A Comparative Analysis of Viral Richness and Viral Sharing in Cave-Roosting Bats
Abstract
1. Introduction
1.1. Caves as Critical Habitat for Bats Globally
1.2. Viral Diversity in Cave- and Non-Cave-Roosting Bats
1.3. Disease Ecology and Unique Traits of Cave-Roosting Bat Species
1.4. Human-Cave Bat Interactions
1.5. Study Objectives
2. Materials and Methods
2.1. Database of Viral Associations and Host Traits
2.2. Viral Richness and Sharing Analyses
3. Results
3.1. Database Summary and Descriptive Statistics
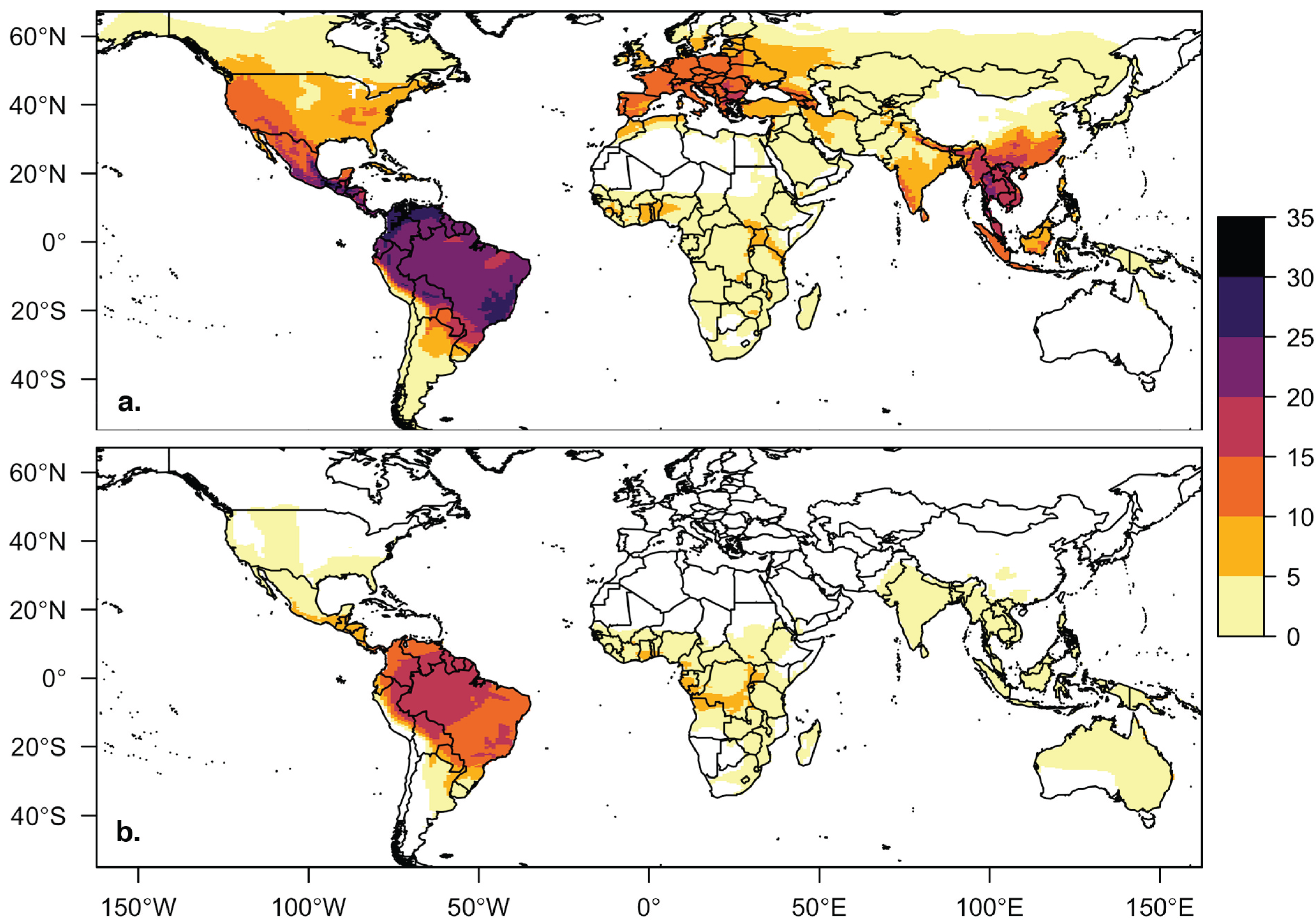
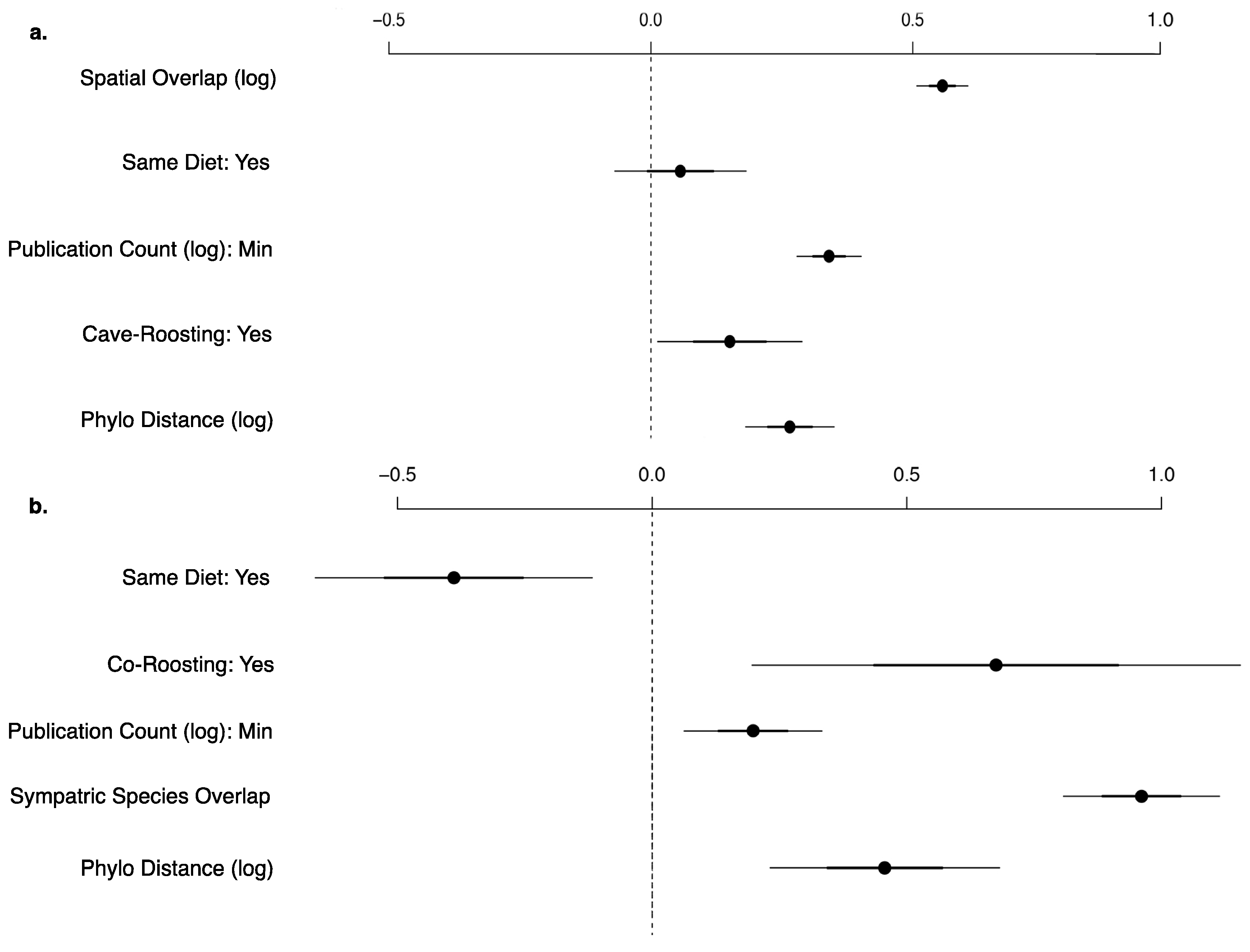
3.2. Viral Richness Analysis
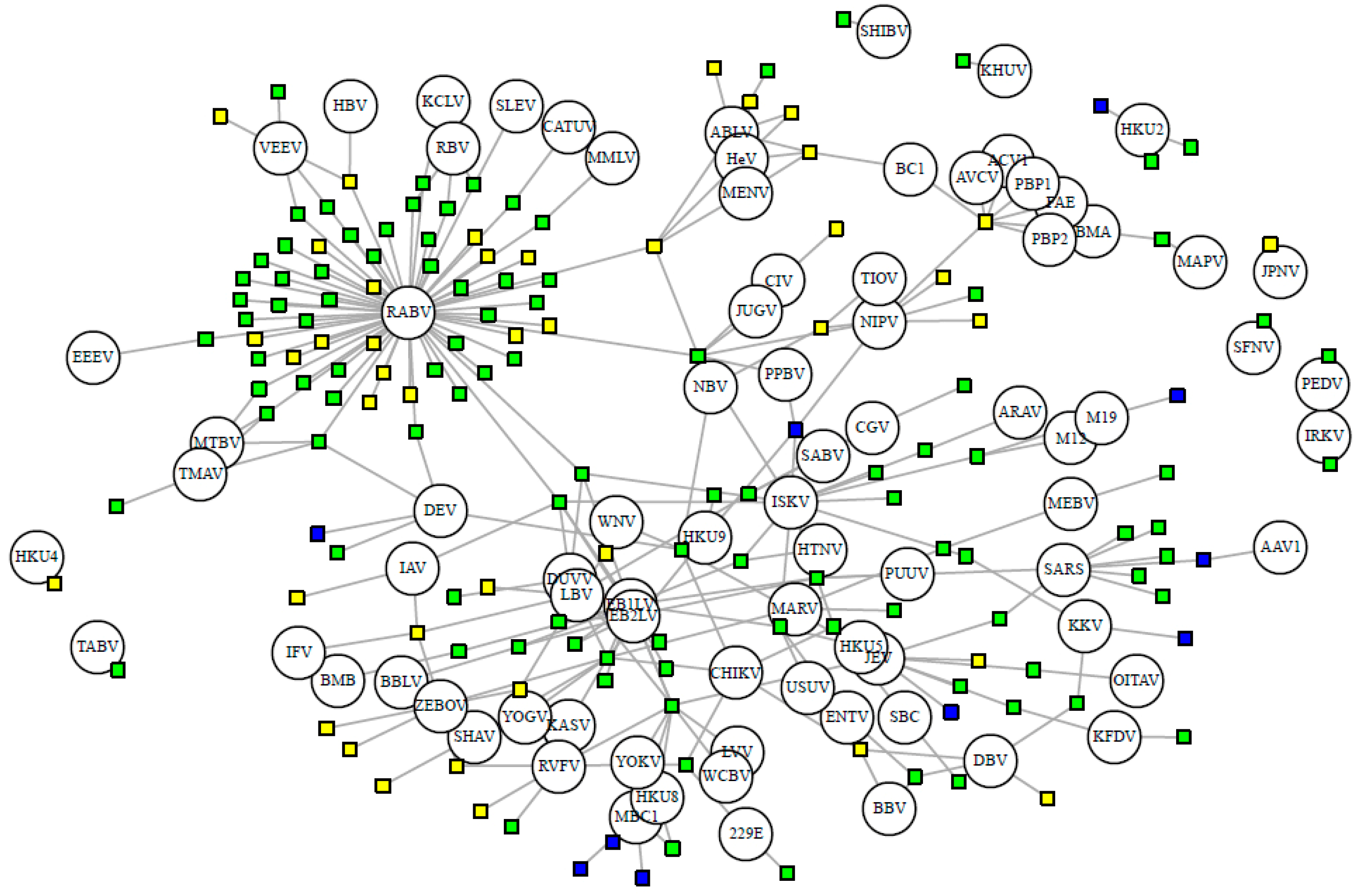
3.3. Viral Sharing Analysis
4. Discussion
4.1. Current State of Knowledge and Data Gaps
4.2. Viral Richness Within Bat Species
4.3. Viral Sharing Among Bat Species
4.4. Caveats and Future Directions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Culver, D.C.; Pipan, T. The Biology of Caves and Other Subterranean Habitats; Oxford University Press: New York, NY, USA, 2009; ISBN 978-0-1992-1993-3. [Google Scholar]
- Kunz, T.H. Roosting ecology of bats. In Ecology of Bats; Kunz, T.H., Ed.; Plenum Publishing Corporation: New York, NY, USA, 1982; pp. 1–55. ISBN 978-1-4613-3423-1. [Google Scholar]
- Arita, H.T. Conservation biology of the cave bats of Mexico. J. Mammal. 1993, 74, 693–702. [Google Scholar] [CrossRef]
- Hutson, A.M.; Mickleburgh, S.P. Microchiropteran Bats: Global Status Survey and Conservation Action Plan; IUCN: Gland, Switzerland, 2001; Volume 56, ISBN 2-8317-0595-9. [Google Scholar]
- Mickleburgh, S.P.; Hutson, A.M.; Racey, P.A. A review of the global conservation status of bats. Oryx 2002, 36, 18–34. [Google Scholar] [CrossRef]
- Furey, N.M.; Racey, P.A. Conservation ecology of cave bats. In Bats in the Anthropocene: Conservation of Bats in a Changing World; Voigt, C., Kingston, T., Eds.; Springer: Cham, Switzerland, 2016; pp. 463–500. ISBN 978-3-319-25220-9. [Google Scholar]
- Brunet, A.K.; Medellín, R.A. The species–area relationship in bat assemblages of tropical caves. J. Mammal. 2001, 82, 1114–1122. [Google Scholar] [CrossRef]
- Phelps, K.; Jose, R.; Labonite, M.; Kingston, T. Correlates of cave-roosting bat diversity as an effective tool to identify priority caves. Biol. Conserv. 2016, 201, 201–209. [Google Scholar] [CrossRef]
- Rodríguez-Durán, A. Bat assemblages in the West Indies: The role of caves. In Island Bats: Evolution, Ecology and Conservation; Fleming, T.H., Racey, P.A., Eds.; University of Chicago: Chicago, IL, USA, 2009; pp. 265–280. ISBN 9780226253305. [Google Scholar]
- Luo, J.; Jiang, T.; Lu, G.; Wang, L.; Wang, J.; Feng, J. Bat conservation in China: Should protection of subterranean habitats be a priority? Oryx 2013, 47, 526–531. [Google Scholar] [CrossRef]
- Olival, K.J.; Hosseini, P.R.; Zambrana-Torrelio, C.; Ross, N.; Bogich, T.L.; Daszak, P. Host and viral traits predict zoonotic spillover from mammals. Nature 2017, 546, 646–650. [Google Scholar] [CrossRef] [PubMed]
- Brook, C.E.; Dobson, A.P. Bats as ‘special’reservoirs for emerging zoonotic pathogens. Trends Microbiol. 2015, 23, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.-F.; Walker, P.J.; Poon, L.L. Mass extinctions, biodiversity and mitochondrial function: Are bats ‘special’as reservoirs for emerging viruses? Curr. Opin. Virol. 2011, 1, 649–657. [Google Scholar] [CrossRef] [PubMed]
- Luis, A.D.; O’Shea, T.J.; Hayman, D.T.; Wood, J.L.; Cunningham, A.A.; Gilbert, A.T.; Mills, J.N.; Webb, C.T. Network analysis of host–virus communities in bats and rodents reveals determinants of cross-species transmission. Ecol. Lett. 2015, 18, 1153–1162. [Google Scholar] [CrossRef] [PubMed]
- Turmelle, A.S.; Olival, K.J. Correlates of viral richness in bats (order Chiroptera). Ecohealth 2009, 6, 522–539. [Google Scholar] [CrossRef] [PubMed]
- Gay, N.; Olival, K.J.; Bumrungsri, S.; Siriaroonrat, B.; Bourgarel, M.; Morand, S. Parasite and viral species richness of Southeast Asian bats: Fragmentation of area distribution matters. Int. J. Parasitol. Parasites Wildl. 2014, 3, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Maganga, G.D.; Bourgarel, M.; Vallo, P.; Dallo, T.D.; Ngoagouni, C.; Drexler, J.F.; Drosten, C.; Nakouné, E.R.; Leroy, E.M.; Morand, S. Bat distribution size or shape as determinant of viral richness in African bats. PLoS ONE 2014, 9, e100172. [Google Scholar] [CrossRef] [PubMed]
- Luis, A.D.; Hayman, D.T.S.; O’Shea, T.J.; Cryan, P.M.; Gilbert, A.T.; Pulliam, J.R.C.; Mills, J.N.; Timonin, M.E.; Willis, C.K.R.; Cunningham, A.A.; et al. A comparison of bats and rodents as reservoirs of zoonotic viruses: Are bats special? Proc. R. Soc. Lond. B Biol. Sci. 2013, 280, 20122753. [Google Scholar] [CrossRef]
- Ge, X.-Y.; Li, J.-L.; Yang, X.-L.; Chmura, A.A.; Zhu, G.; Epstein, J.H.; Mazet, J.K.; Hu, B.; Zhang, W.; Peng, C. Isolation and characterization of a bat SARS-like coronavirus that uses the ACE2 receptor. Nature 2013, 503, 535–538. [Google Scholar] [CrossRef] [PubMed]
- Towner, J.S.; Pourrut, X.; Albariño, C.G.; Nkogue, C.N.; Bird, B.H.; Grard, G.; Ksiazek, T.G.; Gonzalez, J.-P.; Nichol, S.T.; Leroy, E.M. Marburg virus infection detected in a common African bat. PLoS ONE 2007, 2, e764. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-L.; Zhang, Y.-Z.; Jiang, R.-D.; Guo, H.; Zhang, W.; Li, B.; Wang, N.; Wang, L.; Waruhiu, C.; Zhou, J.-H. Genetically diverse filoviruses in Rousettus and Eonycteris spp. bats, China, 2009 and 2015. Emerg. Infect. Dis. 2017, 23, 482–486. [Google Scholar] [CrossRef] [PubMed]
- Negredo, A.; Palacios, G.; Vázquez-Morón, S.; González, F.; Dopazo, H.; Molero, F.; Juste, J.; Quetglas, J.; Savji, N.; de la Cruz Martínez, M. Discovery of an ebolavirus-like filovirus in Europe. PLoS Pathog. 2011, 7, e1002304. [Google Scholar] [CrossRef]
- Anthony, S.J.; Johnson, C.K.; Greig, D.J.; Kramer, S.; Che, X.; Wells, H.; Hicks, A.L.; Joly, D.O.; Wolfe, N.D.; Daszak, P. Global patterns in coronavirus diversity. Virus Evol. 2017, 3, vex012. [Google Scholar] [CrossRef] [PubMed]
- Altringham, J.D. Bats: From Evolution to Conservation; Oxford University Press: Chicago, IL, USA, 2011; ISBN 978-0-1992-0712-1. [Google Scholar]
- Calisher, C.H.; Childs, J.E.; Field, H.E.; Holmes, K.V.; Schountz, T. Bats: Important reservoir hosts of emerging viruses. Clin. Microbiol. Rev. 2006, 19, 531–545. [Google Scholar] [CrossRef] [PubMed]
- Amman, B.R.; Carroll, S.A.; Reed, Z.D.; Sealy, T.K.; Balinandi, S.; Swanepoel, R.; Kemp, A.; Erickson, B.R.; Comer, J.A.; Campbell, S.; et al. Seasonal pulses of Marburg virus circulation in juvenile Rousettus aegyptiacus bats coincide with periods of increased risk of human infection. PLoS Pathog. 2012, 8, e1002877. [Google Scholar] [CrossRef]
- Plowright, R.K.; Eby, P.; Hudson, P.J.; Smith, I.L.; Westcott, D.; Bryden, W.L.; Middleton, D.; Reid, P.A.; McFarlane, R.A.; Martin, G. Ecological dynamics of emerging bat virus spillover. Proc. R. Soc. Lond. B Biol. Sci. 2015, 282, 20142124. [Google Scholar] [CrossRef]
- Streicker, D.G.; Turmelle, A.S.; Vonhof, M.J.; Kuzmin, I.V.; McCracken, G.F.; Rupprecht, C.E. Host phylogeny constrains cross-species emergence and establishment of rabies virus in bats. Science 2010, 329, 676–679. [Google Scholar] [CrossRef] [PubMed]
- Fogarty, R.; Halpin, K.; Hyatt, A.D.; Daszak, P.; Mungall, B.A. Henipavirus susceptibility to environmental variables. Virus Res. 2008, 132, 140–144. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.; Peiris, J.; Lam, S.; Poon, L.; Yuen, K.; Seto, W. The effects of temperature and relative humidity on the viability of the SARS coronavirus. Adv. Virol. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Lorch, J.M.; Muller, L.K.; Russell, R.E.; O’Connor, M.; Lindner, D.L.; Blehert, D.S. Distribution and environmental persistence of the causative agent of white-nose syndrome, Geomyces destructans, in bat hibernacula of the eastern United States. Appl. Environ. Microbiol. 2013, 79, 1293–1301. [Google Scholar] [CrossRef] [PubMed]
- Jurado, V.; Laiz, L.; Rodriguez-Nava, V.; Boiron, P.; Hermosin, B.; Sanchez-Moral, S.; Saiz-Jimenez, C. Pathogenic and opportunistic microorganisms in caves. Int. J. Speleol. 2010, 39, 2. [Google Scholar] [CrossRef]
- Wacharapluesadee, S.; Sintunawa, C.; Kaewpom, T.; Khongnomnan, K.; Olival, K.J.; Epstein, J.H.; Rodpan, A.; Sangsri, P.; Intarut, N.; Chindamporn, A. Group C betacoronavirus in bat guano fertilizer, Thailand. Emerg. Infect. Dis. 2013, 19, 1349. [Google Scholar] [CrossRef] [PubMed]
- Seltmann, A.; Corman, V.M.; Rasche, A.; Drosten, C.; Czirják, G.Á.; Bernard, H.; Struebig, M.J.; Voigt, C.C. Seasonal fluctuations of astrovirus, but not coronavirus shedding in bats inhabiting human-modified tropical forests. Ecohealth 2017, 14, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Clements, R.; Sodhi, N.S.; Schilthuizen, M.; Ng, P.K. Limestone karsts of Southeast Asia: Imperiled arks of biodiversity. AIBS Bull. 2006, 56, 733–742. [Google Scholar] [CrossRef]
- Kingston, T. Research priorities for bat conservation in Southeast Asia: A consensus approach. Biodivers. Conserv. 2010, 19, 471–484. [Google Scholar] [CrossRef]
- Mildenstein, T.; Tanshi, I.; Racey, P.A. Exploitation of bats for bushmeat and medicine. In Bats in the Anthropocene: Conservation of Bats in a Changing World; Voigt, C., Kingston, T., Eds.; Springer: Cham, Switzerland, 2016; pp. 325–375. ISBN 978-3-3192-5220-9. [Google Scholar]
- Mann, S.L.; Steidl, R.J.; Dalton, V.M. Effects of cave tours on breeding Myotis velifer. J. Wildl. Manag. 2002, 66, 618–624. [Google Scholar] [CrossRef]
- Bausch, D.G.; Borchert, M.; Grein, T.; Roth, C.; Swanepoel, R.; Libande, M.L.; Talarmin, A.; Bertherat, E.; Muyembe-Tamfum, J.J.; Tugume, B.; Colebunders, R. Risk factors for Marburg hemorrhagic fever, Democratic Republic of the Congo. Emerg. Infect. Dis. 2003, 9, 1531. [Google Scholar] [CrossRef] [PubMed]
- Albarino, C.G.; Shoemaker, T.; Khristova, M.L.; Wamala, J.F.; Muyembe, J.J.; Balinandi, S.; Campbell, S.; Cannon, D.; Gibbons, A.; Bergeron, E. Genomic analysis of filoviruses associated with four viral hemorrhagic fever outbreaks in Uganda and the Democratic Republic of the Congo in 2012. Virology 2013, 442, 97–100. [Google Scholar] [CrossRef] [PubMed]
- Timen, A.; Koopmans, M.P.; Vossen, A.C.; van Doornum, G.J.; Günther, S.; Van den Berkmortel, F.; Verduin, K.M.; Dittrich, S.; Emmerich, P.; Osterhaus, A.D.; et al. Response to imported case of Marburg hemorrhagic fever, the Netherlands. Emerg. Infect. Dis. 2009, 15, 1171. [Google Scholar] [CrossRef] [PubMed]
- ICTV. Master Species List 2016 v1.3. Available online: https://talk.ictvonline.org/files/master-species-lists/m/msl (accessed on 6 June 2017).
- IUCN. The IUCN Red List of Threatened Species Vol. 2017–1. Available online: http://www.iucnredlist.org/ (accessed on 1 June 2017).
- PREDICT Consortium. PREDICT Preliminary Test Results. Available online: http://data.predict.global/ (accessed on 1 June 2017).
- Jones, K.E.; Bielby, J.; Cardillo, M.; Fritz, S.A.; O’Dell, J.; Orme, C.D.L.; Safi, K.; Sechrest, W.; Boakes, E.H.; Carbone, C. PanTHERIA: A species-level database of life history, ecology, and geography of extant and recently extinct mammals. Ecology 2009, 90, 2648. [Google Scholar] [CrossRef]
- Diniz-Filho, J.A.F.; Bini, L.M.; Rangel, T.F.; Morales-Castilla, I.; Olalla-Tárraga, M.Á.; Rodríguez, M.Á.; Hawkins, B.A. On the selection of phylogenetic eigenvectors for ecological analyses. Ecography 2012, 35, 239–249. [Google Scholar] [CrossRef]
- Diniz-Filho, J.A.F.; Sant’Ana, C.E.R.; Bini, L.M. An eigenvector method for estimating phylogenetic inertia. Evolution 1998, 52, 1247–1262. [Google Scholar] [CrossRef] [PubMed]
- Bininda-Emonds, O.R.; Cardillo, M.; Jones, K.E.; MacPhee, R.D.; Beck, R.M.; Grenyer, R.; Price, S.A.; Vos, R.A.; Gittleman, J.L.; Purvis, A. The delayed rise of present-day mammals. Nature 2007, 446, 507–512. [Google Scholar] [CrossRef] [PubMed]
- Fritz, S.A.; Bininda-Emonds, O.R.; Purvis, A. Geographical variation in predictors of mammalian extinction risk: Big is bad, but only in the tropics. Ecol. Lett. 2009, 12, 538–549. [Google Scholar] [CrossRef] [PubMed]
- Simmons, N.B. Order Chiroptera. In Mammal Species of the World; Wilson, D.E., Reeder, D.M., Eds.; The John Hopkins Press: Baltimore, MD, USA, 2005; pp. 312–529. [Google Scholar]
- Gómez, J.M.; Nunn, C.L.; Verdú, M. Centrality in primate–parasite networks reveals the potential for the transmission of emerging infectious diseases to humans. Proc. Natl. Acad. Sci. USA 2013, 110, 7738–7741. [Google Scholar] [CrossRef] [PubMed]
- Chua, K.B.; Koh, C.L.; Hooi, P.S.; Wee, K.F.; Khong, J.H.; Chua, B.H.; Chan, Y.P.; Lim, M.E.; Lam, S.K. Isolation of Nipah virus from Malaysian island flying-foxes. Microbes Infect. 2002, 4, 145–151. [Google Scholar] [CrossRef]
- Halpin, K.; Young, P.; Field, H.; Mackenzie, J. Isolation of Hendra virus from Pteropid bats: A natural reservoir of Hendra virus. J. Gen. Virol. 2000, 81, 1927–1932. [Google Scholar] [CrossRef] [PubMed]
- Broders, H.G.; Quinn, G.M.; Forbes, G.J. Species status, and the spatial and temporal patterns of activity of bats in southwest Nova Scotia, Canada. Northeast. Nat. (Steuben) 2003, 10, 383–398. [Google Scholar] [CrossRef]
- Kingston, T. Response of bat diversity to forest disturbance in Southeast Asia: Insights from long-term research in Malaysia. In Bat Evolution, Ecology, and Conservation; Adams, R.A., Pedersen, S.C., Eds.; Springer: New York, NY, USA, 2013; pp. 169–185. ISBN 978-1-4614-7397-8. [Google Scholar]
- Fenton, M.B.; Simmons, N.B. Bats: A World of Science and Mystery; University of Chicago Press: Chicago, IL, USA, 2015; ISBN 978-0-2260-6512-0. [Google Scholar]
- Schneeberger, K.; Czirják, G.Á.; Voigt, C.C. Measures of the constitutive immune system are linked to diet and roosting habits of neotropical bats. PLoS ONE 2013, 8, e54023. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Kosoy, M.; Olival, K.J.; Dittmar, K. Horizontal transfers and gene losses in the phospholipid pathway of Bartonella reveal clues about early ecological niches. Genome Biol. Evol. 2014, 6, 2156–2169. [Google Scholar] [CrossRef] [PubMed]
- Muscarella, R.; Fleming, T.H. The role of frugivorous bats in tropical forest succession. Biol Rev Camb Philos. Soc. 2007, 82, 573–590. [Google Scholar] [CrossRef] [PubMed]
- Bumrungsri, S.; Sripaoraya, E.; Chongsiri, T.; Sridith, K.; Racey, P.A. The pollination ecology of durian (Durio zibethinus, Bombacaceae) in southern Thailand. J. Trop. Ecol. 2009, 25, 85–92. [Google Scholar] [CrossRef]
- Bumrungsri, S.; Harbit, A.; Benzie, C.; Carmouche, K.; Sridith, K.; Racey, P. The pollination ecology of two species of parkia (Mimosaceae) in southern Thailand. J. Trop. Ecol. 2008, 24, 467–475. [Google Scholar] [CrossRef]
- Petchmunee, K. Economic Valuation and Learning Process Construction: A Case Study of the Cave Nectarivorous Bat (Eonycteris spelaea dobson). MSc Thesis (Environmental Science), Prince of Songkla University, Hat Yai, Thailand, 2008. (In Thai with English Abstract). [Google Scholar]
- Lau, S.K.; Li, K.S.; Tsang, A.K.; Shek, C.-T.; Wang, M.; Choi, G.K.; Guo, R.; Wong, B.H.; Poon, R.W.; Lam, C.S. Recent transmission of a novel alphacoronavirus, bat coronavirus HKU10, from Leschenault’s rousettes to pomona leaf-nosed bats: First evidence of interspecies transmission of coronavirus between bats of different suborders. J. Virol. 2012, 01305–01312. [Google Scholar] [CrossRef] [PubMed]
- Escalera-Zamudio, M.; Rojas-Anaya, E.; Kolokotronis, S.-O.; Taboada, B.; Loza-Rubio, E.; Méndez-Ojeda, M.L.; Arias, C.F.; Osterrieder, N.; Greenwood, A.D. Bats, primates, and the evolutionary origins and diversification of mammalian gammaherpesviruses. MBio 2016, 7, e01425–e01416. [Google Scholar] [CrossRef] [PubMed]
- Kreuder Johnson, C.; Hitchens, P.L.; Smiley Evans, T.; Goldstein, T.; Thomas, K.; Clements, A.; Joly, D.O.; Wolfe, N.D.; Daszak, P.; Karesh, W.B.; et al. Spillover and pandemic properties of zoonotic viruses with high host plasticity. Sci. Rep. 2015, 5, 14830. [Google Scholar] [CrossRef] [PubMed]
- Longdon, B.; Brockhurst, M.A.; Russell, C.A.; Welch, J.J.; Jiggins, F.M. The evolution and genetics of virus host shifts. PLoS Pathog. 2014, 10, e1004395. [Google Scholar] [CrossRef] [PubMed]
- Poulin, R.; Krasnov, B.R.; Mouillot, D. Host specificity in phylogenetic and geographic space. Trends Parasitol. 2011, 27, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Brierley, L.; Vonhof, M.J.; Olival, K.J.; Daszak, P.; Jones, K.E. Quantifying global drivers of zoonotic bat viruses: A process-based perspective. Am. Nat. 2016, 187, E53–E64. [Google Scholar] [CrossRef] [PubMed]
- Patterson, B.D.; Dick, C.W.; Dittmar, K. Roosting habits of bats affect their parasitism by bat flies (Diptera: Streblidae). J. Trop. Ecol. 2007, 23, 177–189. [Google Scholar] [CrossRef]
- Chen, L.; Liu, B.; Yang, J.; Jin, Q. DBatVir: The database of bat-associated viruses. Database 2014, 2014, bau021. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Yang, L.; Ren, X.; He, G.; Zhang, J.; Yang, J.; Qian, Z.; Dong, J.; Sun, L.; Zhu, Y. Deciphering the bat virome catalog to better understand the ecological diversity of bat viruses and the bat origin of emerging infectious diseases. ISME J. 2016, 10, 609–620. [Google Scholar] [CrossRef] [PubMed]
- Jones, K.E.; Patel, N.G.; Levy, M.A.; Storeygard, A.; Balk, D.; Gittleman, J.L.; Daszak, P. Global trends in emerging infectious diseases. Nature 2008, 451, 990–993. [Google Scholar] [CrossRef] [PubMed]
- Towner, J.S.; Amman, B.R.; Sealy, T.K.; Carroll, S.A.R.; Comer, J.A.; Kemp, A.; Swanepoel, R.; Paddock, C.D.; Balinandi, S.; Khristova, M.L. Isolation of genetically diverse Marburg viruses from Egyptian fruit bats. PLoS Pathog. 2009, 5, e1000536. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Willoughby, A.R.; Phelps, K.L.; PREDICT Consortium; Olival, K.J. A Comparative Analysis of Viral Richness and Viral Sharing in Cave-Roosting Bats. Diversity 2017, 9, 35. https://doi.org/10.3390/d9030035
Willoughby AR, Phelps KL, PREDICT Consortium, Olival KJ. A Comparative Analysis of Viral Richness and Viral Sharing in Cave-Roosting Bats. Diversity. 2017; 9(3):35. https://doi.org/10.3390/d9030035
Chicago/Turabian StyleWilloughby, Anna R., Kendra L. Phelps, PREDICT Consortium, and Kevin J. Olival. 2017. "A Comparative Analysis of Viral Richness and Viral Sharing in Cave-Roosting Bats" Diversity 9, no. 3: 35. https://doi.org/10.3390/d9030035
APA StyleWilloughby, A. R., Phelps, K. L., PREDICT Consortium, & Olival, K. J. (2017). A Comparative Analysis of Viral Richness and Viral Sharing in Cave-Roosting Bats. Diversity, 9(3), 35. https://doi.org/10.3390/d9030035




