2.1. Sample Preparation
Coal particle reactions in conventional coal-fired boilers can generally be divided into two concepts: coal devolatilization and char oxidation [
25]. During the devolatilization of raw coal, most of the moisture and volatile matter in the coal are removed, and the remaining particles, referred to as char, are then employed in oxidization or combustion with oxygen at sufficiently high temperatures (1100–1400 °C). This study focused on char combustion at high temperatures. Coal char production for the experiment was performed using the following process.
Raw low-rank coal (Indonesian sub-bituminous coal,
Figure 1a, was dried for 1 day at 45 °C. The sample powder used as fuel had particle sizes of 75–90 μm. After drying, the sample was heated to 1000 °C at a heating rate of 10 °C/min in a thermogravimetric analyzer (TGA701, LECO Co., St. Joseph, MI, USA) under a nitrogen atmosphere.
Figure 1a,b show the differences between the morphologies of the coal before and after the char production process [
26]. As can be seen in
Figure 1b, some pores developed in the char due to the removal of moisture and volatile matter.
Figure 1c shows graphite powder with particles in the range of 75–90 μm to allow for a comparison with the characteristics of the char.
Table 1 summarizes the results of the proximate and ultimate analyses of the raw coal, char, and graphite.
The initial specific surface area of the char was measured by the Brunauer-Emmett-Teller (BET) method using a Micromeritics ASAP 2020 device. The morphologies of the coal samples were examined by scanning electron microscopy (SEM, S3500N, Hitachi, Tokyo, Japan).
2.2. Coal Heating Reactor
The experimental setup of the coal-heating reactor for the proposed oxygen-enriched combustion is shown in
Figure 2 [
27]. The heating reactor can be used for combustion under high-temperature conditions of up to 1750 K. A Pt wire mesh in the chamber was used to generate heat using a direct current (DC) electrical power supply (OPE-18100S, 4.7 kW, ODA Technologies Co., Ltd., Incheon, Korea) that was controlled using computer software. In this study, the temperatures used for coal heating were measured as 1373, 1473, 1573, and 1673 K.
The Pt mesh (99.9% purity) had a wire diameter of 0.04 mm, and the wire interval was 75 μm. In order to obtain the combustion temperature and reaction time, a thermocouple was located at the center of the Pt mesh.
Coal char samples were loaded onto the wire mesh in a thin layer configuration, as shown in
Figure 2a.
Figure 2b shows the heating state, in which the Pt mesh was heated in the coal-heating reactor. The primary reactant gas was introduced to the chamber during combustion from the atmosphere or from a mixed gas tank for the 21% and 30% oxygen conditions, respectively, and was released back into the atmosphere after combustion.
2.3. Numerical Methods for Kinetics
In this study, a mathematical model was suggested for the calculation of the reaction kinetics of coal char combustion with an oxygen content of 21–30% after devolatilization. The carbon conversion ratio can be expressed as:
where
is the initial mass of the coal char,
is the mass of the coal char at time
t, and
is the mass of the ash content.
The apparent reaction rate (
) is expressed as:
where
is the change of carbon conversion over time.
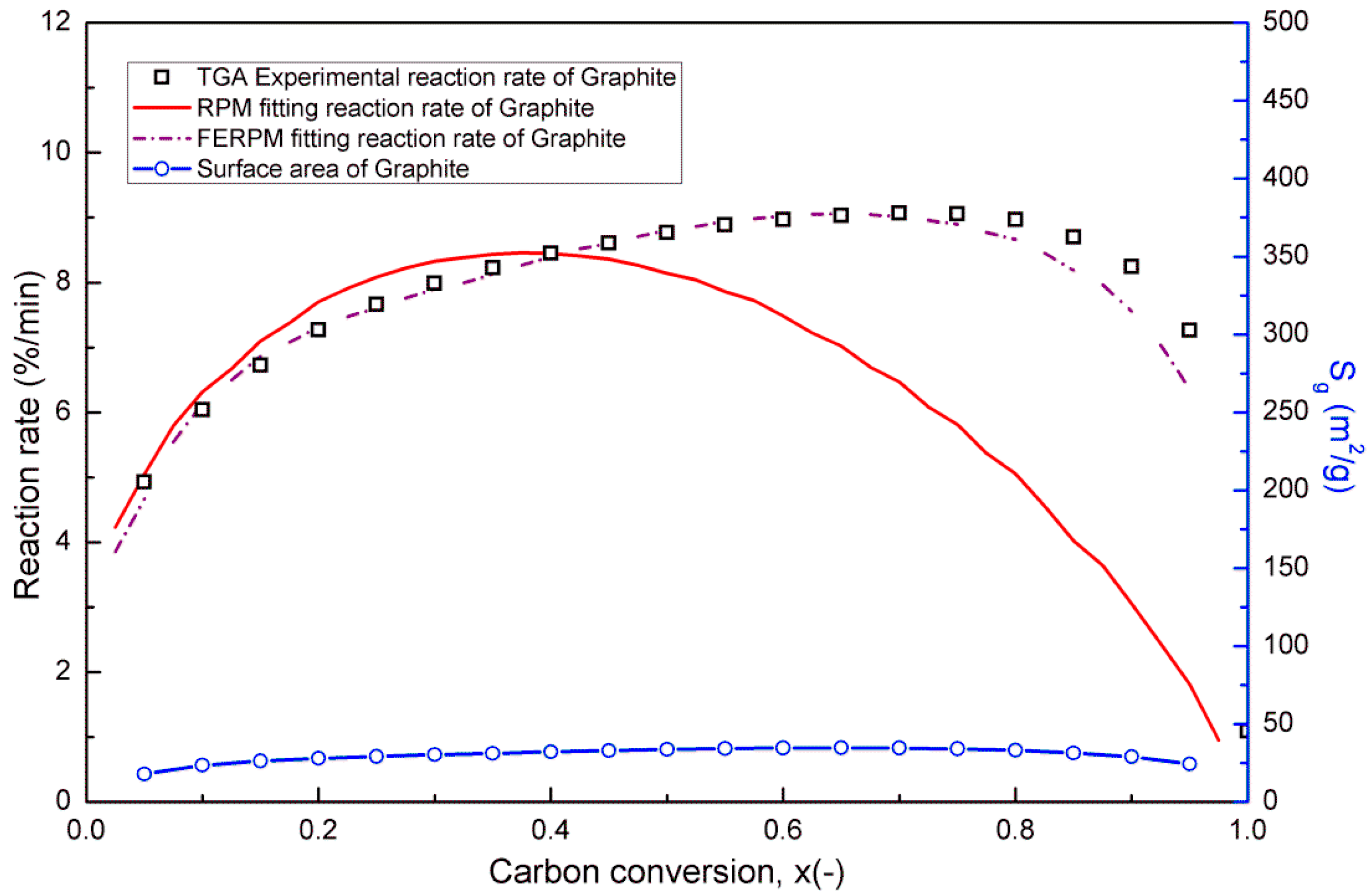
The random pore model (RPM) is widely used to simulate changes in the micropore structure of coal char. However, when considering most low-rank coal or biomass, the random pore model does not fit well with observations. In order to achieve more accurate pore development modeling and reaction rate correlations, the Flexibility-Enhanced Random Pore Model (FERPM) proposed by Lisandy et al. was used in this study [
22]. During the coal char oxidation reaction, the loss of coal char occurs rapidly. Thus, the micropore structure of the coal char also changes rapidly. In order to calculate the pore development (
Sg), the RPM and FERPM were both used in this study, and they are shown below:
where
and
are the initial reaction rates as a function of the temperature, and
and
are the structural parameters.
,
and
are extra parameters for improving the accuracy of the prediction.
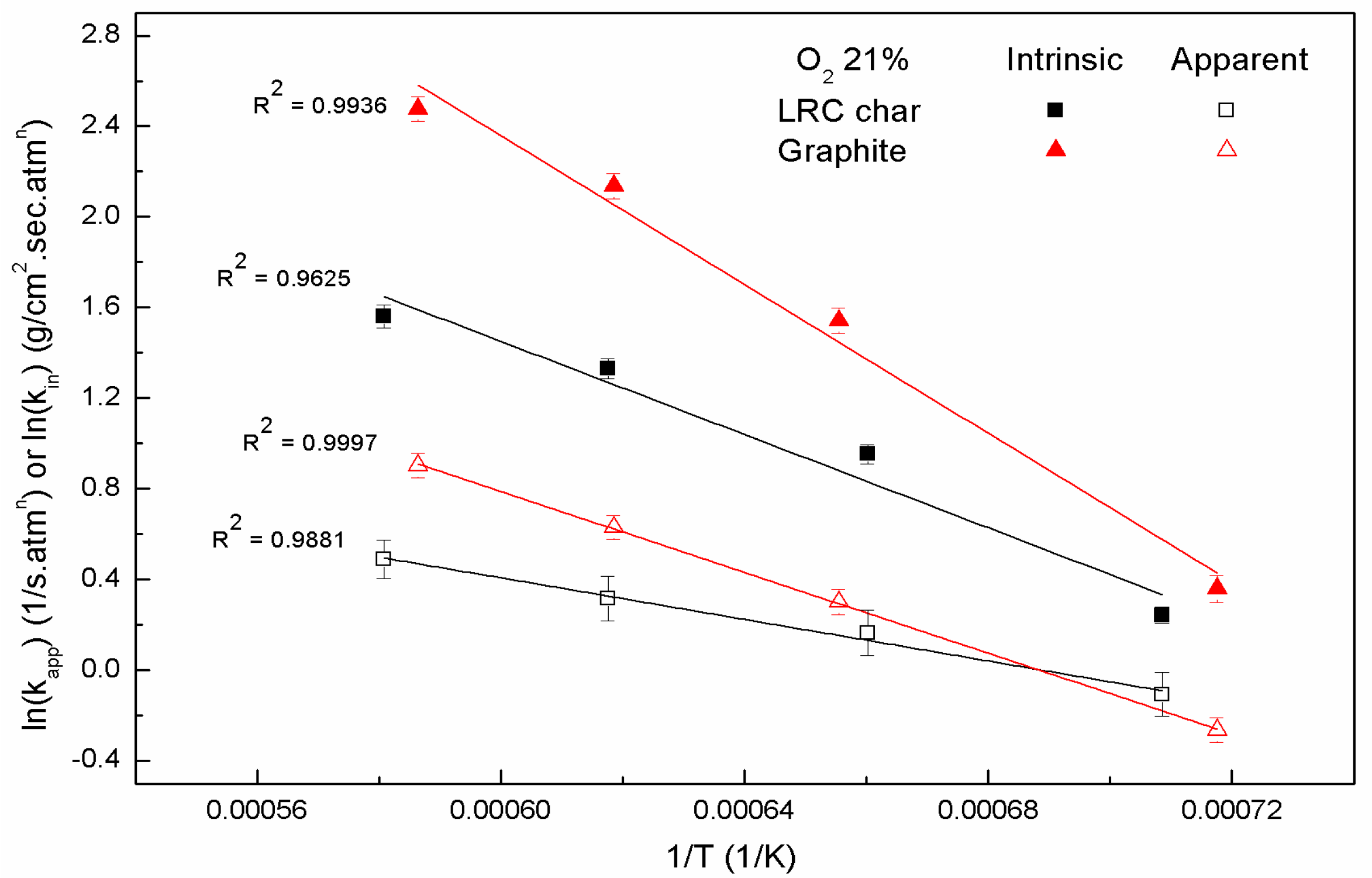
In order to determine the intrinsic reaction rate of the coal char oxidation (
,
), an
nth-order rate equation was used. The
nth-order rate equation of the global reaction can be expressed in its Arrhenius form as:
where
is the intrinsic pre-exponential factor,
is the intrinsic activation energy,
is the universal gas constant,
is the coal char particle temperature,
is the partial pressure of oxygen in the ambient atmosphere, and
is the reaction order, which was derived from the experimental results. This equation is defined by the reaction order and two Arrhenius parameters,
and
.
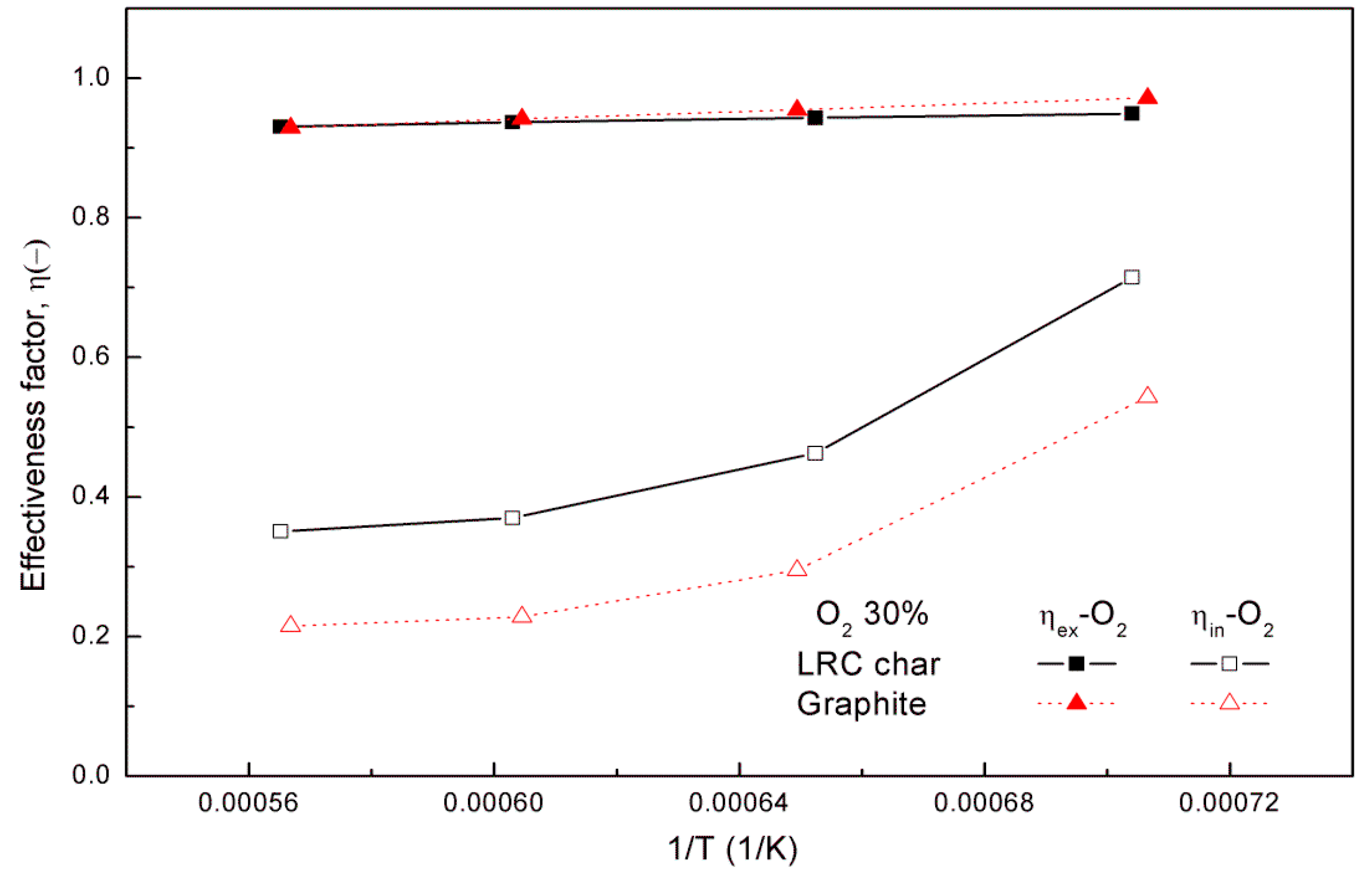
In order to analyze the oxidation of the coal char, the effect of the internal and external effectiveness factors should be considered. The apparent reaction rate is then expressed as [
28]:
The internal effectiveness factor can be defined by the Thiele modulus (
), which was developed to determine the relationship between the diffusion and the reaction rate in a microporous structure [
29,
30]:
where
is the diameter of the coal char,
is the stoichiometric coefficient (i.e., 0.0833 moles of O
2 consumed per gram of reacted carbon),
is the apparent density, and
is the effective O
2 diffusion coefficient in the coal char surface.
is calculated as:
where is the porosity,
is the diffusivity, and
is the tortuosity coefficient of the coal char pore, and its value is assumed to 1.414 [
31]. The diffusivity,
, is calcas:
where
is the binary diffusivity and
is the Knudsen diffusivity, which is the diffusion that occurs when the diameter of the pore is comparable to or smaller than the mean free path of the gas particles involved. These diffusivities are expressed as [
29,
30,
31]:
where
is the diameter of the coal char pore,
is the molar mass,
is the mixed molar mass of A and B,
is the particle collision diameter,
is the collision integral, and
is the total pressure of the system.
The external effectiveness factor (
) is an expression of gas transport to the boundary layer, which is the ratio of the partial pressure of the particle surface to the ambient atmosphere surrounding the coal char particle, and is expressed as:
The kinetic coefficients of the reaction can be considered by the linear regression method as follows: