Astaxanthin Attenuates the Apoptosis of Retinal Ganglion Cells in db/db Mice by Inhibition of Oxidative Stress
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Astaxanthin Improves Oscillatory Potentials(OPs) in db/db Mice

2.1.2. Astaxanthin Inhibits the Apoptosis of Retinal Ganglion Cells (RGCs) in db/db Mice
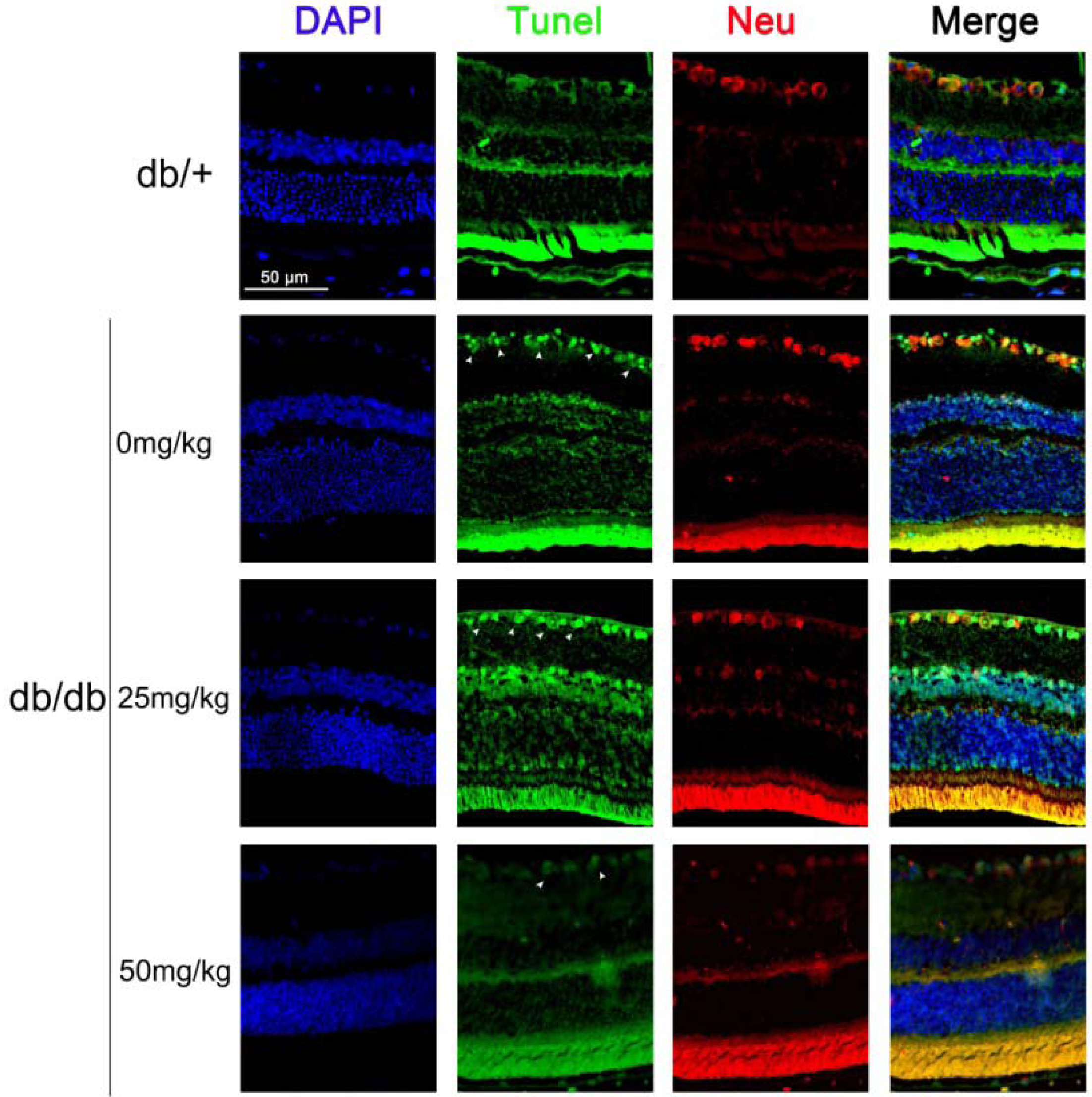
2.1.3. Astaxanthin Reduces Oxidative Stress of the Retina in db/db Mice
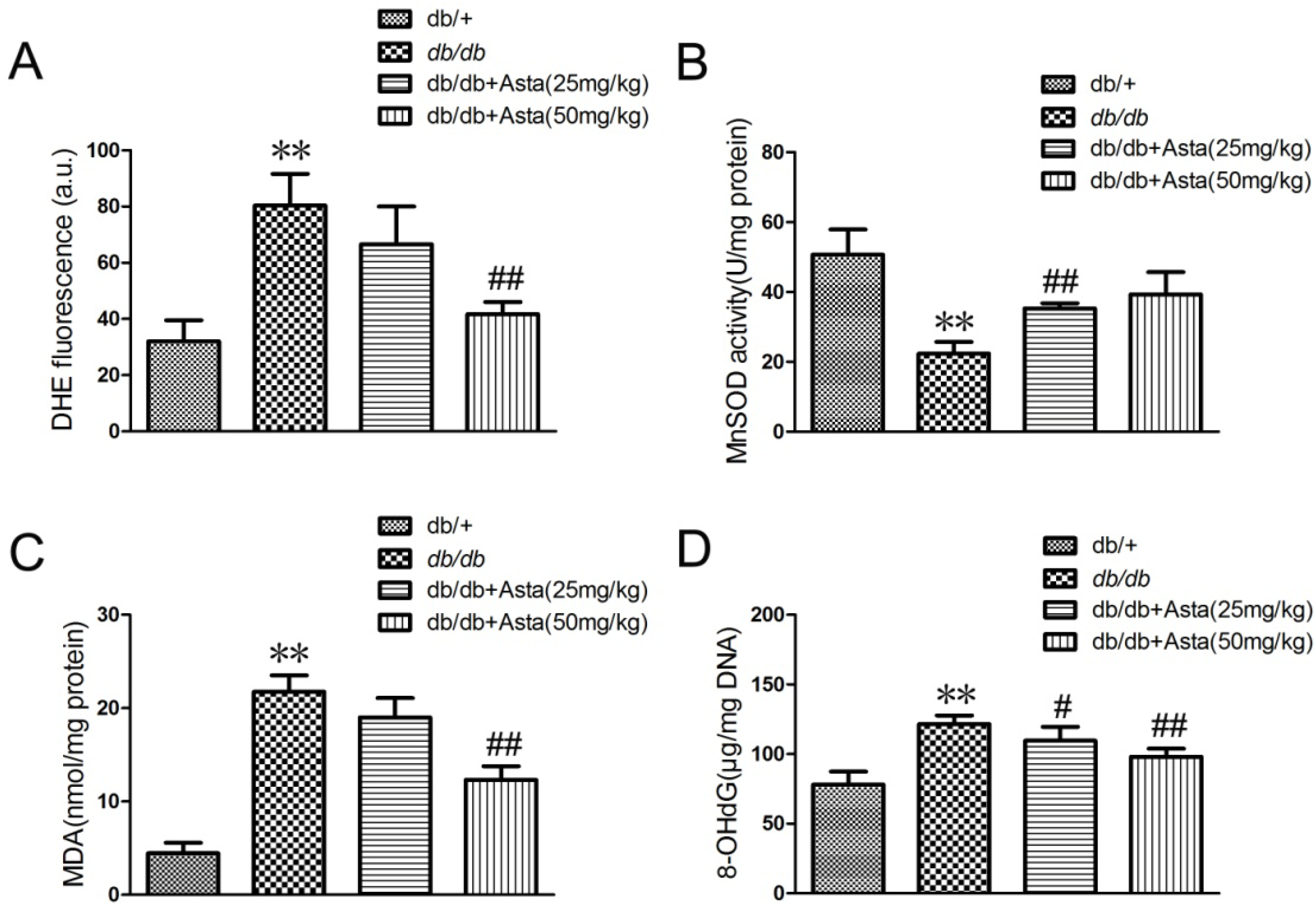
2.1.4. Astaxanthin Attenuates H2O2-Induced Apoptosis in the Transformed Rat Retinal Ganglion Cell Line RGC-5
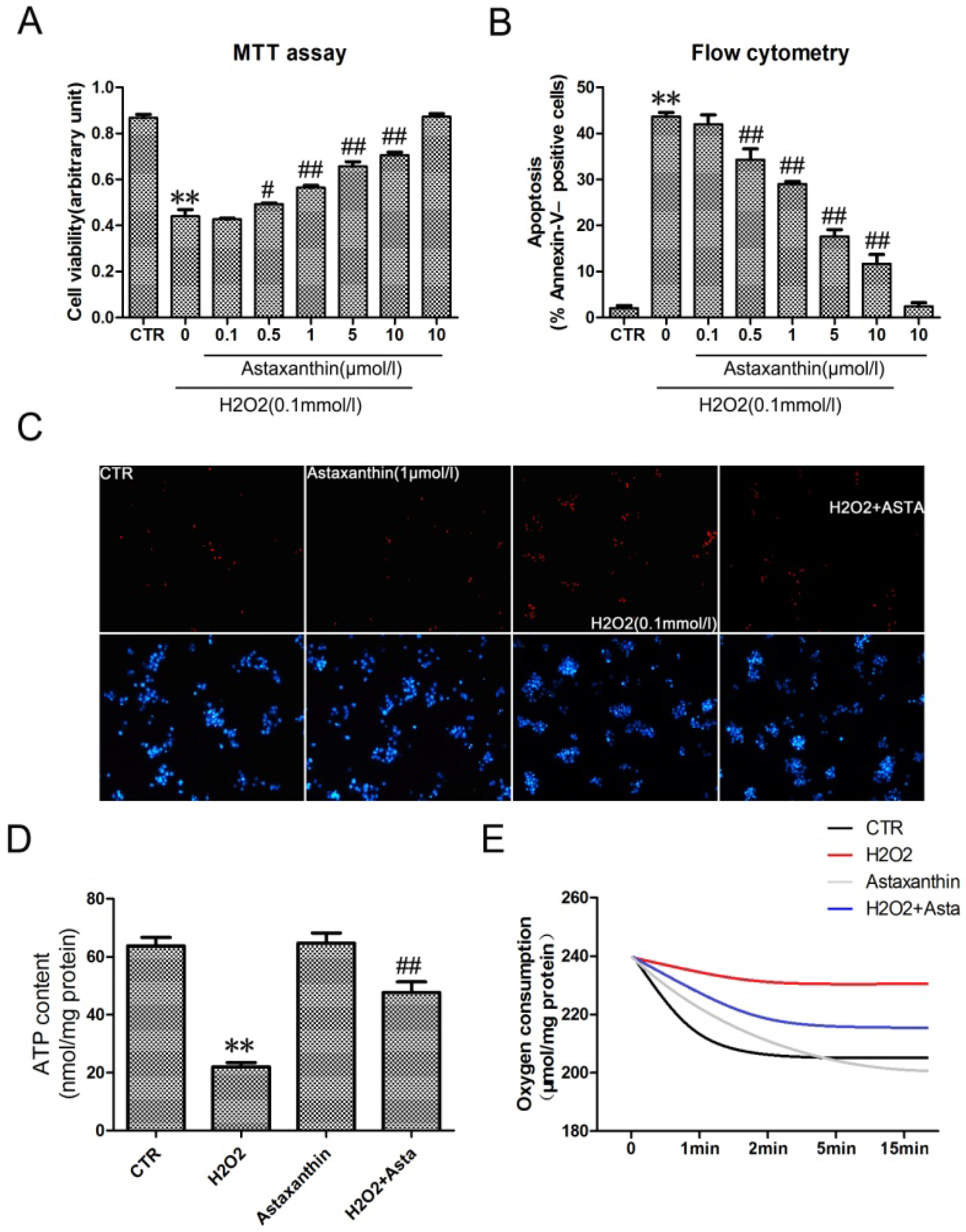
2.2. Discussion
3. Material and Methods
3.1. Reagents
3.2. Animal Treatments
3.3. Electroretinogram Recordings
3.4. Electroretinogram Analysis
3.5. Blood Glucose Test
3.6. Blood Lipid Assay
3.7. Western Blot
3.8. Tunel Staining
3.9. Detection of the Level of Superoxide Anion (O2•−), MDA, 8-OHdG and MnSOD
3.10. Cell Culture
3.11. MTT Assay
3.12. Flow Cytometry
3.13. Hoechst/PI Staining
3.14. ATP Assay
3.15. Oxygen Consumption
3.16. Statistical Analysis
4. Conclusions
Acknowledgements
Conflict of Interests
References
- Antonetti, D.A.; Barber, A.J.; Bronson, S.K.; Freeman, W.M.; Gardner, T.W.; Jefferson, L.S.; Kester, M.; Kimball, S.R.; Krady, J.K.; LaNoue, K.F.; et al. Diabetic retinopathy: Seeing beyond glucose-induced microvascular disease. Diabetes 2006, 55, 2401–2411. [Google Scholar] [CrossRef]
- Ghirlanda, G.; Di Leo, M.A.; Caputo, S.; Cercone, S.; Greco, A.V. From functional to microvascular abnormalities in early diabetic retinopathy. Diabetes Metab. Rev. 1997, 13, 15–35. [Google Scholar] [CrossRef]
- Kowluru, R.A.; Chan, P.S. Oxidative stress and diabetic retinopathy. Exp. Diabetes Res. 2007, 2007, 43603. [Google Scholar]
- Gurler, B.; Vural, H.; Yilmaz, N.; Oguz, H.; Satici, A.; Aksoy, N. The role of oxidative stress in diabetic retinopathy. Eye (Lond.) 2000, 14, 730–735. [Google Scholar] [CrossRef]
- Madsen-Bouterse, S.A.; Kowluru, R.A. Oxidative stress and diabetic retinopathy: Pathophysiological mechanisms and treatment perspectives. Rev. Endocr. Metab. Disord. 2008, 9, 315–327. [Google Scholar] [CrossRef]
- Cui, Y.; Xu, X.; Bi, H.; Zhu, Q.; Wu, J.; Xia, X.; Qiushi, R.; Ho, P.C. Expression modification of uncoupling proteins and MnSOD in retinal endothelial cells and pericytes induced by high glucose: The role of reactive oxygen species in diabetic retinopathy. Exp. Eye Res. 2006, 83, 807–816. [Google Scholar] [CrossRef]
- El-Remessy, A.B.; Bartoli, M.; Platt, D.H.; Fulton, D.; Caldwell, R.B. Oxidative stress inactivates VEGF survival signaling in retinal endothelial cells via PI 3-kinase tyrosine nitration. J. Cell Sci. 2005, 118, 243–252. [Google Scholar] [CrossRef]
- Gil, J.; Almeida, S.; Oliveira, C.R.; Rego, A.C. Cytosolic and mitochondrial ROS in staurosporine-induced retinal cell apoptosis. Free Radic. Biol. Med. 2003, 35, 1500–1514. [Google Scholar] [CrossRef]
- Carmody, R.J.; Cotter, T.G. Oxidative stress induces caspase-independent retinal apoptosis in vitro. Cell Death Differ. 2000, 7, 282–291. [Google Scholar] [CrossRef]
- Sanvicens, N.; Cotter, T.G. Ceramide is the key mediator of oxidative stress-induced apoptosis in retinal photoreceptor cells. J. Neurochem. 2006, 98, 1432–1444. [Google Scholar] [CrossRef]
- Naguib, Y.M. Antioxidant activities of astaxanthin and related carotenoids. J. Agric. Food Chem. 2000, 48, 1150–1154. [Google Scholar] [CrossRef]
- Leite, M.F.; Lima, A.M.; Massuyama, M.M.; Otton, R. Astaxanthin restores the enzymatic antioxidant profile in salivary gland of alloxan-induced diabetic rats. Arch. Oral Biol. 2010, 55, 479–485. [Google Scholar] [CrossRef]
- Nakajima, Y.; Inokuchi, Y.; Shimazawa, M.; Otsubo, K.; Ishibashi, T.; Hara, H. Astaxanthin, a dietary carotenoid, protects retinal cells against oxidative stress in vitro and in mice in vivo. J. Pharm. Pharmacol. 2008, 60, 1365–1374. [Google Scholar] [CrossRef]
- Kim, Y.J.; Kim, Y.A.; Yokozawa, T. Protection against oxidative stress, inflammation, and apoptosis of high-glucose-exposed proximal tubular epithelial cells by astaxanthin. J. Agric. Food Chem. 2009, 57, 8793–8797. [Google Scholar] [CrossRef]
- Moeller, S.M.; Parekh, N.; Tinker, L.; Ritenbaugh, C.; Blodi, B.; Wallace, R.B.; Mares, J.A. Associations between intermediate age-related macular degeneration and lutein and zeaxanthin in the Carotenoids in Age-related Eye Disease Study (CAREDS): Ancillary study of the Women’s Health Initiative. Arch. Ophthalmol. 2006, 124, 1151–1162. [Google Scholar] [CrossRef]
- Krinsky, N.I.; Johnson, E.J. Carotenoid actions and their relation to health and disease. Mol. Aspects Med. 2005, 26, 459–516. [Google Scholar] [CrossRef]
- Winklhofer-Roob, B.M.; Rock, E.; Ribalta, J.; Shmerling, D.H.; Roob, J.M. Effects of vitamin E and carotenoid status on oxidative stress in health and disease. Evidence obtained from human intervention studies. Mol. Aspects Med. 2003, 24, 391–402. [Google Scholar] [CrossRef]
- Li, G.Y.; Fan, B.; Su, G.F. Acute energy reduction induces caspase-dependent apoptosis and activates p53 in retinal ganglion cells (RGC-5). Exp. Eye Res. 2009, 89, 581–589. [Google Scholar] [CrossRef]
- Ji, Y.; Lu, G.; Chen, G.; Huang, B.; Zhang, X.; Shen, K.; Wu, S. Microcystin-LR induces apoptosis via NF-κB/iNOS pathway in INS-1 cells. Int. J. Mol. Sci. 2011, 12, 4722–4734. [Google Scholar] [CrossRef]
- Kowluru, V.; Kowluru, R.A. Increased oxidative stress in diabetes regulates activation of a small molecular weight G-protein, H-Ras, in the retina. Mol. Vis. 2007, 13, 602–610. [Google Scholar]
- Van Reyk, D.M.; Gillies, M.C.; Davies, M.J. The retina: Oxidative stress and diabetes. Redox Rep. 2003, 8, 187–192. [Google Scholar] [CrossRef]
- Burnaugh, L.; Sabeur, K.; Ball, B.A. Generation of superoxide anion by equine spermatozoa as detected by dihydroethidium. Theriogenology 2007, 67, 580–589. [Google Scholar] [CrossRef]
- Pan, H.Z.; Zhang, H.; Chang, D.; Li, H.; Sui, H. The change of oxidative stress products in diabetes mellitus and diabetic retinopathy. Br. J. Ophthalmol. 2008, 92, 548–551. [Google Scholar] [CrossRef]
- Miranda, M.; Muriach, M.; Roma, J.; Bosch-Morell, F.; Genoves, J.M.; Barcia, J.; Araiz, J.; Diaz-Llospis, M.; Romero, F.J. Oxidative stress in a model of experimental diabetic retinopathy: The utility of peroxinytrite scavengers. Arch. Soc. Esp. Oftalmol. 2006, 81, 27–32. [Google Scholar]
- Choi, H.D.; Kim, J.H.; Chang, M.J.; Kyu-Youn, Y.; Shin, W.G. Effects of astaxanthin on oxidative stress in overweight and obese adults. Phytother. Res. 2011, 25, 1813–1818. [Google Scholar] [CrossRef]
- Yuan, J.P.; Peng, J.; Yin, K.; Wang, J.H. Potential health-promoting effects of astaxanthin: A high-value carotenoid mostly from microalgae. Mol. Nutr. Food Res. 2011, 55, 150–165. [Google Scholar] [CrossRef]
- Liu, X.; Osawa, T. Astaxanthin protects neuronal cells against oxidative damage and is a potent candidate for brain food. Forum Nutr. 2009, 61, 129–135. [Google Scholar] [CrossRef]
- Uchiyama, K.; Naito, Y.; Hasegawa, G.; Nakamura, N.; Takahashi, J.; Yoshikawa, T. Astaxanthin protects β-cells against glucose toxicity in diabetic db/db mice. Redox Rep. 2002, 7, 290–293. [Google Scholar] [CrossRef]
- Naito, Y.; Uchiyama, K.; Aoi, W.; Hasegawa, G.; Nakamura, N.; Yoshida, N.; Maoka, T.; Takahashi, J.; Yoshikawa, T. Prevention of diabetic nephropathy by treatment with astaxanthin in diabetic db/db mice. Biofactors 2004, 20, 49–59. [Google Scholar] [CrossRef]
- Poitout, V.; Briaud, I.; Kelpe, C.; Hagman, D. Gluco-lipotoxicity of the pancreatic β cell. Ann. Endocrinol. (Paris) 2004, 65, 37–41. [Google Scholar] [CrossRef]
- Drews, G.; Krippeit-Drews, P.; Dufer, M. Oxidative stress and β-cell dysfunction. Pflugers Arch. 2010, 460, 703–718. [Google Scholar] [CrossRef]
- Yoon, I.H.; Shiroyama, N.; Miyake, Y.; Awaya, S. Oscillatory potentials of local macular ERG in diabetic retinopathy. Korean J. Ophthalmol. 1990, 4, 40–45. [Google Scholar]
- Iizuka, Y.; Hong, S.; Kim, C.Y.; Kim, S.K.; Seong, G.J. Agmatine pretreatment protects retinal ganglion cells (RGC-5 cell line) from oxidative stress in vitro. Biocell 2008, 32, 245–250. [Google Scholar]
- Maher, P.; Hanneken, A. Flavonoids protect retinal ganglion cells from oxidative stress-induced death. Invest. Ophthalmol. Vis. Sci. 2005, 46, 4796–4803. [Google Scholar] [CrossRef]
- Byeon, S.H.; Lee, S.C.; Choi, S.H.; Lee, H.K.; Lee, J.H.; Chu, Y.K.; Kwon, O.W. Vascular endothelial growth factor as an autocrine survival factor for retinal pigment epithelial cells under oxidative stress via the VEGF-R2/PI3K/Akt. Invest. Ophthalmol. Vis. Sci. 2010, 51, 1190–1197. [Google Scholar] [CrossRef]
- Tian, X.Y.; Wong, W.T.; Xu, A.; Chen, Z.Y.; Lu, Y.; Liu, L.M.; Lee, V.W.; Lau, C.W.; Yao, X.; Huang, Y. Rosuvastatin improves endothelial function in db/db mice: Role of angiotensin II type 1 receptors and oxidative stress. Br. J. Pharmacol. 2011, 164, 598–606. [Google Scholar]
- Kitada, M.; Kume, S.; Imaizumi, N.; Koya, D. Resveratrol improves oxidative stress and protects against diabetic nephropathy through normalization of Mn-SOD dysfunction in AMPK/SIRT1-independent pathway. Diabetes 2011, 60, 634–643. [Google Scholar] [CrossRef]
- Xiao, C.; He, M.; Nan, Y.; Zhang, D.; Chen, B.; Guan, Y.; Pu, M. Physiological effects of superoxide dismutase on altered visual function of retinal ganglion cells in db/db mice. PLoS One 2012, 7, e30343. [Google Scholar]
- Martin, B.T.; List, E.O.; Kopchick, J.J.; Sauve, Y.; Harvey, S. Selective inner retinal dysfunction in growth hormone transgenic mice. Growth Horm. IGF Res. 2011, 21, 219–227. [Google Scholar] [CrossRef]
- Rousseau, S.; Lachapelle, P. The electroretinogram recorded at the onset of dark-adaptation: Understanding the origin of the scotopic oscillatory potentials. Doc. Ophthalmol. 1999, 99, 135–150. [Google Scholar] [CrossRef]
- Yu, M.; Peachey, N.S. Attenuation of oscillatory potentials in nob2 mice. Doc. Ophthalmol. 2007, 115, 173–186. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, L.; Li, Y.; Chen, H.; Sun, Z. Nifedipine protects INS-1 β-cell from high glucose-induced ER stress and apoptosis. Int. J. Mol. Sci. 2011, 12, 7569–7580. [Google Scholar] [CrossRef]
- He, S.Q.; Zhang, Y.H.; Venugopal, S.K.; Dicus, C.W.; Perez, R.V.; Ramsamooj, R.; Nantz, M.H.; Zern, M.A.; Wu, J. Delivery of antioxidative enzyme genes protects against ischemia/reperfusion-induced liver injury in mice. Liver Transpl. 2006, 12, 1869–1879. [Google Scholar] [CrossRef]
- Samples Availability: Available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dong, L.-Y.; Jin, J.; Lu, G.; Kang, X.-L. Astaxanthin Attenuates the Apoptosis of Retinal Ganglion Cells in db/db Mice by Inhibition of Oxidative Stress. Mar. Drugs 2013, 11, 960-974. https://doi.org/10.3390/md11030960
Dong L-Y, Jin J, Lu G, Kang X-L. Astaxanthin Attenuates the Apoptosis of Retinal Ganglion Cells in db/db Mice by Inhibition of Oxidative Stress. Marine Drugs. 2013; 11(3):960-974. https://doi.org/10.3390/md11030960
Chicago/Turabian StyleDong, Ling-Yan, Jie Jin, Gao Lu, and Xiao-Li Kang. 2013. "Astaxanthin Attenuates the Apoptosis of Retinal Ganglion Cells in db/db Mice by Inhibition of Oxidative Stress" Marine Drugs 11, no. 3: 960-974. https://doi.org/10.3390/md11030960



