In Vitro Acylation of Okadaic Acid in the Presence of Various Bivalves’ Extracts
Abstract
:1. Introduction

2. Results and Discussion
2.1. Okadaic Acid Binding Proteins in Shellfish

2.2. In Vitro Transformation of OA into 7-O-Palmitoyl OA (DTX3) in M. yessoensis
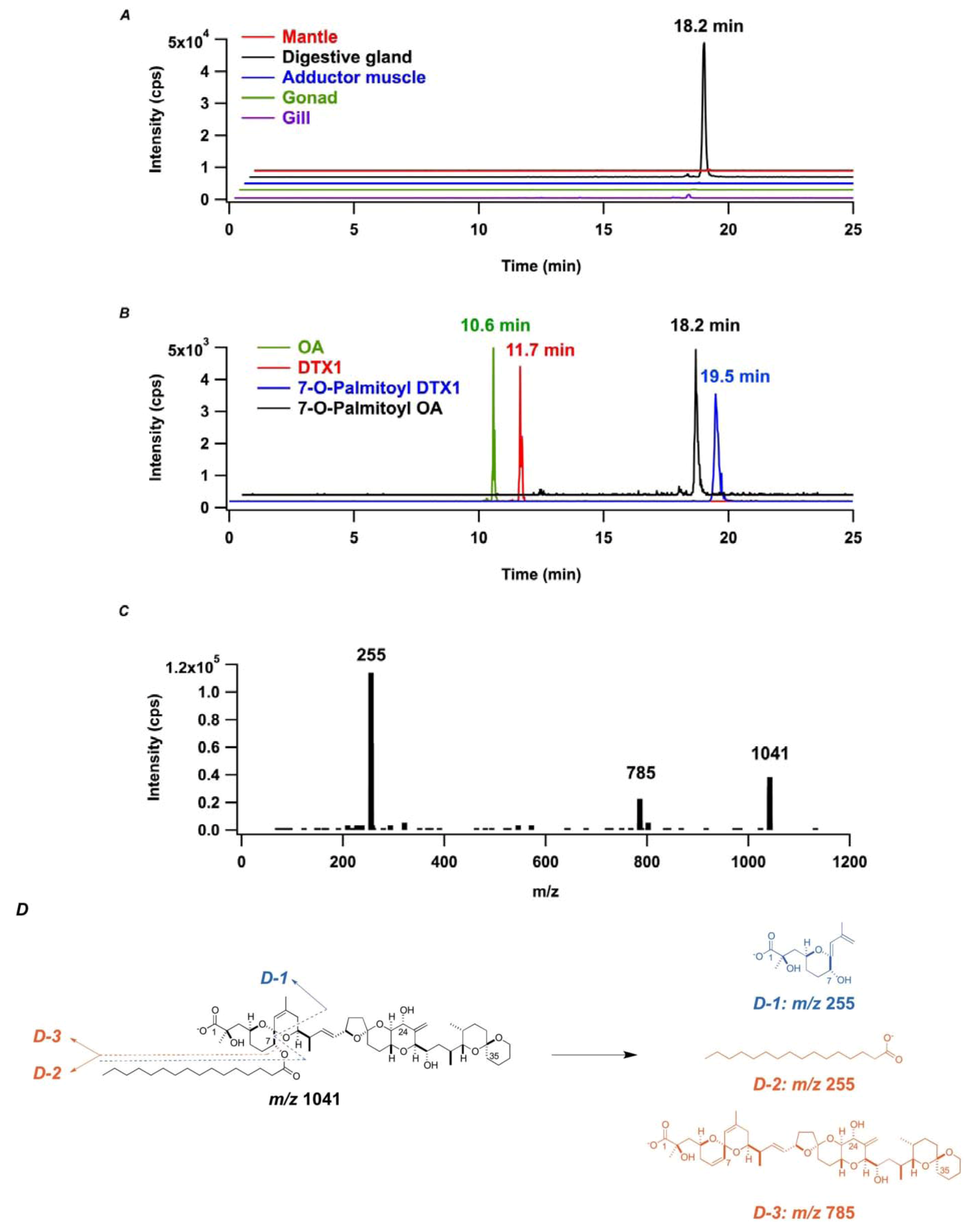

2.3. In Vitro Transformation of OA into 7-O-Palmitoyl OA (DTX3) in Other Bivalve Species
2.4. Characterization of OA-Transforming Enzyme with an Attempt of the Purification
3. Experimental Section
3.1. Materials
3.2. Preparation of the Microsomal and Mitochondrial Fractions from Bivalves
3.3. In Vitro Transformation Reaction
3.4. Binding Assay
4. Conclusions
Acknowledgments
References
- Yasumoto, T.; Oshima, Y.; Yamaguchi, M. Occurrence of a new type of shellfish poisoning in Tohoku District. Bull. Jpn. Soc. Sci. Fish. 1978, 44, 1249–1255. [Google Scholar]
- Yasumoto, T.; Oshima, Y.; Sugawara, W.; Fukuyo, Y.; Oguri, H.; Igarashi, T.; Fujita, N. Identification of Dinophysis fortii as the causative organism of diarrhetic shellfish poisoning. Bull. Jpn. Soc. Sci. Fish. 1980, 46, 1405–1411. [Google Scholar] [CrossRef]
- Murata, M.; Shimatani, M.; Sugitani, H.; Oshima, Y.; Yasumoto, T. Isolation and structural elucidation of the causative toxin of the diarrhetic shellfish poisoning. Bull. Jpn. Soc. Sci. Fish. 1982, 48, 549–552. [Google Scholar] [CrossRef]
- Kumagai, M.; Yanagi, T.; Murata, M.; Yasumoto, T.; Kat, M.; Lassus, P.; Rodriguezvazquez, J.A. Okadaic acid as the causative toxin of diarrhetic shellfish poisoning in Europe. Agric. Biol. Chem. 1986, 50, 2853–2857. [Google Scholar] [CrossRef]
- Tachibana, K.; Scheuer, P.J.; Tsukitani, Y.; Kikuchi, H.; Vanengen, D.; Clardy, J.; Gopichand, Y.; Schmitz, F.J. Okadaic acid, a cytotoxic polyether from marine sponges of the genus Halichondria. J. Am. Chem. Soc. 1981, 103, 2469–2471. [Google Scholar] [CrossRef]
- Hu, T.M.; Doyle, J.; Jackson, D.; Marr, J.; Nixon, E.; Pleasance, S.; Quilliam, M.A.; Walter, J.A.; Wright, J.L.C. Isolation of a new diarrhetic shellfish poison from Irish mussels. J. Chem. Soc. Chem. Commun. 1992, 39–41. [Google Scholar] [CrossRef]
- Yasumoto, T.; Murata, M.; Oshima, Y.; Sano, M.; Matsumoto, G.K.; Clardy, J. Diarrhetic shellfish toxins. Tetrahedron 1985, 41, 1019–1025. [Google Scholar] [CrossRef]
- Suzuki, T.; Mitsuya, T. Comparison of dinophysistoxin-1 and esterified dinophysistoxin-1 (dinophysistoxin-3) contents in the scallop Patinopecten yessoensis and the mussel Mytilus galloprovincialis. Toxicon 2001, 39, 905–908. [Google Scholar] [CrossRef]
- Suzuki, T.; Ota, H.; Yamasaki, M. Direct evidence of transformation of dinophysistoxin-1 to 7-O-Acyl-dinophysistoxin-1 (dinophysistoxin-3) in the scallop Patinopecten yessoensis. Toxicon 1999, 37, 187–198. [Google Scholar] [CrossRef]
- Rossignoli, A.E.; Fernandez, D.; Regueiro, J.; Marino, C.; Blanco, J. Esterification of okadaic acid in the mussel Mytilus galloprovincialis. Toxicon 2011, 57, 712–720. [Google Scholar] [CrossRef]
- Vale, P. Profiles of fatty acids and 7-O-Acyl okadaic acid esters in bivalves: Can bacteria be involved in acyl esterification of okadaic acid? Comp. Biochem. Physiol. C 2010, 151, 18–24. [Google Scholar]
- Schantz, E.J.; Ghazarossian, V.E.; Schnoes, H.K.; Strong, F.M.; Springer, J.P.; Pezzanite, J.O.; Clardy, J. Structure of saxitoxin. J. Am. Chem. Soc. 1975, 97, 1238–1239. [Google Scholar] [CrossRef]
- McFarren, E.F.; Tanabe, H.; Silva, F.J.; Wilson, W.B.; Campbell, J.E.; Lewis, K.H. The occurrence of a ciguatera-like poison in oysters, clams and Gymnodinium breve Cultures. Toxicon 1965, 3, 111–123. [Google Scholar] [CrossRef]
- Wright, J.L.C.; Boyd, R.K.; Defreitas, A.S.W.; Falk, M.; Foxall, R.A.; Jamieson, W.D.; Laycock, M.V.; McCulloch, A.W.; McInnes, A.G.; Odense, P.; et al. Identification of domoic acid, a neuroexcitatory amino acid, in toxic mussels from Eastern Prince Edward Island. Can. J. Chem. 1989, 67, 481–490. [Google Scholar] [CrossRef]
- Toyofuku, H. Joint FAO/WHO/IOC activities to provide scientific advice on marine biotoxins (research report). Mar. Pollut. Bull. 2006, 52, 1735–1745. [Google Scholar] [CrossRef]
- Fast, M.D.; Cembella, A.D.; Ross, N.W. In vitro transformation of paralytic shellfish toxins in the clams Mya arenaria and Protothaca staminea. Harmful Algae 2006, 5, 79–90. [Google Scholar] [CrossRef]
- Hu, T.M.; Marr, J.; Defreitas, A.S.W.; Quilliam, M.A.; Walter, J.A.; Wright, J.L.C.; Pleasance, S. New diol esters isolated from cultures of the dinoflagellates Prorocentrum lima and Prorocentrum concavum. J. Nat. Prod. 1992, 55, 1631–1637. [Google Scholar] [CrossRef]
- MacKenzie, L.A.; Selwood, A.I.; Marshall, C. Isolation and characterization of an enzyme from the Greenshell™ mussel Perna canaliculus that hydrolyses pectenotoxins and esters of okadaic acid. Toxicon 2012, 60, 406–419. [Google Scholar] [CrossRef]
- Hu, T.M.; Curtis, J.M.; Walter, J.A.; Wright, J.L.C. Identification of DTX4, a new water-soluble phosphatase inhibitor from the toxic dinoflagellate Prorocentrum lima. J. Chem. Soc. Chem. Commun. 1995, 597–599. [Google Scholar] [CrossRef]
- Hu, T.M.; Curtis, J.M.; Walter, J.A.; McLachlan, J.L.; Wright, J.L.C. Two new water-soluble DSP toxin derivatives from the dinoflagellate Prorocentrum maculosum: Possible storage and excretion products. Tetrahedron Lett. 1995, 36, 9273–9276. [Google Scholar] [CrossRef]
- Bialojan, C.; Takai, A. Inhibitory effect of a marine-sponge toxin, okadaic acid, on protein phosphatases—Specificity and kinetics. Biochem. J. 1988, 256, 283–290. [Google Scholar]
- Takai, A.; Murata, M.; Torigoe, K.; Isobe, M.; Mieskes, G.; Yasumoto, T. Inhibitory effect of okadaic acid derivatives on protein phosphatases. A study on structure-affinity relationship. Biochem. J. 1992, 284, 539–544. [Google Scholar]
- Yanagi, T.; Murata, M.; Torigoe, K.; Yasumoto, T. Biological-activities of semisynthetic analogs of dinophysistoxin-3, the major diarrhetic shellfish toxin. Agric. Biol. Chem. 1989, 53, 525–529. [Google Scholar]
- Suganuma, M.; Fujiki, H.; Suguri, H.; Yoshizawa, S.; Hirota, M.; Nakayasu, M.; Ojika, M.; Wakamatsu, K.; Yamada, K.; Sugimura, T. Okadaic acid: An additional non-phorbol-12-tetradecanoate-13-acetate-type tumor promoter. Proc. Natl. Acad. Sci. USA 1988, 85, 1768–1771. [Google Scholar]
- Xing, Y.; Xu, Y.; Chen, Y.; Jeffrey, P.D.; Chao, Y.; Lin, Z.; Li, Z.; Strack, S.; Stock, J.B.; Shi, Y. Structure of protein phosphatase 2A core enzyme bound to tumor-inducing toxins. Cell 2006, 127, 341–353. [Google Scholar] [CrossRef]
- Sugiyama, N.; Konoki, K.; Tachibana, K. Isolation and characterization of okadaic acid binding proteins from the marine sponge Halichondria okadai. Biochemistry 2007, 46, 11410–11420. [Google Scholar] [CrossRef]
- Konoki, K.; Saito, K.; Matsuura, H.; Sugiyama, N.; Cho, Y.; Yotsu-Yamashita, M.; Tachibana, K. Binding of diarrheic shellfish poisoning toxins to okadaic acid binding proteins purified from the sponge Halichondria okadai. Bioorg. Med. Chem. 2010, 18, 7607–7610. [Google Scholar] [CrossRef]
- Marston, A. Natural products as a source of protein kinase activators and inhibitors. Curr. Top. Med. Chem. 2011, 11, 1333–1339. [Google Scholar] [CrossRef]
- McCluskey, A.; Sim, A.T.R.; Sakoff, J.A. Serine-threonine protein phosphatase inhibitors: Development of potential therapeutic strategies. J. Med. Chem. 2002, 45, 1151–1175. [Google Scholar] [CrossRef]
- Amzil, Z.; Pouchus, Y.F.; Leboterff, J.; Roussakis, C.; Verbist, J.F.; Marcailloulebaut, C.; Masselin, P. Short-time cytotoxicity of Mussel extracts—A new bioassay for okadaic acid detection. Toxicon 1992, 30, 1419–1425. [Google Scholar]
- Tubaro, A.; Florio, C.; Luxich, E.; Vertua, R.; DellaLoggia, R.; Yasumoto, T. Suitability of the MTT-based cytotoxicity assay to detect okadaic acid contamination of mussels. Toxicon 1996, 34, 965–974. [Google Scholar] [CrossRef]
- Fujiki, H.; Suganuma, M.; Suguri, H.; Yoshizawa, S.; Takagi, K.; Uda, N.; Wakamatsu, K.; Yamada, K.; Murata, M.; Yasumoto, T.; et al. Diarrhetic shellfish toxin, dinophysistoxin-1, is a potent tumor promoter on mouse skin. Jpn. J. Cancer Res. 1988, 79, 1089–1093. [Google Scholar] [CrossRef]
- Taylor, M.W.; Radax, R.; Steger, D.; Wagner, M. Sponge-associated microorganisms: Evolution, ecology and biotechnological potential. Microbiol. Mol. Biol. Rev. 2007, 71, 295–347. [Google Scholar] [CrossRef]
- Kennedy, J.; Codling, C.E.; Jones, B.V.; Dobson, A.D.W.; Marchesi, J.R. Diversity of microbes associated with the marine sponge, Haliclona simulans, isolated from Irish waters and identification of polyketide synthase genes from the sponge metagenome. Environ. Microbiol. 2008, 10, 1888–1902. [Google Scholar] [CrossRef]
- Piel, J. A Polyketide synthase-peptide synthetase gene cluster from an uncultured bacterial symbiont of Paederus beetles. Proc. Natl. Acad. Sci. USA 2002, 99, 14002–14007. [Google Scholar] [CrossRef]
- Rossignoli, A.E.; Blanco, J. Subcellular distribution of okadaic acid in the digestive gland of Mytilus galloprovincialis: First evidences of lipoprotein binding to okadaic acid. Toxicon 2010, 55, 221–226. [Google Scholar] [CrossRef]
- Janer, G.; Mesia-Vela, S.; Porte, C.; Kauffman, F.C. Esterification of vertebrate-type steroids in the eastern oyster (Crassostrea virginica). Steroids 2004, 69, 129–136. [Google Scholar] [CrossRef]
- Suzuki, T.; Kamiyama, T.; Okumura, Y.; Ishihara, K.; Matsushima, R.; Kaneniwa, M. Liquid-chromatographic hybrid triple-quadrupole linear-ion-trap MS/MS analysis of fatty-acid esters of dinophysistoxin-1 in bivalves and toxic dinoflagellates in Japan. Fish. Sci. 2009, 75, 1039–1048. [Google Scholar] [CrossRef]
- Ross, A.C. Retinol esterification by rat liver microsomes—Evidence for a fatty acyl coenzyme A—Retinol acyltransferase. J. Biol. Chem. 1982, 257, 2453–2459. [Google Scholar]
- Yost, R.W.; Harrison, E.H.; Ross, A.C. Esterification by rat-liver microsomes of retinol bound to cellular retinol-binding protein. J. Biol. Chem. 1988, 263, 18693–18701. [Google Scholar]
- Torgersen, T.; Lindegarth, S.; Ungfors, A.; Sandvik, M. Profiles and levels of fatty acid esters of okadaic acid group toxins and pectenotoxins during toxin depuration, Part I: Brown crab (Cancer pagurus). Toxicon 2008, 52, 407–417. [Google Scholar] [CrossRef]
- Torgersen, T.; Sandvik, M.; Lundve, B.; Lindegarth, S. Profiles and levels of fatty acid esters of okadaic acid group toxins and pectenotoxins during toxin depuration. Part II: Blue mussels (Mytilus edulis) and flat oyster (Ostrea edulis). Toxicon 2008, 52, 418–427. [Google Scholar] [CrossRef]
- Suzuki, T.; Mackenzie, L.; Stirling, D.; Adamson, J. Pectenotoxin-2 seco acid: A toxin converted from pectenotoxin-2 by the New Zealand Greenshell mussel, Perna canaliculus. Toxicon 2001, 39, 507–514. [Google Scholar] [CrossRef]
- Cauet, G.; Degryse, E.; Ledoux, C.; Spagnoli, R.; Achstetter, T. Pregnenolone esterification in Saccharomyces cerevisiae—A potential detoxification mechanism. Eur. J. Biochem. 1999, 261, 317–324. [Google Scholar] [CrossRef]
- McLaughlin, G.L.; Saz, H.J.; Debruyn, B.S. Purification and properties of an acyl CoA transferase from Ascaris suum muscle mitochondria. Comp. Biochem. Physiol. B 1986, 83, 523–527. [Google Scholar]
- Spector, A.A.; Mathur, S.N.; Kaduce, T.L. Role of acylcoenzyme A: Cholesterol O-acyltransferase in cholesterol metabolism. Prog. Lipid Res. 1979, 18, 31–53. [Google Scholar] [CrossRef]
- Suzuki, T.; Watanabe, R. Shellfish Toxin Monitoring System in Japan and Some Asian Countries. In New Trends in Marine and Freshwater Toxins, Food and Safety Concerns; Cabado, A.G., Vieites, J.M., Eds.; NOVA: New York, NY, USA, 2012; pp. 305–314. [Google Scholar]
- Samples Availability: Available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Konoki, K.; Onoda, T.; Watanabe, R.; Cho, Y.; Kaga, S.; Suzuki, T.; Yotsu-Yamashita, M. In Vitro Acylation of Okadaic Acid in the Presence of Various Bivalves’ Extracts. Mar. Drugs 2013, 11, 300-315. https://doi.org/10.3390/md11020300
Konoki K, Onoda T, Watanabe R, Cho Y, Kaga S, Suzuki T, Yotsu-Yamashita M. In Vitro Acylation of Okadaic Acid in the Presence of Various Bivalves’ Extracts. Marine Drugs. 2013; 11(2):300-315. https://doi.org/10.3390/md11020300
Chicago/Turabian StyleKonoki, Keiichi, Tatsuya Onoda, Ryuichi Watanabe, Yuko Cho, Shinnosuke Kaga, Toshiyuki Suzuki, and Mari Yotsu-Yamashita. 2013. "In Vitro Acylation of Okadaic Acid in the Presence of Various Bivalves’ Extracts" Marine Drugs 11, no. 2: 300-315. https://doi.org/10.3390/md11020300




