Meroterpenes from Endophytic Fungus A1 of Mangrove Plant Scyphiphora hydrophyllacea
Abstract
:1. Introduction
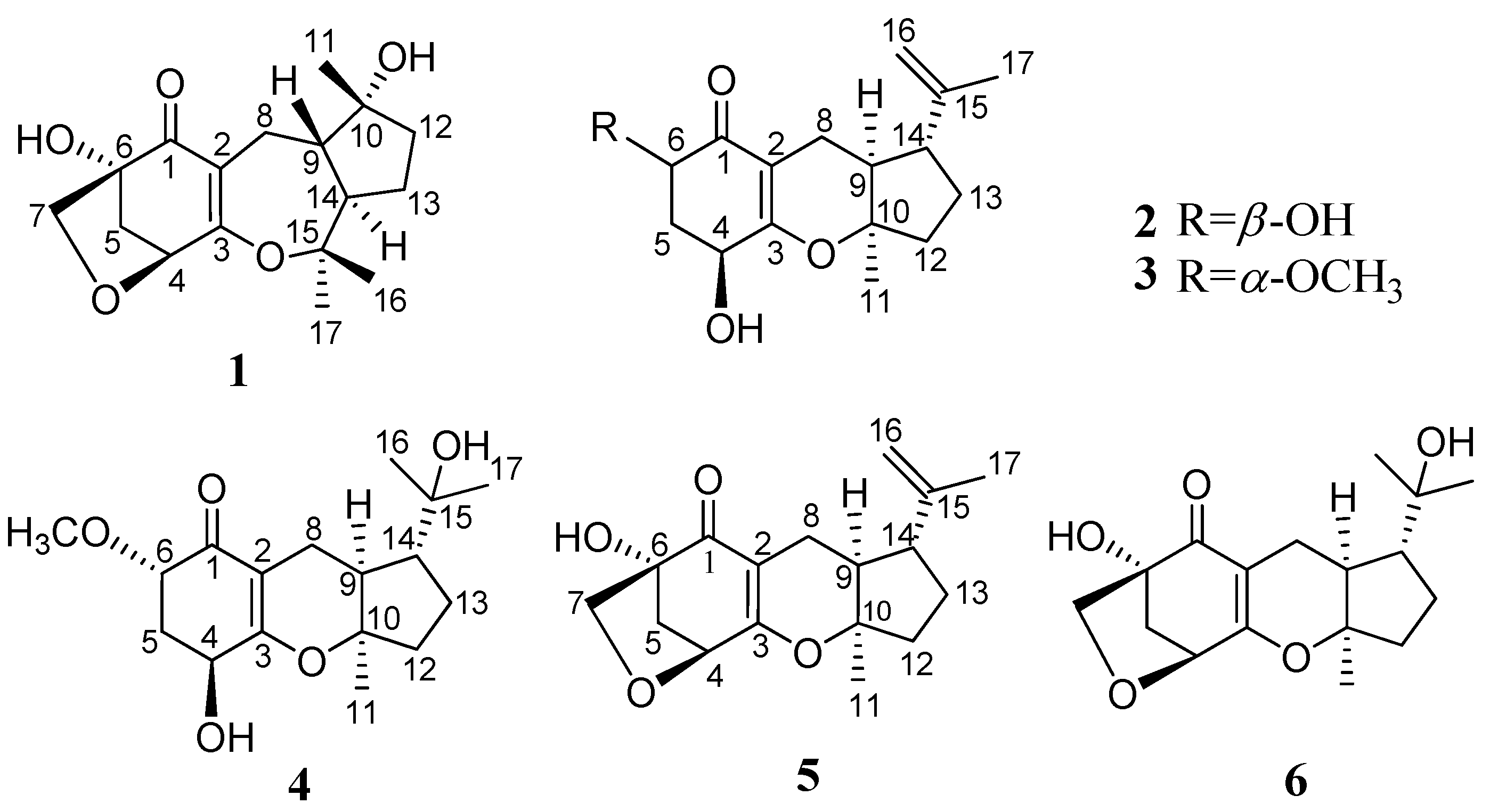
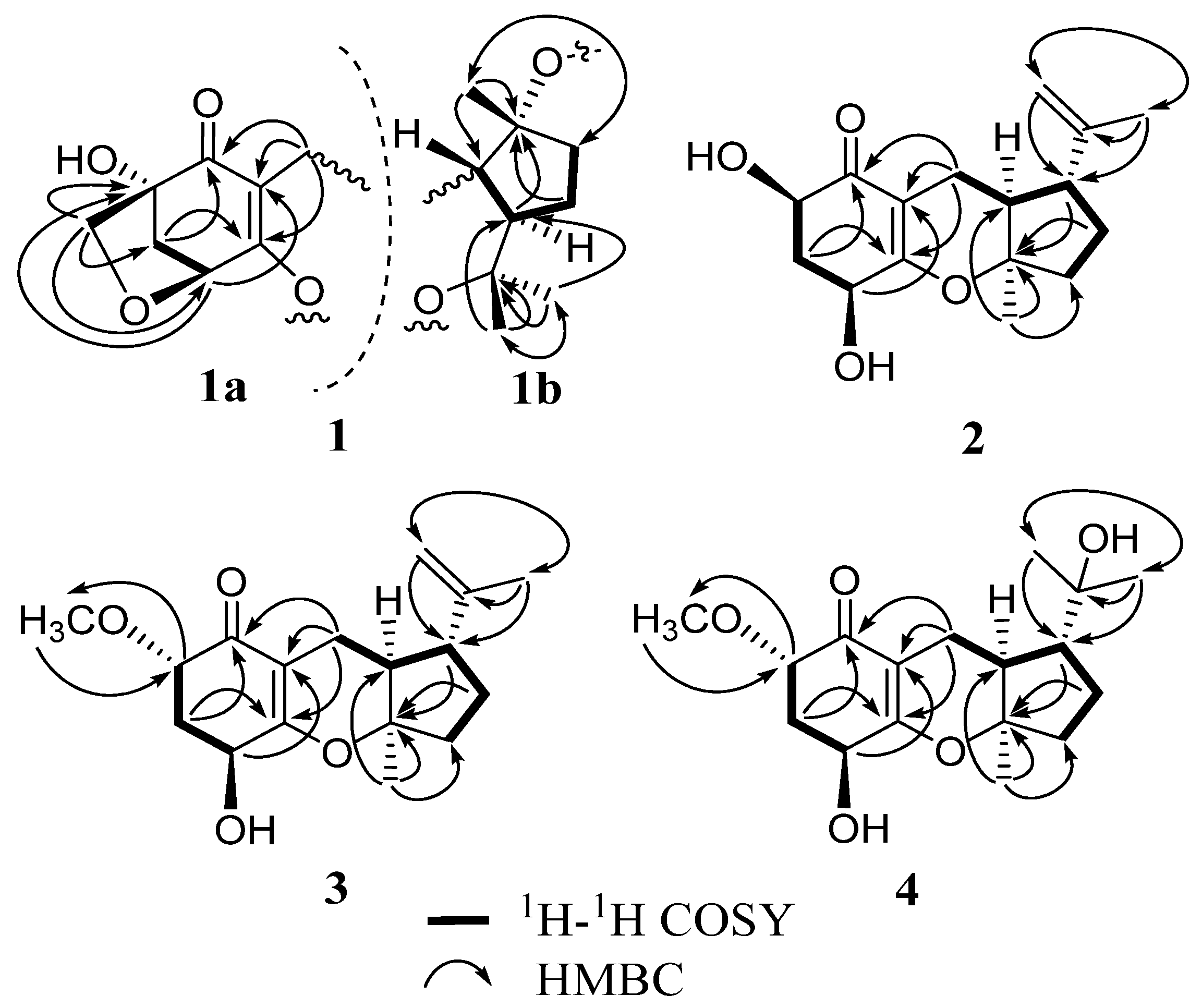
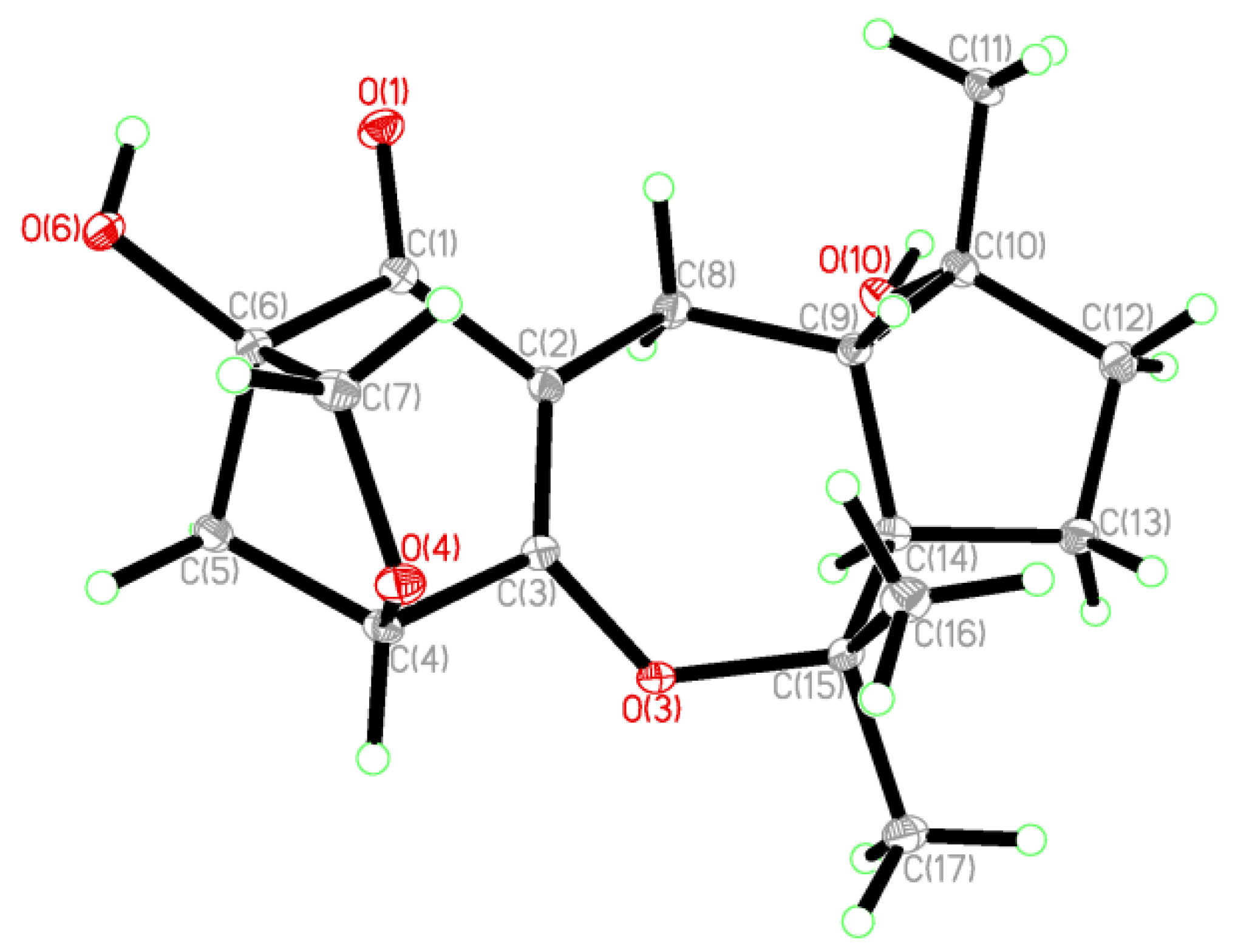
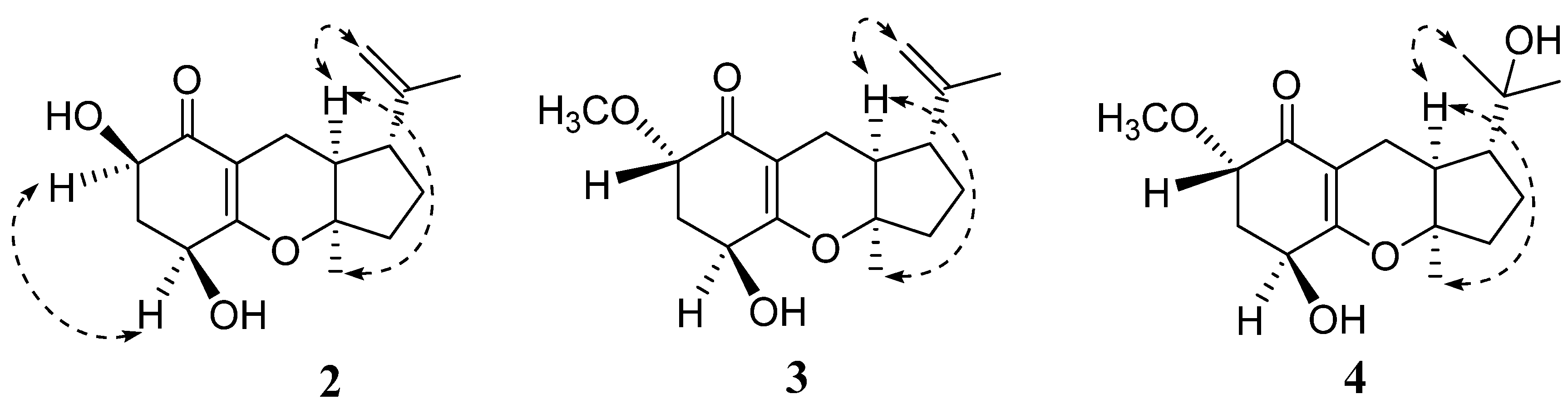
2. Results and Discussion
| position | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 1 | 200.2 s | 198.7 s | 194.9 s | 195.0 s |
| 2 | 114.7 s | 105.0 s | 105.8 s | 105.5 s |
| 3 | 173.9 s | 167.1 s | 167.7 s | 168.5 s |
| 4 | 81.3 d | 66.7 d | 65.7 d | 65.7 d |
| 5 | 42.5 t | 36.9 t | 34.5 t | 34.6 t |
| 6 | 81.8 s | 67.1 d | 79.1 d | 79.0 d |
| 7 | 69.3 t | |||
| 8 | 21.9 t | 16.2 t | 16.1 t | 18.7 t |
| 9 | 46.1 d | 43.3 d | 43.2 d | 41.2 d |
| 10 | 80.5 s | 88.2 s | 87.7 s | 88.9 s |
| 11 | 26.5 q | 23.5 q | 22.3 q | 22.1 q |
| 12 | 39.3 t | 37.7 t | 37.4 t | 38.3 t |
| 13 | 25.9 t | 27.0 t | 26.9 t | 24.5 t |
| 14 | 54.1 d | 48.7 d | 48.9 d | 51.1 d |
| 15 | 86.9 s | 145.4 s | 145.4 s | 72.9 s |
| 16 | 19.2 q | 111.3 t | 111.2 t | 28.6 q |
| 17 | 30.0 q | 19.2 q | 19.2 q | 27.5 q |
| OCH3 | 58.3 q | 58.3 q |
| position | 1 | 2 | 3 | 4 |
|---|---|---|---|---|
| 4 | 4.46, d (5.2) | 4.35, br t (2.8) | 4.24, br s | 4.26, br s |
| 5 | 2.31, m | 2.53, dq (13.4, 5.4) | 2.35, m | 2.38, dt (13.6, 4.2) |
| 1.91, m | 2.01, m | 2.23, m | 2.23, m | |
| 6 | 4.48, dd (11.9, 5.4) | 3.70, dd (6.8, 3.6) | 3.71, dd (7.0, 3.6) | |
| 7 | 3.80, d (8.0) | |||
| 3.45, d (8.0) | ||||
| 8 | 3.03, dd (15.0, 3.2) | 2.32, m | 2.33, d (17.3) | 2.62, d (17.6) |
| 1.92, m | 2.28, m | 2.15, m | 2.26, m | |
| 9 | 1.40, dd (12.0, 3.2) | 1.95, m | 1.94, m | 2.04, m |
| 11 | 1.35, s, 3H | 1.32, s, 3H | 1.33, s, 3H | 1.33, s, 3H |
| 12 | 1.76, m, 2H | 2.10, m; 1.81, m | 2.13, m; 1.79, m | 2.01, m; 1.59, m |
| 13 | 1.95, m; 1.22, m | 1.94, m; 1.57, m | 1.91, m; 1.53, m | 1.79, m; 1.58, m |
| 14 | 2.33, m | 2.27, m | 2.17, m | 1.58, m |
| 16 | 1.17, s, 3H | 4.73, m | 4.72, m | 1.20, s, 3H |
| 4.64, m | 4.62, m | |||
| 17 | 1.45, s, 3H | 1.66, s, 3H | 1.65, s, 3H | 1.18, s, 3H |
| OCH3 | 3.47, s, 3H | 3.47, s, 3H |

3. Experimental Section
3.1. General Experimental Procedures
3.2. Fungal Material and Fermentation
3.3. Extraction and Isolation
3.4. Antibacterial Activity
4. Conclusions
Acknowledgments
References
- Blunt, J.W.; Copp, B.R.; Hu, W.P.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natrual products. Nat. Prod. Rep. 2009, 26, 170–244. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Munro, M.H.G.; Northcote, P.T.; Prinsep, M.R. Marine natrual products. Nat. Prod. Rep. 2010, 27, 165–237. [Google Scholar] [CrossRef]
- Zeng, Y.B.; Mei, W.L.; Zhao, Y.X.; Zhuang, L.; Hong, K.; Dai, H.F. Two new epimeric pairs of iridoid from mangrove plant Scyphiphora hydrophyllacea. Chin. Chem. Lett. 2007, 18, 1509–1511. [Google Scholar] [CrossRef]
- Zeng, Y.B.; Mei, W.L.; Zhao, Y.X.; Dai, H.F. Two new noriridoids from Scyphiphora hydrophyllacea. Z. Naturforschung B 2008, 63, 108–110. [Google Scholar]
- Zeng, Y.B.; Mei, W.L.; Wang, H.; Li, X.N.; Dai, H.F. Scyphiphin D, a new iridoid glucoside dimer from Scyphiphora hydrophyllacea. J. Asian Nat. Prod. Res. 2010, 12, 1010–1014. [Google Scholar] [CrossRef]
- Feng, C.L.; Gong, M.F.; Zeng, Y.B.; Dai, H.F.; Mei, W.L. Scyphiphin C, a new iridoid from Scyphiphora hydrophyllacea. Molecules 2010, 15, 2473–2477. [Google Scholar] [CrossRef]
- Zeng, Y.B.; Dai, H.F.; Huang, J.L.; Mei, W.L. Separation, purification and structural elucidation of the bio-active components from fermentation broth of endophytic fungus C22 from Scyphiphora hydrophyllacea. Chin. J. Antibiot. 2011, 36, 276–279. [Google Scholar]
- Zeng, Y.B.; Wang, H.; Zuo, W.J.; Zheng, B.; Yang, T.; Dai, H.F.; Mei, W.L. A fatty acid glycoside from a marine-derived fungus isolated from mangrove plant Scyphiphora hydrophyllacea. Mar. Drugs 2012, 10, 598–603. [Google Scholar] [CrossRef]
- Zheng, B.; Zeng, Y.B.; Dai, H.F.; Zuo, W.J.; Guo, Z.K.; Yang, T.; Zhong, H.M.; Mei, W.L. Two new meroterpenes from endophytic fungus A1 of Scyphiphora hydrophyllacea. J. Asian Nat. Prod. Res. 2012, 14, 776–779. [Google Scholar] [CrossRef]
- Yuan, W.H.; Liu, M.; Jiang, N.; Guo, Z.K.; Ma, J.; Zhang, J.; Song, Y.C.; Tan, R.X. Guignardones A–C: Three meroterpenes from Guignardia mangiferae. Eur. J. Org. Chem. 2010, 33, 6348–6353. [Google Scholar]
- Xu, S.Y.; Bian, R.L.; Chen, X. Methods of Pharmacology Experiment; People’s Sanitation Press: Beijing, China, 2003; pp. 1651–1653. [Google Scholar]
- Molinar, E.; Rios, N.; Spadafora, C.; Arnold, A.E.; Coley, P.D.; Kursar, T.A.; Gerwick, W.H.; Cubilla-Rios, L. Coibanoles, a new class of meroterpenoids produced by Pycnoporus sanguineus. Tetrahedron Lett. 2012, 53, 919–922. [Google Scholar] [CrossRef]
- Denise, O.G.; Norberto, P.L.; Monica, T.P. Meroterpenes isolated from the endophytic fungus Guignardia mangiferae. Phytochem. Lett. 2012, 5, 519–523. [Google Scholar] [CrossRef]
- Samples Availability: Available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mei, W.-L.; Zheng, B.; Zhao, Y.-X.; Zhong, H.-M.; Chen, X.-L.W.; Zeng, Y.-B.; Dong, W.-H.; Huang, J.-L.; Proksch, P.; Dai, H.-F. Meroterpenes from Endophytic Fungus A1 of Mangrove Plant Scyphiphora hydrophyllacea. Mar. Drugs 2012, 10, 1993-2001. https://doi.org/10.3390/md10091993
Mei W-L, Zheng B, Zhao Y-X, Zhong H-M, Chen X-LW, Zeng Y-B, Dong W-H, Huang J-L, Proksch P, Dai H-F. Meroterpenes from Endophytic Fungus A1 of Mangrove Plant Scyphiphora hydrophyllacea. Marine Drugs. 2012; 10(9):1993-2001. https://doi.org/10.3390/md10091993
Chicago/Turabian StyleMei, Wen-Li, Bo Zheng, You-Xing Zhao, Hui-Ming Zhong, Xun-Li Wu Chen, Yan-Bo Zeng, Wen-Hua Dong, Jiu-Li Huang, Peter Proksch, and Hao-Fu Dai. 2012. "Meroterpenes from Endophytic Fungus A1 of Mangrove Plant Scyphiphora hydrophyllacea" Marine Drugs 10, no. 9: 1993-2001. https://doi.org/10.3390/md10091993








