Air Gasification of Agricultural Waste in a Fluidized Bed Gasifier: Hydrogen Production Performance
Abstract
:1. Introduction
| Oxidation reaction: | C + O2 → CO2 | (1) |
| C + ½ O2 → CO | (2) | |
| Boudouard: | C + CO2 → 2CO | (3) |
| Water gas: | C + H2O → CO + H2 | (4) |
| Methanation: | C + 2H2 → CH4 | (5) |
| Water gas shift: | CO + 2H2O → CO2 + H2 | (6) |
2. Experimental Section
2.1. Experimental Set Up
| Palm kernel shell | Coconut shell | |
|---|---|---|
| Moisture content (% wet basis) | 7.96 | 4.89 |
| Proximate analysis (wt % dry basis) | ||
| Volatile matter | 72.47 | 30.62 |
| Fixed carbon | 18.56 | 26.41 |
| Ash | 8.97 | 42.98 |
| Ultimate analysis (wt% dry basis) | ||
| Carbon | 51.63 | 45.24 |
| Hydrogen | 5.52 | 5.04 |
| Nitrogen | 1.89 | 1.46 |
| Sulfur | 0.05 | 0.06 |
| Oxygen (by different) | 40.91 | 48.2 |
| Higher Heating Values (HHV) (MJ/kg) | 22.97 | 16.07 |
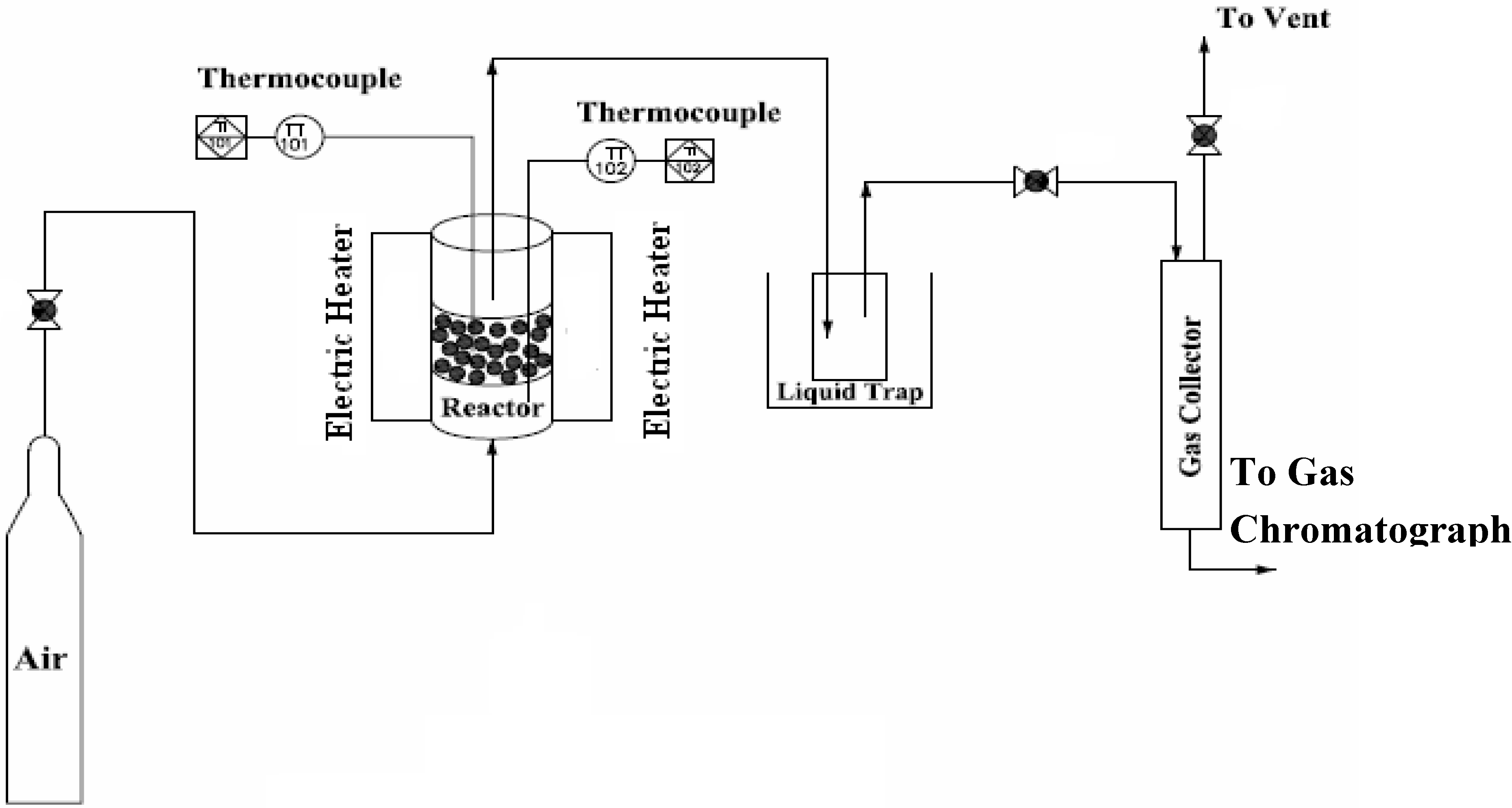
2.3. Experimental Procedures
3. Results and Discussion
3.1. The Effect of Temperature
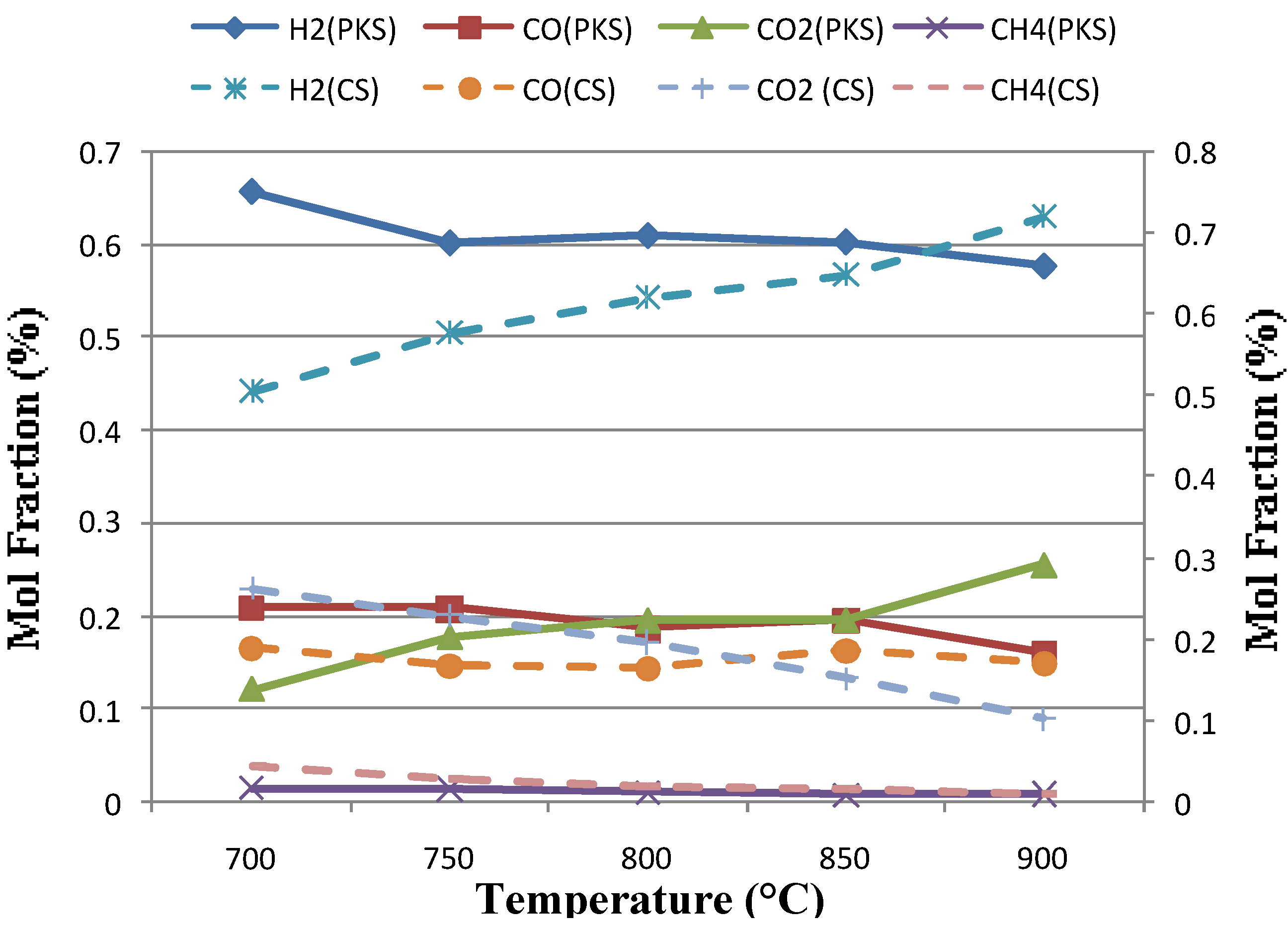
| Reactor temperature (°C) | 700 | 750 | 800 | 850 | 900 |
|---|---|---|---|---|---|
| Gas yield (mol/kg biomass) | |||||
| a) coconut shell | 0.91 | 1.81 | 1.42 | 2.12 | 2.95 |
| b) palm kernel shell | 2.36 | 3.46 | 4.37 | 5.25 | 5.90 |
| Gas LHV (kJ/NM3) | |||||
| a) coconut shell | 930 | 1033 | 1174 | 1874 | 2384 |
| b) palm kernel shell | 2783 | 3276 | 3711 | 4451 | 3720 |
3.2. Effect of Fluidization Ratio
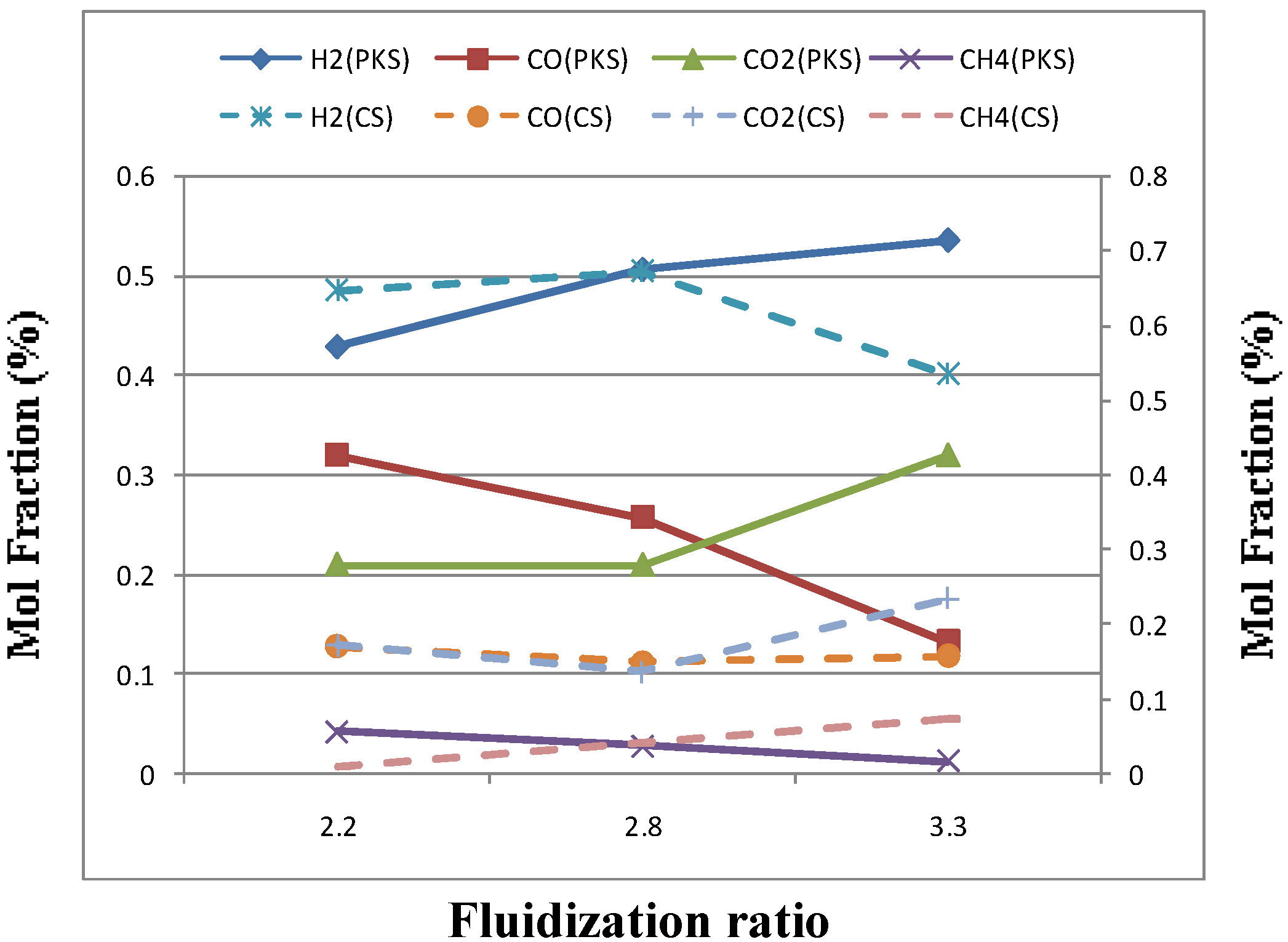
| Fluidization ratio (μ/ μmf) | 2.20 | 2.80 | 3.33 |
|---|---|---|---|
| Gas yield (mol/kg biomass) | |||
| a) coconut shell | 0.60 | 0.32 | 0.26 |
| b) palm kernel shell | 3.15 | 3.57 | 4.42 |
| Gas LHV (kJ/NM3) | |||
| a) coconut shell | 484 | 292 | 279 |
| b) palm kernel shell | 4607 | 1756 | 1961 |
3.3. Effect of Static Bed Height
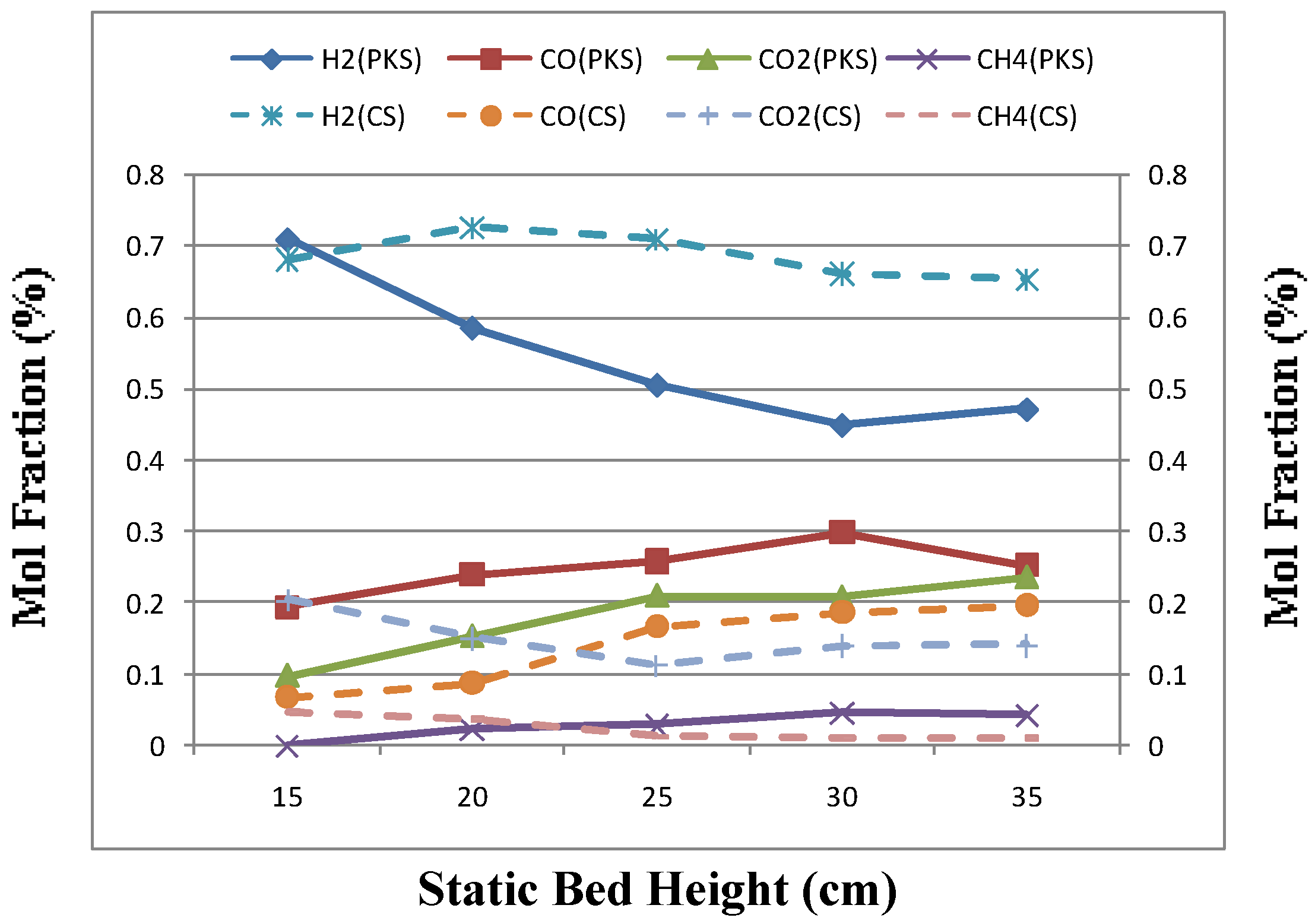
| Bed Height (mm) | 15 | 20 | 25 | 30 | 35 |
|---|---|---|---|---|---|
| Gas yield (mol/kg biomass) | |||||
| a) coconut shell | 0.49 | 0.54 | 1.21 | 1.38 | 1.19 |
| b) palm kernel shell | 3.43 | 3.41 | 3.57 | 3.95 | 3.97 |
| Gas LHV (kJ/NM3) | |||||
| a) coconut shell | 323 | 369 | 995 | 1204 | 1067 |
| b) palm kernel shell | 2877 | 3758 | 3427 | 5578 | 4887 |
3.4. Effect of Equivalence ratio (ER)

| Bed Height (mm) | 0.15 | 0.20 | 0.25 | 0.30 | 0.45 |
|---|---|---|---|---|---|
| Gas yield (mol/kg biomass) | |||||
| a) coconut shell | 0.31 | 0.41 | 1.08 | 0.64 | 0.25 |
| b) palm kernel shell | 2.7 | 4.02 | 2.81 | 1.86 | 1.08 |
| Gas LHV (kJ/NM3) | |||||
| a) coconut shell | 117 | 190 | 473 | 261 | 85 |
| b) palm kernel shell | 2286 | 2863 | 3467 | 1812 | 1482 |
4. Conclusions
Acknowledgements
References
- Chuah, T.G.; Wan Azlina, A.G.K.; Robiah, Y.; Omar, R. Biomass as the renewable energy sources in Malaysia. Int. J. Green Ener. 2006, 3, 323–346. [Google Scholar] [CrossRef]
- Bridgwater, A.V. Renewable fuels and chemicals by thermal processing of biomass. J. Chem. Eng. 2003, 91, 87–102. [Google Scholar] [CrossRef]
- Lapuerta, M.; Hernandez, J.J.; Pozoz, A.; Lopez, J. Gasification and co-gasification of biomass wastes:effect of the biomass origin and the gasifier operating conditions. Fuel Process. Technol. 2008. [Google Scholar] [CrossRef]
- Xiao, R.; Jin, B.; Zhou, H.; Zhong, Z.; Zhang, M. Air gasification of polyproplynen plastic waste in fluidized bed gasifier. Energy Convers. Manage. 2007, 48, 778–786. [Google Scholar] [CrossRef]
- Gonzalez, J.F.; Roman, S.; Bragado, D.; Calderon, M. Investigation on the reactions influencing biomass air and air/steam gasification for hydrogen production. Fuel Process. Technol. 2008. [Google Scholar] [CrossRef]
- Lv, P.M.; Xiong, Z.; Chang, H.J.; Chen, Y.; Zhu, J.X. An experimental study on biomass air-steam gasification in a fluidized bed. Bioresour. Technol. 2004, 95, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Lim, M.T.; Alimuddin, Z. Bubbling fluidized bed biomass gasification—Performance, process findings and energy analysis. Renewable Energy 2008, 33, 2339–2343. [Google Scholar] [CrossRef]
- Hoque, M.M.; Bhattacharya, S.C. Fuel characteristics of gasified coconut shell in a fluidized and a spouted bed reactor. Energy 2001, 26, 101–110. [Google Scholar] [CrossRef]
- Ravelli, S.; Perdichizzi, A.; Barigozzi, G. Descriptions, application and numerical modelling of bubbling fluiidzed bed combustion in waste-to-energy plants. Prog. Energy Combust. Sci. 2008, 34, 224–253. [Google Scholar] [CrossRef]
- Dermibas, A. Gaseous products from biomass by pyrolysis and gasification : effects of catalyst on hydrogen yield. Energy Convers. Manage. 2002, 43, 897–909. [Google Scholar]
- Warnecke, R. Gasification of biomasss: comparison of fixed bed and fluidized bed. Biomass Bioenergy 2000, 18, 489–497. [Google Scholar] [CrossRef]
- Gil, J.; Corella, J.; Aznar, M.P.; Caballero, M.A. Biomass gasification in atmospheric and bubbling fluidized bed: effect of type gasifying agent on the product distribution. Biomass Bioenergy 1999, 17, 389–403. [Google Scholar] [CrossRef]
- Andre, R.N.; Pinto, F.; Franco, C.; Dias, M.; Gulyurtlu, I.; Matos, M.A.A.; Cabrita, I. Fluidised bed co-gasification of coal and olive oil industry wastes. Fuel 2005, 84, 1635–1644. [Google Scholar] [CrossRef]
- Dogru, M. gasification of hazelnut shells. Energy 2000, 27, 415–427. [Google Scholar] [CrossRef]
- Stiegel, G.J.; Ramezan, M. Hydrogen from coal gasification: an economical pathway to a sustainable future. Int. J. Coal Geol. 2006, 65, 173–190. [Google Scholar] [CrossRef]
- Van Der Drift, A.; Doorn, J.V.; Vermulen, J.W. Ten residual biomass fuels for circulating fluidized bed gasification. Biomass Bioenergy 2001, 20, 45–56. [Google Scholar] [CrossRef]
- Tillman, D.A. Biomass Cofiring: The Technology, The experience, The Combustion Consequences. Biomass Bioenergy 2000, 19, 365–384. [Google Scholar] [CrossRef]
- Mansary, K.G.; Ghaly, A.E.; Al –Taweel, A.M.; hamdullahpur, F.; Ugursal, V.I. Air gasification of rice husk in a dual distributor type fluidized bed gasifier. Biomass Bioenergy 1999, 4, 315–332. [Google Scholar] [CrossRef]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wan Ab Karim Ghani, W.A.; Moghadam, R.A.; Salleh, M.A.M.; Alias, A.B. Air Gasification of Agricultural Waste in a Fluidized Bed Gasifier: Hydrogen Production Performance. Energies 2009, 2, 258-268. https://doi.org/10.3390/en20200258
Wan Ab Karim Ghani WA, Moghadam RA, Salleh MAM, Alias AB. Air Gasification of Agricultural Waste in a Fluidized Bed Gasifier: Hydrogen Production Performance. Energies. 2009; 2(2):258-268. https://doi.org/10.3390/en20200258
Chicago/Turabian StyleWan Ab Karim Ghani, W. A., Reza Alipour Moghadam, M. A. Mohd Salleh, and A. B. Alias. 2009. "Air Gasification of Agricultural Waste in a Fluidized Bed Gasifier: Hydrogen Production Performance" Energies 2, no. 2: 258-268. https://doi.org/10.3390/en20200258
APA StyleWan Ab Karim Ghani, W. A., Moghadam, R. A., Salleh, M. A. M., & Alias, A. B. (2009). Air Gasification of Agricultural Waste in a Fluidized Bed Gasifier: Hydrogen Production Performance. Energies, 2(2), 258-268. https://doi.org/10.3390/en20200258




