Chemical Characteristics and NaCl Component Behavior of Biochar Derived from the Salty Food Waste by Water Flushing
Abstract
:1. Introduction
2. Experimental
2.1. Raw Material
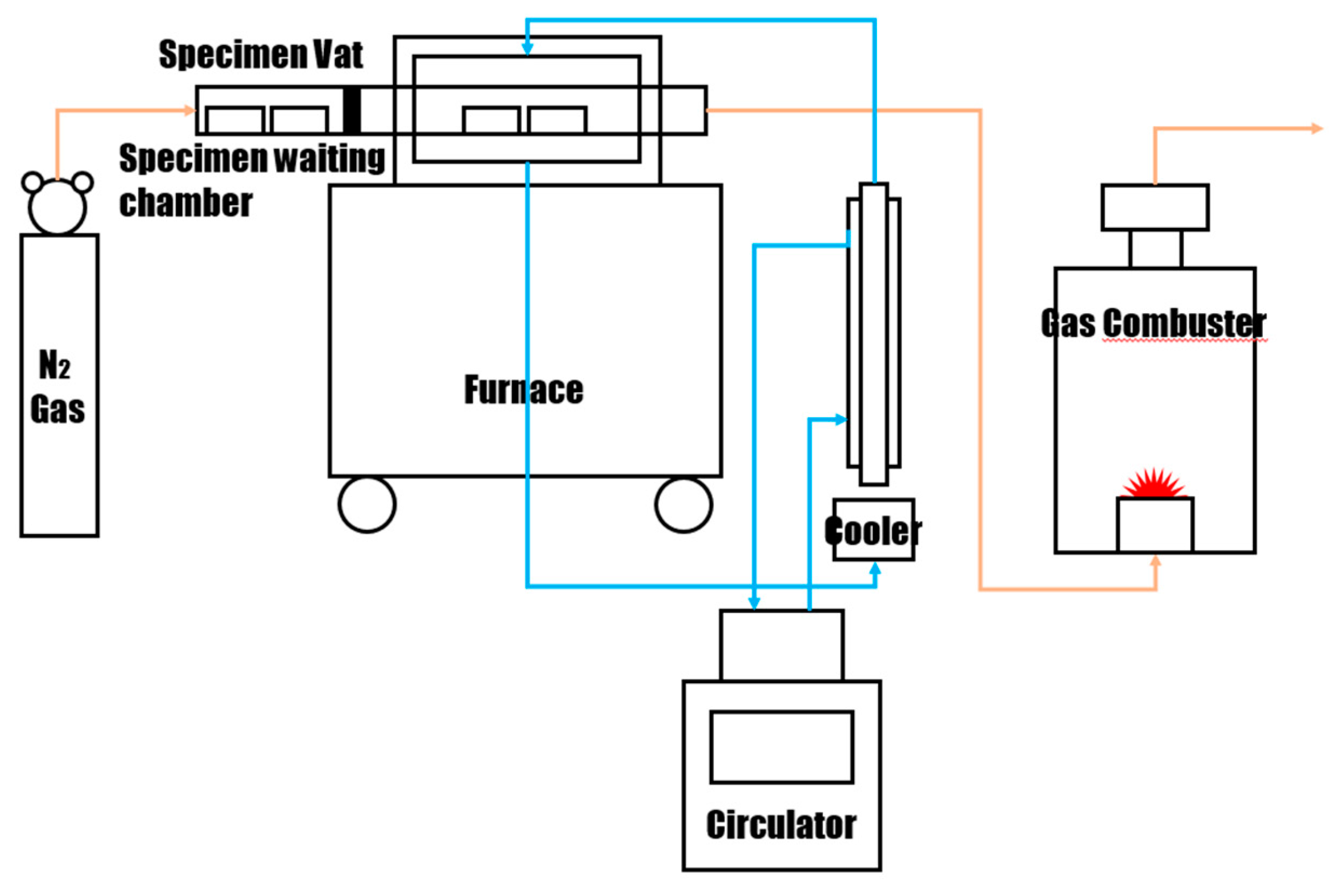
2.2. Experimental Methods
2.3. Analytical Methods
2.3.1. Characterization
2.3.2. X-ray Diffraction Spectroscopy
2.3.3. FT-IR
2.3.4. NMR
3. Results and Discussion
3.1. Chemical Composition of Salty Food-Waste-Derived Biochar
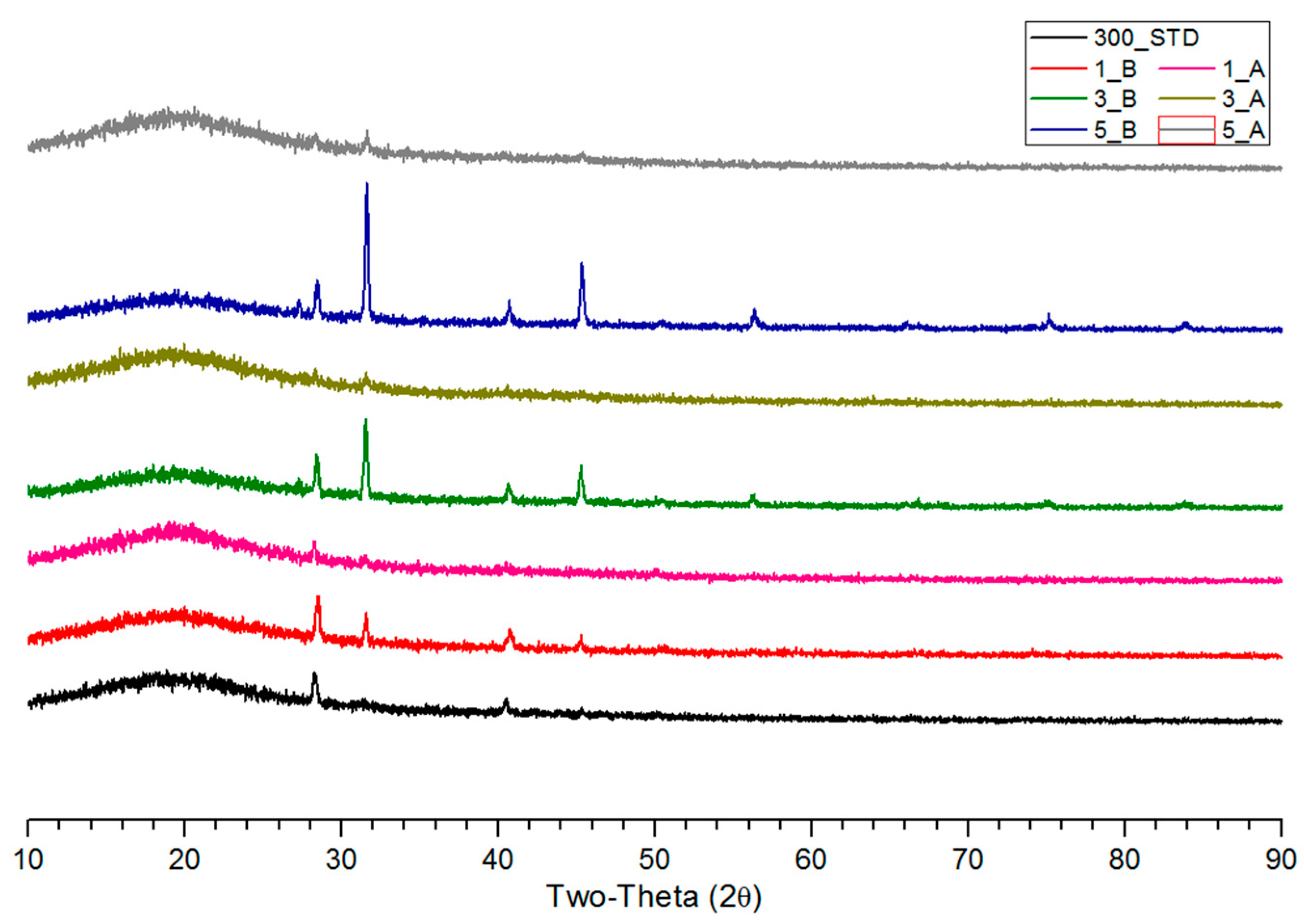
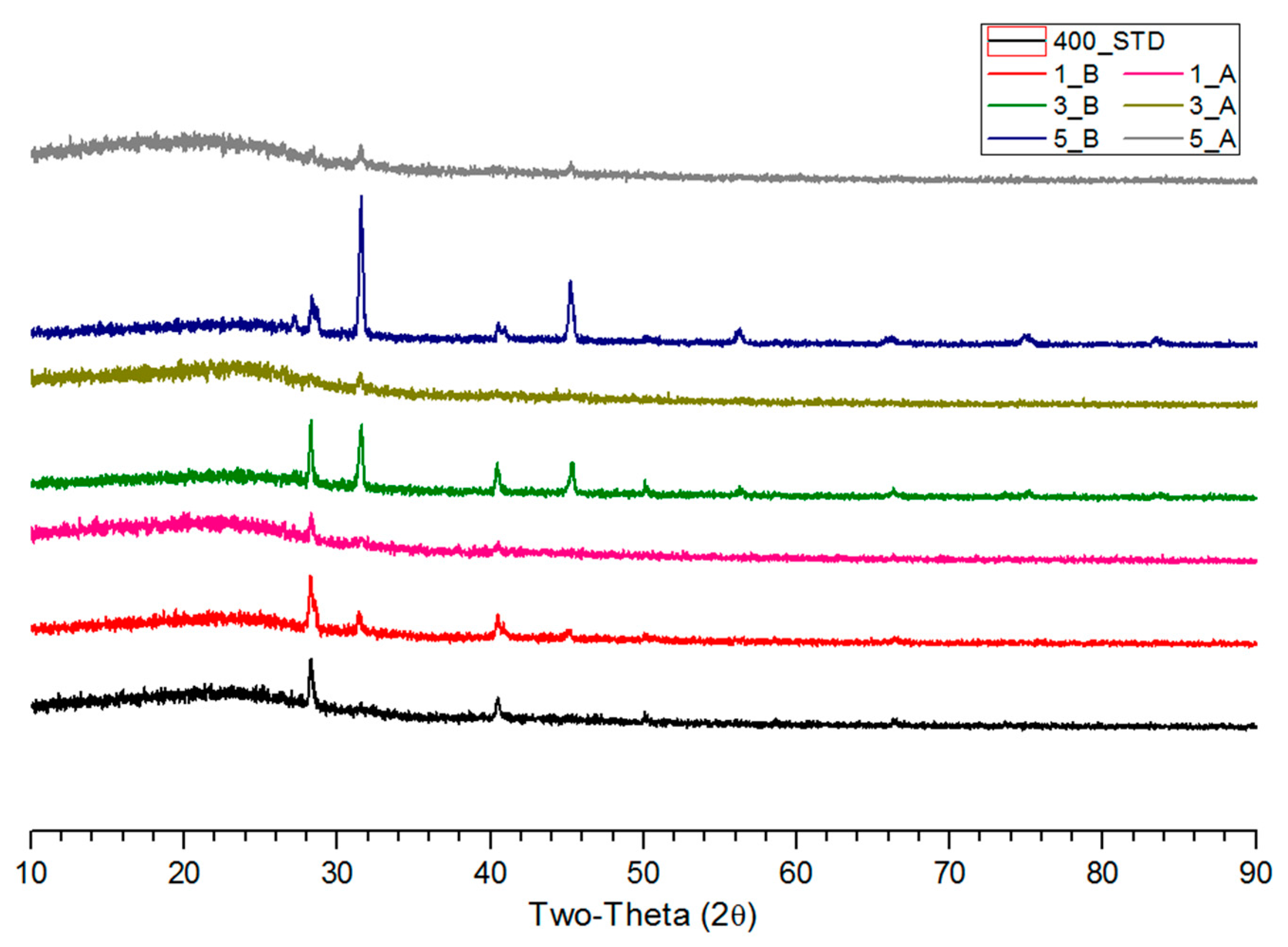
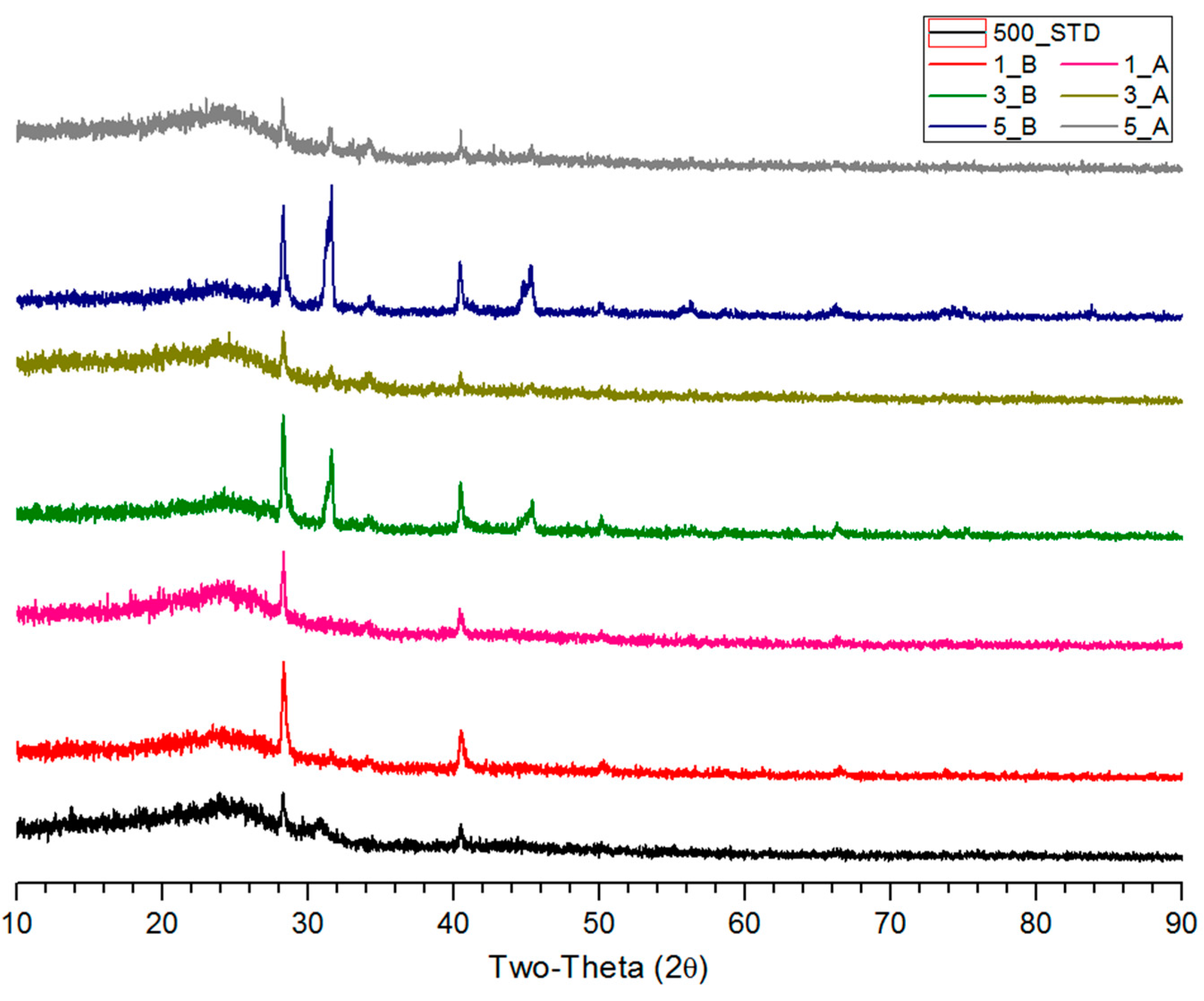
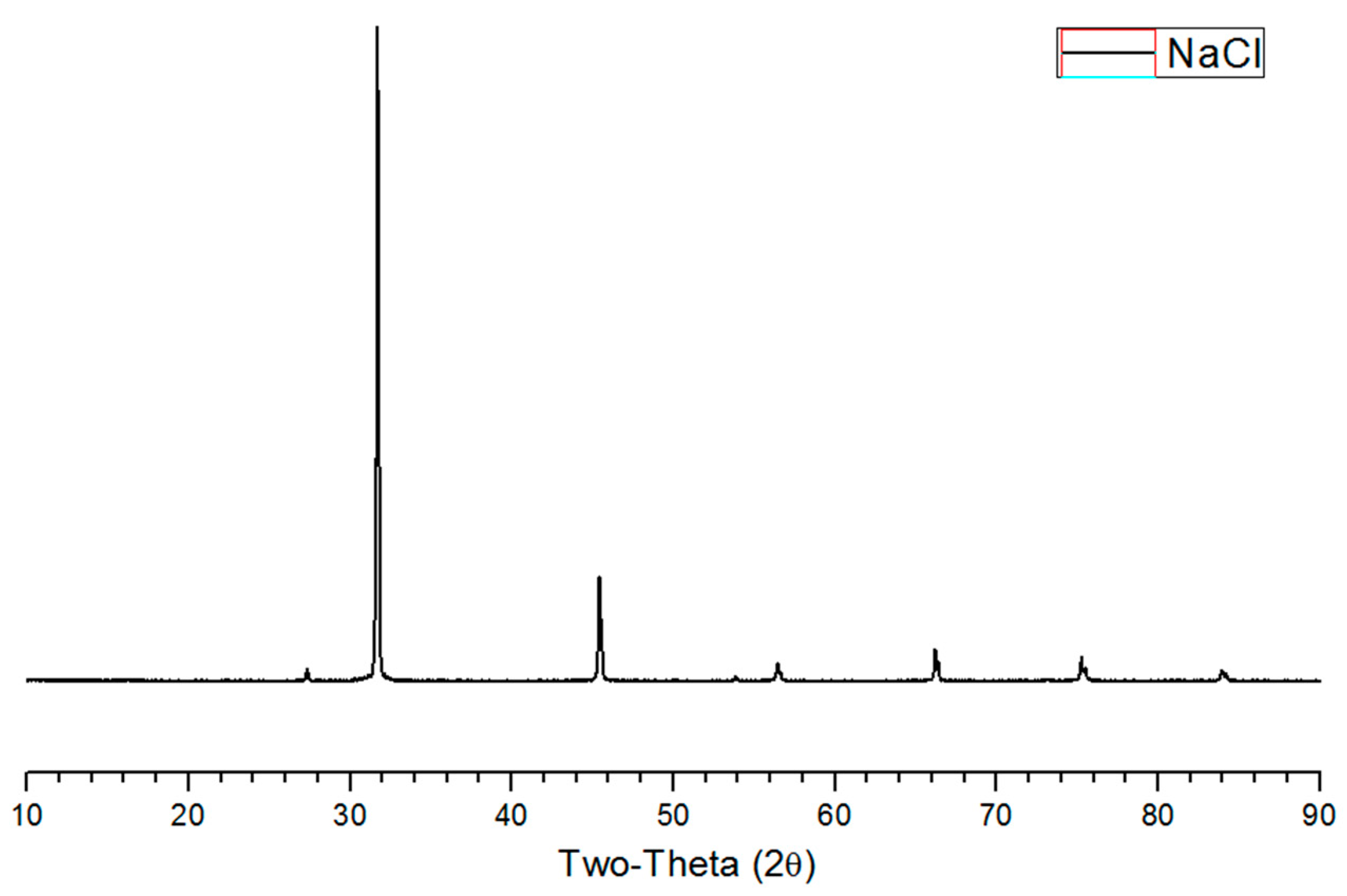
3.2. XRD Analysis of Salty Food-Waste-Derived Biochar
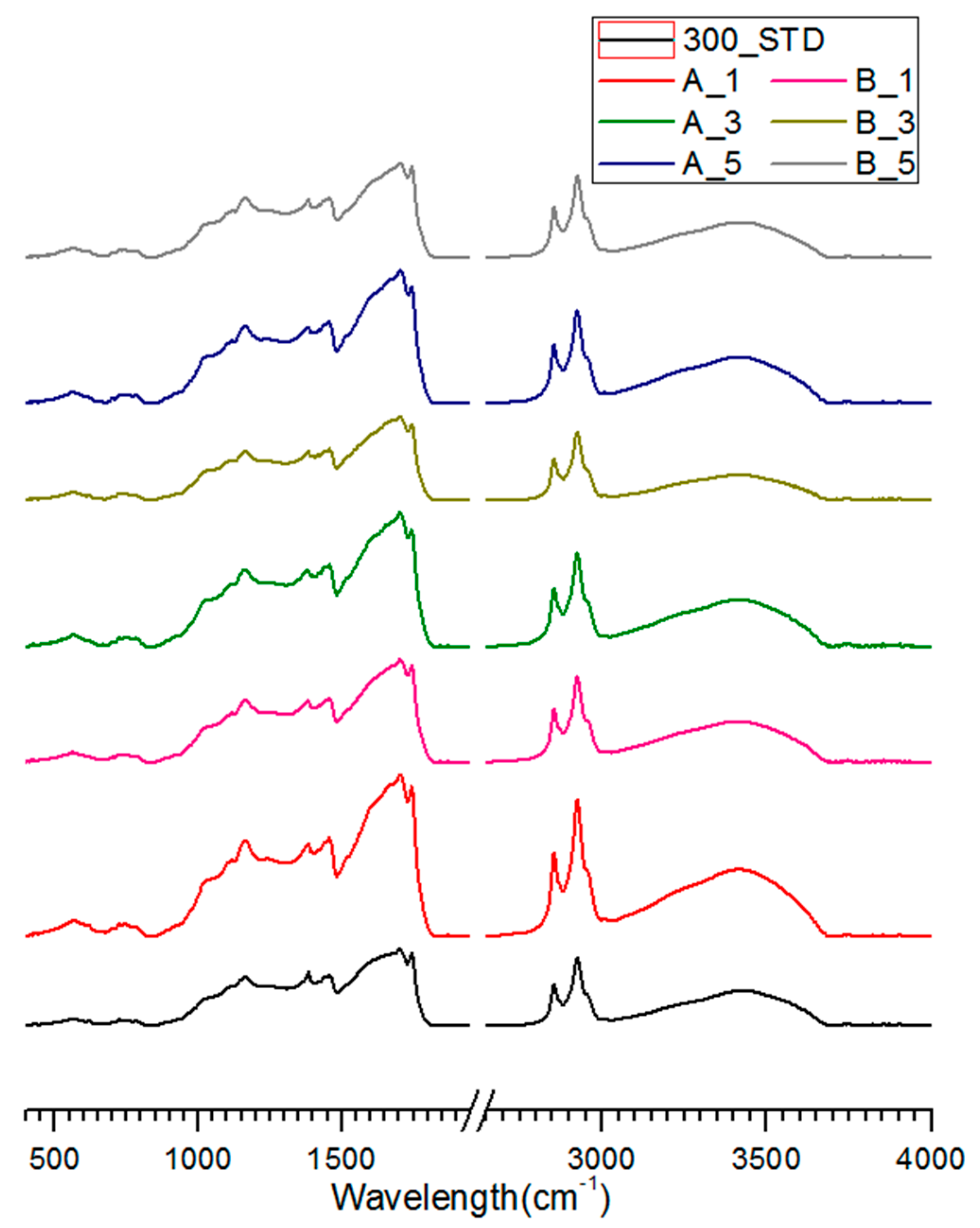
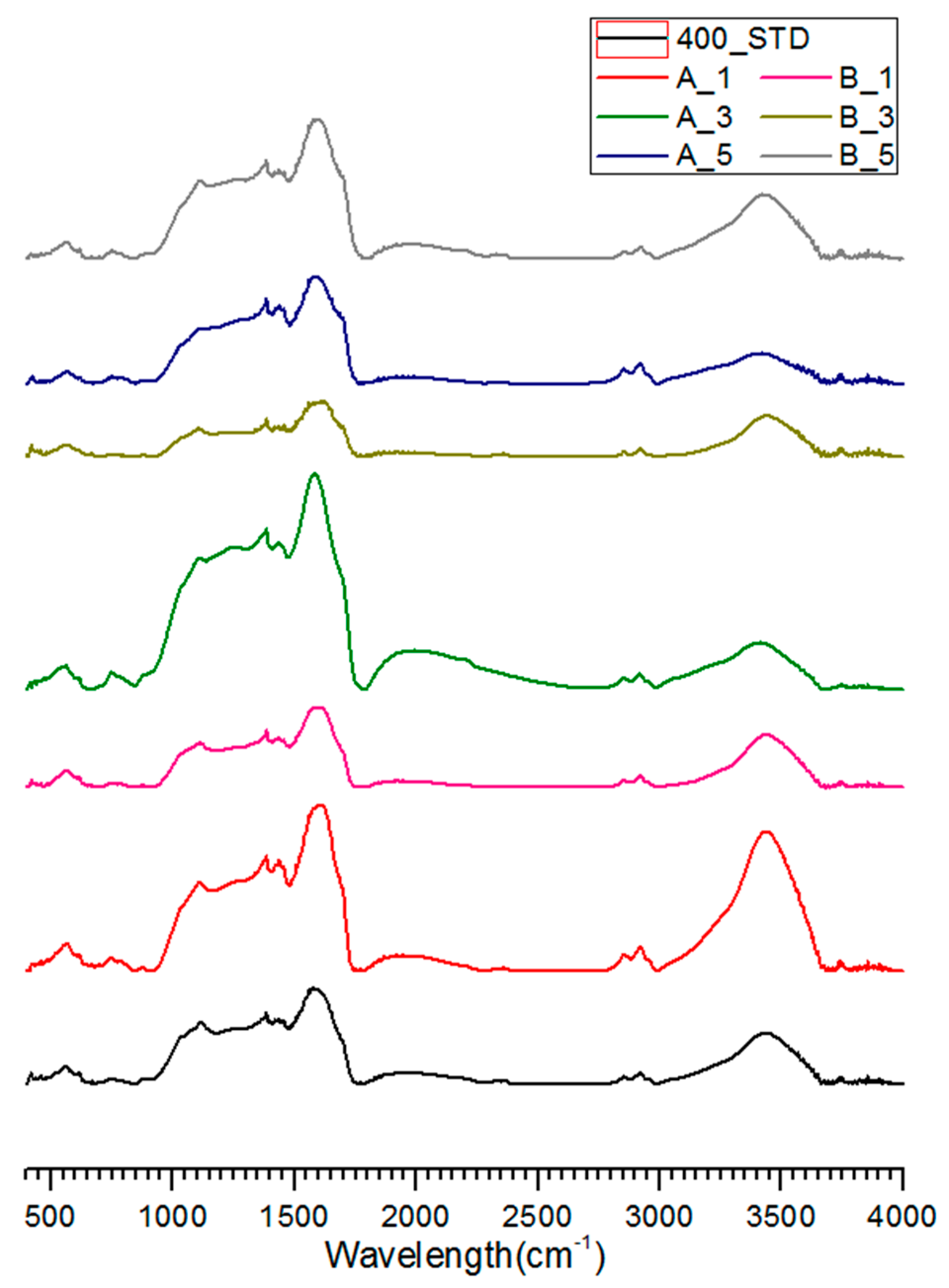
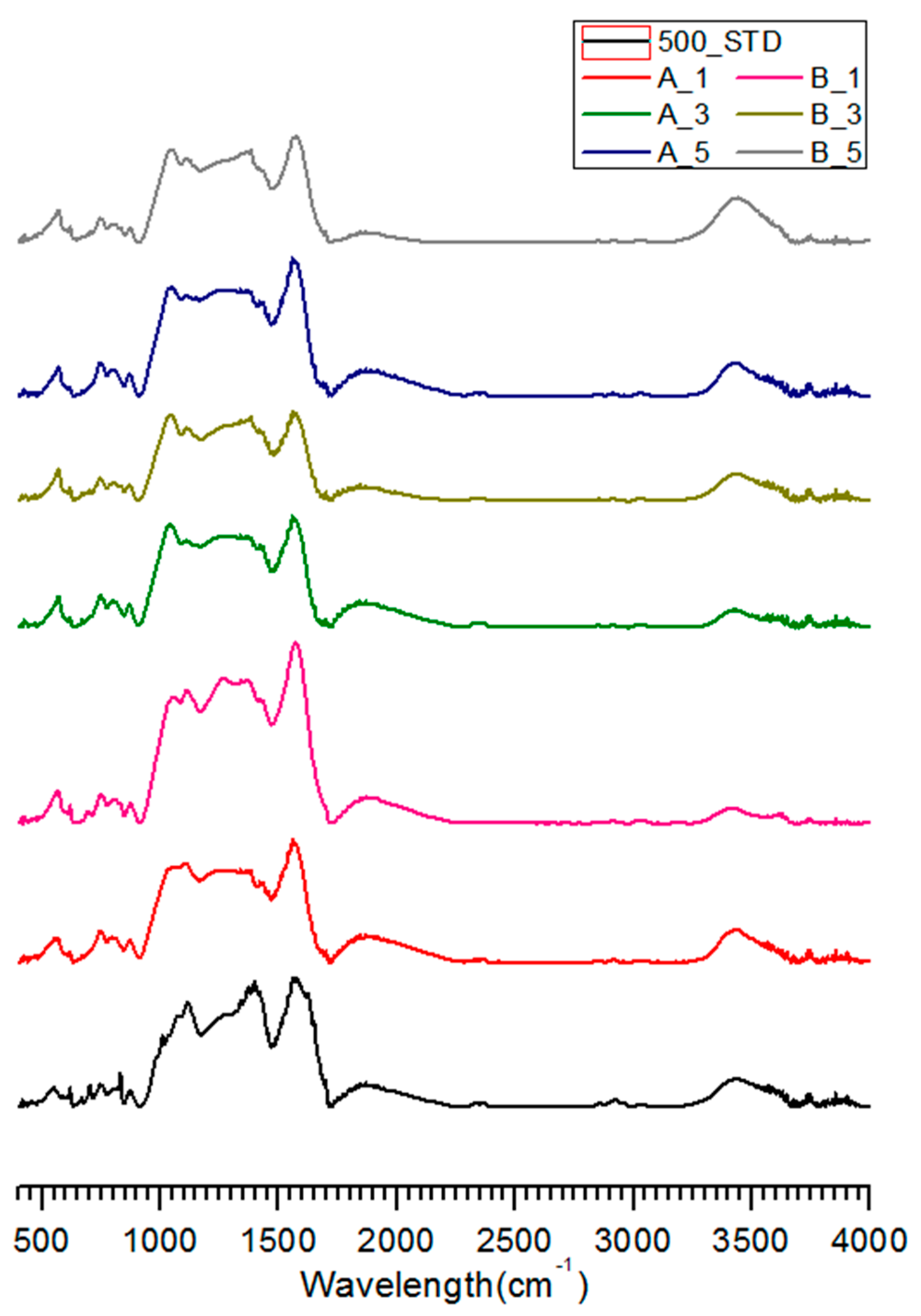
3.3. FT-IR Analysis of Salty Food-Waste-Derived Biochar
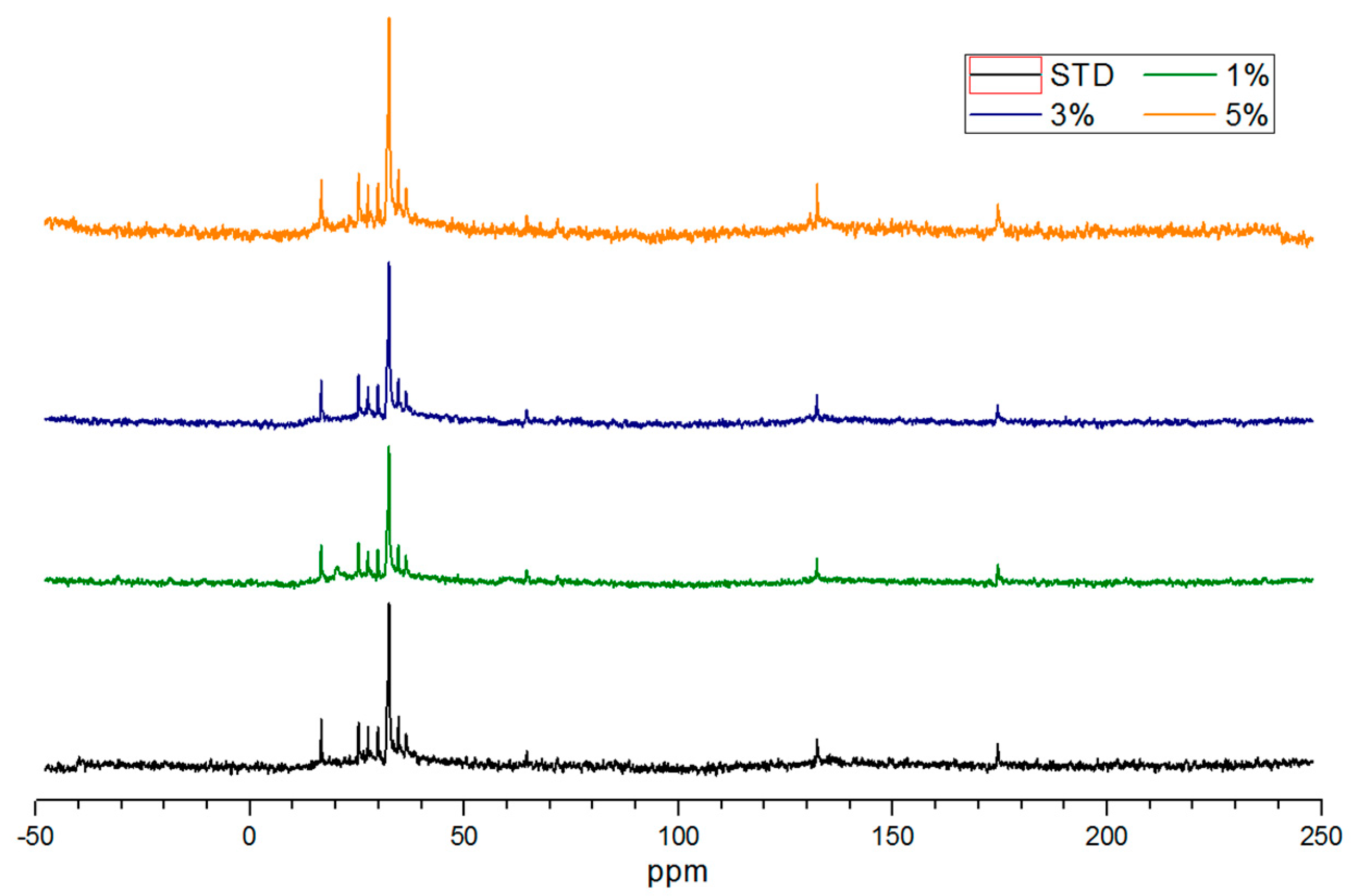
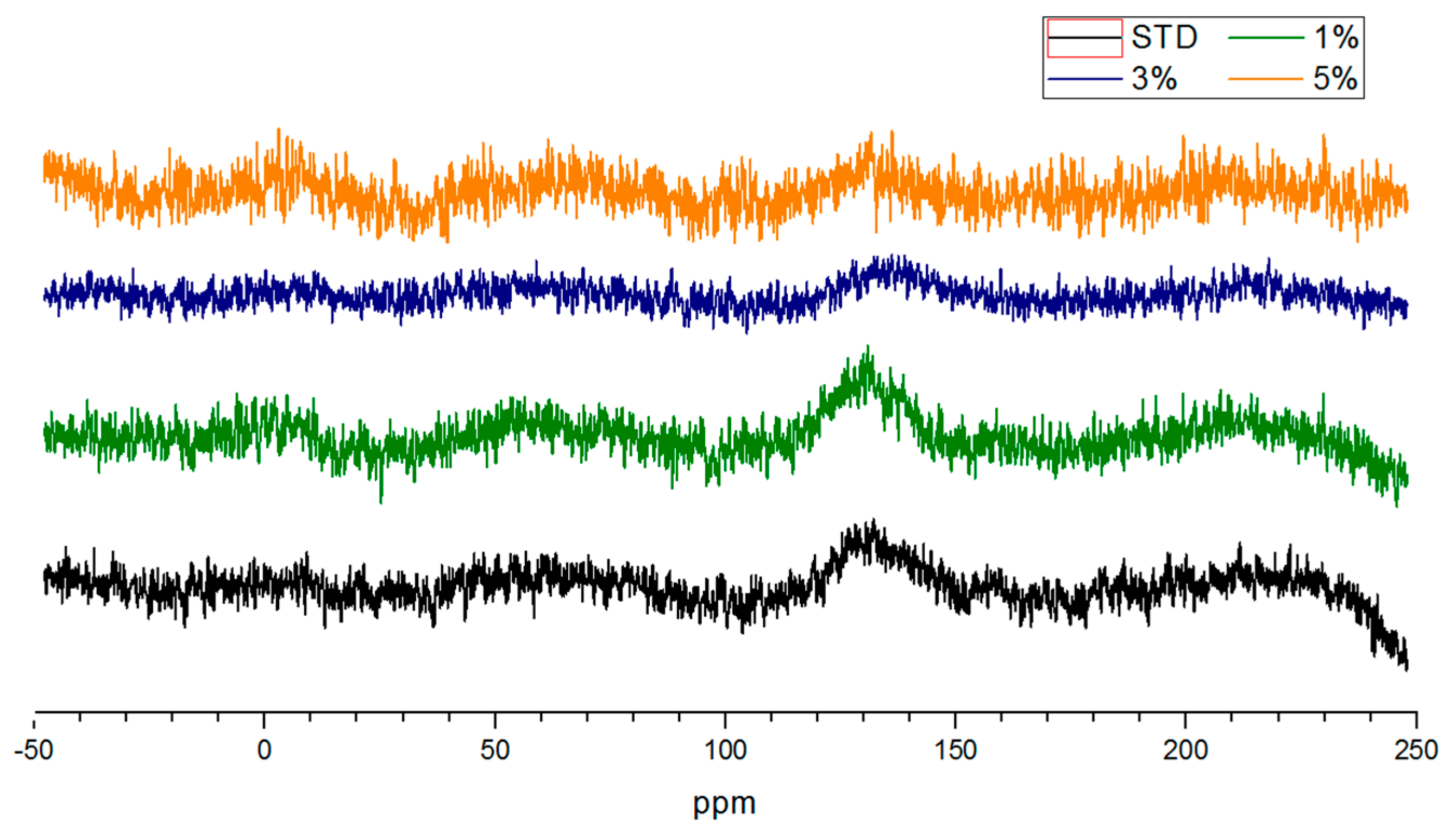
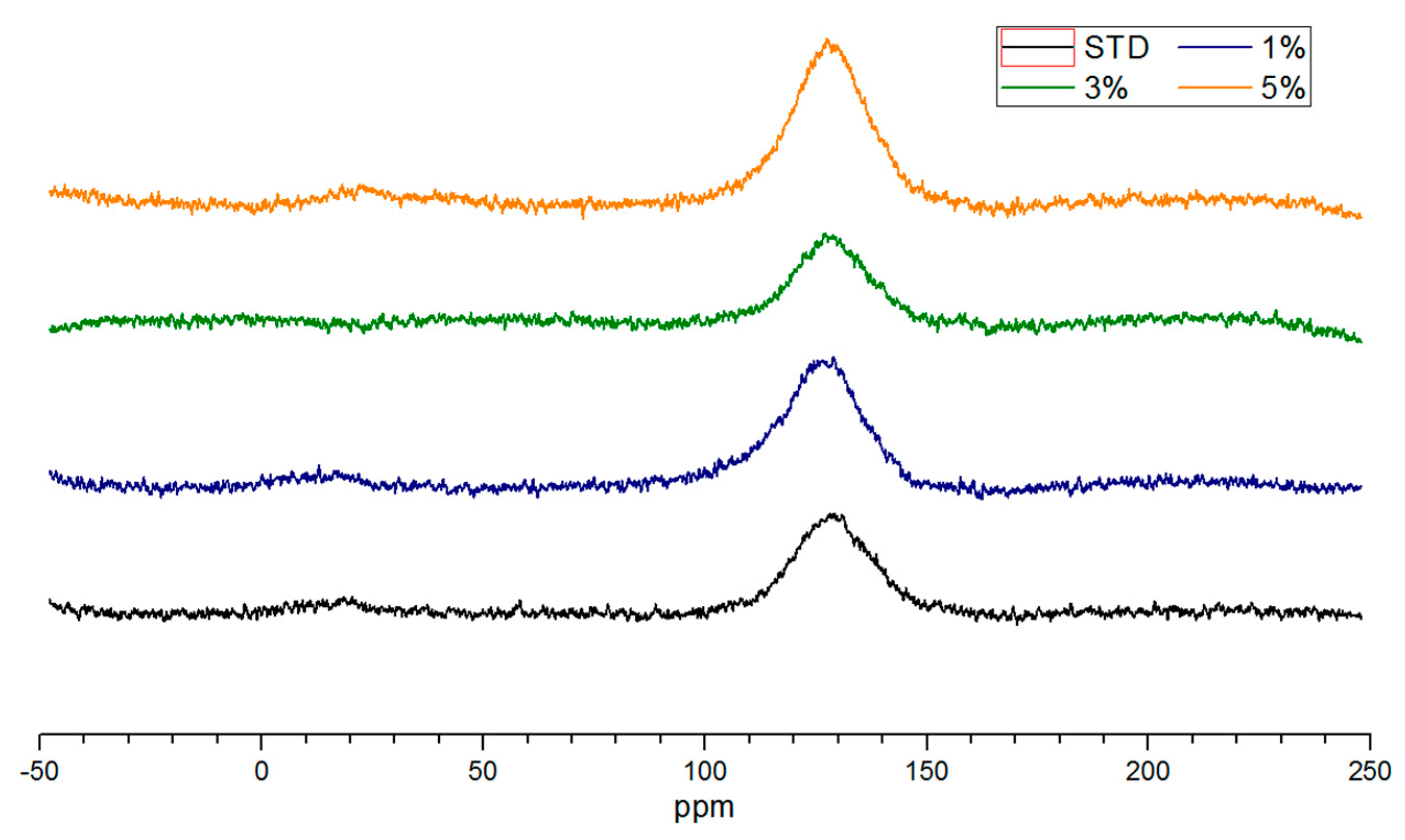
3.4. NMR Analysis of Salty Food Waste Derived Biochar
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Hall, K.D.; Guo, J.; Dore, M.; Chow, C.C. The progressive increase of food waste in America and its environmental impact. PLoS ONE 2009, 4, e7940. [Google Scholar] [CrossRef] [PubMed]
- Venkat, K. The climate change and economic impacts of food waste in the United States. Int. J. Food Syst. Dyn. 2011, 2, 431–446. [Google Scholar]
- Lehmann, J.; Joseph, S. Biochar for environmental management: An introduction. In Biochar for Environmental Management: Science and Technology; Lehmann, J., Joseph, S., Eds.; Earthscan: London, UK, 2009. [Google Scholar]
- Ioannidou, O.; Zabaniotou, A. Agricultural residues as precursors for activated carbon production—A review. Renew. Sustain. Energy Rev. 2007, 11, 1966–2005. [Google Scholar] [CrossRef]
- Lehmann, J. A handful of carbon. Nature 2007, 447, 143–144. [Google Scholar] [CrossRef] [PubMed]
- Brassard, P.; Godbout, S.; Raghavan, V.G.S.; Palacios, J.H.; Grenier, M.; Zegan, D. The production of engineered biochars in a vertical auger pyrolysis reactor for carbon sequestration. Energies 2017, 10, 288. [Google Scholar] [CrossRef]
- Taghizadeh-Toosi, A.; Clough, T.J.; Condron, L.M.; Sherlock, R.R.; Anderson, C.R.; Craigie, R.A. Biochar incorporation into pasture soil suppresses in situ nitrous oxide emissions from ruminant urine patches. J. Environ. Qual. 2011, 40, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, P.J.; Dalley, T.S.L.; Helleur, R.J. Preliminary laboratory production and characterization of biochars from lignocellulosic municipal waste. J. Anal. Appl. Pyrolysis 2013, 99, 71–78. [Google Scholar] [CrossRef]
- Prakongkep, N.; Gilkes, R.J.; Wiriyakitnateekul, W. Forms and solubility of plant nutrient elements in tropical plant waste biochars. J. Plant Nutr. Soil Sci. 2015, 178, 732–740. [Google Scholar] [CrossRef]
- Kwapinski, W.; Byrne, C.M.P.; Kryachko, E.; Wolfram, P.; Adley, C.; Leahy, J.J.; Novotny, E.H.; Hayes, M.H.B. Biochar from biomass and waste. Waste Biomass Valoriz. 2010, 1, 177–189. [Google Scholar] [CrossRef]
- Nguyen, B.T.; Lehmann, J.; Hockaday, W.C.; Joseph, S.; Masiello, C.A. Temperature sensitivity of black carbon decomposition and oxidation. Environ. Sci. Technol. 2010, 44, 3324–3331. [Google Scholar] [CrossRef] [PubMed]
- Downie, A.; Alan, C.; Paul, M. Physical properties of biochar. In Biochar for Environmental Management: Science and Technology; Earthscan: London, UK, 2009. [Google Scholar]
- Jin, H. Characterization of Microbial Life Colonizing Biochar and Biochar-Amended Soils. Ph.D. Thesis, Cornell University, Ithaca, NY, USA, 2010. [Google Scholar]
- Lehmann, J.; Rillig, M.C.; Thies, J.; Masiello, C.A.; Hockaday, W.C.; Crowleye, D. Biochar effects on soil biota—A review. Soil Biol. Biochem. 2011, 43, 1812–1836. [Google Scholar] [CrossRef]
- Deenik, J.L.; McClellan, T.; Uehara, G.; Antal, M.J.; Campbell, S. Charcoal volatile matter content influences plant growth and soil nitrogen transformations. Soil Sci. Soc. Am. J. 2010, 74, 1259–1270. [Google Scholar] [CrossRef]
- Guizani, C.; Jeguirim, M.; Valin, S.; Limousy, L.; Salvador, S. Biomass Chars: The Effects of Pyrolysis Conditions on Their Morphology, Structure, Chemical Properties and Reactivity. Energies 2017, 10, 796. [Google Scholar] [CrossRef]
- Hossain, M.K.; Strezov, V.; Chan, K.Y.; Ziolkowski, A.; Nelsona, P.F. Influence of pyrolysis temperature on production and nutrient properties of wastewater sludge biochar. J. Environ. Manag. 2011, 92, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Cantrell, K.B.; Hunt, P.G.; Uchimiya, M.; Novak, J.M.; Ro, K.S. Impact of pyrolysis temperature and manure source on physicochemical characteristics of biochar. Bioresour. Technol. 2012, 107, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Selvam, A.; Chan, M.; Wong, J.W.C. Nitrogen conservation and acidity control during food wastes composting through struvite formation. Bioresour. Technol. 2013, 147, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Chan, M.T.; Selvam, A.; Wong, J.W.C. Reducing nitrogen loss and salinity during ‘struvite’ food waste composting by zeolite amendment. Bioresour. Technol. 2016, 200, 838–844. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Environment (MOE). A Study on FoodWaste Reduction Equipment Guidelines and Quality Standard P; Ministry of Environment: Sejong City, Korea, 2009.
- Yang, X.; Wang, H.; Strong, P.J.; Xu, S.; Liu, S.; Lu, K.; Sheng, K.; Guo, J.; Che, L.; He, L.; et al. Thermal Properties of Biochars Derived from Waste Biomass Generated by Agricultural and Forestry Sectors. Energies 2017, 10, 469. [Google Scholar] [CrossRef]
- Quyn, D.M.; Wu, H.; Li, C.-Z. Volatilisation and catalytic effects of alkali and alkaline earth metallic species during the pyrolysis and gasification of Victorian brown coal. Part I. Volatilisation of Na and Cl from a set of NaCl-loaded samples. Fuel 2002, 81, 143–149. [Google Scholar] [CrossRef]
- Korea Rural Development Administration, National Academy of Agricultural Science. A Method of Inspection of Physical and Chemical Fertilizers; Rural Development Administration: Jeonju-si, Korea, 2016.
- Korea Rural Development Administration, National Academy of Agricultural Science. Methods of Soil Chemical Analysis; Rural Development Administration: Jeonju-si, Korea, 2010.
- Zambon, I.; Colosimo, F.; Monarca, D.; Cecchini, M.; Gallucci, F.; Proto, A.R.; Lord, R.; Colantoni, A. An innovative agro-forestry supply chain for residual biomass: Physicochemical characterisation of biochar from olive and hazelnut pellets. Energies 2016, 9, 526. [Google Scholar] [CrossRef] [Green Version]
- Spokas, K.A. Review of the stability of biochar in soils: Predictability of O:C molar ratios. Carbon Manag. 2010, 1, 289–303. [Google Scholar] [CrossRef]
- Mukome, F.N.D.; Zhang, X.; Silva, L.C.R.; Six, J.; Parikh, S.J. Use of chemical and physical characteristics to investigate trends in biochar feedstocks. J. Agric. Food Chem. 2013, 61, 2196–2204. [Google Scholar] [CrossRef] [PubMed]
- Mukome, F.N.D.; Parikh, S.J. Chemical, Physical, and Surface characterization of Biochar. In Biochar: Production, Characterization, and Applications; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Ok, Y.S.; Uchimiya, S.M.; Chang, S.X.; Bolan, N. (Eds.) Biochar: Production, Characterization, and Applications; CRC Press: Boca Raton, FL, USA, 2015. [Google Scholar]
- Yuan, J.-H.; Xu, R.-K.; Zhang, H. The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresour. Technol. 2011, 102, 3488–3497. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Yang, M.; Feng, Q.; McGrouther, K.; Wang, H.; Lu, H.; Chen, Y. Chemical characterization of rice straw-derived biochar for soil amendment. Biomass Bioenergy 2012, 47, 268–276. [Google Scholar] [CrossRef]
- Sohi, S.P.; Krull, E.; Lopez-Capel, E.; Bol, R. A review of biochar and its use and function in soil. Adv. Agron. 2010, 105, 47–82. [Google Scholar]
- Zhao, S.-X.; Ta, N.; Wang, X.-D. Effect of Temperature on the Structural and Physicochemical Properties of Biochar with Apple Tree Branches as Feedstock Material. Energies 2017, 10, 1293. [Google Scholar] [CrossRef]
- Van Zwieten, L.; Kimber, S.; Morris, S.; Chan, K.Y.; Downie, A.; Rust, J.; Joseph, S.; Cowie, A. Effects of biochar from slow pyrolysis of papermill waste on agronomic performance and soil fertility. Plant Soil 2010, 327, 235–246. [Google Scholar] [CrossRef]
- Ketterings, Q.; Reid, S.; Rao, R. Cornell University Agronomy Fact Sheet #22: Cation Exchange Capacity (CEC); Cornell University: Ithaca, NY, USA, 2007. [Google Scholar]
- Bourke, J.; Manley-Harris, M.; Fushimi, C.; Dowaki, K.; Nunoura, T.; Antal, M.J., Jr. Do all carbonized charcoals have the same chemical structure? 2. A model of the chemical structure of carbonized charcoal. Ind. Eng. Chem. Res. 2007, 46, 5954–5967. [Google Scholar] [CrossRef]
- Guerrero, M.; Ruiz, M.P.; Millera, Á.; Alzueta, M.U.; Bilbao, R. Characterization of biomass chars formed under different devolatilization conditions: Differences between rice husk and eucalyptus. Energy Fuels 2008, 22, 1275–1284. [Google Scholar] [CrossRef]
- Keiluweit, M.; Nico, P.S.; Johnson, M.G.; Kleber, M. Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Herbert, L.; Hosek, I.; Kripalani, R. The Characterization and Comparison of Biochar Produced from a Decentralized Reactor Using Forced Air and Natural Draft Pyrolysis; California Polytechnic State University: San Luis Obispo, CA, USA, 2012. [Google Scholar]
- Cheng, C.-H.; Lehmann, J.; Thies, J.E.; Burton, S.D.; Engelhardb, M.H. Oxidation of black carbon by biotic and abiotic processes. Org. Geochem. 2006, 37, 1477–1488. [Google Scholar] [CrossRef]
- Boeriu, C.G.; Bravo, D.; Gosselink, R.J.A.; van Dam, J.E.G. Characterisation of structure-dependent functional properties of lignin with infrared spectroscopy. Ind. Crop. Prod. 2004, 20, 205–218. [Google Scholar] [CrossRef]
- Kačuráková, M.; Wilson, R.H. Developments in mid-infrared FT-IR spectroscopy of selected carbohydrates. Carbohydr. Polym. 2001, 44, 291–303. [Google Scholar] [CrossRef]
- Lammers, K.; Arbuckle-Keil, G.; Dighton, J. FT-IR study of the changes in carbohydrate chemistry of three New Jersey pine barrens leaf litters during simulated control burning. Soil Biol. Biochem. 2009, 41, 340–347. [Google Scholar] [CrossRef]
- Kubo, S.; Kadla, J.F. Hydrogen bonding in lignin: A Fourier transform infrared model compound study. Biomacromolecules 2005, 6, 2815–2821. [Google Scholar] [CrossRef] [PubMed]
- Parimala, K.; Balachandran, V. Structural study, NCA, FT-IR, FT-Raman spectral investigations, NBO analysis and thermodynamic properties of 2′, 4′-difluoroacetophenone by HF and DFT calculations. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 110, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Porchelvi, E.E.; Muthu, S. The spectroscopic (FT-IR, FT-Raman and NMR), NCA, Fukui function analysis first order hyperpolarizability, TGA of 6-chloro-3, 4dihydro-2H-1, 2, 4-benzothiazine-7-sulphonamide1, 1-dioxide by ab initio HF and Density Functional method. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 123, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Mcbeath, A.V.; Smernik, R.J.; Krull, E.S.; Lehmann, J. The influence of feedstock and production temperature on biochar carbon chemistry: A solid-state 13 C NMR study. Biomass Bioenergy 2014, 60, 121–129. [Google Scholar] [CrossRef]
- Knicker, H.; Hilscher, A.; González-Vila, F.J.; Almendros, G. A new conceptual model for the structural properties of char produced during vegetation fires. Org. Geochem. 2008, 39, 935–939. [Google Scholar] [CrossRef]
- Brewer, C.E.; Unger, R.; Schmidt-Rohr, K.; Brown, R.C. Criteria to select biochars for field studies based on biochar chemical properties. BioEnergy Res. 2011, 4, 312–323. [Google Scholar] [CrossRef]
| Classification | Composition Ratio (wt %) | Methods of Food Ingredient Processing | |
|---|---|---|---|
| Food Ingredients | Processing Method | ||
| Grains | 16 | Rice (16) | |
| Vegetables | 51 | Napa cabbage (9) | Cutting width less than 100 mm. |
| Potato (20) | Chop into 5 mm size pieces. | ||
| Onion (20) | |||
| Daikon (2) | |||
| Fruits | 14 | Apple (7) | Split into 8 pieces in lengthwise. |
| Mandarin/Orange (7) | |||
| Meat and Fish | 19 | Meat (4) | Cutting width around 3 cm. |
| Fish (12) | Split into 4 pieces. | ||
| Egg shell (3) | |||
| Total | 100 | 100 | |
| Sample ID | Component, wt % | Atomic Ratio | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| C | H | N | O | Na | Cl | Ash | C/N | H/C | O/C | |
| Dry | 51.46 | 13.22 | 3.14 | 32.18 | - | - | - | 16.38 | 0.257 | 0.63 |
| 300 °C_STD | 60.97 | 5.37 | 5.28 | 23.66 | 1.21 | 1.56 | 4.72 | 11.55 | 0.088 | 0.39 |
| 300 °C_1%_B | 60.18 | 5.32 | 5.29 | 25.88 | 3.08 | 3.17 | 3.33 | 11.38 | 0.088 | 0.43 |
| 300 °C_1%_A | 61.70 | 5.59 | 5.24 | 25.19 | 2.02 | 1.51 | 2.28 | 11.77 | 0.091 | 0.41 |
| 300 °C_3%_B | 58.96 | 5.17 | 5.09 | 22.53 | 6.63 | 6.26 | 8.25 | 11.58 | 0.088 | 0.38 |
| 300 °C_3%_A | 63.21 | 5.46 | 5.16 | 24.34 | 2.67 | 1.50 | 1.83 | 12.25 | 0.086 | 0.39 |
| 300°C_5%_B | 57.29 | 5.04 | 4.98 | 20.83 | 9.30 | 8.74 | 11.86 | 11.5 | 0.088 | 0.36 |
| 300 °C_5%_A | 62.08 | 5.81 | 5.42 | 23.03 | 2.62 | 1.50 | 3.66 | 11.45 | 0.094 | 0.37 |
| 400 °C_STD | 64.69 | 3.64 | 5.09 | 17.82 | 1.92 | 1.90 | 8.76 | 12.71 | 0.056 | 0.28 |
| 400 °C_1%_B | 63.27 | 3.60 | 5.38 | 17.98 | 4.74 | 4.18 | 9.77 | 11.76 | 0.057 | 0.28 |
| 400 °C_1%_A | 68.71 | 4.28 | 5.04 | 17.60 | 4.01 | 1.20 | 4.37 | 13.63 | 0.062 | 0.26 |
| 400 °C_3%_B | 57.85 | 3.24 | 4.16 | 21.08 | 9.22 | 8.25 | 13.67 | 13.91 | 0.056 | 0.36 |
| 400 °C_3%_A | 65.43 | 3.70 | 5.26 | 20.94 | 3.43 | 1.19 | 4.67 | 12.44 | 0.057 | 0.32 |
| 400 °C_5%_B | 56.15 | 3.20 | 3.99 | 18.74 | 16.53 | 12.66 | 17.92 | 14.07 | 0.057 | 0.33 |
| 400 °C_5%_A | 68.43 | 4.34 | 4.51 | 19.47 | 4.64 | 1.36 | 3.25 | 15.17 | 0.063 | 0.28 |
| 500 °C_STD | 70.11 | 2.81 | 5.23 | 8.83 | 0.51 | 0.90 | 13.02 | 13.41 | 0.04 | 0.13 |
| 500 °C_1%_B | 68.82 | 2.71 | 5.22 | 10.45 | 3.98 | 3.74 | 12.80 | 13.18 | 0.039 | 0.15 |
| 500 °C_1%_A | 72.07 | 2.93 | 5.13 | 9.43 | 3.43 | 1.03 | 10.44 | 14.05 | 0.041 | 0.13 |
| 500 °C_3%_B | 65.72 | 2.69 | 4.63 | 10.33 | 9.65 | 9.42 | 16.63 | 14.19 | 0.041 | 0.16 |
| 500 °C_3%_A | 72.10 | 2.98 | 5.13 | 11.32 | 5.14 | 1.49 | 8.47 | 14.05 | 0.041 | 0.16 |
| 500 °C_5%_B | 61.78 | 2.43 | 4.44 | 10.50 | 15.56 | 13.80 | 20.85 | 13.91 | 0.039 | 0.17 |
| 500 °C_5%_A | 73.28 | 2.92 | 4.92 | 11.80 | 5.35 | 1.45 | 7.08 | 14.89 | 0.04 | 0.16 |
| Sample ID | CEC (cmol/kg) | Extractable Cations (cmolc/kg) | |||
|---|---|---|---|---|---|
| Ca | K | Mg | Na | ||
| 300 °C_STD | 25.57 | 0.39 | 12.68 | 0.34 | 5.56 |
| 300 °C_1%_B | 22.20 | 0.29 | 7.77 | 0.12 | 6.99 |
| 300 °C_1%_A | 18.14 | 0.45 | 4.95 | 0.15 | 4.67 |
| 300 °C_3%_B | 22.42 | 0.17 | 4.06 | 0.06 | 11.08 |
| 300 °C_3%_A | 20.35 | 0.47 | 3.99 | 0.16 | 8.04 |
| 300 °C_5%_B | 110.71 | 0.35 | 16.59 | 0.24 | 86.26 |
| 300 °C_5%_A | 22.67 | 0.58 | 3.89 | 0.22 | 10.07 |
| 400 °C_STD | 62.94 | 1.10 | 44.07 | 1.37 | 14.21 |
| 400 °C_1%_B | 120.39 | 1.19 | 62.59 | 1.15 | 49.98 |
| 400 °C_1%_A | 36.65 | 0.90 | 18.00 | 0.64 | 12.71 |
| 400 °C_3%_B | 163.48 | 0.96 | 52.17 | 1.23 | 108.91 |
| 400 °C_3%_A | 52.60 | 1.30 | 16.17 | 1.00 | 24.68 |
| 400 °C_5%_B | 250.72 | 0.65 | 53.19 | 1.29 | 185.03 |
| 400 °C_5%_A | 39.46 | 1.06 | 9.90 | 0.86 | 22.80 |
| 500 °C_STD | 54.91 | 0.89 | 60.38 | 1.58 | 1.07 |
| 500 °C_1%_B | 75.22 | 0.83 | 62.37 | 1.10 | 14.88 |
| 500 °C_1%_A | 24.79 | 1.07 | 18.23 | 0.68 | 7.67 |
| 500 °C_3%_B | 156.81 | 0.76 | 67.59 | 1.22 | 92.30 |
| 500 °C_3%_A | 18.90 | 1.29 | 10.63 | 1.00 | 9.27 |
| 500 °C_5%_B | 262.08 | 0.63 | 73.55 | 1.31 | 189.90 |
| 500 °C_5%_A | 24.86 | 1.34 | 10.29 | 1.07 | 14.58 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.-E.; Jo, J.-H.; Kim, I.-T.; Yoo, Y.-S. Chemical Characteristics and NaCl Component Behavior of Biochar Derived from the Salty Food Waste by Water Flushing. Energies 2017, 10, 1555. https://doi.org/10.3390/en10101555
Lee Y-E, Jo J-H, Kim I-T, Yoo Y-S. Chemical Characteristics and NaCl Component Behavior of Biochar Derived from the Salty Food Waste by Water Flushing. Energies. 2017; 10(10):1555. https://doi.org/10.3390/en10101555
Chicago/Turabian StyleLee, Ye-Eun, Jun-Ho Jo, I-Tae Kim, and Yeong-Seok Yoo. 2017. "Chemical Characteristics and NaCl Component Behavior of Biochar Derived from the Salty Food Waste by Water Flushing" Energies 10, no. 10: 1555. https://doi.org/10.3390/en10101555




