Chitosan Composites for Bone Tissue Engineering—An Overview
Abstract
:1. Introduction
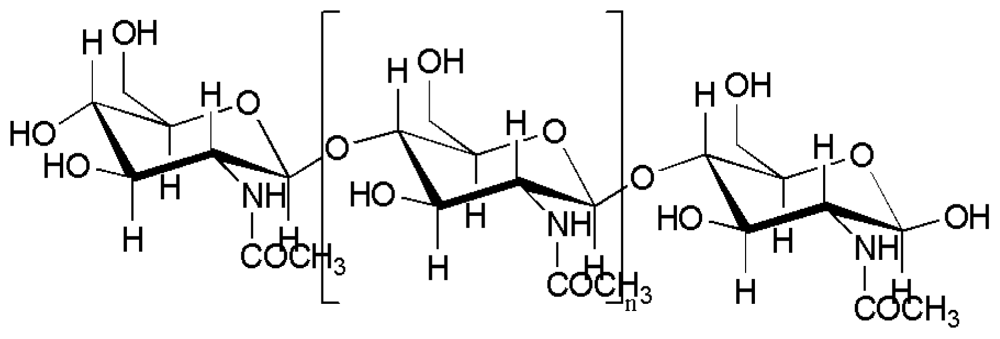
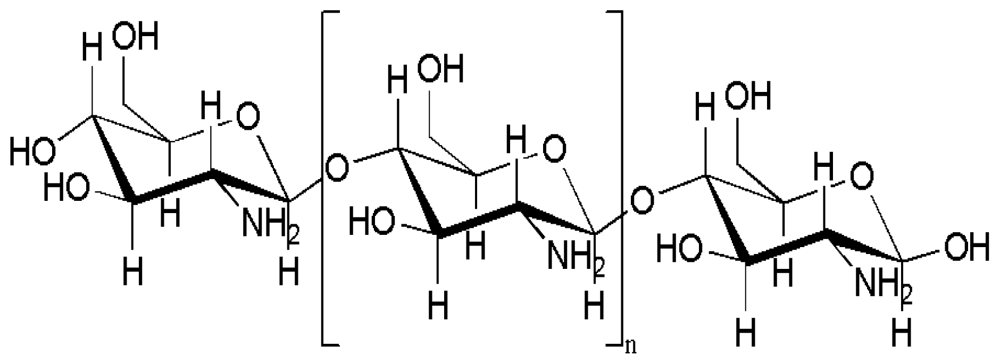
2. Chitosan for Bone Tissue Engineering
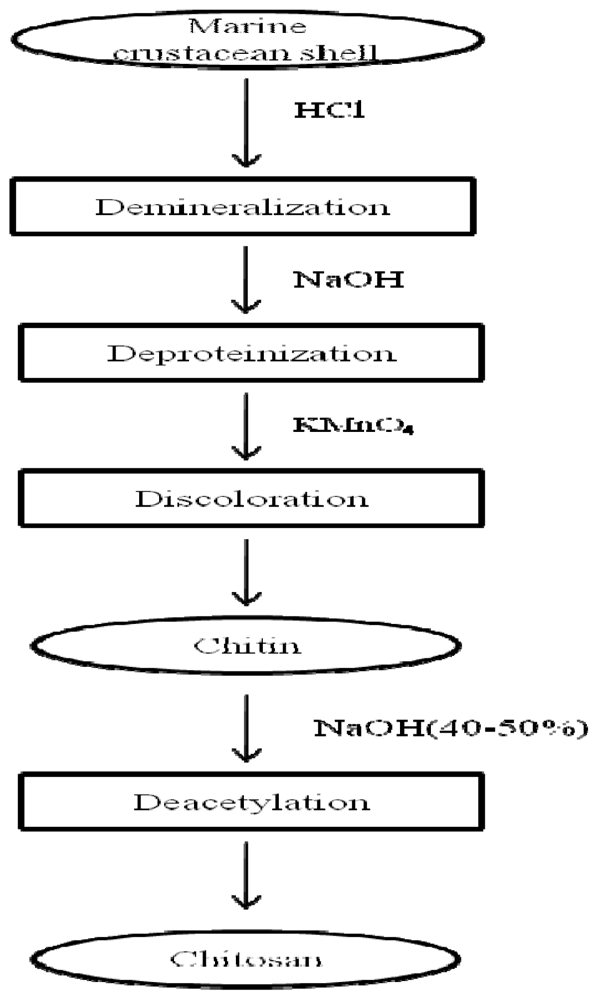
2.1. Preparation of chitosan by chemical methods
2.2. Enzymatic hydrolysis method
2.3. Properties and application of chitosan
3. Composite Materials for Bone Tissue Engineering
3.1. Preparation of CTS/HAp composite materials
3.2. Mechanical properties of CTS/HAp composite
3.3. In vitro study of CTS/HAp composites
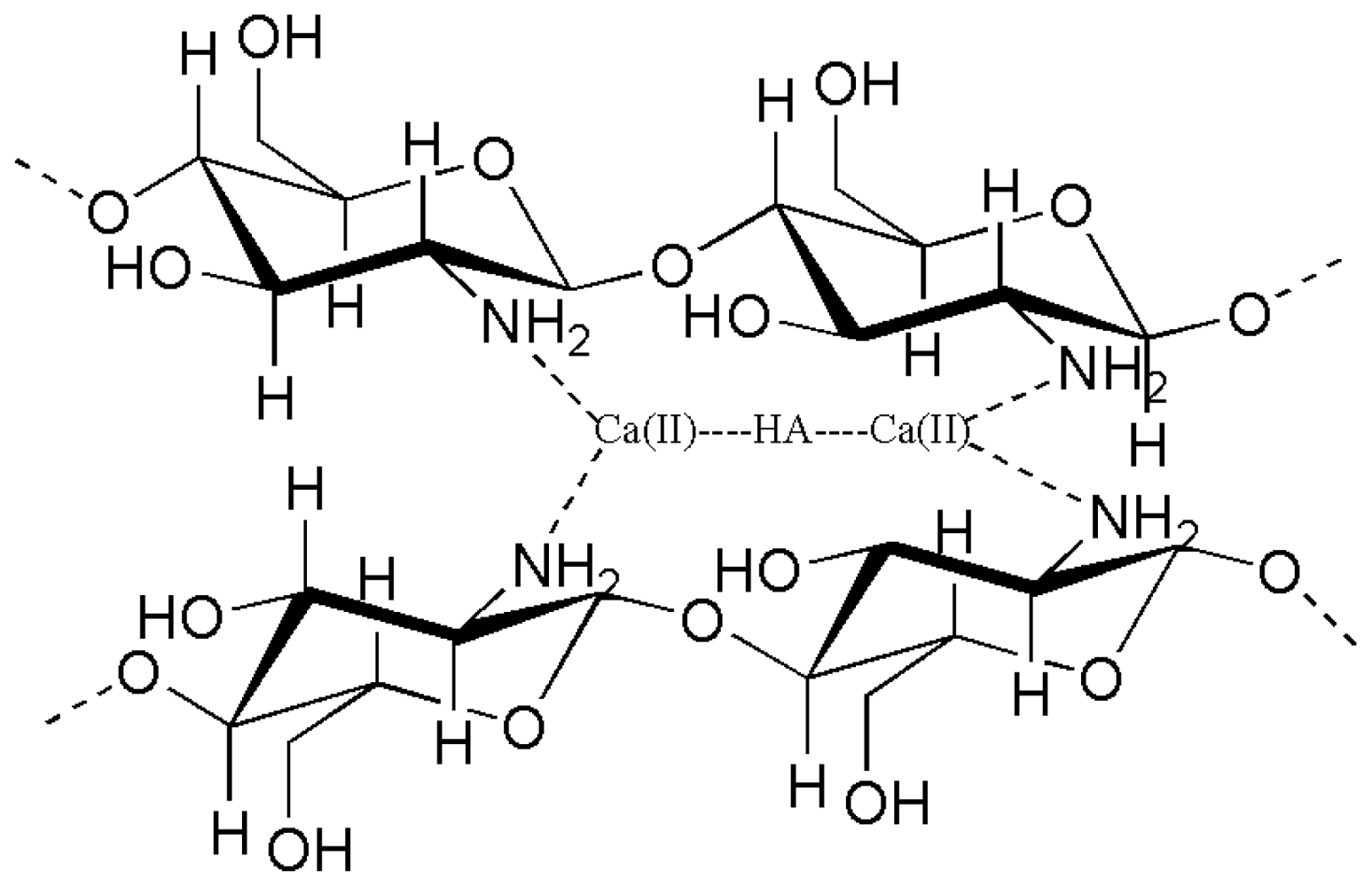
3.4. Chemical interaction of CTS/HAp composites
4. Carbon Nano Tubes for Bone Tissue Engineering
4.1. Preparation of CTS/CNT composite
4.2. Mechanical properties of CTS/CNT composite
4.3. In vitro study of CTS/CNT composites
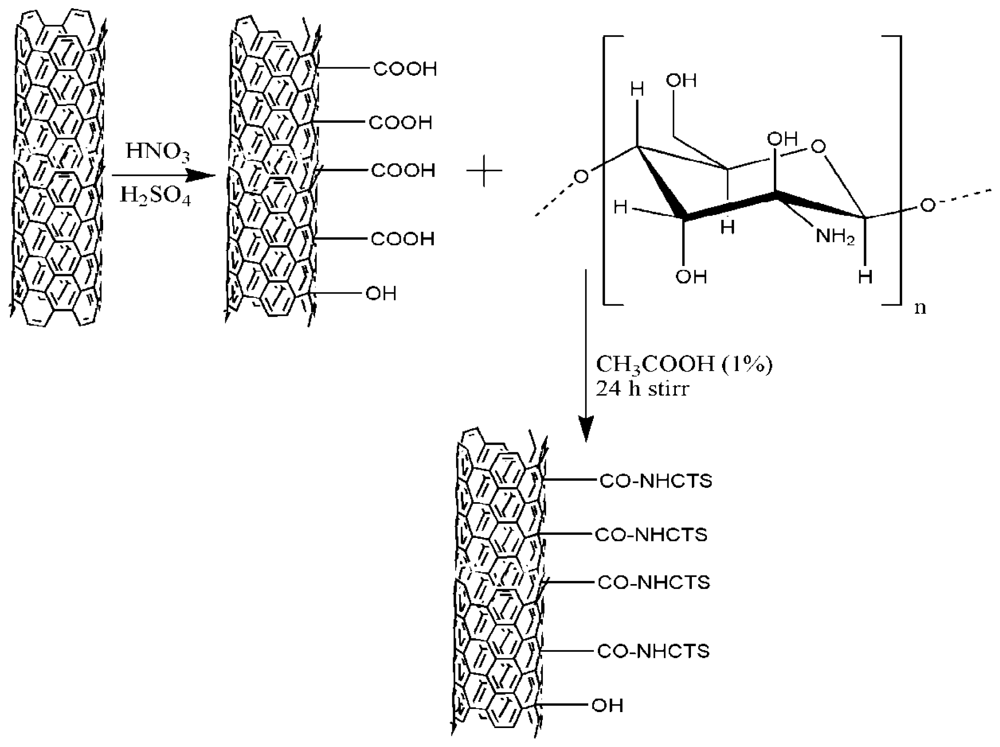
4.4. Chemical interactions between CTS/CNT
5. Remaining Challenges and Future Directions for Bone Biomaterials
6. Conclusions
Acknowledgements
References
- Pizzoferrato, A; Cenni, E; Ciapetti, G; Granchi, D; Savarino, L; Stea, S. Inflammatory response to metals and ceramics. In Integrated Biomaterials Science; Kluwer Academic/Plenum Publishers: New York, NY, USA, 2002; pp. 735–791. [Google Scholar]
- Wagh, A. Chemically Bonded Phosphate Ceramics: Twenty-First Century Materials with Diverse Applications; Elsevier Science: New York, NY, USA, 2004. [Google Scholar]
- Giannoudis, P; Dinopoulos, H; Tsiridis, E. Bone substitutes: an update. Injury 2005, 36, 20–27. [Google Scholar]
- Wang, M. Developing bioactive composite materials for tissue replacement. Biomaterials 2003, 24, 2133–2151. [Google Scholar]
- Iwata, H; Yana, S; Nasu, M; Yosue, T. Effects of chitosan oligosaccharides on the femur trabecular structure in ovariectomized rats. Oral Radiol 2005, 21, 19–22. [Google Scholar]
- Porporatto, C; Canali, MM; Bianco, ID; Correa, SG. The biocompatible polysaccharide chitosan enhances the oral tolerance to type II collagen. Clin. Exp. Immunol 2009, 155, 79–87. [Google Scholar]
- Murakami, K; Aoki, H; Nakamura, S; Nakamura, S-I; Takikawa, M; Hanzawa, M; Kishimoto, S; Hattori, H; Tanaka, Y; Kiyosawa, T; Sato, Y; Ishihara, M. Hydrogel blends of chitin/chitosan, fucoidan and alginate as healing impaired wound dressings. Biomaterials 2010, 31, 83–90. [Google Scholar]
- Madhavan, P; Nair, K. Utilization of prawn waste: Isolation of chitin and its conversion to chitosan. Fish. Technol 1974, 11, 50–53. [Google Scholar]
- Shahidi, F; Abuzaytoun, R. Chitin, chitosan, and co products: chemistry, production, applications, and health effects. Adv. Food Nutr. Res 2005, 49, 93–135. [Google Scholar]
- Cai, J; Yang, J; Du, Y; Fan, L; Qiu, Y; Li, J; Kennedy, JF. Enzymatic preparation of chitosan from the waste Aspergillus niger mycelium of citric acid production plant. Carbohydr. Polymers 2006, 64, 151–157. [Google Scholar]
- Kim, S-K; Rajapakse, N. Enzymatic production and biological activities of chitosan oligosaccharides (COS): A review. Carbohydr. Polymers 2005, 62, 357–368. [Google Scholar]
- No, HK; Meyers, SP. Preparation and characterization of chitin and chitosan—A Review. J. Aquat. Food Prod. Technol 1995, 4, 27–52. [Google Scholar]
- Je, J; Kim, S. Water soluble chitosan derivatives as a BACE1 inhibitor. Bioorg. Med. Chem 2005, 13, 6551–6555. [Google Scholar]
- Jeon, Y; Shahidi, F; Kim, S. Preparation of chitin and chitosan oligomers and their applications in physiological functional foods. Food Rev. Int 2000, 16, 159–176. [Google Scholar]
- Di Martino, A; Sittinger, M; Risbud, MV. Chitosan: A versatile biopolymer for orthopaedic tissue-engineering. Biomaterials 2005, 26, 5983–5990. [Google Scholar]
- Francis Suh, J; Matthew, H. Application of chitosan-based polysaccharide biomaterials in cartilage tissue engineering: a review. Biomaterials 2000, 21, 2589–2598. [Google Scholar]
- Ueno, H; Mori, T; Fujinaga, T. Topical formulations and wound healing applications of chitosan. Adv. Drug Delivery Rev 2001, 52, 105–115. [Google Scholar]
- Hu, Q; Li, B; Wang, M; Shen, J. Preparation and characterization of biodegradable chitosan/hydroxyapatite nanocomposite rods via in situ hybridization: a potential material as internal fixation of bone fracture. Biomaterials 2004, 25, 779–785. [Google Scholar]
- Madihally, S; Matthew, H. Porous chitosan scaffolds for tissue engineering. Biomaterials 1999, 20, 1133–1142. [Google Scholar]
- Austin, K. Scaffold Design: Use of Chitosan in cartilage tissue engineering. MMG 445 Basic Biotech. eJ 2007, 3, 62. [Google Scholar]
- Teng, S; Lee, E; Yoon, B; Shin, D; Kim, H; Oh, J. Chitosan/nanohydroxyapatite composite membranes via dynamic filtration for guided bone regeneration. J. Biomed. Mater. Res. Part A 2009, 88, 569–580. [Google Scholar]
- Chen, F; Wang, Z; Lin, C. Preparation and characterization of nano sized hydroxyapatite particles and hydroxyapatite/chitosan nano-composite for use in biomedical materials. Mater. Lett 2002, 57, 858–861. [Google Scholar]
- Zhang, Y; Venugopal, JR; El-Turki, A; Ramakrishna, S; Su, B; Lim, CT. Electrospun biomimetic nanocomposite nanofibers of hydroxyapatite/chitosan for bone tissue engineering. Biomaterials 2008, 29, 4314–4322. [Google Scholar]
- Xianmiao, C; Yubao, L; Yi, Z; Li, Z; Jidong, L; Huanan, W. Properties and in vitro biological evaluation of nano-hydroxyapatite/chitosan membranes for bone guided regeneration. Mater. Sci. Eng. C 2009, 29, 29–35. [Google Scholar]
- Kong, L; Gao, Y; Cao, W; Gong, Y; Zhao, N; Zhang, X. Preparation and characterization of nano-hydroxyapatite/chitosan composite scaffolds. J. Biomed. Mater. Res. Part A 2005, 75A, 275–282. [Google Scholar]
- Manjubala, I; Ponomarev, I; Wilke, I; Jandt, K. Growth of osteoblast like cells on biomimetic apatite-coated chitosan scaffolds. J. Biomed. Mater. Res. Part A: Appl. Biomater 2008, 8, 7–16. [Google Scholar]
- Thein-Han, W; Misra, R. Biomimetic chitosan–nanohydroxyapatite composite scaffolds for bone tissue engineering. Acta Biomater 2009, 5, 1182–1197. [Google Scholar]
- Oliveira, J; Rodrigues, M; Silva, S; Malafaya, P; Gomes, M; Viegas, C; Dias, I; Azevedo, J; Mano, J; Reis, R. Novel hydroxyapatite/chitosan bilayered scaffold for osteochondral tissue-engineering applications: Scaffold design and its performance when seeded with goat bone marrow stromal cells. Biomaterials 2006, 27, 6123–6137. [Google Scholar] [Green Version]
- Ding, S. Biodegradation behavior of chitosan/calcium phosphate composites. J. Non-Cryst. Solids 2007, 353, 2367–2373. [Google Scholar]
- Yamaguchi, I; Tokuchi, K; Fukuzaki, H; Koyama, Y; Takakuda, K; Monma, H; Tanaka, J. Preparation and microstructure analysis of chitosan/hydroxyapatite nanocomposites. J. Biomed. Mater. Res 2001, 55, 20–27. [Google Scholar]
- Kim, S-K; Mendis, E. Bioactive compounds from marine processing byproducts-A review. Food Res. Int 2006, 39, 383–393. [Google Scholar]
- Nath, S; Dey, A; Mukhopadhyay, AK; Basu, B. Nanoindentation response of novel hydroxyapatite-mullite composites. Mater. Sci. Eng. A 2009, 513–514, 197–201. [Google Scholar]
- Wang, X; Tan, Y; Zhang, B; Gu, Z; Li, X. Synthesis and evaluation of collagen-chitosan-hydroxyapatite nanocomposites for bone grafting. J. Biomed. Mater. Res. Part A 2009, 89, 1079–1087. [Google Scholar]
- Wang, X; Ma, J; Wang, Y; He, B. Bone repair in radii and tibias of rabbits with phosphorylated chitosan reinforced calcium phosphate cements. Biomaterials 2002, 23, 4167–4176. [Google Scholar]
- Kuo, Y; Lin, C. Effect of genipin-crosslinked chitin-chitosan scaffolds with hydroxyapatite modifications on the cultivation of bovine knee chondrocytes. Biotechnol. Bioeng 2006, 95, 132–144. [Google Scholar]
- Pena, J; Izquierdo-Barba, I; García, M; Vallet-Regí, M. Room temperature synthesis of chitosan/apatite powders and coatings. J. Eur. Ceram. Soc 2006, 26, 3631–3638. [Google Scholar]
- Madhumathi, K; Shalumon, K; Rani, V; Tamura, H; Furuike, T; Selvamurugan, N; Nair, S; Jayakumar, R. Wet chemical synthesis of chitosan hydrogel-hydroxyapatite composite membranes for tissue engineering applications. Int. J. Biol. Macromol 2009, 45, 12–15. [Google Scholar]
- Liuyun, J; Yubao, L; Chengdong, X. A novel composite membrane of chitosan-carboxymethyl cellulose polyelectrolyte complex membrane filled with nano-hydroxyapatite I. Preparation and properties. J. Mater. Sci.: Mater. Med 2009, 20, 1645–1652. [Google Scholar]
- Li, Q; Chen, Z; Darvell, B; Zeng, Q; Li, G; Ou, G; Wu, M. Biomimetic synthesis of the composites of hydroxyapatite and chitosan–phosphorylated chitosan polyelectrolyte complex. Mater. Lett 2006, 60, 3533–3536. [Google Scholar]
- Verma, D; Katti, K; Katti, D. Effect of Biopolymers on structure of hydroxyapatite and interfacial interactions in biomimetically synthesized hydroxyapatite/biopolymer nanocomposites. Ann. Biomed. Eng 2008, 36, 1024–1032. [Google Scholar]
- Davidenko, N; Carrodeguas, R; Peniche, C; Solís, Y; Cameron, R. Chitosan/apatite composite beads prepared by in situ generation of apatite or Si-apatite nanocrystals. Acta Biomater 2010, 6, 466–476. [Google Scholar]
- Murugan, R; Ramakrishna, S. Bioresorbable composite bone paste using polysaccharide based nano hydroxyapatite. Biomaterials 2004, 25, 3829–3835. [Google Scholar]
- Zhang, Y; Zhang, M. Cell growth and function on calcium phosphate reinforced chitosan scaffolds. J. Mater. Sci.: Mater. Med 2004, 15, 255–260. [Google Scholar]
- Ehrlich, H; Krajewska, B; Hanke, T; Born, R; Heinemann, S; Knieb, C; Worch, H. Chitosan membrane as a template for hydroxyapatite crystal growth in a model dual membrane diffusion system. J. Membr. Sci 2006, 273, 124–128. [Google Scholar]
- Redepenning, J; Venkataraman, G; Chen, J; Stafford, N. Electrochemical preparation of chitosan/hydroxyapatite composite coatings on titanium substrates. J. Biomed. Mater. Res 2003, 66, 411–416. [Google Scholar]
- Pang, X; Zhitomirsky, I. Electrodeposition of composite hydroxyapatite–chitosan films. Mater. Chem. Phys 2005, 94, 245–251. [Google Scholar]
- Huang, Z; Dong, Y; Chu, C; Lin, P. Electrochemistry assisted reacting deposition of hydroxyapatite in porous chitosan scaffolds. Mater. Lett 2008, 62, 3376–3378. [Google Scholar]
- Pang, X; Casagrande, T; Zhitomirsky, I. Electrophoretic deposition of hydroxyapatite–CaSiO3– chitosan composite coatings. J. Colloid Interface Sci 2009, 330, 323–329. [Google Scholar]
- Pang, X; Zhitomirsky, I. Electrophoretic deposition of composite hydroxyapatite-chitosan coatings. Mater. Charact 2007, 58, 339–348. [Google Scholar]
- Yuan, H; Chen, N; Lü, X; Zheng, B. Experimental study of natural hydroxyapatite/chitosan composite on reconstructing bone defects. J. Nanjing Med. Univ 2008, 22, 372–375. [Google Scholar]
- Manjubala, I; Scheler, S; Bössert, J; Jandt, K. Mineralisation of chitosan scaffolds with nano-apatite formation by double diffusion technique. Acta Biomater 2006, 2, 75–84. [Google Scholar]
- Matsuda, A; Ikoma, T; Kobayashi, H; Tanaka, J. Preparation and mechanical property of core-shell type chitosan/calcium phosphate composite fiber. Mater. Sci. Eng. C 2004, 24, 723–728. [Google Scholar]
- Li, Z; Yubao, L; Aiping, Y; Xuelin, P; Xuejiang, W; Xiang, Z. Preparation and in vitro investigation of chitosan/nano-hydroxyapatite composite used as bone substitute materials. J. Mater. Sci. Mater. Med 2005, 16, 213–219. [Google Scholar]
- Li, B; Hu, Q; Qian, X; Fang, Z; Shen, J. Bioabsorbable chitosan/hydroxyapatite composite rod for internal fixation of bone fracture prepared by in situ precipitation. Acta Polym. Sin 2002, 6, 828–833. [Google Scholar]
- Aronow, M; Gerstenfeld, L; Owen, T; Tassinari, M; Stein, G; Lian, J. Factors that promote progressive development of the osteoblast phenotype in cultured fetal rat calvaria cells. J. Cell. Physiol 1990, 143, 213–221. [Google Scholar]
- Li, Q; Wu, M; Tang, L. Bioactivity of a Novel Nano composite of hydroxyapatite and chitosan phosphorylated chitosan polyelectrolyte complex. J. Bioact. Compat. Polym 2008, 23, 520. [Google Scholar]
- Xu, H; Simon, C. Fast setting calcium phosphate–chitosan scaffold: mechanical properties and biocompatibility. Biomaterials 2005, 26, 1337–1348. [Google Scholar]
- Zhang, Y; Ni, M; Zhang, M; Ratner, B. Calcium phosphate chitosan composite scaffolds for bone tissue engineering. Tissue Eng 2003, 9, 337–345. [Google Scholar]
- Mukherjee, D; Tunkle, A; Roberts, R; Clavenna, A; Rogers, S; Smith, D. An animal evaluation of a paste of chitosan glutamate and hydroxyapatite as a synthetic bone graft material. J. Biomed. Mater. Res. Part B: Appl. Biomater 2003, 67, 603–609. [Google Scholar]
- Kikuchi, M; Ikoma, T; Itoh, S; Matsumoto, H; Koyama, Y; Takakuda, K; Shinomiya, K; Tanaka, J. Biomimetic synthesis of bone like nanocomposites using the self-organization mechanism of hydroxyapatite and collagen. Compos. Sci. Technol 2004, 64, 819–825. [Google Scholar]
- Ajayan, P; Zhou, O. Applications of carbon nanotubes. Carbon Nanotubes 2001, 80, 391–425. [Google Scholar]
- Samal, S; Bal, S. Carbon Nanotube Reinforced Ceramic Matrix Composites-A Review. J. Miner. Mater. Charact. Eng 2008, 7, 355–370. [Google Scholar]
- Wang, SF; Shen, L; Zhang, WD; Tong, YJ. Preparation and Mechanical Properties of Chitosan/Carbon Nanotubes Composites. Biomacromolecules 2005, 6, 3067–3072. [Google Scholar]
- Usui, Y; Aoki, K; Narita, N; Murakami, N; Nakamura, I; Nakamura, K; Ishigaki, N; Yamazaki, H; Horiuchi, H; Kato, H. Carbon nanotubes with high bone-tissue compatibility and bone-formation acceleration effects. Small 2008, 4, 240. [Google Scholar]
- Terada, M; Abe, S; Akasaka, T; Uo, M; Kitagawa, Y; Watari, F. Development of a multiwalled carbon nanotube coated collagen dish. Dental Mater. J 2009, 28, 82–88. [Google Scholar]
- Liu, Y; Tang, J; Chen, X; Xin, JH. Decoration of carbon nanotubes with chitosan. Carbon 2005, 43, 3178–3180. [Google Scholar]
- Lau, C; Cooney, MJ; Atanassov, P. Conductive macroporous composite chitosan carbon nanotube scaffolds. Langmuir 2008, 24, 7004–7010. [Google Scholar]
- Qiu, JD; Xie, HY; Liang, RP. Preparation of porous chitosan/carbon nanotubes film modified electrode for biosensor application. Microchim. Acta 2008, 162, 57–64. [Google Scholar]
- Kumar, B; Feller, J-F; Castro, M; Lu, J. Conductive bio polymer nano composites (CPC): Chitosan carbon nanotube transducers assembled via spray layer by layer for volatile organic compound sensing. Talanta 2010, 81, 908–915. [Google Scholar]
- Ozarkar, S; Jassal, M; Agrawal, A. pH and electrical actuation of single walled carbon nanotube/chitosan composite fibers. Smart Mater. Struct 2008, 17, 055016. [Google Scholar]
- Spinks, GM; Shin, SR; Wallace, GG; Whitten, PG; Kim, SI; Kim, SJ. Mechanical properties of chitosan/CNT microfibers obtained with improved dispersion. Sens. Actuat. B: Chem 2006, 115, 678–684. [Google Scholar]
- Zanello, LP; Zhao, B; Hu, H; Haddon, RC. Bone cell proliferation on carbon nanotubes. Nano Lett 2006, 6, 562–567. [Google Scholar]
- Zhang, X; Meng, L; Lu, Q. Cell Behaviors on polysaccharide wrapped single wall carbon nanotubes: A quantitative study of the surface properties of biomimetic nanofibrous scaffolds. ACS Nano 2009, 3, 3200–3206. [Google Scholar]
- Wörle-Knirsch, JM; Pulskamp, K; Krug, HF. Oops they did it again! carbon nanotubes hoax scientists in viability assays. Nano Lett 2006, 6, 1261–1268. [Google Scholar]
- Spitalsky, Z; Tasis, D; Papagelis, K; Galiotis, C. Carbon nanotube polymer composites: Chemistry, processing, mechanical and electrical properties. Prog. Polym. Sci 2010, 35, 357–401. [Google Scholar]
- Ashok Kumar, N; Ganapathy, HS; Kim, JS; Jeong, YS; Jeong, YT. Preparation of poly 2-hydroxyethyl methacrylate functionalized carbon nanotubes as novel biomaterial nanocomposites. Eur. Polym. J 2008, 44, 579–586. [Google Scholar]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Venkatesan, J.; Kim, S.-K. Chitosan Composites for Bone Tissue Engineering—An Overview. Mar. Drugs 2010, 8, 2252-2266. https://doi.org/10.3390/md8082252
Venkatesan J, Kim S-K. Chitosan Composites for Bone Tissue Engineering—An Overview. Marine Drugs. 2010; 8(8):2252-2266. https://doi.org/10.3390/md8082252
Chicago/Turabian StyleVenkatesan, Jayachandran, and Se-Kwon Kim. 2010. "Chitosan Composites for Bone Tissue Engineering—An Overview" Marine Drugs 8, no. 8: 2252-2266. https://doi.org/10.3390/md8082252
APA StyleVenkatesan, J., & Kim, S.-K. (2010). Chitosan Composites for Bone Tissue Engineering—An Overview. Marine Drugs, 8(8), 2252-2266. https://doi.org/10.3390/md8082252




