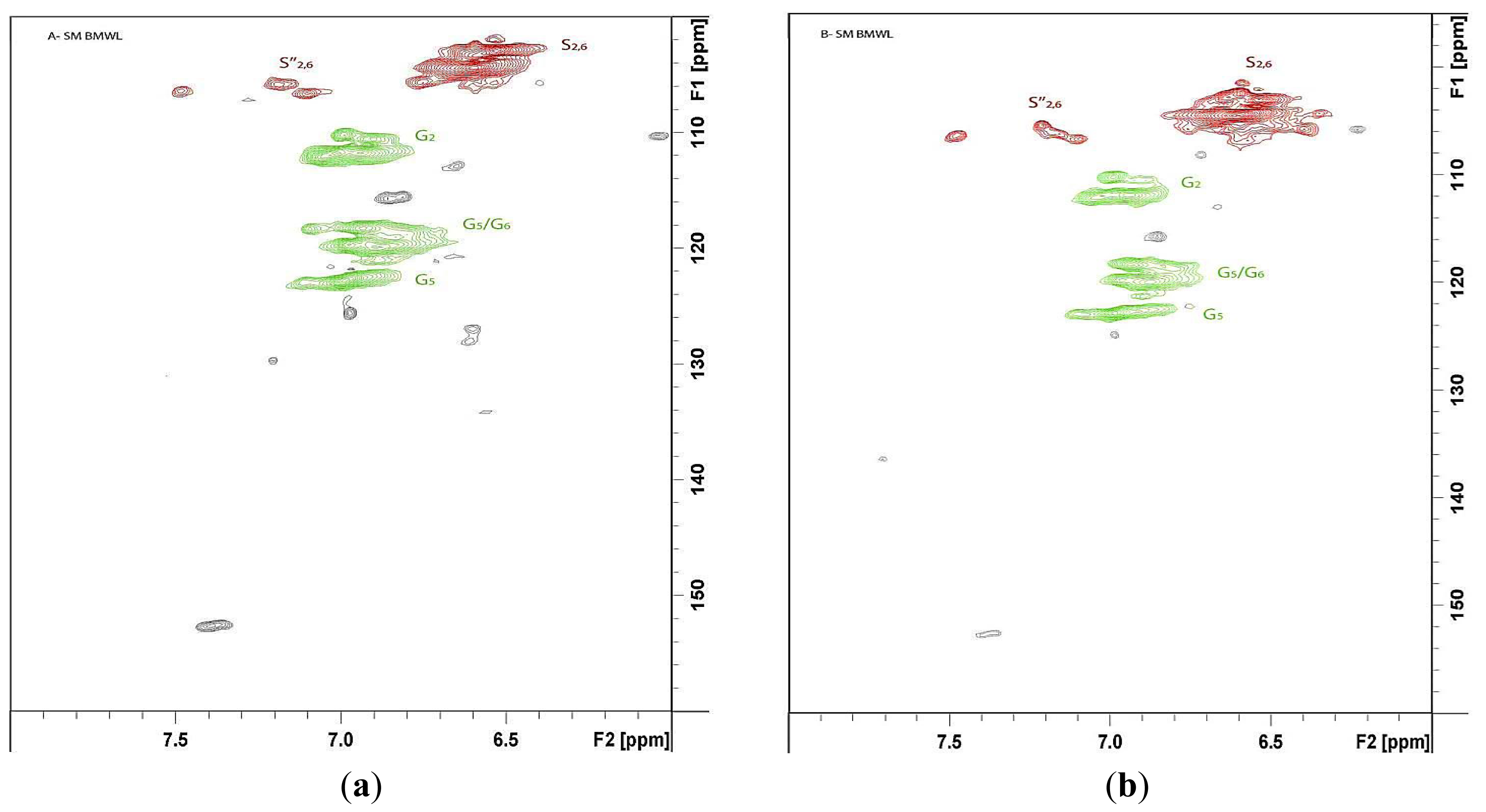
The HSQC spectra were analyzed and volume integration was performed to evaluate the differences in the contents of observed lignin substructures (β-O-4, β-5, β-β, dibenzodioxocin (DB), and spirodienone (SD)) (Figure 3a and Table 4) and main lignin units (S/G ratio) (Figure 3b and Table 4) between MWLpurs isolated from SM_AW and ESM_DCM. The volume integration results indicated the β-O-4 as a main substructure in both MWLpurs. The signals of DB and SD substructures were observed in the HSQC spectrum of MWLpur of SM_AW with estimated amounts equal to 1.9% and 2.8%, respectively. It should be noted that the presence of DB substructure is reported sporadically for hardwood lignin [20,21]; the formation of DB in lignin depends strongly on the amounts of lignin precursors, coniferyl (converts to G-unit) and sinapyl (converts to S-unit) alcohol present during lignification of the cell wall (and hence, strong dependence of the amount of DB on the S/G ratio) [21,22]. The presence of DB substructure is observed more commonly in hardwood lignins formed as a result of genetically-modified biosynthesis [23]. However, based on our NMR results, the DB substructure is also present in the lignin of ML of native SM. The absence of DB and SD correlations from HSQC of MWLpur of hot-water ESM (ESM_DCM; note that HWE is performed in mild acid conditions) may serve as an additional evidence of cleavage of these structures in mild acid conditions [22,24].
The cleavage of SD substructure during HWE supports the presence of β-1 substructure in MWL of ESM_DCM whose Cβ-Hβ correlation is indicated in the HSQC (Figure 4b, Eβ) [24]. A direct comparison of the amounts of lignin substructures in lignin samples is hindered by the different yields of isolated lignin from native and hot-water ESM. Nevertheless, an increase in the amount of β-O-4 substructures in lignin isolated from ESM_DCM is consistent with a higher S/G ratio (confirmed with NBO and HSQC results, Table 4) of this MWL extracted mainly from SW compared to that of MWL extracted mainly from ML and SM_AW. The results of determination of the PhOH group content of MWLpurs indicate an increase of the PhOH groups during HWE and corroborate previous results [5,7] suggesting the cleavage of aryl-ether bonds in lignin and decrease in molecular weight of lignin. It should be noted that the cleavage of DB substructure brings about new PhOH groups into lignin [22], contributing to the observed increase.
Based on the results of this study, a more efficient delignification of hot-water ESM may be attributed to a significant increase in accessibility of lignin in SW resulting from a range of reactions including the cleavage of bonds in lignin and in LCC.
3. Material and Methods
SM obtained from Heiberg Forest (State University of New York College of Environmental Science and Forestry (SUNY-ESF), Syracuse, NY, USA) was debarked and chipped at the Department of Paper and Bioprocess Engineering, SUNY-ESF. The chips were ground in a Wiley mill (30 mesh) prior to the Soxhlet extraction. A 4 L M/K type digester (M/K systems Inc., Peabody, MA, USA), which is basically a pressure cooker capable of circulating liquor while cooking, was used for HWE.
HWE: SM (Acer saccharum) chips were extracted in water in an M/K digester: 200 g wood chips; liquor-to-wood ratio = 8; ramp temperature increase to 160 °C; 2 h at 160 °C; P-factor = 543.7 (P-factor as described by Sixta [1]; final pH = 3.5). Hot-water extracted chips (ESM) were washed twice with water for 15 min at 80 °C. HWE was performed in duplicate with an average yield of extracted chips at 76.3% of original wood (based on oven-dried (OD) wood).
MWL isolation: SM wood chips (SM and ESM) were ground in a Wiley mill (30 mesh, Thomas Scientific, Swedesboro, NJ, USA) and pre-extracted by AW (SM_AW and ESM_AW) and DCM (ESM_DCM) for 8 h or 6 h using a Soxhlet apparatus prior to ball-milling. The ESM_AW extract was further analyzed by HSQC.
After vacuum drying at 40 °C for seven days, samples were transferred into the dry 500 mL stainless steel ball-mill jar (a kind donation of the United States Department of Agriculture (USDA)-FPL, Madison, WI, USA; Schutz-O'Neill Co., Minneapolis, MN, USA; 500 mL; 2/3 of the volume stainless steel balls, d = 6 mm) and the jar was flushed with N2 before sealing. Ball-milling was performed on a vibratory ball mill with two jars in a cold room for 6 h (30-min break at every hour). After separation from the balls, ball-milled wood was treated in toluene (2 mL/g) with magnetic stirring for 24 h (in attempt to collect iron if present). The toluene was removed by evaporation. Lignin was extracted by a modified Björkman procedure (following the procedure described by Ikeda et al. [14], except that lignin dissolution was performed by ultrasonication): ball-milled wood was sonicated (ultrasonic bath Branson 3510, Branson Ultrasonics Corporation, Danbury, CT, USA) in DW (96:4) for 2 h twice (fresh dioxane, Mallinckrodt Chemicals 4937-04, St. Louis, MO, USA; 10 mL DW/g wood). After extraction, DW was evaporated in vacuum (42 °C). The solid residue was dissolved in acetic acid:water (9:1) (20 mL/g) and added gradually into a 10 times larger volume of distilled water (stored in the cold room over-night). The solid precipitate was removed by centrifugation and remaining acetic acid was removed by multiple addition and removal of ethanol (evaporation). The yield of so-called crude MWL (MWLcrude) was recorded and the lignin content was determined following the Klason lignin procedure, including the acid-soluble lignin [25]. Subsequently, the MWLcrude (SM_AW and ESM_DCM) was dissolved in 1,2-dichloroethane:ethanol (2:1, v/v, minimal amount required for dissolution) followed by precipitation in ether (10 times volume of dichloroethane/ethanol) to further purify the samples (MWLpur). The isolation of MWL was performed in duplicates.
MWL acetylation: MWLpur (SM_AW and ESM_DCM) samples were acetylated with acetic anhydride in the presence of pyridine, following procedure reported in literature [26].
PSD of ball-milled samples: PSD testing of ball-milled samples (SM_AW and ESM_AW) before and after DW treatment was performed on a Horiba PSD Analyzer CAPA-700 instrument (Edison, NJ, USA) using centrifugal sedimentation in water at 1500 rpm. In accordance with this method PSD is calculated by combining the Stokes' sedimentation equation with the light absorption-concentration relationship through the Beer's law. The Stokes equation requires that particles be spherical; inspection with a light microscope revealed spherical shape of ball-milled particles. Particle size range of 0–14 μm was found to be sufficient to include all particles. For comparison, ball-milled aspen (Aspen_FPL—source: the FPL) was analyzed too. The results represent average of at least three measurements. Coefficient of variation (cV) of the replicates for each individual particle size was averaged for the entire range of particle sizes, yielding a representation of the average quality of the data.
NMR analysis: All HSQC experiments were acquired at 30 °C on a Bruker AVANCE III 600 spectrometer (600 MHz 1H frequency, Bruker Biospin Corporation, Billerica, MA, USA) equipped with a 5 mm triple resonance z-gradient probe. Data was processed in Topspin v. 3.2 from Bruker Biospin. Acetylated samples (MWLpur) were dissolved in CDCl3 and the central chloroform solvent peak was used as internal reference (δC 77.0 and δH 7.26 ppm). Non-acetylated sample of the AW extract of hot-water ESM (AW_ESM) was dissolved in acetone-d6/D2O (9:1) and the central acetone solvent peak was used as internal reference (δC 29.8 and δH 2.04 ppm).
The distortionless enhancement by polarization transfer (DEPT) edited HSQC experiments had the following parameters: acquired from 11.8 ppm to −0.2 ppm in F2 (1H) using 1024 data points (acquisition time of 71 ms), 220 ppm to 0 ppm in F1(13C) using 400 increments, 32 scans with 1.5 s interscan delay, total acquisition time of 2 h and 48 min, or 5 h and 40 min. Processing used typical squared sine-bell apodization in F2 and F1.
For quantification of the main interunit linkages characteristic of the β-O-4, β-5, β-β, DB, and SD substructures, the integration of the well resolved contours assigned in accordance with the previous data on NMR of lignin [18–21,23,24,27,28] was performed (integral correction factor was equal to 1) [28].
S/G ratio: 1. NBO was performed to determine the S/G ratio of wood and MWLpur samples according to the method of Bose et al. [29]. 2. 2D 13C-1H correlation HSQC NMR spectroscopy data was also used to determine the S/G ratio of MWLpur and AW_ESM samples based on the integral volume of S2/6 and G2 from the aromatic region (G2 integral value was multipled by two) [28].
PhOH group content: the determination of the PhOH group content in MWLpur samples was performed in accordance with the UV ionization difference method recommended by Gärtner [30].
4. Conclusions
Based on the comparison of the ball-milling of SM and hot-water ESM and the yield and properties of milled-wood lignin isolated from ball-milled wood samples, a more efficient delignification of hot-water ESM may be attributed to a significant increase in the accessibility of lignin in SW. The cleavage of DB and SD lignin substructures taking place during HWE is observed. After HWE, SM contains a fraction of lignin easily accessible and soluble in AW (9:1). To facilitate subsequent processing of wood, this lignin should be removed from hot-water extracted wood in a separate operation in a HWE based hardwood biorefinery.
Acknowledgments
This research work was financially supported by the USDA-McIntire-Stennis “Enhancing production of uniform high purity lignin” and National Science Foundation (NSF): “Technology Enhancement of Hot Water Extraction” research grants. We wish to extend our sincere gratitude to Thomas E. Amidon for his guidance and leadership in pioneering the field of HWE technology at SUNY-ESF. We also wish to thank our colleagues Sally Ralph and Alan Rudie at the FPL in Madison, WI for their continued support and assistance. Finally, we would like to thank David J. Kiemle (Department of Chemistry, SUNY-ESF) and Aditi Nagardeolekar for assistance on NMR and UV-Vis experiments respectively.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sixta, H. Handbook of Pulp; Wiley-VCH: Weinheim, Germany, 2006. [Google Scholar]
- Ye, P.X.; Cheng, L.; Ma, H.; Bujanovic, B.; Goundalkar, M.; Amidon, T.E. Biorefinery with Water. In The Role of Green Chemistry in Biomass Processing and Conversion; Xie, H., Gathergood, N., Eds.; Wiley John & Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 135–180. [Google Scholar]
- Amidon, T.E.; Wood, C.D.; Shupe, A.M.; Wang, Y.; Graves, M.; Liu, S. Biorefinery: Conversion of woody biomass to chemicals, energy and materials. J. Biobased Mater. Bioenergy 2008, 2, 100–120. [Google Scholar]
- Amidon, T.E.; Bujanovic, B.; Liu, S.; Howard, J.R. Commercializing biorefinery technology: A case for the multi-product pathway to a viable biorefinery. Forests 2011, 2, 929–947. [Google Scholar]
- Gong, C.; Goundalkar, M.J.; Bujanovic, B.; Amidon, T.E. Evaluation of different sulfur-free delignification methods for hot-water extracted hardwood. J. Wood Chem. Technol. 2012, 32, 93–104. [Google Scholar]
- Bolton, T.S.; Francis, R.C.; Amidon, T.E. Chemical and physical changes due to acidolysis of chips ahead of alkaline pulping. Proceedings of the 14th International Symposium on Wood, Fiber, and Pulping Chemistry (ISWFPC), Durban, South Africa, 25–28 June 2007.
- Leschinsky, M.; Zuckerstätter, G.; Weber, H.K.; Patt, R.; Sixta, H. Effect of autohydrolysis of Eucalyptus globulus wood on lignin structure. Part 1: Comparison of different lignin fractions formed during water prehydrolysis. Holzforschung 2008, 62, 645–652. [Google Scholar]
- Guo, X.-P.; Lai, Y.-Z. Variation of softwood and hardwood lignin in the reactivity of aryl ether units under acidic conditions. Proceedings of the 14th International Symposium on Wood, Fiber, and Pulping Chemistry (ISWFPC), Durban, South Africa, 25–28 June 2007.
- Li, J.; Gellerstedt, G. Improved lignin properties and reactivity by modifications in the autohydrolysis process of aspen wood. Ind. Crops Prod. 2008, 27, 175–181. [Google Scholar]
- Schuerch, C. The solvent properties of liquids and their relation to the solubility, swelling, isolation and fractionation of lignin. J. Am. Chem. Soc. 1952, 74, 5061–5067. [Google Scholar]
- Holmbom, B. Extractives. In Analytical Methods in Wood Chemistry, Pulping & Papermaking; Sjostrom, A., Ed.; Springer: Berlin/Heidelberg, Germany, 1999; pp. 125–148. [Google Scholar]
- Fasching, M.; Schröder, P.; Wollboldt, R.P.; Weber, H.K.; Sixta, H. A new and facile method for isolation of lignin from wood based on complete wood dissolution. Holzforschung 2008, 62, 15–23. [Google Scholar]
- Leschinsky, M.; Zuckerstätter, G.; Weber, H.K.; Patt, R.; Sixta, H. Effect of autohydrolysis of Eucalyptus globulus wood on lignin structure. Part 2: Influence of autohydrolysis intensity. Holzforschung 2008, 62, 653–658. [Google Scholar]
- Ikeda, T.; Holtman, K.; Kadla, J.F.; Chang, H.-M.; Jameel, H. Studies on the effect of ball milling on lignin structure using a modified DFRC method. J. Agric. Food Chem. 2002, 50, 129–135. [Google Scholar]
- Riddick, J.A.; Bunger, W.B.; Sakano, T.K. Organic Solvents: Physical Properties and Methods of Purification. In Techniques of Chemistry, 4th ed.; Weissberger, A., Ed.; John Wiley & Sons: New York, NY, USA, 1986; Volume II. [Google Scholar]
- Joseleau, J.-P.; Faix, O.; Kuroda, K.-I.; Ruel, K. A polyclonal antibody directed against syringylpropane epitopes of native lignins. C. R. Biol. 2004, 327, 809–815. [Google Scholar]
- Björkman, A. Studies of finely divided wood. Part 5. The effect of ball-milling. Svensk. Paperstidn. 1957, 60, 329–335. [Google Scholar]
- Yuan, T.-Q.; Sun, S.-N.; Xu, F.; Sun, R.-C. Structural characterization of lignin from triploid of Populus tomentosa Carr. J. Agric. Food Chem. 2011, 59, 6605–6615. [Google Scholar]
- Yuan, T.-Q.; Sun, S.-N.; Xu, F.; Sun, R.-C. Characterization of lignin structures and lignin-carbohydrate complex (LCC) linkages by quantitative 13C and 2D HSQC NMR spectroscopy. J. Agric. Food Chem. 2011, 59, 10604–10614. [Google Scholar]
- Capanema, A.E.; Balakshin, M.Y.; Kadla, J.F. Quantitative characterization of a hardwood milled wood lignin by nuclear magnetic resonance spectroscopy. J. Agric. Food Chem. 2005, 53, 9639–9649. [Google Scholar]
- Sette, M.; Wechselberger, R.; Crestini, C. Elucidation of lignin structure by quantitative NMR. Chem. Eur. J. 2011, 17, 9529–9535. [Google Scholar]
- Brunow, G.; Kilpeläinen, I.; Sipilä, J.; Syrjanen, K.; Karhunen, P.; Setälä, H.; Rummakko, P. Oxidative Coupling of Phenols and the Biosynthesis of Lignin. In Lignin and Lignan Biosynthesis; Lewis, N.G., Sarkanen, S., Eds.; American Chemical Society: Washington, DC, USA, 1998; Chapter 10; pp. 131–147. [Google Scholar]
- Ralph, J.; Lapierre, C.; Lu, F.; Marita, J.M.; Pilate, G.; Van Doorsselaere, J.; Boerjan, W.; Jouanin, L. NMR evidence for benzodioxane structures resulting from incorporation of 5-hydroxyconiferyl alcohol into lignins of O-methyltransferase-deficient poplars. J. Agric. Food Chem. 2001, 49, 86–91. [Google Scholar]
- Zhang, L.; Gellerstedt, G. NMR studies on the occurrence of spirodienone structures in lignins. J. Wood. Chem. Technol. 2006, 26, 65–67. [Google Scholar]
- Dence, C.W. The Determination of Lignin. In Methods in Lignin Chemistry; Lin, S.Y., Dence, C.W., Eds.; Springer-Verlag: Berlin/Heidelberg, Germany, 1992; pp. 33–61. [Google Scholar]
- Lundquist, K. Proton (1H) NMR Spectroscopy. In Methods in Lignin Chemistry; Lin, S.Y., Dence, C.W., Eds.; Springer-Verlag: Berlin/Heidelberg, Germany, 1992; pp. 242–249. [Google Scholar]
- Ralph, S.A.; Ralph, J.; Landucci, L.L. NMR Database of Lignin and Cell Wall Model Compounds. Available online: http://ars.usda.gov/Services/docs.htm?docid=10491_Retrieved (accessed on 16 November 2013).
- Stewart, J.J.; Akiyama, T.; Chapple, C.; Ralph, J.; Mansfield, S.D. The effects on lignin structure of overexpression of ferulate 5-hydroxylase in hybrid poplar. Plant. Physiol. 2009, 150, 621–635. [Google Scholar]
- Bose, S.K.; Francis, R.C.; Govender, M.; Bush, T.; Spark, A. Lignin content versus syringyl to guaiacyl ratio amongst poplars. Bioresour. Technol. 2009, 100, 1628–1633. [Google Scholar]
- Gärtner, A.; Gellerstedt, G.; Tamminen, T. Determination of phenolic hydroxyl groups in residual lignin using a modified UV-method. Nord. Pulp Pap. Res. J. 1999, 14, 163–170. [Google Scholar]
Nomenclature
| HWE | Hot-water extraction |
| SM | Sugar maple (Acer saccharum) |
| ESM | Hot-water extracted sugar maple |
| MWL | Milled wood lignin |
| PSD | Particle size distribution |
| PhOH | Free phenolic hydroxyl group content |
| AW | 9:1 Acetone:water |
| DCM | Dichloromethane (methylene chloride) |
| DW | 96:4 Dioxane:water |
| SM_AW | SM pre-extracted with AW |
| ESM_AW | Hot-water ESM that is further pre-extracted with AW |
| ESM_DC | Hot-water ESM that is further pre-extracted with DCM |
| AW_ESM | Extract obtained during AW pre-extraction of ESM |
| S/G | Ratio of syringyl:guaiacyl units |
| NBO | Nitrobenzene oxidation |
| MWLcrude | MWL not treated with 1,2-dichloroethane:ethanol (2:1); crude MWL |
| MWLpur | MWL treated with 1,2-dicholoroethane:ethanol (2:1); purified MWL |
| DB | Dibenzodioxocin lignin substructure |
| SD | Spirodienone lignin substructure |
| DEPT | Distortionless Enhancement by Polarization Transfer |
| HSQC | Heteronuclear Single Quantum Coherence Spectroscopy |

| Wood samples | Extractives in (% OD SM/ESM wood) | Total lignin (Klason + acid insoluble) | ||
|---|---|---|---|---|
| AW | DCM | Wood | % OD wood | |
| SM | 3.2 | - | SM_AW | 22.0 |
| ESM | 11.4 | - | ESM_AW | 16.4 |
| ESM | - | 3.9 | ESM_DCM | 25.9 |
| Ball-milled wood | SM_AW | ESM_AW | Aspen_FPL | |||
|---|---|---|---|---|---|---|
| Dav, μm | cV | Dav, μm | cV | Dav, μm | cV | |
| Before DW | 9.898 | 0.371 | 8.438 | 0.183 | 8.751 | 0.307 |
| After DW | 6.601 | 0.319 | 6.173 | 0.398 | - | - |
| Wood sample | Crude MWL yield% wood | Lignin in crude MWL% MWL | Lignin in MWL% Lignin in Wood | Purification yield% crude MWL | ||
|---|---|---|---|---|---|---|
| Klason | Acid soluble | Total | ||||
| SM_AW | 1.98 | 78.60 | 2.60 | 81.20 | 7.30 | 85.6 |
| ESM_DCM | 9.74 | 85.30 | 2.64 | 87.94 | 33.10 | 91.3 |
| ESM_AW | 5.44 | 80.49 | 2.59 | 83.08 | 27.55 | n.d. |
| MWLpur | % of total bonds in lignin | PhOH | S/G | |||||
|---|---|---|---|---|---|---|---|---|
| β-O-4 | β-β | β-5 | DB | SD | mmol/g lignin | 2D NMR | NBO | |
| SM_AW | 68.7 | 18.9 | 7.8 | 1.9 | 2.8 | 0.632 | 1.04 | 0.85 |
| ESM_DCM | 78.4 | 15.4 | 6.2 | - | - | 2.670 | 2.02 | 1.36 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Goundalkar, M.J.; Corbett, D.B.; Bujanovic, B.M. Comparative Analysis of Milled Wood Lignins (MWLs) Isolated from Sugar Maple (SM) and Hot-Water Extracted Sugar Maple (ESM). Energies 2014, 7, 1363-1375. https://doi.org/10.3390/en7031363
Goundalkar MJ, Corbett DB, Bujanovic BM. Comparative Analysis of Milled Wood Lignins (MWLs) Isolated from Sugar Maple (SM) and Hot-Water Extracted Sugar Maple (ESM). Energies. 2014; 7(3):1363-1375. https://doi.org/10.3390/en7031363
Chicago/Turabian StyleGoundalkar, Mangesh J., Derek B. Corbett, and Biljana M. Bujanovic. 2014. "Comparative Analysis of Milled Wood Lignins (MWLs) Isolated from Sugar Maple (SM) and Hot-Water Extracted Sugar Maple (ESM)" Energies 7, no. 3: 1363-1375. https://doi.org/10.3390/en7031363




