Is Yessotoxin the Main Phycotoxin in Croatian Waters?
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Sampling
3.2. Mouse Bio-assay (MBA)
3.2.1. Sample Preparation
3.2.2. Extraction Procedure
3.3. Enzyme-Linked Immunosorbent Assay Method (ELISA) for the Determination of Yessotoxin (Ytx)
3.4. Liquid Chromatography-Mass Spectrometry (LC-MS/MS)
3.4.1. Chemicals and reagents
3.4.2. Alkaline Hydrolysis
3.5. Phytoplankton Analysis
4. Conclusions
Acknowledgements
- Samples Availability: Available from the authors.
References and Notes
- Sournia, A; Chretiennot-Dinet, MJ; Ricard, M. Marine phytoplankton: How many species in the world ocean. J Plankon Res 1991, 13, 1093–1099. [Google Scholar]
- Hallegraeff, GM. Harmful algal blooms: a global overview. In Manual on Harmful Marine Microalgae; Hallegraeff, GM, Anderson, DM, Cembella, AD, Eds.; UNESCO: Paris, France, 2003; pp. 25–49. [Google Scholar]
- Ninčević-Gladan, Ž; Marasović, I; Skejić, S; Bužančić, M; Ujević, I; Arapov, J. Harmful algal bloom monitoring program at the shell-fish breading areas through the eastern Adriatic coast (an overview). In Book of Proceedings 2nd Congress of the Alps-Adria Working Community on Maritime, Undersea, and Hyperbaric Medicine; Petri, N, Ed.; Naval Medical Institute of the Croatian Navy: Split, Croatia, 2006; pp. 145–156. [Google Scholar]
- Marasović, I; Ninčević-Gladan, Ž; Skejić, S; Bužančić, M; Ujević, I. Temporal distribution of Dinophysis spp. in relation with DSP shellfish toxicity. J Environ Health 2007, 1, 493–506. [Google Scholar]
- Boni, L; Mancini, L; Milandri, A; Poletti, R; Pompei, M; Viviani, R. First cases of diarrhetic shellfish poisoning in the Northern Adriatic Sea. In Marine Coastal Eutrophication. Sci Total Environ Suppl; Vollenweider, RA, Marchetti, R, Viviani, R, Eds.; Elsevier: Amsterdam, The Netherland, 1992; pp. 419–426. [Google Scholar]
- Orhanović, S; Ninčević, Ž; Marasović, I; Pavela-Vrančić, M. Phytoplankton toxins in the Central Adriatic Sea. Croat Chem Acta 1996, 69, 291–303. [Google Scholar]
- Marasović, I; Ninčević, Ž; Pavela-Vrančić, M; Orhanović, S. A Survey of shellfish toxicity in the Central Adriatic Sea. J Mar Biol Assoc UK 1998, 78, 745–754. [Google Scholar]
- Pavela-Vrančić, M; Meštrović, V; Marasović, I; Gillman, M; Furey, A; James, KJ. DSP toxin profile in the coastal waters of the Central Adriatic Sea. Toxicon 2008, 40, 1601–1607. [Google Scholar]
- Ninčević Gladan, Ž; Skejić, S; Bužančić, M; Marasović, I; Arapov, J; Ujević, I; Bojanić, N; Grbec, B; Kušpilić, G; Vidjak, O. Seasonal variability in Dinophysis spp. abundances and DSP outbreaks along the eastern Adriatic coast. Bot Mar 2008, 51, 449–463. [Google Scholar]
- Draisci, R; Lucentini, L; Giannetti, L; Boria, P; Poletti, R. First report of pectenotoxin-2 (PTX-2) in algae (Dinophysis fortii) related to seafood poisoning in Europe. Toxicon 1996, 34, 923–935. [Google Scholar]
- Van Egmond, HP; Aune, T; Lassus, P; Speijers, G; Waldock, M. Paralytic and diarrhoeic shellfish poisons: occurrence in Europe, toxicity, analysis and regulation. J Nat Toxins 1993, 2 , 41–83. [Google Scholar]
- Čustović, S; Orhanovic, S; Ninčević Gladan, Ž; Josipović, T; Pavela-Vrančić, M. Occurence of yessotoxin (YTX) in the coastal waters of the eastern-mid Adriatic Sea (Croatia). Fresenius Environ Bull 2009, 18, 1452–1455. [Google Scholar]
- Yasumoto, T; Murata, M; Oshima, Y; Matsumoto, GL; Clardy, J. Diarrhetic shellfish poisoning. In Seafood Toxins; Ragelis, EP, Ed.; American Chemical Society Symposium Series: Washington DC, USA, 1984; pp. 207–214. [Google Scholar]
- Fernández, ML; Míguez, A; Cacho, E; Martínez, A; Diogéne, J; Yasumoto, T. Procedimiento para detección de ao, dtxs, ptxs y azas en el extracto de diclorometano y detección de ytxs en extracto metanólico. In Floraciones algales nocivas en el cono sur americano; Sar, EA, Ferrario, ME, Reguera, B, Eds.; Instituto spanol de Oceanografia: Madrid, Spain, 2002; pp. 77–120. [Google Scholar]
- Samadal, IA; Aasen, JAB; Briggs, LR; Dahl, E; Miles, CO. Comparison of ELISA and LC-MS analyses for yessotoxins in blue mussels (Mytilus edulis). Toxicon 2005, 46, 7–15. [Google Scholar]
- Ciminiello, P; Fattorusso, E. Yessotoxins: chemistry, metabolism and chemical analysis. In Seafood and Freshwater Toxins; Botana, LM, Ed.; CRC Press: New York NY, USA, 2008; pp. 285–314. [Google Scholar]
- Satake, M; Ichimura, T; Sekiguchi, K; Yoshimatsu, S; Oshima, Y. Confirmation of yessotoxin and 45, 46, 47-trinoryessotoxin production by Protoceratium reticulatum collected in Japan. Nat Toxins 1999, 7, 147–150. [Google Scholar]
- Ciminiello, P; Fattorusso, E; Forino, M; Magno, S; Poletti, R. 42, 43, 44, 45, 46, 47, 55-Heptanor-41-oxohomoyessotoxin, a new biotoxin from mussels of the northern Adriatic sea. Chem Res Toxicol 2001, 14, 596–599. [Google Scholar]
- Miles, CO; Samdal, IA; Aasen, JAG; Jensen, DJ; Quilliam, MA; Petersen, D; Briggs, LM; Wilkins, AL; Rise, F; Cooney, JM; MacKenzie, AL. Evidence for numerous analogs of yessotoxin in Protoceratium reticulatum. Harmful Algae 2005, 4, 1075–1091. [Google Scholar]
- Paz, B; Riobo, P; Souto, ML; Gil, LV; Norte, M; Fernandez, JJ; Franco, JM. Detection and identification of glycoyessotoxin A in a culture of the dinoflagellate Protoceratium reticulatum. Toxicon 2006, 48, 611–619. [Google Scholar]
- Aasen, J; Samdal, IA; Miles, CO; Dahl, E; Briggs, LR; Aune, T. Yessotoxins in Norwegian blue mussels (Mytilus edulis): uptake from Protoceratium reticulatum, metabolism and depuration. Toxicon 2005, 45, 265–272. [Google Scholar]
- Howard, MDA; Silver, M; Kudela, RM. Yessotoxin detected in mussel (Mytilus californicus) and phytoplankton samples from the US west coast. Harmful Algae 2008, 7, 646–652. [Google Scholar]
- Rhodes, L; McNabb, P; de Salas, M; Briggs, L; Beuzenberg, V; Gladstone, M. Yessotoxin production by Gonyaulax spinifera. Harmful Algae 2006, 5, 148. [Google Scholar]
- Riccardi, M; Guerrini, F; Roncarati, F; Milandri, A; Cangini, M; Pigozzi, S; Riccardi, E; Ceredi, A; Ciminiello, P; Dell’Aversano, C; Fattorusso, E; Forino, M; Tartaglione, L; Pistocchi, R. Gonyaulax spinifera from the Adriatic sea: Toxin production and phylogenetic analysis. Harmful Algae 2009, 8, 279–290. [Google Scholar]
- Aune, T; Sørby, R; Yasumoto, T; Ramstad, H; Landsverk, T. Comparison of oral and intraperitoneal toxicity of yessotoxin towards mice. Toxicon 2002, 40, 77–82. [Google Scholar]
- Tubaro, A; Sosa, S; Altinier, G; Soranzo, MR; Satake, M; Della Loggia, R; Yasumoto, T. Short-term oral toxicity of homoyessotoxins, yessotoxin and okadaic acid in mice. Toxicon 2004, 43, 439–445. [Google Scholar]
- EC. Corrigendum to Regulation No 853/2004 of the European Parliament and of the Council of the 29 April 2004 laying down specific hygiene rules for food of animal origin. Official Journal of the European Union L 139 of 30 April 2004.
- Üthermöhl, H. Zur Vervollkommnung der quantitativen Phytoplankton Methodik. Mitt Int Ver Theor Angew Limnol 1958, 9, 1–38. [Google Scholar]
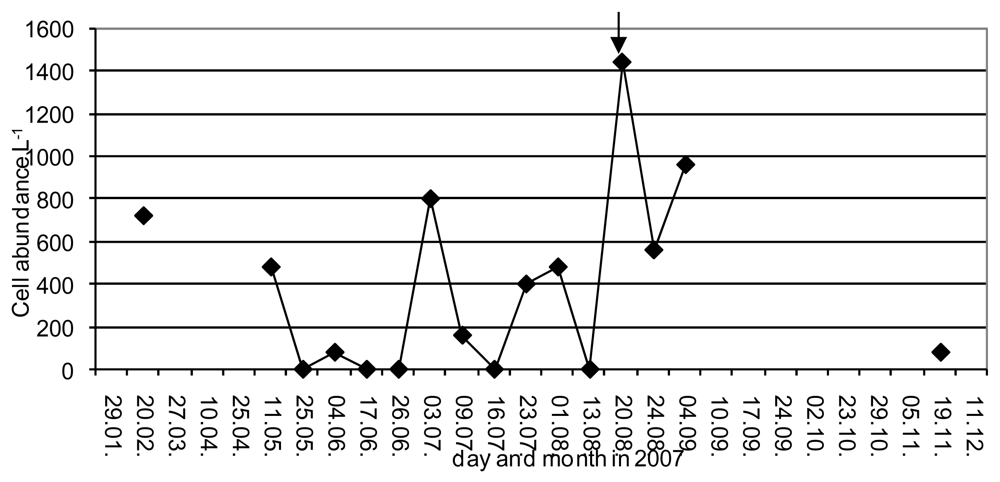
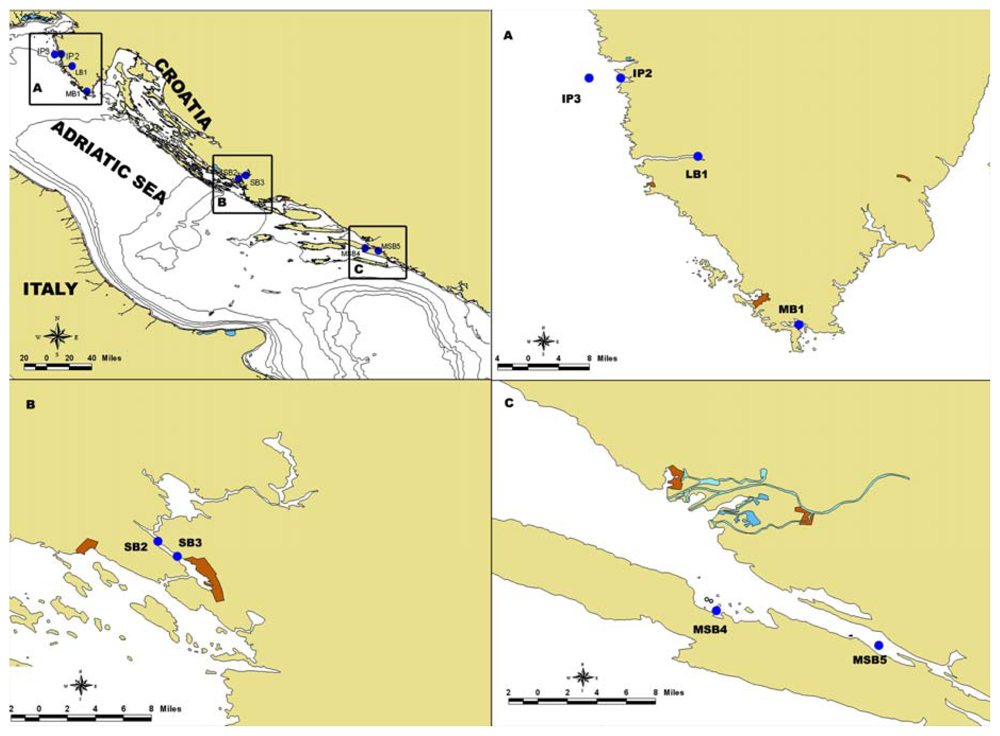
| Date | Station | MBA for DSP | YTXs by ELISA (μg kg−1) |
|---|---|---|---|
| 24. April | SB 3 | Positive | 939 |
| 24. May | IP 3 | Positive | 1894 |
| 04. June | SB 2 | Positive | n.a |
| 27. November | MSB 5 | Positive | n.a. |
| Date | Station | MBA for DSP [13] | MBA for YTX [14] | YTXs by ELISA (μg kg−1) |
|---|---|---|---|---|
| 20. August | MB 1 | Negative | Positive | 7900 |
| 10. September | MSB 4 | Negative | Positive | 42 |
| 17. September | IP 2 | Negative | Positive | 194 |
| 02. October | IP 2 | Negative | Positive | 567 |
| 02. October | LB 1 | Positive | Negative | 105 |
| 22. October | LB 1 | Positive | Negative | n.a. |
| Station | date | YTX | Homo YTX | 45- OH- YTX | 45- OH- homo | Carboxy -YTX | Carboxy-homo YTX | PTX-2 | PTXs | PTX2-SA | 7-epi- PTX2-SA | OA | Total OA | AZAs |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SB 3 | 24.04 | 131 | 89 | traces | traces | traces | <25 | traces | traces | <38 | ||||
| IP 3 | 24.05. | traces | 72 | traces | <25 | traces | <38 | |||||||
| MB1 | 20.08. | 244 | 59 | 144 | 794 | traces | <25 | traces | traces | 26 | <38 | |||
| MSB 4 | 10.09. | traces | <25 | traces | traces | <38 | ||||||||
| IP 2 | 17.09 | traces | <25 | <38 | ||||||||||
| IP2 | 02.10. | 77 | traces | <25 | traces | <38 | ||||||||
| LB 1 | 02.10. | 72 | traces | <25 | 32 | traces | 341 | 787 | <38 | |||||
| LB 1 | 22.10. | traces | <25 | 38 | 13 | 462 | 1041 | <38 | ||||||
| MSB 5 | 27.11. | traces | <25 | 27 | 37 | 92 | <38 |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gladan, Ž.N.; Ujević, I.; Milandri, A.; Marasović, I.; Ceredi, A.; Pigozzi, S.; Arapov, J.; Skejić, S.; Orhanović, S.; Isajlović, I. Is Yessotoxin the Main Phycotoxin in Croatian Waters? Mar. Drugs 2010, 8, 460-470. https://doi.org/10.3390/md8030460
Gladan ŽN, Ujević I, Milandri A, Marasović I, Ceredi A, Pigozzi S, Arapov J, Skejić S, Orhanović S, Isajlović I. Is Yessotoxin the Main Phycotoxin in Croatian Waters? Marine Drugs. 2010; 8(3):460-470. https://doi.org/10.3390/md8030460
Chicago/Turabian StyleGladan, Živana Ninčević, Ivana Ujević, Anna Milandri, Ivona Marasović, Alfiero Ceredi, Silvia Pigozzi, Jasna Arapov, Sanda Skejić, Stjepan Orhanović, and Igor Isajlović. 2010. "Is Yessotoxin the Main Phycotoxin in Croatian Waters?" Marine Drugs 8, no. 3: 460-470. https://doi.org/10.3390/md8030460
APA StyleGladan, Ž. N., Ujević, I., Milandri, A., Marasović, I., Ceredi, A., Pigozzi, S., Arapov, J., Skejić, S., Orhanović, S., & Isajlović, I. (2010). Is Yessotoxin the Main Phycotoxin in Croatian Waters? Marine Drugs, 8(3), 460-470. https://doi.org/10.3390/md8030460





