Antimetastatic Effect of Halichondramide, a Trisoxazole Macrolide from the Marine Sponge Chondrosia corticata, on Human Prostate Cancer Cells via Modulation of Epithelial-to-Mesenchymal Transition
Abstract
:1. Introduction
2. Results
2.1. Growth Inhibitory Activity of Halichondramide in PC3 Prostate Cancer Cells
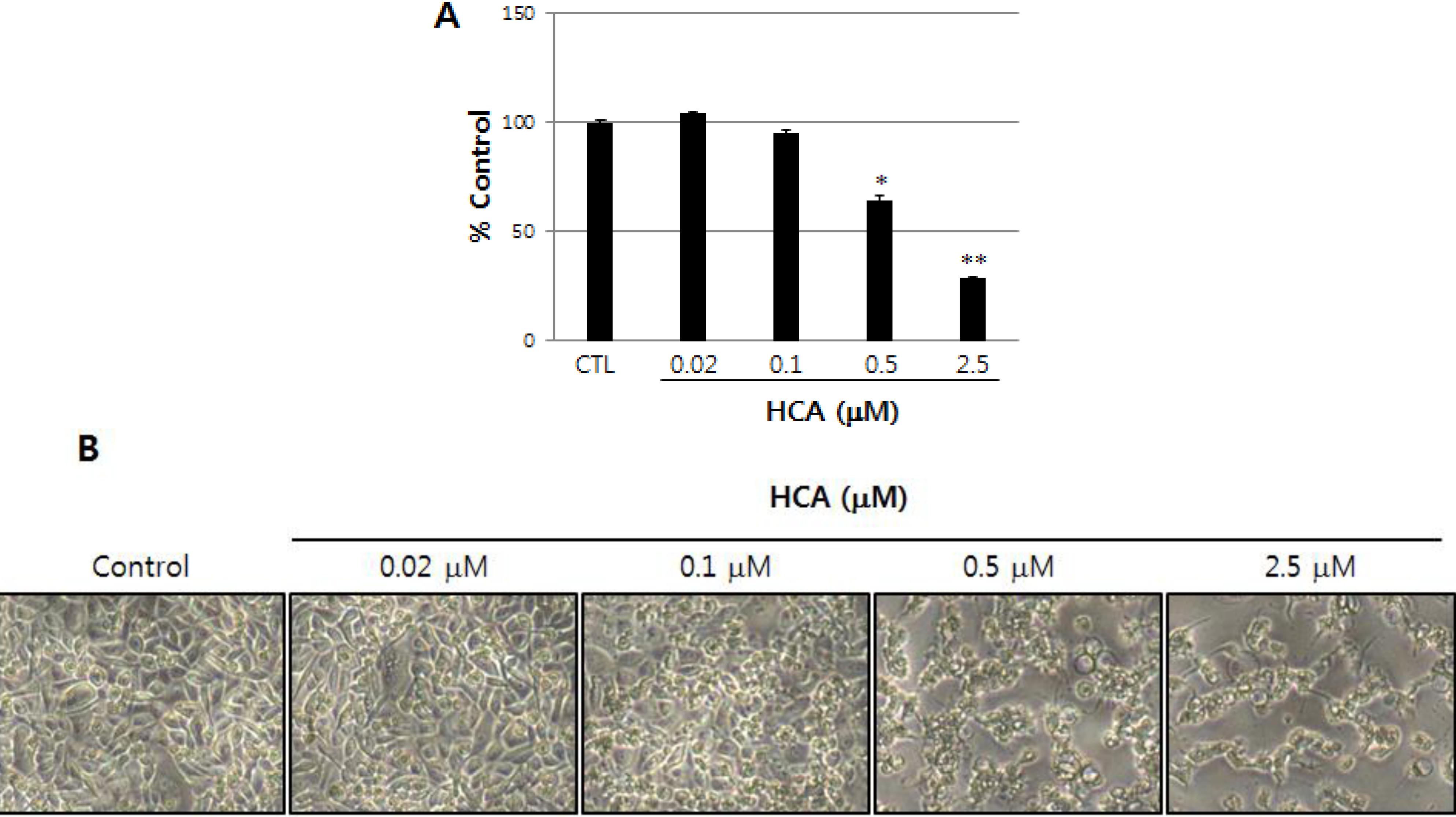
2.2. Modulation of the Gene Expression Levels of PRL-3, MMPs, and Cadherins by HCA

2.3. Suppressive Expression of PRL-3 and Its Associated Proteins by HCA

2.4. Inhibitory Effect of HCA on Cell Migration and Invasion
2.5. Suppressive Effect of HCA on the MMPs in Gelatin Zymography
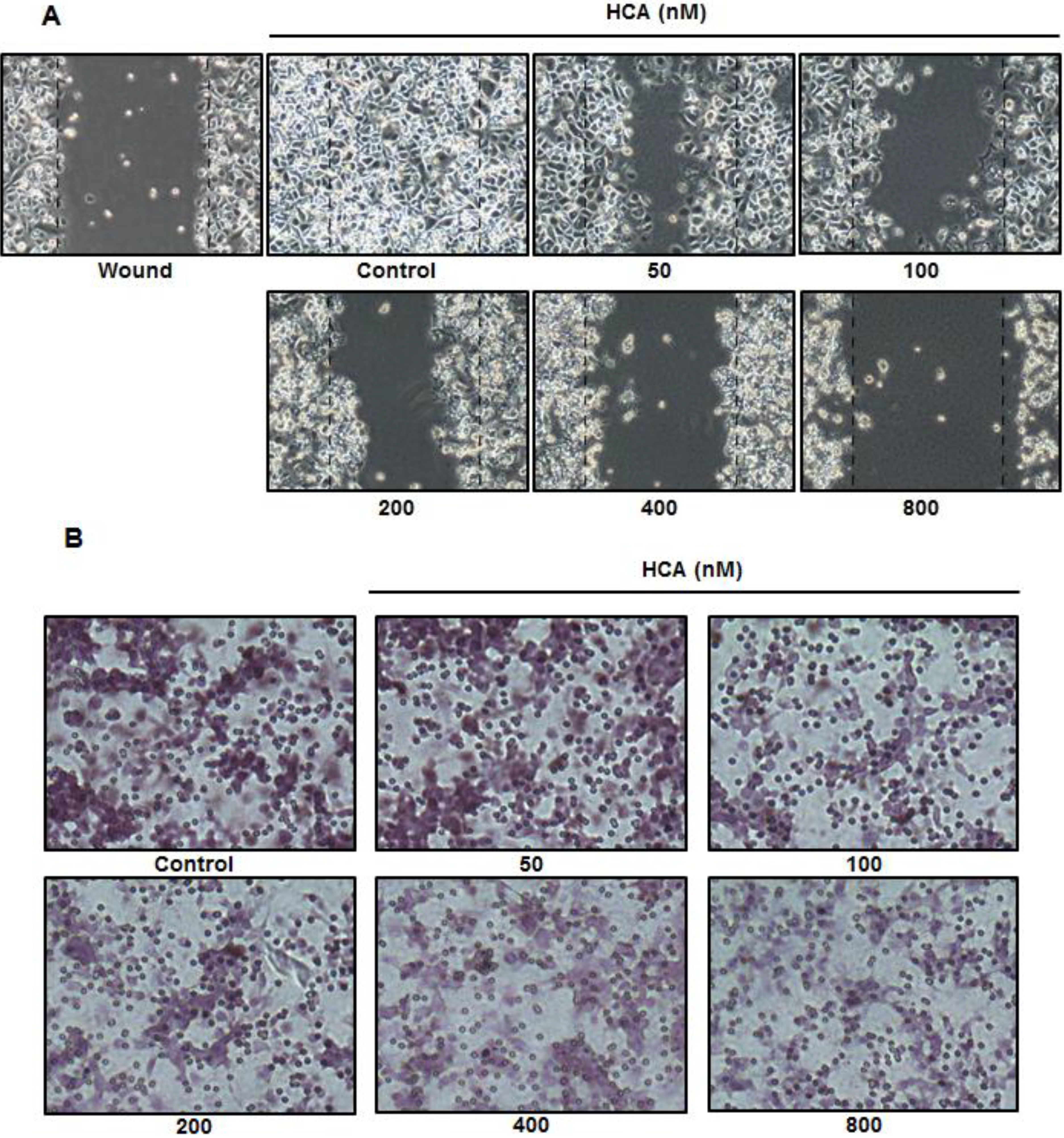
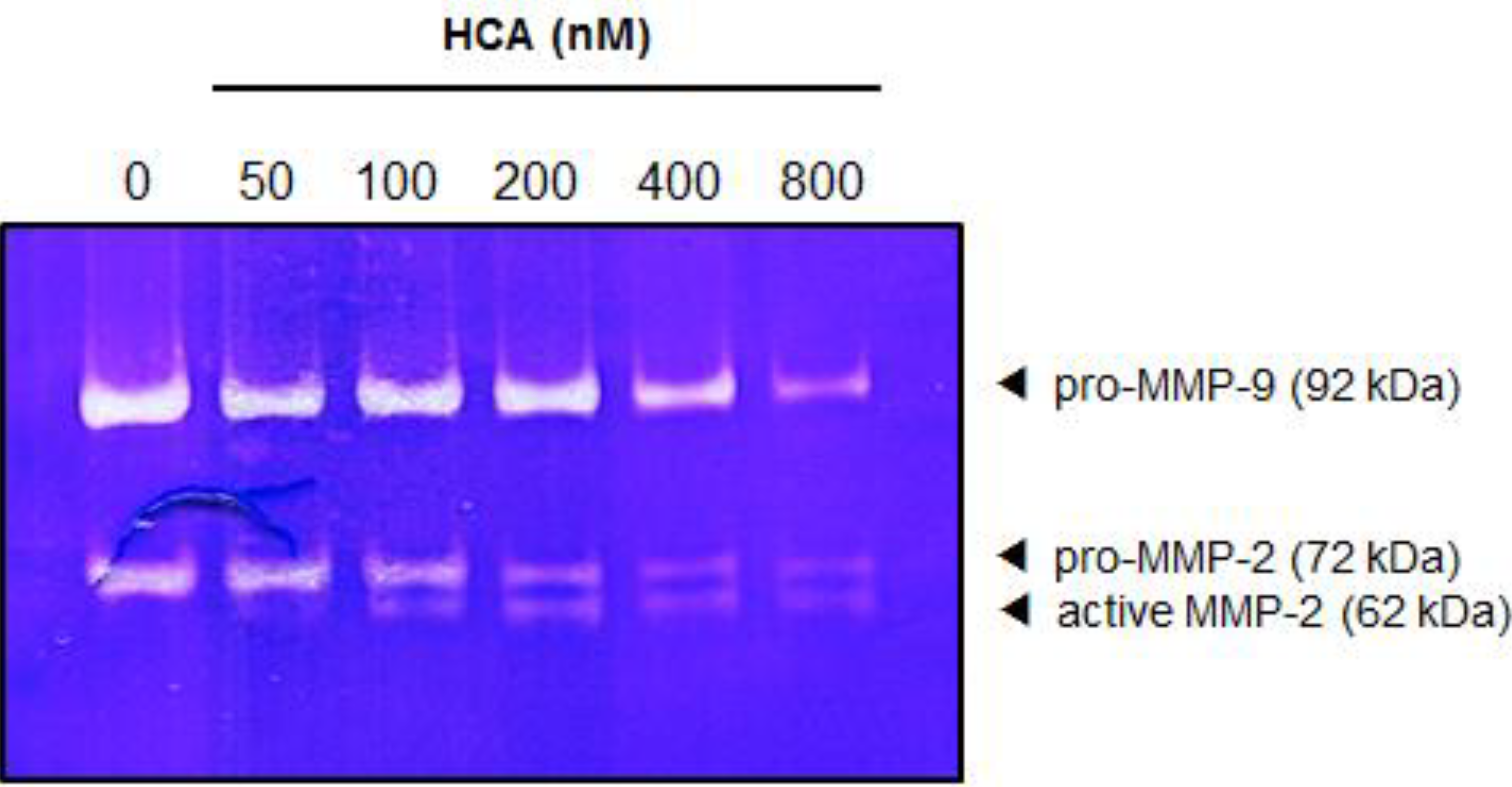
3. Discussion
4. Experimental Section
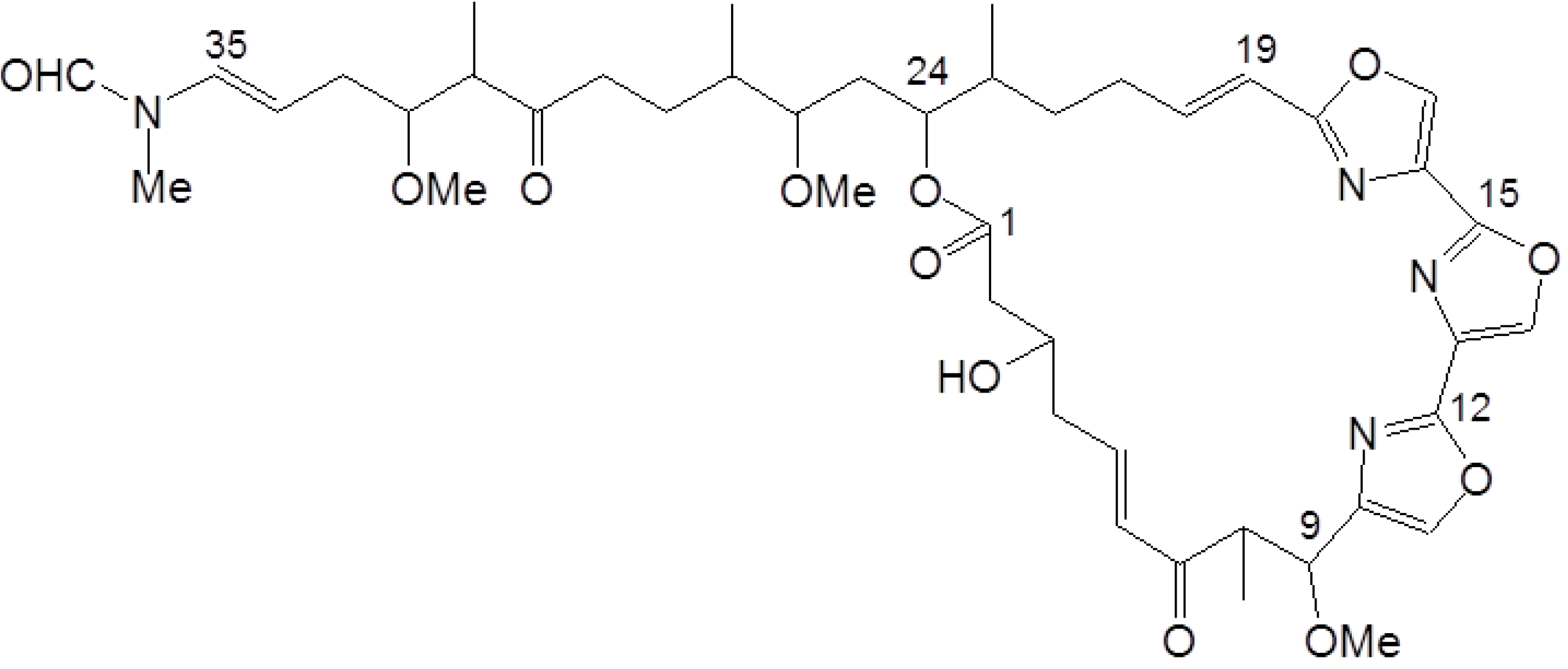
4.1. Reagents
4.2. Cell Culture
4.3. The Evaluation of Antiproliferation Activity
4.4. Analysis of Gene Expression by Real-Time RT-PCR
| Target gene | Sequences | |
|---|---|---|
| PRL-3 | Sense | 5′-AGA AGT ACG GGG CTA CCA CTG-3′ |
| Antisense | 5′-CAC AAC GGT GAT GCC ATC CTT-3′ | |
| p85 | Sense | 5′-CTG CCT CCT AAA CCA CCA AAA-3′ |
| Antisense | 5′-TTC ATA CCG TTG TTG GCT ACA G-3′ | |
| p110 | Sense | 5′-CCA CGA CCA TCA TCA GGT GAA-3′ |
| Antisense | 5′-CCT CAC GGA GGC ATT CTA AAG T-3′ | |
| E-cadherin | Sense | 5′-CGA GAG CTA CAC GTT CAC GG-3′ |
| Antisense | 5′-GGG TGT CGA GGG AAA AAT AGG-3′ | |
| N-cadherin | Sense | 5′-AGC CAA CCT TAA CTG AGG AGT-3′ |
| Antisense | 5′-GGC AAG TTG ATT GGA GGG ATG-3′ | |
| β-Actin | Sense | 5′-AGC ACA ATG AAG ATC AAG AT-3′ |
| Antisense | 5′-TGT AAC GCA ACT AAG TCA TA-3′ | |
| MMP2 | Sense | 5′-GAT ACC CCT TTG ACG GTA AGG A-3′ |
| Antisense | 5′-CCT TCT CCC AAG GTC CAT AGC-3′ | |
| MMP9 | Sense | 5′-TGT ACC GCT ATG GTT ACA CTC G-3′ |
| Antisense | 5′-GGC AGG GAC AGT TGC TTC T-3′ | |
4.5. Western Blot Analysis
4.6. Wound Healing Assay
4.7. Cell Migration Assay
4.8. Gelatin Zymography
4.9. Statistics
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2012, 29, 144–222. [Google Scholar]
- Montaser, R.; Luesch, H. Marine natural products: A new wave of drugs? Future Med. Chem. 2011, 3, 1475–1489. [Google Scholar] [CrossRef]
- Halim, H.; Chunhacha, P.; Suwanborirux, K.; Chanvorachote, P. Anticancer and antimetastatic activities of Renieramycin M, a marine tetrahydroisoquinoline alkaloid, in human non-small cell lung cancer cells. Anticancer Res. 2011, 31, 193–201. [Google Scholar]
- Nguyen, H.T.; Chau, V.M.; Tran, T.H.; Phan, V.K.; Hoang, T.H.; Nguyen, T.D.; Nguyen, X.N.; Tai, B.H.; Hyun, J.H.; Kang, H.K.; et al. C29 sterols with a cyclopropane ring at C-25 and 26 from the Vietnamese marine sponge Ianthella sp. and their anticancer properties. Bioorg. Med. Chem. Lett. 2009, 19, 4584–4588. [Google Scholar] [CrossRef]
- Kijjoa, A.; Wattanadilok, R.; Campos, N.; Nascimento, M.S.; Pinto, M.; Herz, W. Anticancer activity evaluation of kuanoniamines A and C isolated from the marine sponge Oceanapia sagittaria, collected from the Gulf of Thailand. Mar. Drugs 2007, 5, 6–22. [Google Scholar] [CrossRef]
- Jain, S.; Vahdat, L.T. Eribulin mesylate. Clin. Can. Res. 2011, 17, 6615–6622. [Google Scholar] [CrossRef]
- Shin, J.; Lee, H.S.; Kim, J.Y.; Shin, H.J.; Ahn, J.W.; Paul, V.J. New macrolides from the sponge Chondrosia corticata. J. Nat. Prod. 2004, 67, 1889–1892. [Google Scholar] [CrossRef]
- Bae, S.Y.; Kim, G.D.; Jeon, J.E.; Shin, J.; Lee, S.K. Anti-proliferative effect of (19Z)-halichondramide, a novel marine macrolide isolated from the sponge Chondrosia corticata, is associated with G2/M cell cycle arrest and suppression of mTOR signaling in human lung cancer cells. Toxicol. In Vitro 2012, 27, 694–699. [Google Scholar]
- Yoon, S.O.; Park, S.J.; Yun, C.H.; Chung, A.S. Roles of matrix metalloproteinases in tumor metastasis and angiogenesis. J. Biochem. Mol. Biol. 2003, 36, 128–137. [Google Scholar] [CrossRef]
- Diamond, R.H.; Cressman, D.E.; Laz, T.M.; Abrams, C.S.; Taub, R. PRL-1, a unique nuclear protein tyrosine phosphatase, affects cell growth. Mol. Cell. Biol. 1994, 14, 3752–3762. [Google Scholar]
- Cates, C.A.; Michael, R.L.; Stayrook, K.R.; Harvey, K.A.; Burke, Y.D.; Randall, S.K.; Crowell, P.L.; Crowell, D.N. Prenylation of oncogenic human PTP(CAAX) protein tyrosine phosphatases. Cancer Lett. 1996, 110, 49–55. [Google Scholar]
- Zeng, Q.; Hong, W.; Tan, Y.H. Mouse PRL-2 and PRL-3, two potentially prenylated protein tyrosine phosphatases homologous to PRL-1. Biochem. Biophys. Res. Commun. 1998, 244, 421–427. [Google Scholar]
- Stephens, B.J.; Han, H.; Gokhale, V.; von Hoff, D.D. PRL phosphatases as potential molecular targets in cancer. Mol. Cancer Ther. 2005, 4, 1653–1661. [Google Scholar] [CrossRef]
- Sager, J.A.; Benvenuti, S.; Bardelli, A. PRL-3: A phosphatase for metastasis? Cancer Biol. Ther. 2004, 3, 952–953. [Google Scholar] [CrossRef]
- Bardelli, A.; Saha, S.; Sager, J.A.; Romans, K.E.; Xin, B.; Markowitz, S.D.; Lengauer, C.; Velculescu, V.E.; Kinzler, K.W.; Vogelstein, B. PRL-3 expression in metastatic cancers. Clin. Cancer Res. 2003, 9, 5607–5615. [Google Scholar]
- Wang, L.; Shen, Y.; Song, R.; Sun, Y.; Xu, J.; Xu, Q. An anticancer effect of curcumin mediated by down-regulating phosphatase of regenerating liver-3 expression on highly metastatic melanoma cells. Mol. Pharmacol. 2009, 76, 1238–1245. [Google Scholar] [CrossRef]
- Wu, X.; Zeng, H.; Zhang, X.; Zhao, Y.; Sha, H.; Ge, X.; Zhang, M.; Gao, X.; Xu, Q. Phosphatase of regenerating liver-3 promotes motility and metastasis of mouse melanoma cells. Am. J. Pathol. 2004, 164, 2039–2054. [Google Scholar] [CrossRef]
- Zeng, Q.; Dong, J.M.; Guo, K.; Li, J.; Tan, H.X.; Koh, V.; Pallen, C.J.; Manser, E.; Hong, W. PRL-3 and PRL-1 promote cell migration, invasion, and metastasis. Cancer Res. 2003, 63, 2716–2722. [Google Scholar]
- Saha, S.; Bardelli, A.; Buckhaults, P.; Velculescu, V.E.; Rago, C.; St Croix, B.; Romans, K.E.; Choti, M.A.; Lengauer, C.; Kinzler, K.W.; et al. A phosphatase associated with metastasis of colorectal cancer. Science 2001, 294, 1343–1346. [Google Scholar] [CrossRef]
- Araki, K.; Shimura, T.; Suzuki, H.; Tsutsumi, S.; Wada, W.; Yajima, T.; Kobayahi, T.; Kubo, N.; Kuwano, H. E/N-cadherin switch mediates cancer progression via TGF-beta-induced epithelial-to-mesenchymal transition in extrahepatic cholangiocarcinoma. Br. J. Cancer 2011, 105, 1885–1893. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Thiery, J.P.; Sleeman, J.P. Complex networks orchestrate epithelial-mesenchymal transitions. Nat. Rev. Mol. Cell Biol. 2006, 7, 131–142. [Google Scholar] [CrossRef]
- Kiyoto, S.; Kawai, Y.; Kawakita, T.; Kino, E.; Okuhara, M.; Uchida, I.; Tanaka, H.; Hashimoto, M.; Terano, H.; Kohsaka, M.; et al. A new antitumor complex, WF-1360, WF-1360A, B, C, D, E and F. J. Antibiot. 1986, 39, 762–772. [Google Scholar] [CrossRef]
- McParland, V.; Varsano, G.; Li, X.; Thornton, J.; Baby, J.; Aravind, A.; Meyer, C.; Pavic, K.; Rios, P.; Kohn, M. The metastasis-promoting phosphatase PRL-3 shows activity toward phosphoinositides. Biochemistry 2011, 50, 7579–7590. [Google Scholar] [CrossRef]
- Matter, W.F.; Estridge, T.; Zhang, C.; Belagaje, R.; Stancato, L.; Dixon, J.; Johnson, B.; Bloem, L.; Pickard, T.; Donaghue, M.; et al. Role of PRL-3, a human muscle-specific tyrosine phosphatase, in angiotensin-II signaling. Biochem. Biophys. Res. Commun. 2001, 283, 1061–1068. [Google Scholar] [CrossRef]
- Werner, S.R.; Lee, P.A.; DeCamp, M.W.; Crowell, D.N.; Randall, S.K.; Crowell, P.L. Enhanced cell cycle progression and down regulation of p21(Cip1/Waf1) by PRL tyrosine phosphatases. Cancer Lett. 2003, 202, 201–211. [Google Scholar] [CrossRef]
- Ming, J.; Liu, N.; Gu, Y.; Qiu, X.; Wang, E.H. PRL-3 facilitates angiogenesis and metastasis by increasing ERK phosphorylation and up-regulating the levels and activities of Rho-A/C in lung cancer. Pathology 2009, 41, 118–126. [Google Scholar] [CrossRef]
- Cully, M.; You, H.; Levine, A.J.; Mak, T.W. Beyond PTEN mutations: The PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nat. Rev. Cancer 2006, 6, 184–192. [Google Scholar] [CrossRef]
- Tripathi, P.; Kamarajan, P.; Somashekar, B.S.; MacKinnon, N.; Chinnaiyan, A.M.; Kapila, Y.L.; Rajendiran, T.M.; Ramamoorthy, A. Delineating metabolic signatures of head and neck squamous cell carcinoma: Phospholipase A2, a potential therapeutic target. Int. J. Biochem. Cell Biol. 2012, 44, 1852–1861. [Google Scholar] [CrossRef]
- MacKinnon, N.; Khan, A.P.; Chinnaiyan, A.M.; Rajendrian, T.M.; Ramamoorthy, A. Androgen receptor activation results in metabolite signatures of an aggressive prostate cancer phenotype: An NMR-based metabolomics study. Metabolomics 2012, 8, 1026–1036. [Google Scholar] [CrossRef]
- Cho, S.K.; Hong, J.Y.; Park, H.J.; Lee, S.K. Anticancer activity of novel daphnane diterpenoids from Daphne genkwa through cell-cycle arrest and suppression of Akt/STAT/Src signalings in human lung cancer cells. Biomol. Ther. 2012, 20, 513–519. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Kim, G.D.; Cheong, O.J.; Bae, S.Y.; Shin, J.; Lee, S.K. 6″-debromohamacanthin A, a Bis (Indole) alkaloid, inhibits angiogenesis by targeting the VEGFR2-mediated PI3K/AKT/mTOR signaling pathways. Mar. Drugs 2013, 11, 1087–1103. [Google Scholar] [CrossRef]
- Hwang, H.J.; Park, H.J.; Chung, H.J.; Min, H.Y.; Park, E.J.; Hong, J.Y.; Lee, S.K. Inhibitory effects of caffeic acid phenethyl ester on cancer cell metastasis mediated by the down-regulation of matrix metalloproteinase expression in human HT1080 fibrosarcoma cells. J. Nutr. Biochem. 2006, 17, 356–362. [Google Scholar] [CrossRef]
- Park, E.J.; Park, H.J.; Chung, H.J.; Shin, Y.; Min, H.Y.; Hong, J.Y.; Kang, Y.J.; Ahn, Y.H.; Pyee, J.H.; Lee, S.K. Antimetastatic activity of pinosylvin, a natural stilbenoid, is associated with the suppression of matrix metalloproteinases. J. Nutr. Biochem. 2012, 23, 946–952. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shin, Y.; Kim, G.D.; Jeon, J.-e.; Shin, J.; Lee, S.K. Antimetastatic Effect of Halichondramide, a Trisoxazole Macrolide from the Marine Sponge Chondrosia corticata, on Human Prostate Cancer Cells via Modulation of Epithelial-to-Mesenchymal Transition. Mar. Drugs 2013, 11, 2472-2485. https://doi.org/10.3390/md11072472
Shin Y, Kim GD, Jeon J-e, Shin J, Lee SK. Antimetastatic Effect of Halichondramide, a Trisoxazole Macrolide from the Marine Sponge Chondrosia corticata, on Human Prostate Cancer Cells via Modulation of Epithelial-to-Mesenchymal Transition. Marine Drugs. 2013; 11(7):2472-2485. https://doi.org/10.3390/md11072472
Chicago/Turabian StyleShin, Yoonho, Gi Dae Kim, Ju-eun Jeon, Jongheon Shin, and Sang Kook Lee. 2013. "Antimetastatic Effect of Halichondramide, a Trisoxazole Macrolide from the Marine Sponge Chondrosia corticata, on Human Prostate Cancer Cells via Modulation of Epithelial-to-Mesenchymal Transition" Marine Drugs 11, no. 7: 2472-2485. https://doi.org/10.3390/md11072472





