1. Introduction
Sesterterpenoids are a group of compounds with C
25 carbon frameworks derived from five isoprene units, and exhibit a variety of biological activities including cytotoxic and antimicrobial functions [
1,
2,
3]. They have been found from widespread sources including fungi, bacteria, plants, insects and marine organisms [
4,
5]. Marine organisms represent a broad source of potential sesterterpenoids. For example, genus
Phyllospongia, one of the most common marine sponges, has been shown to be a prolific producer of various scalarane sesterterpenoids [
6], and marine-derived fungi belong to the genus of
Aspergillus produced sesterterpenoid ophiobolins [
7].
Ophiobolins (Ophs) are a class of sesterpenoids and characterized by the structure of a tricyclic 5-8-5 ring system derived from head to tail linkages of five isoprene units (
Figure 1). These small molecules, ranged in molecular weight from 338 to 432, are produced by fungi mainly belong to the genus
Bipolaris and
Aspergillus. The first member of this family named ophiobolin A (compound
1,
Figure 2) was isolated from the pathogenic plant fungus
Ophiobolus miyabeanus by Nakamura and Ishibashi in 1958 [
8]. Numerous additional analogs have been reported since then. Here, we summarize 49 natural products of Ophs, which are assigned into 23 subgroups (A–W).
There are enormous potential applications of Ophs in different fields due to their wide biological effects. Initially, the researchers focused on the investigations of phytotoxic properties of Ophs, which are produced by pathogenic fungi attacking agricultural crops [
9,
10,
11,
12,
13,
14,
15]. Later, Ophs have been proven to possess a broad spectrum of other biological properties, such as antimicrobial [
15,
16], nematocidal [
17,
18,
19] and cytotoxic activities [
1,
5,
9]. Additionally, MDR reversal effects of Ophs on multiple tumor cells with a low concentration were proven by both in vitro and in vivo experiments [
20,
21]. Furthermore, Ophs also possess anti-influenza [
22] and inflammation-promoting activities [
23]. With their diverse pharmacological properties, Ophs are shown to be a group of promising drug candidates. Although a variety of potential biological activities of Ophs have been widely investigated, their molecular targets, the functional mechanisms, and structure–activity relationships are still obscure. After Au et al. (2000) reviewed the biology of 23 Ophs [
9], 26 more Ophs have been reported/added to the family. In this review, we summarize the chemical structures, sources, and biological properties of 49 reported Ophs. Furthermore, we will focus on the cytotoxic properties, mechanism of action, and structure–activity relationship of Ophs to demonstrate their pervasiveness and importance in drug discovery.
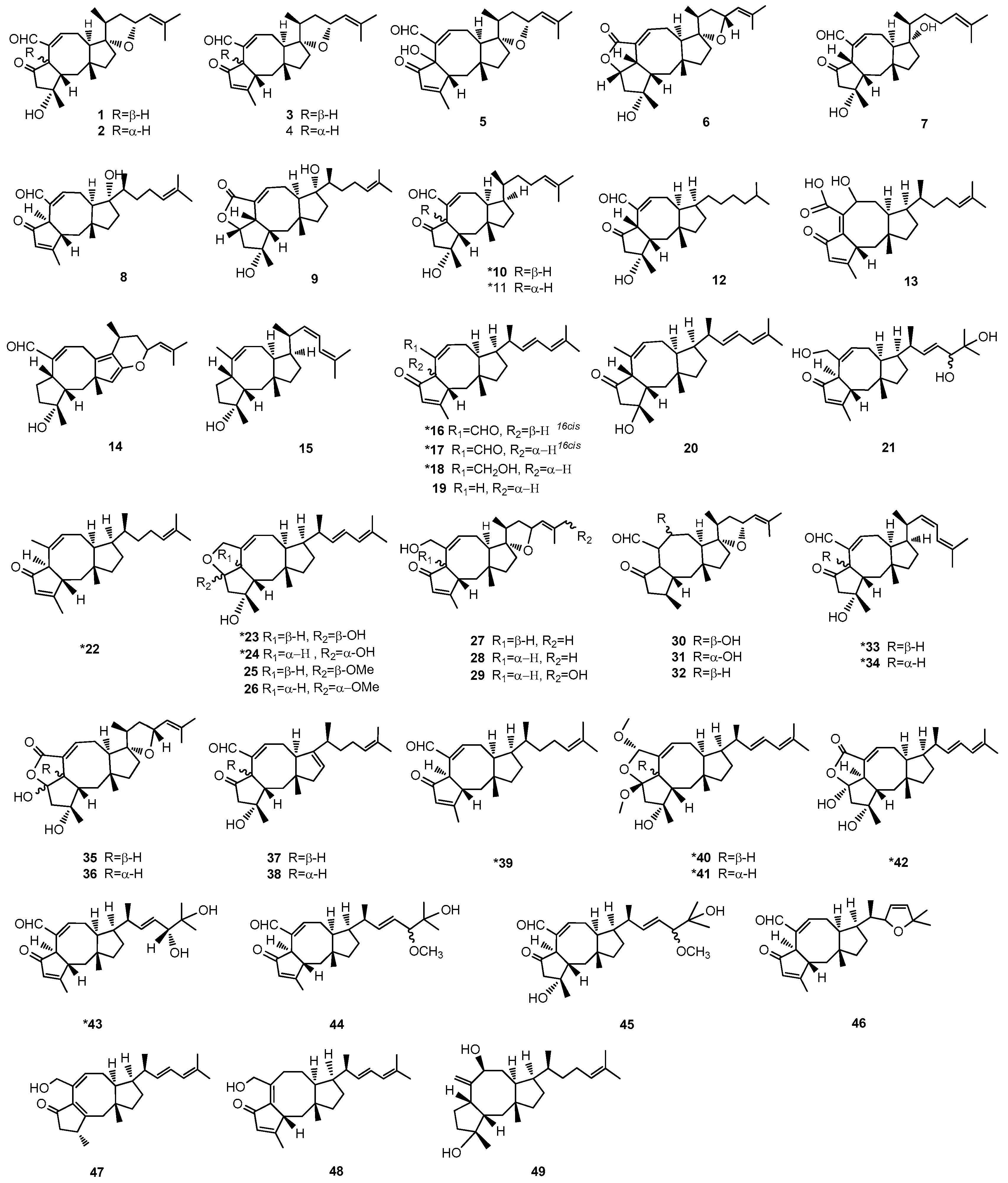
2. Chemical Structure and Source of Ophs
These small molecules are characterized by a core structure of 5-8-5 tricyclic carbon skeletons (
Figure 1) and assigned into A–W subgroups. All reported Ophs, numbered from 1–49, are shown in
Figure 2. The original names of the biological sources of Ophs are listed in
Table 1.
With many structurally similar analogs, Ophs family needed an approach to distinguish between them [
24,
25]. Kildgaard et al. (2014) reported that Ophs can be dereplicated accurately by using ultra-high performance liquid chromatography–diode array detection–quadrupole time of flight mass spectrometry (UHPLC-DAD-QTOFMS) with a tandem high resolution MS (MS/HRMS) Library and eight Ophs were available as standards and included in the library at present [
25].
In the earlier studies, different research groups adopted different taxonomical nomenclatures for the same pathogenic fungi, which caused confusions. Therefore, some of the fungi have been reclassified. For example, Drechslera oryzae and Helminthosporium oryzae have been reclassified in Bipolaris oryzae.
The majority of Ophs were discovered from the fungi genera of
Bipolaris and
Aspergillus. For example, genus
Bipolaris produces ophiobolins A–B, I–J, and L–M, while genus
Aspergillus produces ophiobolins C, F–H, K, N, and U–W (
Table 1). Marine-derived fungi belong to
Aspergillus produce various Ophs. For example,
Aspergillus sp. (Taxonomy ID: 5065) produces ophiobolins G, H, K and O [
4] (
Table 1); and
Emericella variecolor (reclassified in
Aspergillus stellatu, Taxonomy ID: 1549217) produces ophiobolins C, G, H, K and N [
26,
27] (
Table 1). The other genera including
Drechslera [
28],
Ulocladium [
29],
Penicillium [
30],
Mollisia [
31], and
Cephalosporium [
32], were producers of a part of the discovered structures. Moreover, ophiobolin A lactone and ophiobolin B lactone were transformed from ophiobolin A by the bacteria
Polyangium cellulosum and
Pseudomonas aeruginosa, respectively [
30].
It is not yet known whether the similarities in structures of Ophs are drastically associated with genus. The Ophs (compounds 1–5, 27–32, 35, and 36) with furan structure at C14-17 are majorly produced by Bipolaris, while the Ophs without this structure are majorly produced by Aspergillus. On the other hand, Cochliobolus heterostrophus, which was later classified to Bipolaris, can produce both structures with (compounds 1–4, 27, and 35) or without (compounds 7, 8, 10–12, 15, 23, 33, 34, 37, and 38) the furan ring.
It is worth noting that the biosynthetic production of Ophs is influenced by the different culture conditions. For example,
Drechslera gigantean (Taxonomy ID: 1937655), a global fungal plant pathogen, was able to produce
1,
2,
4, and
27 in liquid cultures. However, in solid conditions, it produced
7,
14,
30, and
31 [
26]. While all the compounds produced from liquid culture and some compounds from solid culture have the furan structure,
7 and
14 did not have the ring. Before confirmation of the genus position of
Cochliobolus heterostrophus from different reports, the correlation between the Ophs production and the taxonomic position of their source strains or their culture conditions is still uncertain.
3. Phytotoxic Activities
Early research considered Ophs as the metabolites of pathogenic fungi, which attacked various crops by inducing brown spot lesions on the leaves [
9]. These crops included monocotyledons and dicotyledons species, especially grass weeds [
12]. Furthermore, compound
1 inhibited the growth of Tobacco Bright Yellow-2 (TBY-2) at concentrations of 2–5 μM [
21,
50].
Table 2 lists the phytotoxic activities of Ophs on 12 tested plants.
Compound
1 was proven to be phytotoxic to almost all of the tested plants, even at the lowest concentration. Compound
1 gave necrotic legions on cabbage and corn while
2,
3,
4,
7, and
27 did not (
Table 2) [
13,
14,
15]. Ophs (
1,
2,
3,
4,
7,
27) showed phytotoxic properties by causing necrosis on the plants except tobacco [
15]. Furthermore, the Ophs exhibited other phytotoxic effects:
1 was effective in reducing sheath blight incidence under field conditions [
51], and both
16 and
23 inhibited growth of etiolated wheat coleoptile at 10 μM [
44]. Furthermore, while Xiao et al. (1991) had illustrated that
27 (1.2 × 10
3 μM) was inactive to corn leaves [
14], Sugawara et al. (1987) and Kim et al. (1999) demonstrated it was active to corn leaves at a lower concentration (1.0 × 10
3 μM) [
13,
15].
The ophiobolin A-series
1,
2, and
3 appeared to be more active on the tested plants than other Ophs, but
4 displayed low activity at the same concentration (
Table 2). Compounds
1 and
3 were β-H isomers of
2 and
4 at C-6, respectively. Comparing phytotoxic effects between the two groups of isomers, β-H (C-6) of compounds enhanced the action on barley, corn, goose grass, and velvet-leaf [
14,
15]. However, β-H (C-6) of
1 suppressed the action on cocklebur and Johnson grass, and there was no influence on sorghum and soybean [
13,
15] (
Table 2). However, β-H (C-6) of
3 improved activity on cocklebur, rice and soybean [
15] (
Table 2). Therefore, the structure-phytotoxic activity of stereochemistry at C6 was obscure, as β-H (C-6) did not always elevate phytotoxic effect.
The structures of
3 and
4 lacked a hydroxy group at C-3 on the basis of
1 and
2, respectively. The hydroxy (C-3) of
1 improved the inhibition against barley and cabbage, reduced the activity on cocklebur and tested plants, while the activity on the remaining tested plants was unchanged (
Table 2) [
13,
14,
15]. The hydroxy (C-3) of
2 elevated phytotoxic effects on all the tested plants except grain sorghum and velvet-leaf, whose growth had no distinction between
2 and
4 (
Table 2) [
13,
15]. At this moment, it is impossible to determine whether the hydroxy group at C-3 of Ophs improved their phytotoxic activity or not. For hydroxyl at C-14, there is no difference between the activities of
7 and
10 on corn, Johnson grass, and sorghum (
Table 2) [
13]. However, comparing with
1, when there was no hydroxy group at C-3 and α-H existed at C-6 in
4, the phytotoxic action was significantly decreased in all the test plants [
13,
14,
15] except goose grass (
Table 2) [
15].
Sugawara et al. (1987) demonstrated that compound
1 was much more phytotoxic than
10 against corn, Johnson grass, and sorghum, which suggested that the lack of the configuration of the tetrahydrofuran ring between C14 to C17 possibly reduced phytotoxicities (
Table 2) [
13]. The structural difference between
1 and
27 was that hydroxy (C3) and aldehyde (C21) attached to
1, double bond (C3) and hydroxy (C21) attached to
27. Compound
1 visibly elevated the activities on all the experimental plants except cocklebur, goose grass, and velvet-leaf [
15], whose growth had no difference between them (
Table 2) [
13,
14,
15]. The carbonyl group (C21) of
3 significantly improved the phytotoxic effect on all the tested plants except cabbage [
14] and goose grass [
15], the growth of which had no difference (
Table 2) [
13,
14,
15].
Besides the phytotoxic effects displayed in
Table 2, molecules
1,
2 and
4 were proven to be phytotoxic to several monocotyledon and dicotyledon weeds [
12]. The structure–activity relationship was similar to the above characterizations. β-H (C-6) did not always enhance phytotoxic effect when comparing
1 with
2. Whereas
1 displayed strong activities on nearly all the tested weeds in comparison with
4, the possible structural features for reducing phytotoxic activities could be a combination of the lack of the hydroxy group at C-3 and the stereochemistry of hydrogen atom at C-6 (from β-H to α-H) [
12].
Ophs produce phytotoxic activities in plants through multiple mechanisms of action. Early studies have demonstrated that both
1 and
7 could inhibit the germination and growth of
Oryzo sativa seeds [
11], infect rice leaves, and cause leaf brown spot through reducing the photosynthetic leaf area and the efficient use of nutrients [
52]. It is observed that
1 have stimulated the net leakage of electrolytes from roots, leaves, and coleoptiles of rice plants and loss of β-cyanin from beetroot tissue at 250 μM [
53]. Furthermore, the molecule induced a loss of osmotic pressure in the guard cells, ultimately resulted in stomatal closure and decreases of
gs [
54], and caused loss of electrolytes and glucose from maize and carrot roots [
10]. Treat tissues with
1 effectively inhibited the uptake of exogenous
32PO
4 and 2-deoxyglucose at 25 and 250 μM, respectively [
10,
53]. In addition,
7 exhibited inhibitory effects of proton extrusion from maize coleoptiles [
39]. The release of electrolytes could explain that the phytotoxicity of Ophs, in stimulating intracellular potassium, phosphate, glucose, carbohydrates, amino acids, and proteins leakage, influenced the transport processes of plasma membrane systems [
10,
39,
53]. Later, it has been demonstrated that the activity of
1 associating with calmodulin played a critical role in the development of cell cycle [
55]. Moreover, lysine 75 was the primary inhibitory site for
1, other amino acids including lysine 75, 77, and 148 acted as the binding sites of calmodulin antagonism [
56,
57]. Furthermore, compound
1 performed the inhibitory of TBY-2 through arresting the cell cycle and altering the intracellular partitioning of glutathione between nuclei and cytoplasm [
21,
50].
10. Conclusions and Future Perspectives
Belonging to the tricarbocyclic sesterterpenoids with a 5-8-5 ring system structure, 49 natural Ophs have been reported and assigned into A–W in order of discovery. They display potential applications in various fields due to their broad spectrum of biological characteristics such as phytotoxic, antimicrobial, nematocidal, cytotoxic, anti-influenza, and inflammation-promoting activities. Particularly, these sesterterpenoid-type molecules exhibit anti-proliferative activities against a vast number of cancer cell lines mainly including breast, cervical, colon, glioblastoma multiforme (GBM), lung, leukemia, and melanoma carcinoma. They also display potent cytotoxic properties against multidrug resistance (MDR) and cancer stem cells (CSCs). The anti-proliferative activity against a broad range of cell lines entitle the sesterterpenoid-type Ophs with great possibilities in the utilization of their diverse pharmacological properties in drug development especially in cancer drug discovery. Actually, numerous drugs in clinical have been proven to exhibit broad biological activities. For example, recent investigation illustrated that clarithromycin, a well-known antibiotic drug, possessed anti-cancer effects in multiple tumor types on the base of its extensive preclinical and clinical data [
79]. Indeed, the sesterterpenoid-type compounds are promising drug candidates, as current studies are attempting to prepare chemoembolization particles possessing the sustained release effects for the delivery of compound
1 to the target locations [
63].
More studies are required for obtaining biological Ophs. Although many researchers have studied their chemical synthesis, merely few Ophs can be totally synthesized [
80,
81,
82,
83]. Traditional total synthesis of Ophs needs a complex multistep reaction. However, Brill et al. (2016) have discovered that 6-epi-ophiobolin-N could be totally synthesized in only nine steps, which is one of the shortest total synthesis steps for sesterterpenoids by far, through the “biomimetic synthetic strategy” [
84]. However, there are very few reports on Ophs biosynthesis. Recent investigations demonstrate that the biosynthesis of Ophs was related to the first sesterterpene synthase (AcOS) [
85] and the
oblBAc gene (cytochrome P450) from the cryptic gene cluster [
86], and the biosynthesis of Ophs skeleton involves multiple gene clusters, which are in charge of C15, C20, C25, and C30 terpenoid biosynthesis [
87].
Despite the vast number of investigations on biological and pharmacological properties of Ophs in the recent years, we still have only preliminary understanding of the structure–activity relationship and the functional mechanism of Ophs, which are crucial for uncovering novel molecular candidates. As discussed in this review, the structure–activity relationship is still not established. However, under many circumstances, it can be deduced that Ophs with a 6-alpha (6R) stereochemistry have weaker biological activities including phytotoxic, antibacterial, and nematocidal activities than Ophs with a 6S stereochemistry; Ophs with a tetrahydrofuran ring between C14 to C17 have stronger activities including phytotoxic, antifungal, and anticancer activities; Ophs with a tetrahydrofuran ring between C5 to C7 and C21 have significant weaker anti-CSC activities. Furthermore, the dicarbonyl at C5 and C21, and the hydroxyl group attached to C-3, C-6 or C14, are critical for Ophs’ anticancer activities.
The biological and pharmacological characteristics of Ophs are intrinsically associated with influencing the process of proliferation and mediating different pathways as a sure Ca2+/calmodulin antagonist. The bioactive molecules induce cell cycle arrest, apoptosis, paraptosis, and autophagic process by different mechanisms depending on the cell type, which revealed that Ophs acted on multiple targets. Additionally, the PE non-protein target related to lipid membrane may explain the broad bioactivities of Ophs on plants, microorganisms, nematode, trypanosome, and mammalian cells, since PE is ubiquitously found in the nature. Further investigations are needed to clarify whether this covalent reaction exists not only in membrane PE but also in the other proteins such as the calmodulin that possesses the primary amines. Furthermore, Additional research is needed in order to illustrate how this non-protein target connected with the other observations such as the pathways and the different morphologies of cell death such as apoptosis, paraptosis, and necrosis. More evidence is still required to elucidate whether Ophs are multi-targets compounds or a single target accounts for their biological activity, regardless of structure.