Cnidarian Toxins Acting on Voltage-Gated Ion Channels
Abstract
:Introduction
Cnidarian Ion Channel Toxins
NaV Channel Toxins
KV Channel Toxins
Concluding remarks
Acknowledgments
Abbreviations
| KV channel | voltage-gated potassium channel |
| NaV | voltage-gated sodium channel |
| CaV | voltage-gated calcium channel |
| ApA | Anthopleurin A |
| ApB | Anthopleurin B |
| ATX II | Anemone sulcata toxin II |
| Bg II | Bunodosoma granulifera toxin II |
| Sh I | peptide neurotoxin I from Stichodactyla helianthus |
| RP II | polypeptide toxin II from Radianthus paumotensis |
| RP III | polypeptide toxin III from Radianthus paumotensis |
| RTX I | neurotoxin I from Radianthus macrodactylus |
| PaTX | toxin from Paracicyonis actinostoloides |
| Er I | peptide toxin I from Entacmaea ramsayi |
| Da I | peptide toxin I from Dofleinia armata |
| ATX III | Anemone sulcata toxin III |
| ShK | potassium channel toxin from Stichodactyla helianthus |
| BgK | potassium channel toxin from Bunodosoma granulifera |
| AsKS | kalciceptine from Anemonia sulcata |
| HmK | potassium channel toxin from Heteractis magnifica |
| AeK | potassium channel toxin from Actinia equina |
| AsKC 1-3 | kalcicludines 1-3 from Anemonia sulcata |
| BDS-I, BDS-II | blood depressing toxins I and II from Anemonia sulcata |
| APETx1 | potassium channel toxin 1 from Anthopleura elegantissima |
| SNTX | sea nettle toxin |
| E | glutamic acid |
| D | aspartic acid |
| K | lysine |
| R | arginine |
| Y | tyrosine |
| HERG | human ether-a-go-go-related gene |
- Samples Availability: Available from the authors.
References
- Yu, F. H.; Yarov-Yarovoy, V.; Gutman, G. A.; Catterall, W. A. Overview of molecular relationships in the voltage-gated ion channel superfamily. Pharmacol. Rev 2005, 57, 387–395. [Google Scholar]
- MacKinnon, R. Potassium channels. FEBS Lett 2003, 555, 62–65. [Google Scholar]
- Gutman, G. A.; Chandy, K. G.; Grissmer, S.; Lazdunski, M.; McKinnon, D.; Pardo, L. A.; Robertson, G. A.; Rudy, B.; Sanguinetti, M. C.; Stuhmer, W.; Wang, X. International Union of Pharmacology. LIII. Nomenclature and molecular relationships of voltage-gated potassium channels. Pharmacol. Rev 2005, 57, 473–508. [Google Scholar]
- Catterall, W. A.; Goldin, A. L.; Waxman, S. G. International Union of Pharmacology. XLVII. Nomenclature and structure-function relationships of voltage-gated sodium channels. Pharmacol. Rev 2005, 57, 397–409. [Google Scholar]
- Catterall, W. A.; Perez-Reyes, E.; Snutch, T. P.; Striessnig, J. International Union of Pharmacology. XLVIII. Nomenclature and structure-function relationships of voltage-gated calcium channels. Pharmacol. Rev 2005, 57, 411–425. [Google Scholar]
- Doyle, D. A.; Morais Cabral, J.; Pfuetzner, R. A.; Kuo, A.; Gulbis, J. M.; Cohen, S. L.; Chait, B. T.; MacKinnon, R. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 1998, 280, 69–77. [Google Scholar]
- Strong, M.; Chandy, K. G.; Gutman, G. A. Molecular evolution of voltage-sensitive ion channel genes: on the origins of electrical excitability. Mol. Biol. Evol 1993, 10, 221–242. [Google Scholar]
- Anderson, P. A.; Greenberg, R. M. Phylogeny of ion channels: clues to structure and function. Comp. Biochem. Physiol. B Biochem. Mol. Biol 2001, 129, 17–28. [Google Scholar]
- Dolphin, A. C. A short history of voltage-gated calcium channels. Br. J. Pharmacol 2006, 147, S56–S62. [Google Scholar]
- Richards, M. W.; Butcher, A. J.; Dolphin, A. C. Ca2+ channel β-subunits: structural insights AID our understanding. Trends Pharmacol. Sci 2004, 25, 626–632. [Google Scholar]
- Dolphin, A. C. Beta subunits of voltage-gated calcium channels. J. Bioenerg. Biomembr 2003, 35, 599–620. [Google Scholar]
- Hanlon, M. R.; Wallace, B. A. Structure and function of voltage-dependent ion channel regulatory beta subunits. Biochemistry 2002, 41, 2886–2894. [Google Scholar]
- Walker, D.; De Waard, M. Subunit interaction sites in voltage-dependent Ca2+ channels: role in channel function. Trends Neurosci 1998, 21, 148–154. [Google Scholar]
- Birnbaumer, L.; Qin, N.; Olcese, R.; Tareilus, E.; Platano, D.; Costantin, J.; Stefani, E. Structures and functions of calcium channel β subunits. J. Bioenerg. Biomembr 1998, 30, 357–375. [Google Scholar]
- Joseph, R.; Pahari, S.; Hodgson, W. C.; Kini, R. M. Hypotensive agents from snake venoms. Curr. Drug Targets Cardiovasc. Haematol. Disord 2004, 4, 437–459. [Google Scholar]
- Kudo, Y.; Shibata, S. The potent excitatory effect of a novel polypeptide, anthopleurin-B, isolated from a sea anemone (Anthopleura xanthogrammica) on the frog spinal cord. J. Pharmacol. Exp. Ther 1980, 214, 443–448. [Google Scholar]
- French, R. J.; Terlau, H. Sodium channel toxins--receptor targeting and therapeutic potential. Curr. Med. Chem 2004, 11, 3053–3064. [Google Scholar]
- Terlau, H.; Olivera, B. M. Conus venoms: a rich source of novel ion channel-targeted peptides. Physiol. Rev 2004, 84, 41–68. [Google Scholar]
- Layer, R.T.; McIntosh, J.M. Conotoxins: therapeutic potential and application. Mar. Drugs 2006, 4, 82–118. [Google Scholar]
- Honma, T.; Shiomi, K. Peptide Toxins in Sea Anemones: Structural and Functional Aspects. Mar. Biotechnol 2006, 8, 1–10. [Google Scholar]
- Geffeney, S. L.; Fujimoto, E.; Brodie, E. D., 3rd; Brodie, E. D.; Ruben, P. C. Evolutionary diversification of TTX-resistant sodium channels in a predator-prey interaction. Nature 2005, 434, 759–763. [Google Scholar]
- Wen, L.; Yang, S.; Zhou, W.; Zhang, Y.; Huang, P. New conotoxin SO-3 targeting N-type voltage-sensitive calcium channels. Mar. Drugs 2006, 4, 215–227. [Google Scholar]
- Watters, M. R. Tropical marine neurotoxins: venoms to drugs. Semin. Neurol 2005, 25, 278–289. [Google Scholar]
- Kem, W. R.; Pennington, M. W.; Dunn, B. M. Sea anemone polypeptide toxins affecting sodium channels: Initial structure-activity investigations. Hall, S., Strichartz, G., Eds.; In Marine Toxins: Origin, Structure, and Molecular Pharmacology; Washington, D.C; American Chemical Society, 1990; pp. 279–289. [Google Scholar]
- Norton, R. S. Structure and structure-function relationships of sea anemone proteins that interact with the sodium channel. Toxicon 1991, 29, 1051–1084. [Google Scholar]
- Ahmed, A-S.; McArthur, J.; Ostroumov, V.; French, R.J. Marine toxins that target voltage-gated sodium channels. Mar. Drugs 2006, 4, 157–192. [Google Scholar]
- Wunderer, G.; Fritz, H.; Wachter, E.; Machleidt, W. Amino-acid sequence of a coelenterate toxin: toxin II from Anemonia sulcata. Eur. J. Biochem 1976, 68, 193–198. [Google Scholar]
- Tanaka, M.; Hainu, M.; Yasunobu, K. T.; Norton, T. R. Amino acid sequence of the Anthopleura xanthogrammica heart stimulant, anthopleurin A. Biochemistry 1977, 16, 204–208. [Google Scholar]
- Reimer, N. S.; Yasunobu, C. L.; Yasunobu, K. T.; Norton, T. R. Amino acid sequence of the Anthopleura xanthogrammica heart stimulant, anthopleurin-B. J. Biol. Chem 1985, 260, 8690– 8693. [Google Scholar]
- Cunha, R. B.; Santana, A. N.; Amaral, P. C.; Carvalho, M. D.; Carvalho, D. M.; Cavalheiro, E. A.; Maigret, B.; Ricart, C. A.; Cardi, B. A.; Sousa, M. V.; Carvalho, K. M. Primary structure, behavioral and electroencephalographic effects of an epileptogenic peptide from the sea anemone Bunodosoma cangicum. Toxicon 2005, 45, 207–217. [Google Scholar]
- Cariello, L.; de Santis, A.; Fiore, F.; Piccoli, R.; Spagnuolo, A.; Zanetti, L.; Parente, A. Calitoxin, a neurotoxic peptide from the sea anemone Calliactis parasitica: amino acid sequence and electrophysiological properties. Biochemistry 1989, 28, 2484–2489. [Google Scholar]
- Spagnuolo, A.; Zanetti, L.; Cariello, L.; Piccoli, R. Isolation and characterization of two genes encoding calitoxins, neurotoxic peptides from Calliactis parasitica (Cnidaria). Gene 1994, 138, 187–191. [Google Scholar]
- Shiomi, K.; Honma, T.; Ide, M.; Nagashima, Y.; Ishida, M.; Chino, M. An epidermal growth factor-like toxin and two sodium channel toxins from the sea anemone Stichodactyla gigantea. Toxicon 2003, 41, 229–236. [Google Scholar]
- Norton, T. R.; Shibata, S.; Kashiwagi, M.; Bentley, J. Isolation and characterization of the cardiotonic polypeptide anthopleurin-A from the sea anemone Anthopleura xanthogrammica. J. Pharm. Sci 1976, 65, 1368–1374. [Google Scholar]
- Beress, L.; Beress, R.; Wunderer, G. Isolation and characterisation of three polypeptides with neurotoxic activity from Anemonia sulcata. FEBS Lett 1975, 50, 311–314. [Google Scholar]
- Bergman, C.; Dubois, J. M.; Rojas, E.; Rathmayer, W. Decreased rate of sodium conductance inactivation in the node of Ranvier induced by a polypeptide toxin from sea anemone. Biochim. Biophys. Acta 1976, 455, 173–184. [Google Scholar]
- Catterall, W. A.; Beress, L. Sea anemone toxin and scorpion toxin share a common receptor site associated with the action potential sodium ionophore. J. Biol. Chem 1978, 253, 7393–7396. [Google Scholar]
- Vincent, J. P.; Balerna, M.; Barhanin, J.; Fosset, M.; Lazdunski, M. Binding of sea anemone toxin to receptor sites associated with gating system of sodium channel in synaptic nerve endings in vitro. Proc. Natl. Acad. Sci. U.S.A 1980, 77, 1646–1650. [Google Scholar]
- Schweitz, H.; Vincent, J. P.; Barhanin, J.; Frelin, C.; Linden, G.; Hugues, M.; Lazdunski, M. Purification and pharmacological properties of eight sea anemone toxins from Anemonia sulcata, Anthopleura xanthogrammica, Stoichactis giganteus, and Actinodendron plumosum. Biochemistry 1981, 20, 5245–5252. [Google Scholar]
- Warashina, A.; Ogura, T.; Fujita, S. Binding properties of sea anemone toxins to sodium channels in the crayfish giant axon. Comp. Biochem. Physiol. C 1988, 90, 351–359. [Google Scholar]
- Cestele, S.; Catterall, W. A. Molecular mechanisms of neurotoxin action on voltage-gated sodium channels. Biochimie 2000, 82, 883–892. [Google Scholar]
- Rogers, J. C.; Qu, Y.; Tanada, T. N.; Scheuer, T.; Catterall, W. A. Molecular determinants of high affinity binding of alpha-scorpion toxin and sea anemone toxin in the S3-S4 extracellular loop in domain IV of the Na+ channel alpha subunit. J. Biol. Chem 1996, 271, 15950–15962. [Google Scholar]
- Benzinger, G. R.; Kyle, J. W.; Blumenthal, K. M.; Hanck, D. A. A specific interaction between the cardiac sodium channel and site-3 toxin anthopleurin B. J. Biol. Chem 1998, 273, 80–84. [Google Scholar]
- Oliveira, J. S.; Redaelli, E.; Zaharenko, A. J.; Cassulini, R. R.; Konno, K.; Pimenta, D. C.; Freitas, J. C.; Clare, J. J.; Wanke, E. Binding specificity of sea anemone toxins to Nav 1.1–1.6 sodium channels: unexpected contributions from differences in the IV/S3–S4 outer loop. J. Biol. Chem 2004, 279, 33323–33335. [Google Scholar]
- Tejedor, F. J.; Catterall, W. A. Site of covalent attachment of alpha-scorpion toxin derivatives in domain I of the sodium channel alpha subunit. Proc. Natl. Acad. Sci. U.S.A 1988, 85, 8742–8746. [Google Scholar]
- Thomsen, W. J.; Catterall, W. A. Localization of the receptor site for alpha-scorpion toxins by antibody mapping: implications for sodium channel topology. Proc. Natl. Acad. Sci. U.S.A 1989, 86, 10161–10165. [Google Scholar]
- Smith, J. J.; Alphy, S.; Seibert, A. L.; Blumenthal, K. M. Differential phospholipid binding by site 3 and site 4 toxins. Implications for structural variability between voltage-sensitive sodium channel domains. J. Biol. Chem 2005, 280, 11127–11133. [Google Scholar]
- Monks, S. A.; Pallaghy, P. K.; Scanlon, M. J.; Norton, R. S. Solution structure of the cardiostimulant polypeptide anthopleurin-B and comparison with anthopleurin-A. Structure 1995, 3, 791–803. [Google Scholar]
- Seibert, A. L.; Liu, J.; Hanck, D. A.; Blumenthal, K. M. Arg-14 loop of site 3 anemone toxins: effects of glycine replacement on toxin affinity. Biochemistry 2003, 42, 14515–14521. [Google Scholar]
- Khera, P. K.; Benzinger, G. R.; Lipkind, G.; Drum, C. L.; Hanck, D. A.; Blumenthal, K. M. Multiple cationic residues of anthopleurin B that determine high affinity and channel isoform discrimination. Biochemistry 1995, 34, 8533–8541. [Google Scholar]
- Loret, E. P.; del Valle, R. M.; Mansuelle, P.; Sampieri, F.; Rochat, H. Positively charged amino acid residues located similarly in sea anemone and scorpion toxins. J. Biol. Chem 1994, 269, 16785–16788. [Google Scholar]
- Kem, W. R.; Parten, B.; Pennington, M. W.; Price, D. A.; Dunn, B. M. Isolation, characterization, and amino acid sequence of a polypeptide neurotoxin occurring in the sea anemone Stichodactyla helianthus. Biochemistry 1989, 28, 3483–3489. [Google Scholar]
- Schweitz, H.; Bidard, J. N.; Frelin, C.; Pauron, D.; Vijverberg, H. P.; Mahasneh, D. M.; Lazdunski, M.; Vilbois, F.; Tsugita, A. Purification, sequence, and pharmacological properties of sea anemone toxins from Radianthus paumotensis. A new class of sea anemone toxins acting on the sodium channel. Biochemistry 1985, 24, 3554–3561. [Google Scholar]
- Metrione, R. M.; Schweitz, H.; Walsh, K. A. The amino acid sequence of toxin RpIII from the sea anemone, Radianthus paumotensis. FEBS Lett 1987, 218, 59–62. [Google Scholar]
- Zykova, T. A.; Kozlovskaia, E. P.; Eliakov, G. B. Amino acid sequence of neurotoxin II from the sea anemone Radianthus macrodactylus. Bioorg. Khim 1988, 14, 878–882. [Google Scholar]
- Nishida, S.; Fujita, S.; Warashina, A.; Satake, M.; Tamiya, N. Amino acid sequence of a sea anemone toxin from Parasicyonis actinostoloides. Eur. J. Biochem 1985, 150, 171–173. [Google Scholar]
- Martinez, G.; Kopeyan, C. Toxin III from Anemonia sulcata: primary structure. FEBS Lett 1977, 84, 247–252. [Google Scholar]
- Tudor, J. E.; Pallaghy, P. K.; Pennington, M. W.; Norton, R. S. Solution structure of ShK toxin, a novel potassium channel inhibitor from a sea anemone. Nat. Struct. Biol 1996, 3, 317–320. [Google Scholar]
- Dauplais, M.; Lecoq, A.; Song, J.; Cotton, J.; Jamin, N.; Gilquin, B.; Roumestand, C.; Vita, C.; de Medeiros, C. L.; Rowan, E. G.; Harvey, A. L.; Menez, A. On the convergent evolution of animal toxins. Conservation of a diad of functional residues in potassium channel-blocking toxins with unrelated structures. J. Biol. Chem 1997, 272, 4302–4309. [Google Scholar]
- Pennington, M. W.; Mahnir, V. M.; Khaytin, I.; Zaydenberg, I.; Byrnes, M. E.; Kem, W. R. An essential binding surface for ShK toxin interaction with rat brain potassium channels. Biochemistry 1996, 35, 16407–16411. [Google Scholar]
- Alessandri-Haber, N.; Lecoq, A.; Gasparini, S.; Grangier-Macmath, G.; Jacquet, G.; Harvey, A. L.; de Medeiros, C.; Rowan, E. G.; Gola, M.; Menez, A.; Crest, M. Mapping the functional anatomy of BgK on Kv1.1, Kv1.2, and Kv1.3. Clues to design analogs with enhanced selectivity. J. Biol. Chem 1999, 274, 35653–35661. [Google Scholar]
- Gilquin, B.; Racape, J.; Wrisch, A.; Visan, V.; Lecoq, A.; Grissmer, S.; Menez, A.; Gasparini, S. Structure of the BgK-Kv1.1 complex based on distance restraints identified by double mutant cycles. Molecular basis for convergent evolution of Kv1 channel blockers. J. Biol. Chem 2002, 277, 37406–37413. [Google Scholar]
- Norton, R. S.; Pennington, M. W.; Wulff, H. Potassium channel blockade by the sea anemone toxin ShK for the treatment of multiple sclerosis and other autoimmune diseases. Curr. Med. Chem 2004, 11, 3041–3052. [Google Scholar]
- Schweitz, H.; Bruhn, T.; Guillemare, E.; Moinier, D.; Lancelin, J. M.; Beress, L.; Lazdunski, M. Kalicludines and kaliseptine. Two different classes of sea anemone toxins for voltage sensitive K+ channels. J. Biol. Chem 1995, 270, 25121–25126. [Google Scholar]
- Diochot, S.; Schweitz, H.; Beress, L; Lazdunski, M. Sea anemone peptides with a specific blocking activity against the fast inactivating potassium channel Kv3.4. J. Biol. Chem. 1998, 273, 6744–6749. [Google Scholar]
- Diochot, S.; Loret, E.; Bruhn, T.; Beress, L.; Lazdunski, M. APETx1, a new toxin from the sea anemone Anthopleura elegantissima, blocks voltage-gated human ether-a-go-go-related gene potassium channels. Mol. Pharmacol 2003, 64, 59–69. [Google Scholar]
- Castaneda, O.; Sotolongo, V.; Amor, A. M.; Stocklin, R.; Anderson, A. J.; Harvey, A. L.; Engstrom, A.; Wernstedt, C.; Karlsson, E. Characterization of a potassium channel toxin from the Caribbean Sea anemone Stichodactyla helianthus. Toxicon 1995, 33, 603–613. [Google Scholar]
- Aneiros, A.; Garcia, I.; Martinez, J. R.; Harvey, A. L.; Anderson, A. J.; Marshall, D. L.; Engstrom, A.; Hellman, U.; Karlsson, E. A potassium channel toxin from the secretion of the sea anemone Bunodosoma granulifera. Isolation, amino acid sequence and biological activity. Biochim. Biophys. Acta 1993, 1157, 86–92. [Google Scholar]
- Cotton, J.; Crest, M.; Bouet, F.; Alessandri, N.; Gola, M.; Forest, E.; Karlsson, E.; Castaneda, O.; Harvey, A. L.; Vita, C.; Menez, A. A potassium-channel toxin from the sea anemone Bunodosoma granulifera, an inhibitor for Kv1 channels. Revision of the amino acid sequence, disulfide-bridge assignment, chemical synthesis, and biological activity. Eur. J. Biochem 1997, 244, 192–202. [Google Scholar]
- Gendeh, G. S.; Young, L. C.; de Medeiros, C. L.; Jeyaseelan, K.; Harvey, A. L.; Chung, M. C. A new potassium channel toxin from the sea anemone Heteractis magnifica: isolation, cDNA cloning, and functional expression. Biochemistry 1997, 36, 11461–11471. [Google Scholar]
- Minagawa, S.; Ishida, M.; Nagashima, Y.; Shiomi, K. Primary structure of a potassium channel toxin from the sea anemone Actinia equina. FEBS Lett 1998, 427, 149–151. [Google Scholar]
- Salinas, E. M.; Cebada, J.; Valdes, A.; Garateix, A.; Aneiros, A.; Alvarez, J. L. Effects of a toxin from the mucus of the Caribbean sea anemone (Bunodosoma granulifera) on the ionic currents of single ventricular mammalian cardiomyocytes. Toxicon 1997, 35, 1699–1709. [Google Scholar]
- Burnett, J.W.; Weinrich, D.; Williamson, J.A.; Fenner, P.J.; Lutz, L.L.; Bloom, D.A. Autonomic neurotoxicity of jellyfish and marine animal venoms. Clin. Autonomic Res 1998, 8, 125–130. [Google Scholar]
- Rojas, A.; Torres, M.; Rojas, J.I; Feregrino, A.; Heimer-de la Cotera, E. Calcium-dependent smooth muscle excitatory effect elicited by the venom of the hydrocoral Millepora complanata. Toxicon 2002, 40, 777–785. [Google Scholar]
- Lin, W.W; Lee, C.Y.; Burnett, J.W. Effect of sea nettle (Chrysaora quinquecirrha) venom on isolated rat aorta. Toxicon 1988, 26, 1209–1212. [Google Scholar]
- Dubois, J.M.; Tanguy, J; Burnett, J.W. Ionic channels induced by sea nettle toxin in the nodal membrane. Biophys. J. 1983, 42, 199–202. [Google Scholar]
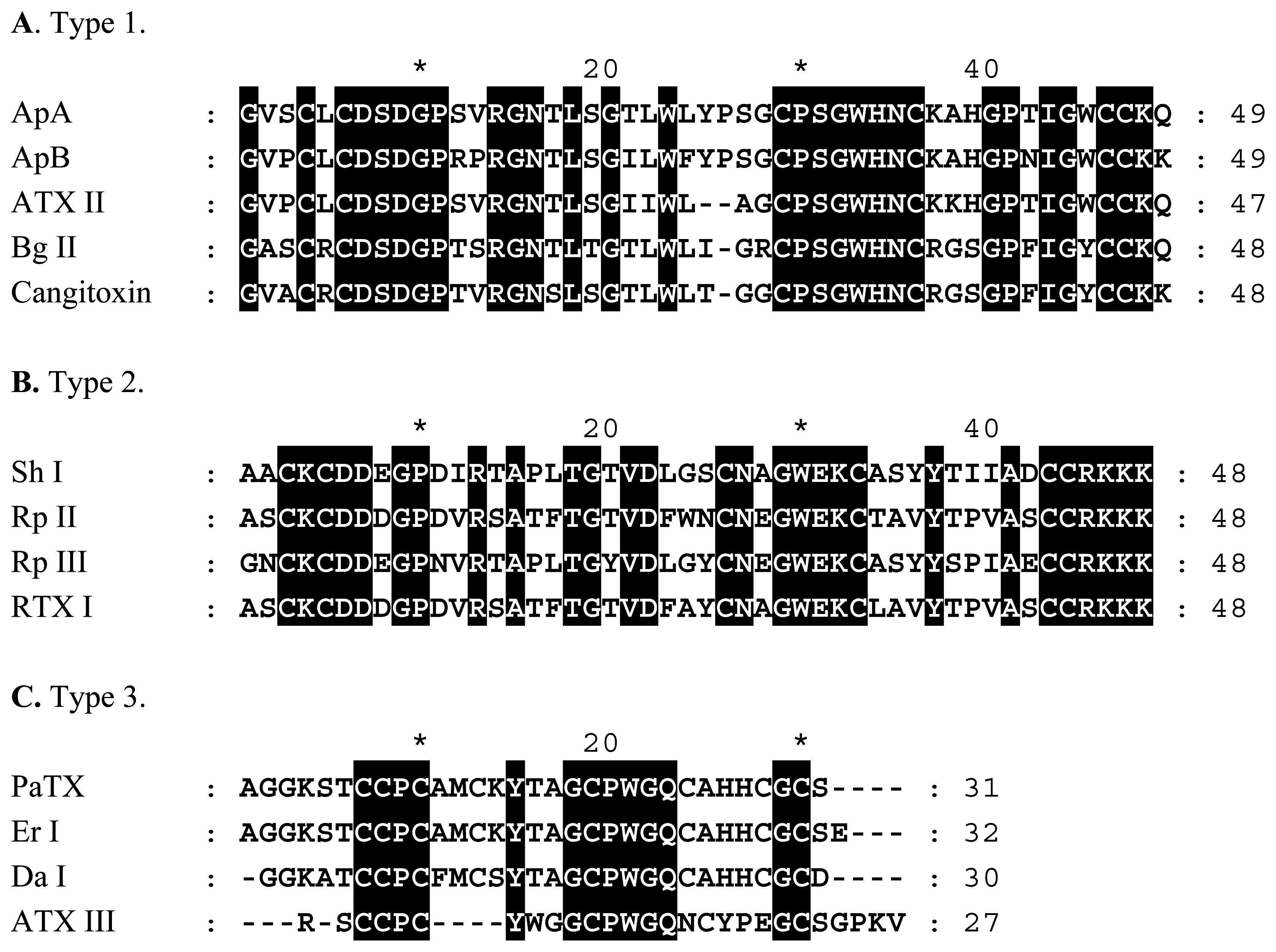
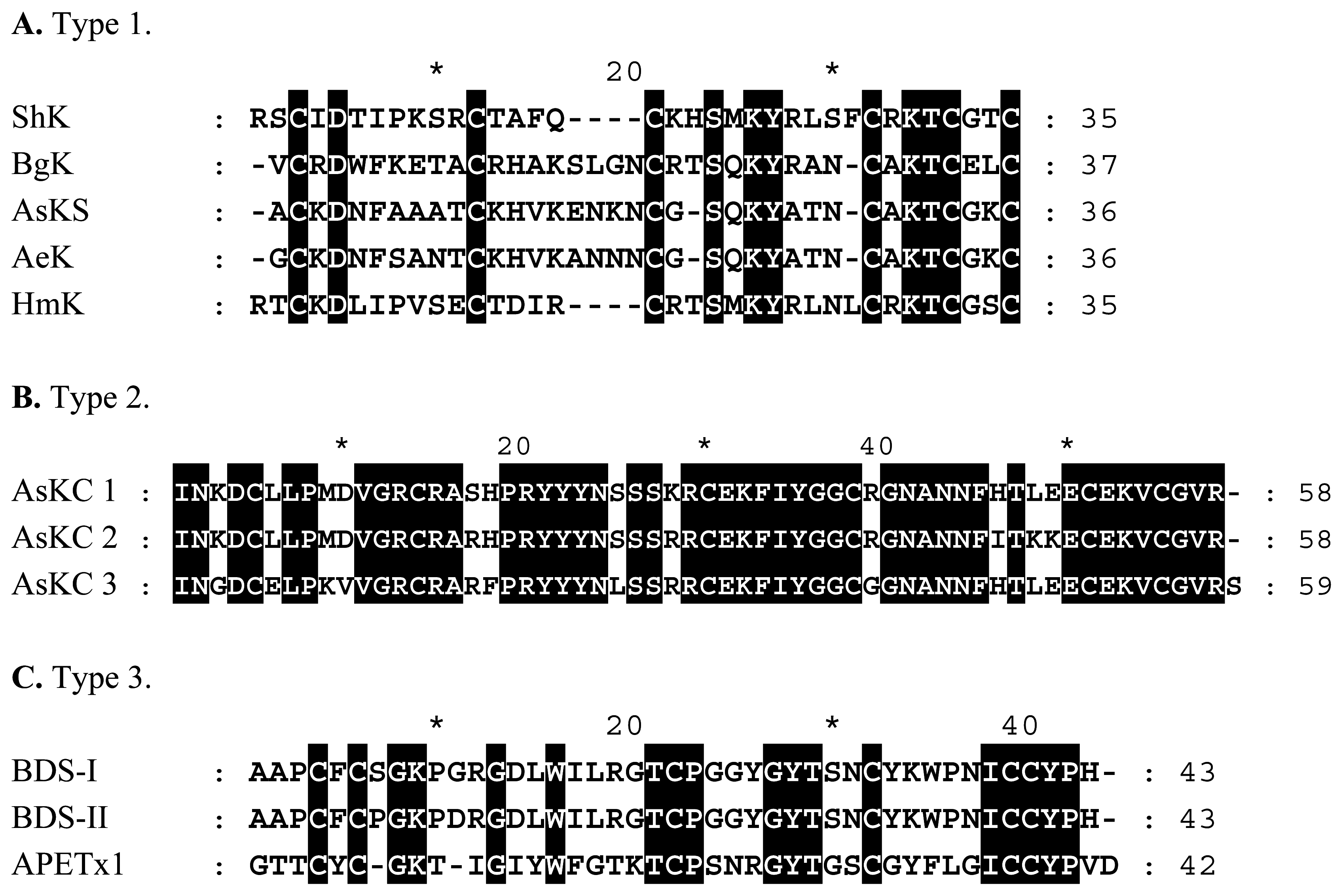
| Toxin | Species | Type | Reference |
|---|---|---|---|
| ShK | Stichodactyla helianthus | 1 | [67] |
| AsKS (kaliseptine) | Anemonia sulcata | 1 | [64] |
| BgK | Bunodosoma granulifera | 1 | [68,69] |
| HmK | Heteractis magnifica | 1 | [70] |
| AeK | Actinia equine | 1 | [71] |
| AsKC 1–3 (kalcicludines 1–3) | Anemonia sulcata | 2 | [64] |
| BDS-I, BDS-II | Anemonia sulcata | 3 | [65] |
| APETx1 | Anthopleura elegantissima | 3 | [66] |
© 2006 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Messerli, S.M.; Greenberg, R.M. Cnidarian Toxins Acting on Voltage-Gated Ion Channels. Mar. Drugs 2006, 4, 70-81. https://doi.org/10.3390/md403070
Messerli SM, Greenberg RM. Cnidarian Toxins Acting on Voltage-Gated Ion Channels. Marine Drugs. 2006; 4(3):70-81. https://doi.org/10.3390/md403070
Chicago/Turabian StyleMesserli, Shanta M., and Robert M. Greenberg. 2006. "Cnidarian Toxins Acting on Voltage-Gated Ion Channels" Marine Drugs 4, no. 3: 70-81. https://doi.org/10.3390/md403070
APA StyleMesserli, S. M., & Greenberg, R. M. (2006). Cnidarian Toxins Acting on Voltage-Gated Ion Channels. Marine Drugs, 4(3), 70-81. https://doi.org/10.3390/md403070




