3.1. Fourier Transform Infrared (FTIR) Spectroscopy Analysis of Transformer Paper
During operation, the oil–paper composite insulation within a transformer experiences an aging process that is caused by a combination of thermal, electrical, and mechanical stresses. Thermal stress in oil–paper composite insulation is the most significant factor in insulation aging [
26]. Due to thermal stress, the degradation process of insulation materials within transformers leads to changes in the chemical structure of the insulation material. A chemical reaction takes place during the degradation process.
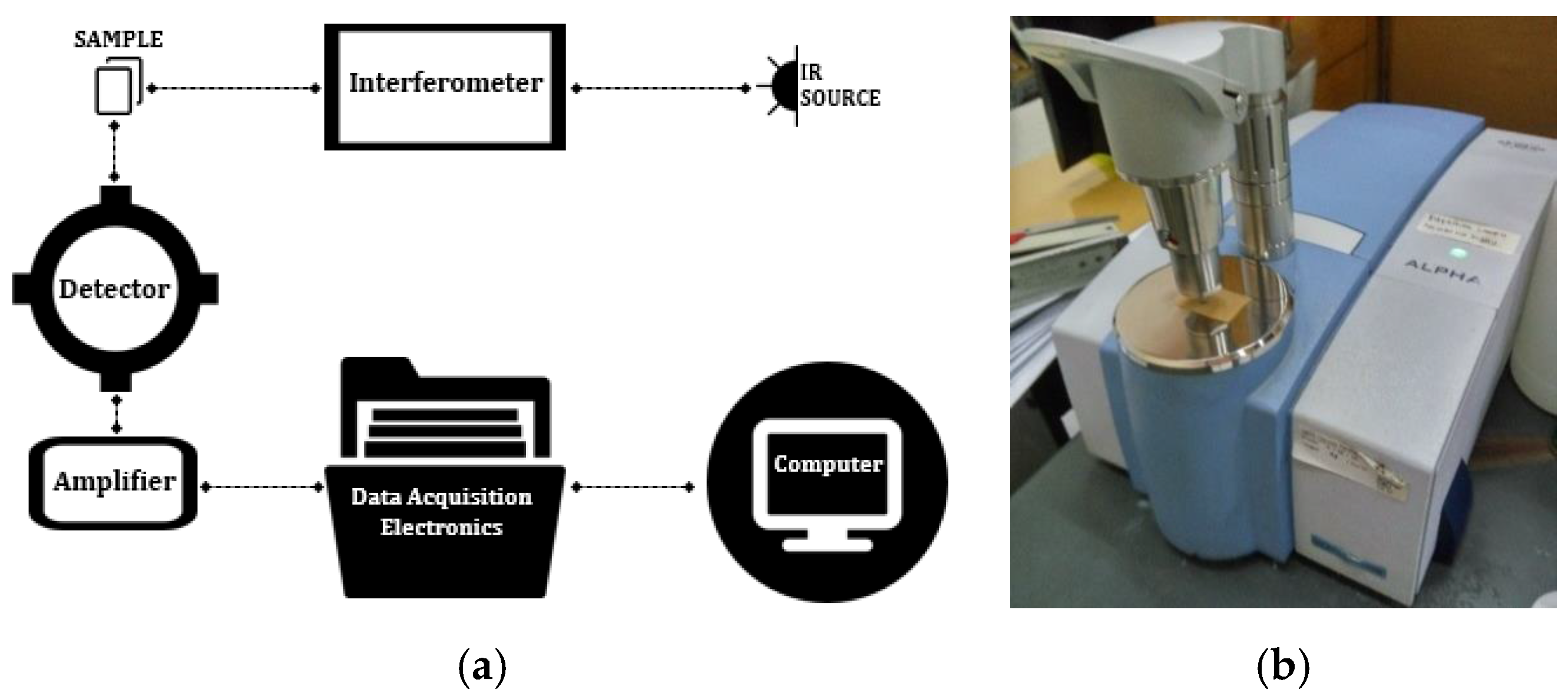
Table 2 shows the visual appearances of paper surfaces aged in mineral oil. The color of the paper surface becomes darker with aging. Solid dielectrics become darker as the aging time and temperature increase. It can be stated that an old transformer might have darker solid dielectrics compared to a new one. When higher temperatures were applied, the insulation paper became darker faster. The visual appearance of the paper sample changed with aging, which indicates changes in the microstructure and chemical structure of the cellulose paper. The chemical structure of the transformer paper aged in mineral oil can be identified by using Fourier transform infrared (FTIR) spectroscopy. FTIR was used to identify the functional groups of the investigated material, which is shown by the intensity of peak absorbance.
Figure 2 shows the FTIR spectra of transformer paper aged in mineral oil for 1008 h.
Table 3 and
Table 4 show the peak absorbance values of the paper samples. There is a significant difference between FTIR spectra, especially the peak absorbance value of the infrared spectra of the paper sample at aging temperatures of 120 °C and 150 °C of the same aging times. This confirms that thermal stress influences the chemical structure of paper samples.
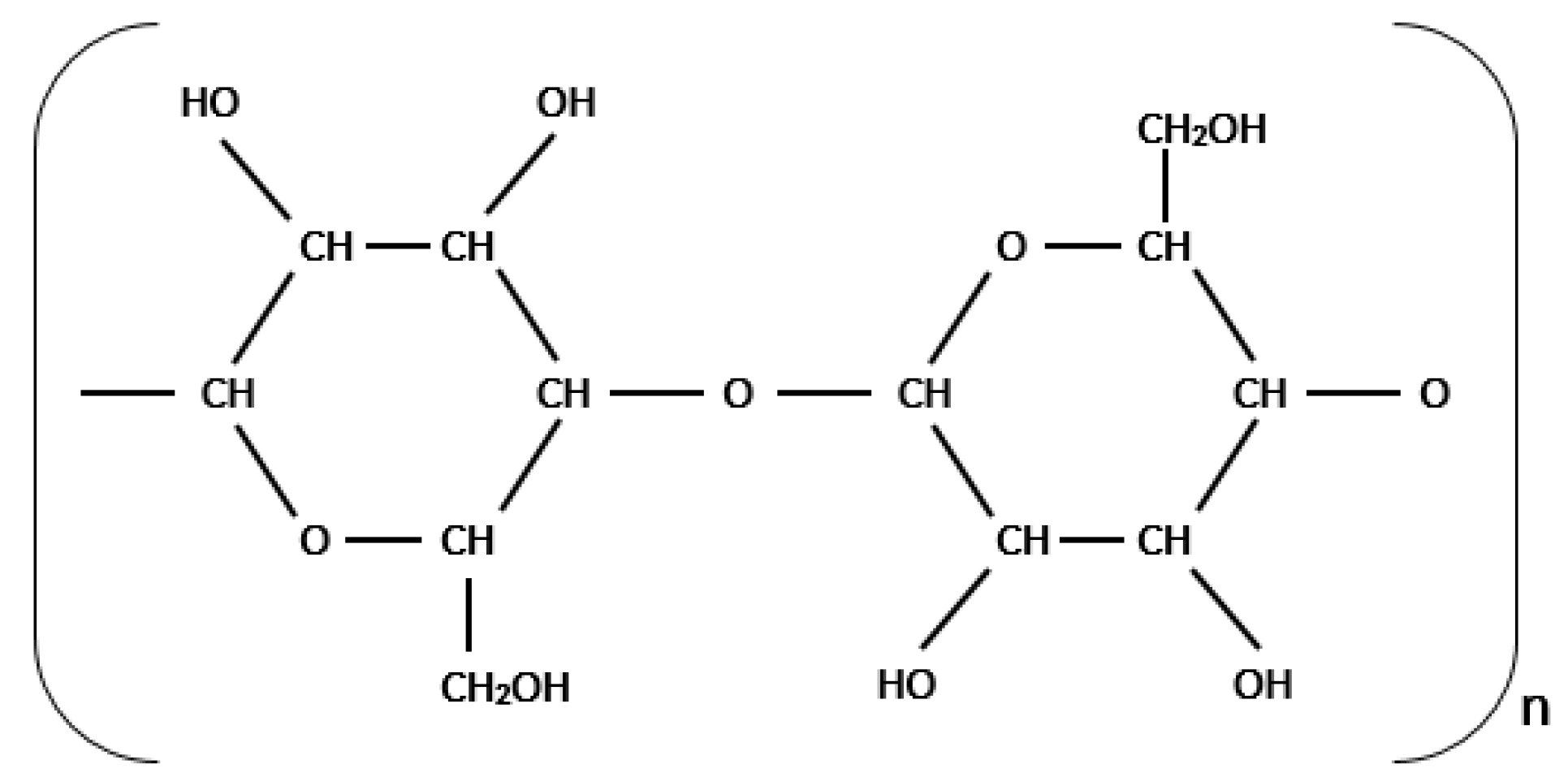
From
Figure 2, the FTIR spectrum of new cellulose paper shows absorbance peaks at wavenumbers 3700–3000 cm
−1 (which is an O–H functional group), at wavenumbers 3000–2700 cm
−1 (which is a C–H functional group), and at wavenumbers 1500–900 cm
−1 (which is a C–O functional group). These functional groups identified are consistent with the basic structure of cellulose as shown in
Figure 3.
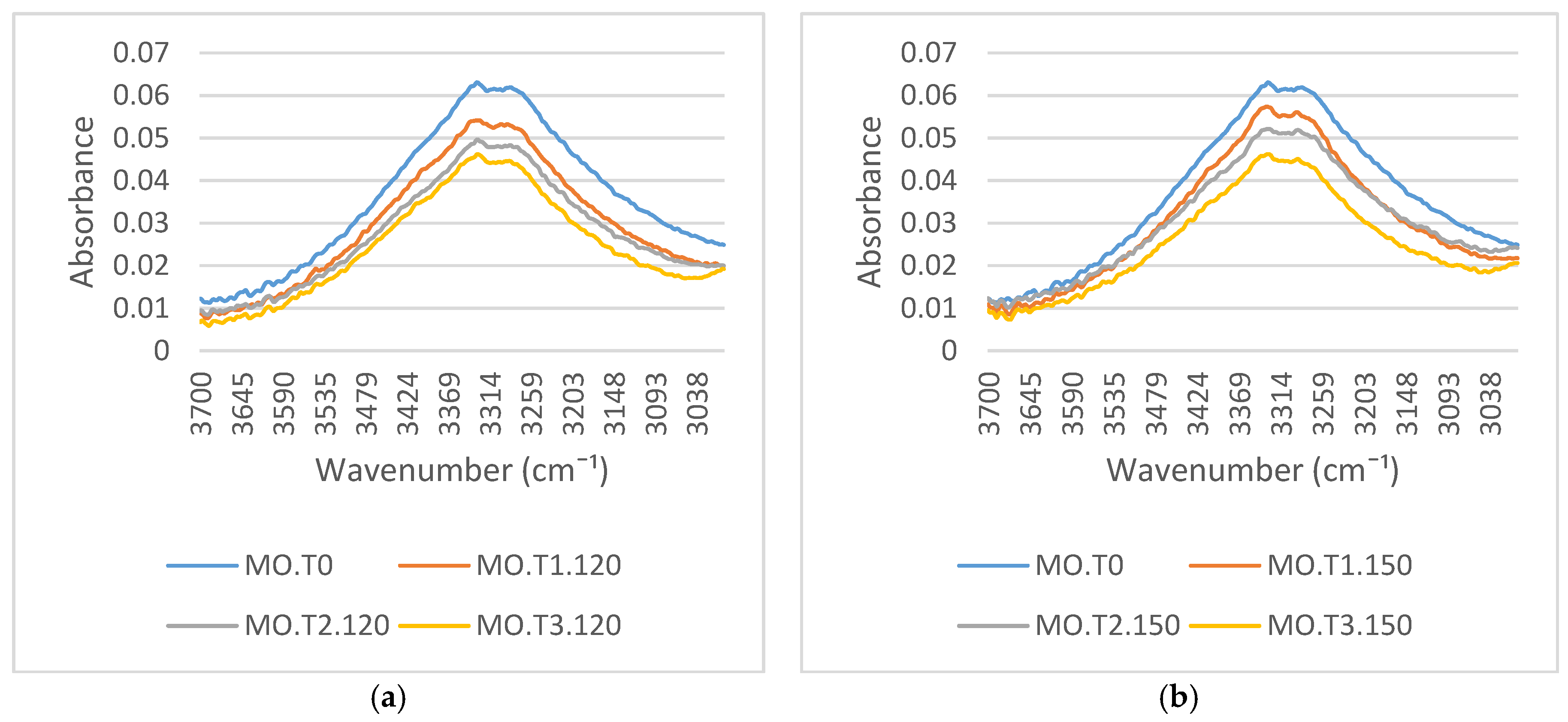
Figure 4a,b presents the FTIR spectra of transformer paper aged in mineral oil at aging temperatures of 120 °C and 150 °C. These figures relate to the hydroxyl groups of the spectra. The peak absorbance at 3329 cm
−1 represents the O–H functional group. The peak absorbance located close to 3340 cm
−1 is a typical characteristic of cellulose [
27]. As shown in these figures, it is clear that the peak absorbance value of the O–H functional group located close to 3329 cm
−1 decreases with aging due to the oxidation process. The products of degradation of cellulose paper are CO, CO
2, H
2O, H
2, CH
4, and furans, which are dissolved in the mineral oil [
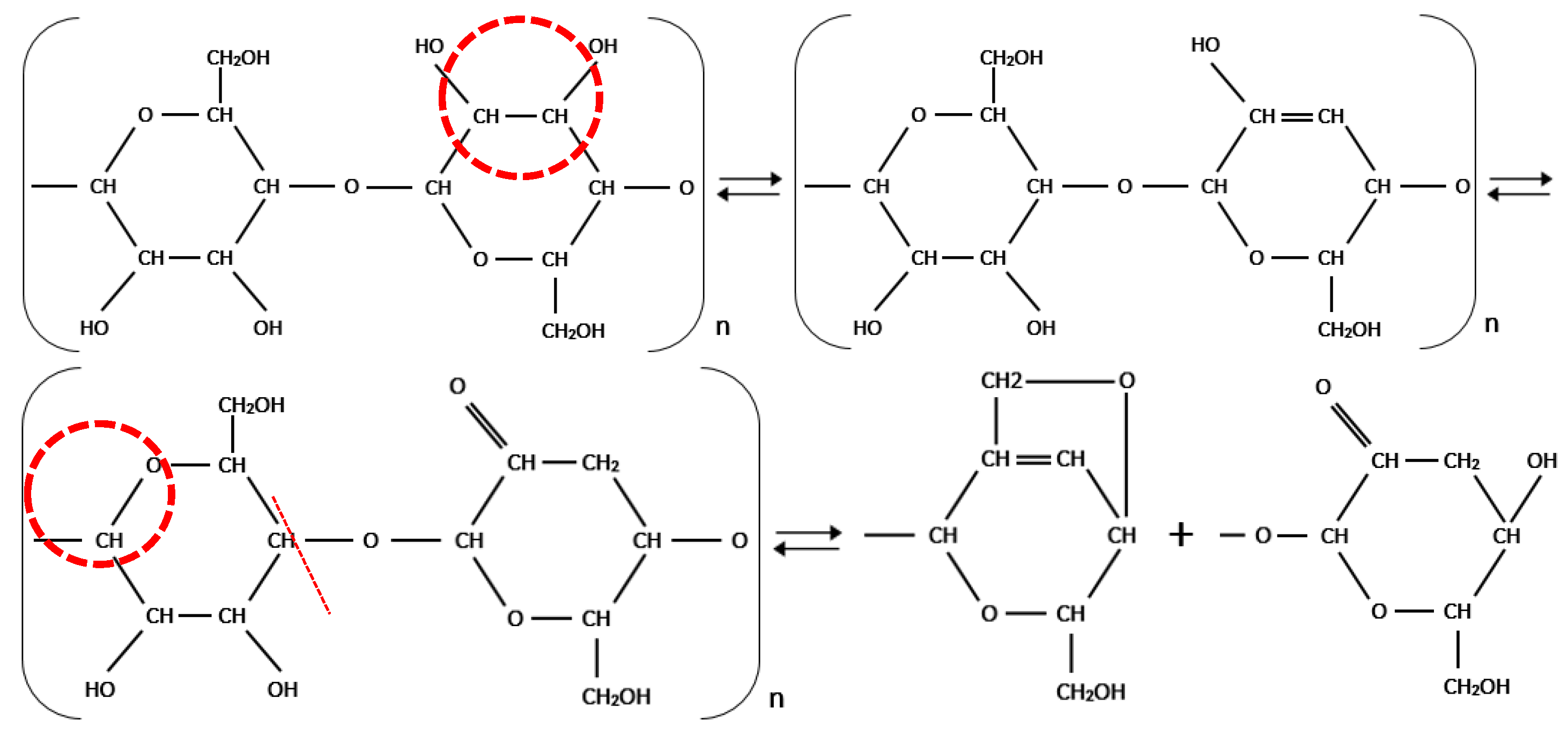
28]. During thermal aging, the cleavage of cellulose chains involves an O–H functional group. An O–H group reacts with a hydrogen atom generating a water molecule. An alcohol is transformed into ketone by an oxidation process. In this process, the hydrogen of the O–H group is displaced toward the carbon group and the CH
2OH groups react with oxygen linking the glucose molecules, thereby cleaving the chain as shown in
Figure 5 [
29,
30]. This intensity of peak absorbance located close to 3329 cm
−1 can be associated with a reduction in molecular weight or the DP value [
7].
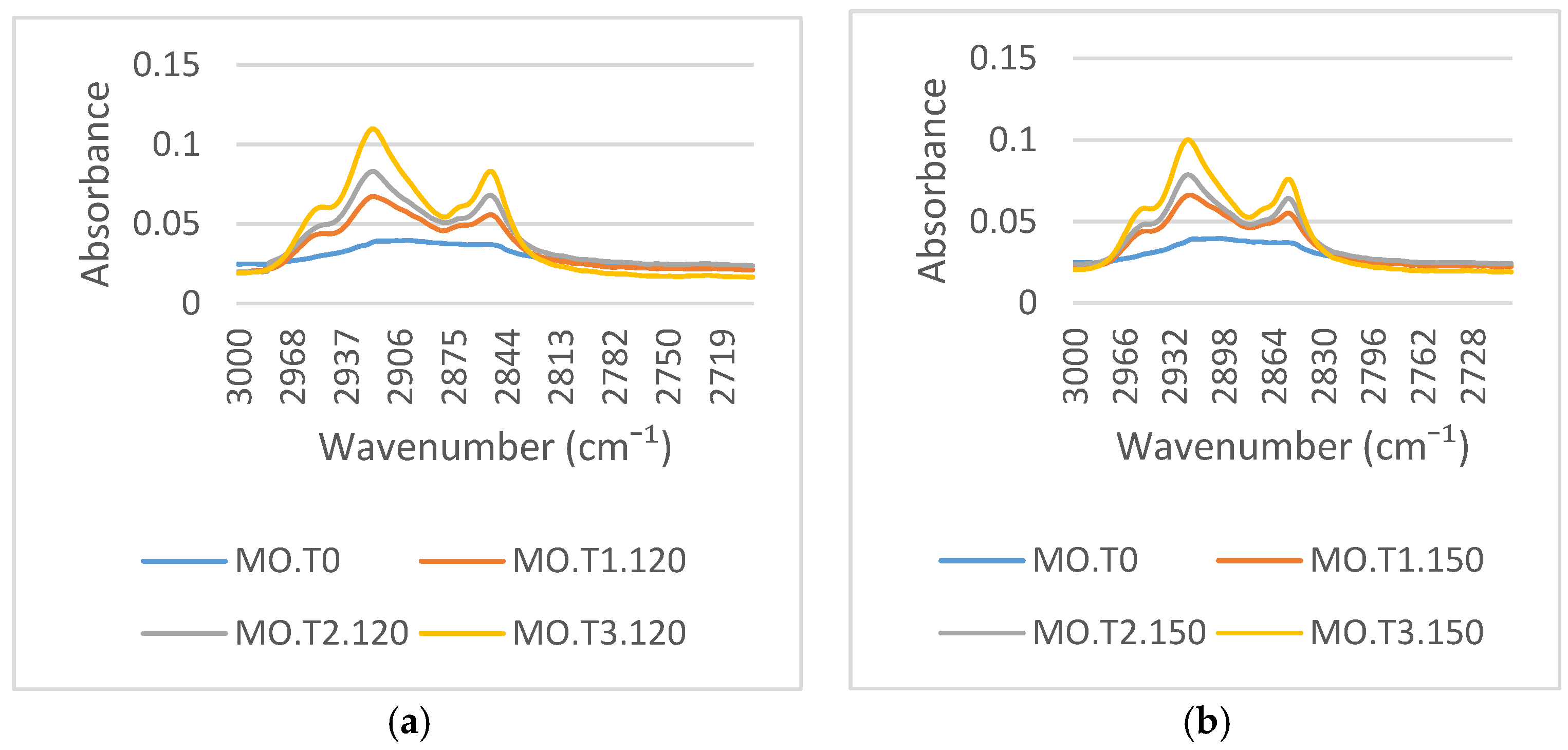
Figure 6a,b illustrates the variations of the FTIR spectra around 2900 cm
−1, which represent a C–H functional group. The intensity of these absorbance peaks at 2922 cm
−1 and 2854 cm
−1 increase with aging due to the adsorption process of the mineral oil to the paper surface during aging [
7]. The major degradation of the aging mechanism in mineral oil is oxidation [
7,
29]. Oxidation decomposes the hydrocarbon molecules into other substances: hydroperoxide, alcohol, aldehyde, ketones, and esters. The oxidation reaction of mineral oil also generates carboxylic acids (R–COOH), which bind to alcohol thereby generating esters. The degradation process of transformer paper aged in mineral oil is due to the combined effects of pyrolysis, hydrolysis, and oxidation [
7]. Temperature, moisture, and oxygen are the key factors that determine the aging rate of cellulose paper [
7]. CO
2, water, furanic compounds, and carboxyl acids (R–COOH) form through the oxidative degradation of cellulose paper [
7]. During thermal aging, the carboxylic acids generated from the oxidation process of mineral oil can react with hydroxyl groups on the cellulose forming a cellulose graft polymer via a condensation reaction [
31]:
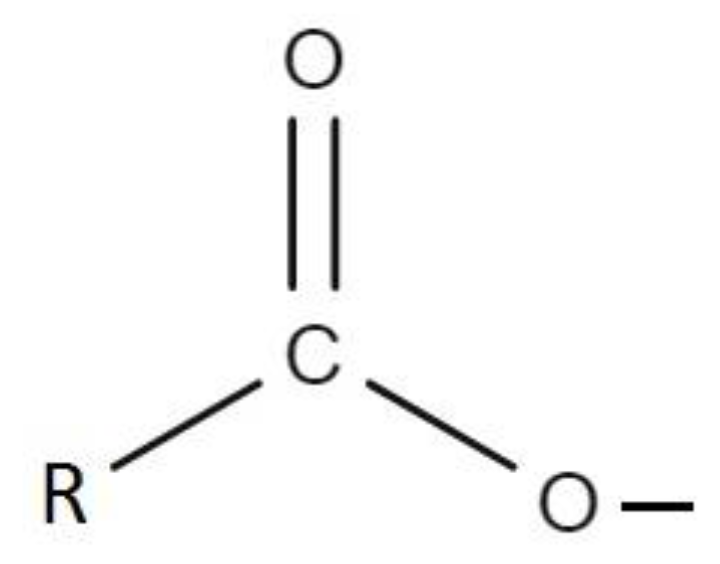
Carboxylic acids are an organic compound that contain a carbon–oxygen double bond and an oxygen–hydrogen single bond. R is a hydrocarbon group that has a lot of C–H functional groups. The reaction between the carboxylic acids in the mineral oil and the hydroxyl groups on the cellulose paper is the cause of the increasing C–H functional group on the cellulose paper after a hydrocarbon group (-R) in the mineral oil is moved to the cellulose paper. The radicals generated on the cellulose surface facilitate the physical attachment of low molecular weight oil molecules and lead to the formation of a layer of hydrocarbon lamination on the cellulose surface and an increase in the intensity of the corresponding to C–H vibrations [
7]. Carboxyl acids in the mineral oil and the hydroxyl groups of cellulose paper are not only the main factors increasing the C–H functional group but are also the main factors in the emergence of the C=O functional group on the cellulose paper surface.
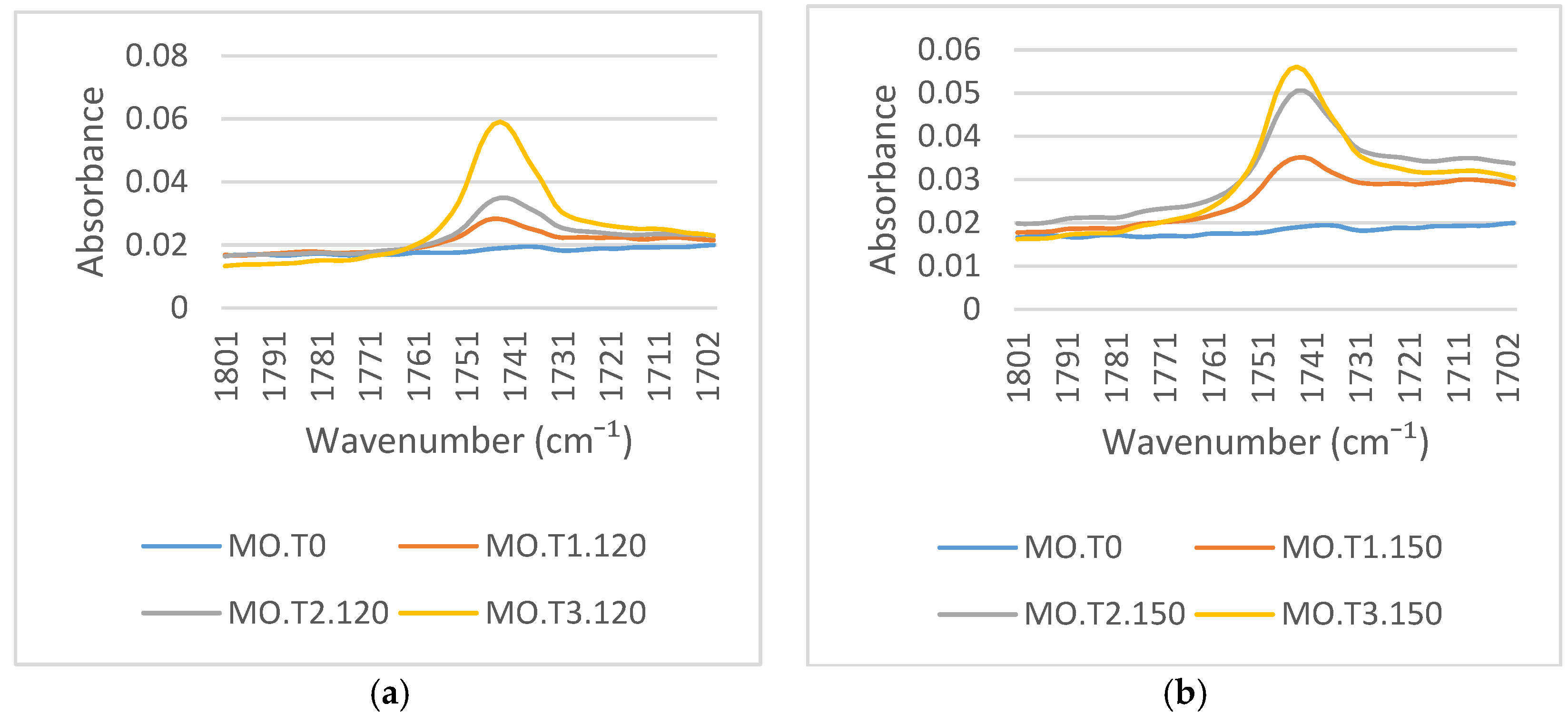
Figure 7a,b presents the FTIR spectra around 1700 cm
−1, which represents the C=O functional group. The intensity of this absorbance peak at 1745 cm
−1 increases with aging. This peak emerges on the cellulose paper after thermal aging in mineral oil. There is no absorbance peak at this functional group region for new transformer paper. The emergence of the C=O functional group is due to interactions between the low molecular weight acids dissolved in the mineral oil and the cellulose [
31]. Carboxylic acids containing a carbon–oxygen double functional group react with the hydroxyl groups on the cellulose, forming a cellulose graft polymer and H
2O. A carboxylate of the carboxylic acid (RCOO-) in mineral oil binds to the cellulose; this creates the emergence of the C=O functional group on the cellulose paper surface, as shown in
Figure 8.
3.2. Correlation between Average Number of Chain Scissions and the Structural Changes of Transformer Paper Aged in Mineral Oil
The intensity of the peak absorbance of the functional groups within transformer paper changes during thermal aging due to the chemical process. The higher the temperature and the longer the duration of thermal aging of transformer paper in mineral oil, the higher is the thermal energy value. The chemical reaction is faster in systems with higher thermal energy. Svante Arrhenius showed the relationship between temperature and the rate constant by the reaction in Equation (1):
where
k is the rate constant at temperature
T (K),
A is a constant called ‘frequency factor’,
Ea is the activation energy for the reaction (J/mol), and
R is the universal gas constant (8.314 J/mole/K). The activation energy represents the energy that the molecule in the initial state of the process must have before it can take part in the reaction. The reaction rate at any time is assumed to be proportional to the number of unbroken polymer chain bonds in the aging of paper [
8,
32]. The Arrhenius equation about the thermal aging of transformer paper in mineral oil is shown in Equation (2) [
33]:
In this calculation of the average number of chain scissions, the value of
A depends on the initial moisture content within the paper sample and the type of insulating paper. The value of
A used for this calculation was 21 × 10
8 for non-upgraded kraft paper with a high moisture content [
6,
34]. For the expected lifetime of the transformer, the energy activation of 111 kJ/mol was used in this calculation [
6,
34,
35].
Table 5 shows the average number of chain scissions of each paper sample. The average number of chain scissions of the paper sample increases with an increasing aging temperature at the same aging time. The probability of molecules colliding with each other increases with aging temperature. As a result, the kinetic energy of the molecules increases, making the effect to the activation energy of a reaction. The thermal energy increased due to the kinetic energy of the molecules making the degradation process at a temperature of 150 °C higher than at a temperature of 120 °C.
Figure 9 shows the correlation between the intensity of the absorbance peaks of the functional groups at 3329 cm
−1, 2922 cm
−1, 2854 cm
−1, and 1745 cm
−1 and the average chain scissions of the paper samples. The intensity of the peak absorbance of the functional group within the transformer paper that changes with aging correlates with the aging mechanisms during thermal aging. The intensity of the O–H functional group correlates with the average chain scissions due to the breakage of the hydrogen bonds of cellulose. The reduced intensity of the O–H functional group can be attributed to the reduction of the molecular weight/degree of polymerization [
7]. The average number of chain scissions correlates with the molecular weight as shown in Equation (3) [
7]:
DP0 represents the DP value of new transformer paper and DPt represents the DP value of transformer paper after thermal aging. From these figures, it can be seen that there is a linear correlation between the intensity of the peak absorbance of the O–H functional group and the average number of chain scissions. Those figures also show that there is a correlation between the intensity of the peak absorbance of the C–H and the C=O functional groups and the average number of chain scissions. It indicates that the reaction of the carboxylic acids in the mineral oil with the hydroxyl groups on the cellulose occurred at the same time as the process of chain scission on the cellulose.
Table 6 shows the value of R
2 from the correlation between the functional group and the average number of chain scissions of transformer paper. R
2 is the coefficient of determination, which has a range of 0–1 and measures the closeness of the data to the regression line. A value of R
2 of 1 means that the correlation has a linear relationship. This result gives initial information that there is a correlation between the structural changes of transformer paper during thermal aging with the average number of chain scissions.