On the History of Cattle Genetic Resources
Abstract
:1. Introduction
2. Wild ancestors and Sites of Domestication





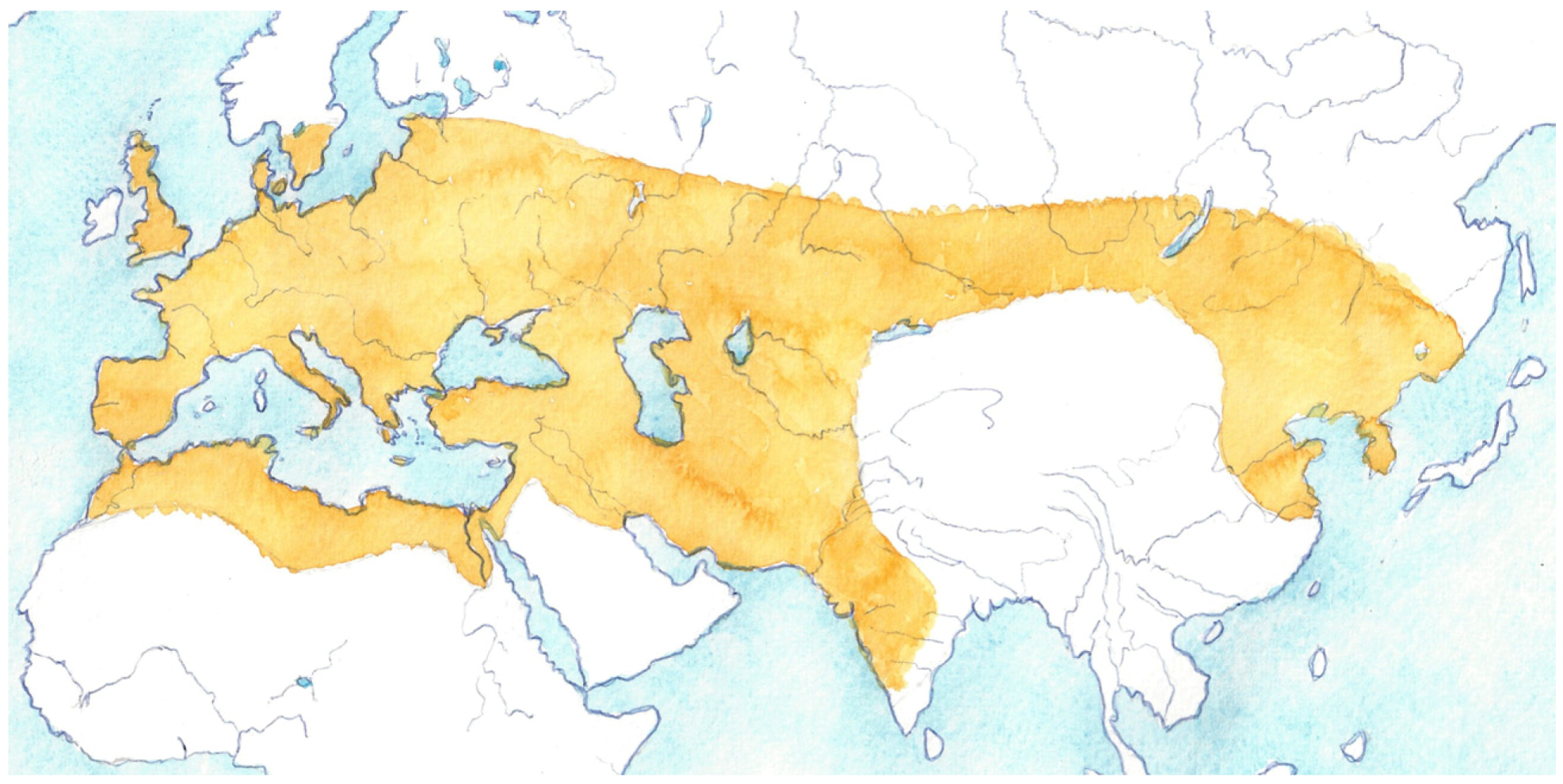
3. Early Taurine Dispersal
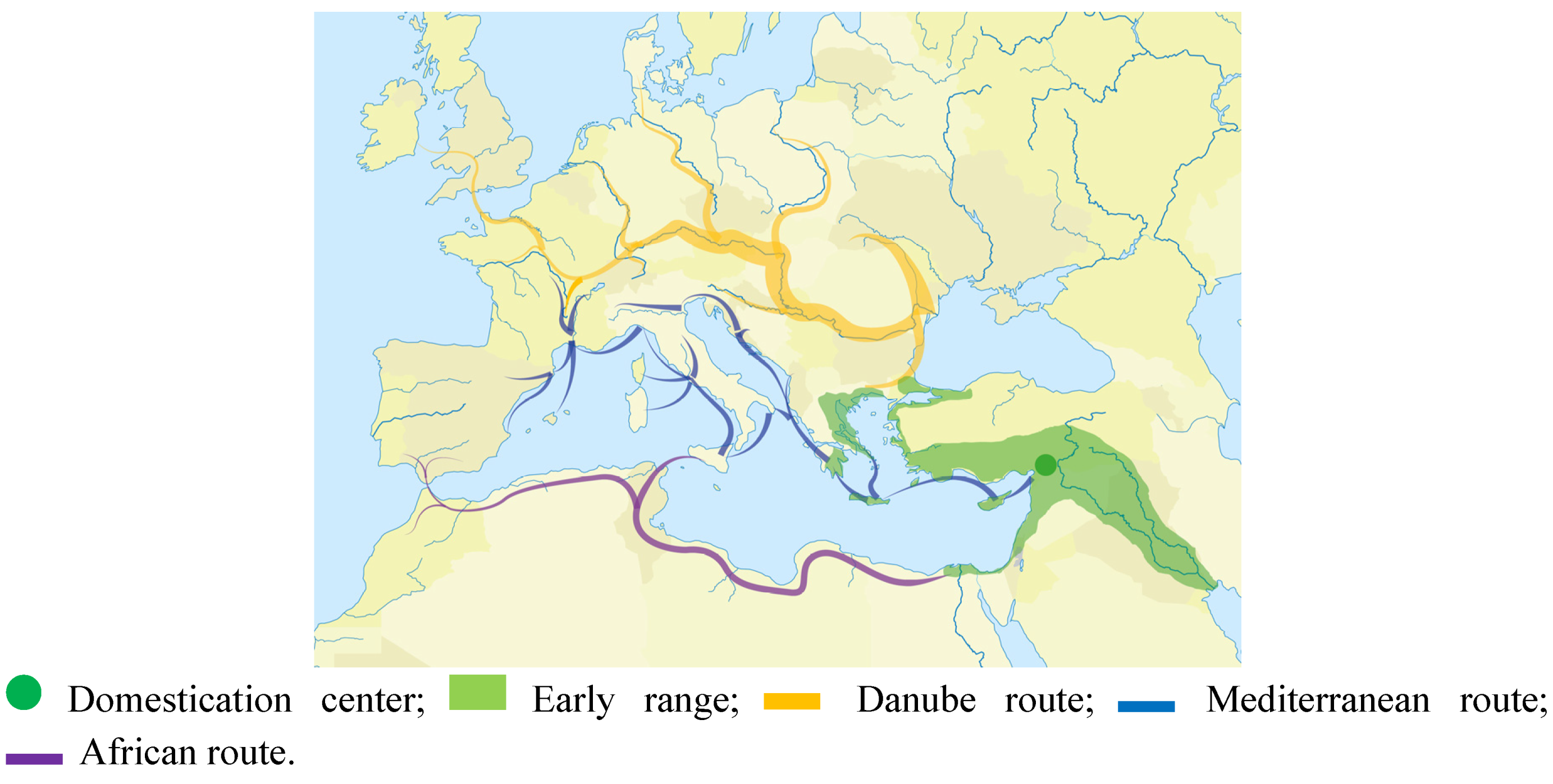
4. Early Zebu Dispersal

5. Short-Horned and Small Taurine Cattle in the Bronze and Iron Age
6. Large Greek and Roman Cattle

7. Medieval Cattle and Catastrophes


8. Recovery of Cattle
9. Preindustrial Progress

10. The First Breeds



11. European Breeds of the 19th Century: Tour of the Continent



12. Breed Groups and Clusters
- (1)
- North-European cattle, comprising the following breed clusters:
- (a)
- Four clusters corresponding to the expansion of popular dairy breeds (black-pied, red-pied, Baltic red and Nordic Ayrshire);
- (b)
- Three regional clusters of related but diverse breeds (British, Nordic and Russian-Siberian); and
- (c)
- A loose cluster of Shorthorn with several Belgian and North-French, dairy-beef and beef breeds influenced by the Shorthorn, including the Maine-Anjou and Charolais.
- (2)
- Central European cattle, with many dual purpose (beef-work or dairy-work) and triple-purpose breeds, comprising three major and two minor breed clusters:
- (a)
- Two breed clusters corresponding to the expansion of the Simmental and Swiss Brown breeds, respectively. The Simmental cluster also contains related Swiss, French and Italian cattle from the western Alps, the German and Austrian yellow and blond breeds and the German Hinterwälder and Vorderwälder;
- (b)
- The unicolored beef and beef-work breeds from South France;
- (c)
- Two minor clusters of Alpine Grey cattle and of the spotted dairy cattle from the eastern Alps (Pinzgauer, Pustertaler and Čika). The Piemontese beef breed also belongs to the Central-European cattle, but does not belong to a breed cluster.
- (3)
- Iberian cattle, with a large variety of coat colors and horn morphology and mainly used as beef, work and fighting cattle.
- (4)
- The mostly long-horned and grey Podolian cattle, primarily developed as beef and work animals.
- (5)
- The genetically diverse breeds of the Balkans and Anatolian breeds, still representing the undeveloped taurine cattle.
13. Asian Cattle

14. African Cattle

15. Cattle in the New World



16. Cattle without Borders


17. New Life for Local Breeds
- In the 1960s the Greek Shorthorn was described as one landrace population [209], but by 2010 eight distinct local varieties were recognized [210]. In addition to the 33 recognized indigenous breeds of Ethiopia, several more have been identified and reported in recent years [211,212,213,214,215,216].
- For Fipa cattle, a zenga type of southwest Tanzania, two varieties were described in 2011 [217].
- Recently the formal national recognition of several Indian desi (local’) breeds has increased the number of zebu breeds in India to more than 30.
- For the all but vanished Florida Cracker and Pineywoods cattle of the southern United States, 5 and 15 distinct local lines and herds, respectively, are now recognized [218].
- In Ecuador four local types of Sierra Criollos, kept at different altitude, have been differentiated [219].
- Rescue and maintenance of the remaining populations (on the hoof). An extreme example is the rescue of the feral cattle on Enderby Island [220]. Using oocytes and clones from the single surviving cow and semen collected from one of the bulls shot in 1991, resulted in six calves being born in 2006.
- Sustainable conservation, for instance by advertising (branding) presumed unique qualities of a local breed (e.g., grass finished beef or slow food): “If you want to save a breed, they have to have a job.” [221].
- Selection of animals from related breeds that resemble the animals from the endangered breed. Examples:
- -
- In 1986 the Austrian Tux-Zillertaler was reconstituted by crossings the approximately 30 remaining females with Swiss Hérens sires.
- -
- Rebreeding of the Ansbach-Triesdorfer, which had vanished in 1940, began in 1987 by selection of German Fleckvieh cows from the Ansbach region with the characteristic speckled color patterns on head and feet.
- -
- The French Bordelaise was considered extinct in 1960, but has been reconstituted since 1987 by using crossbred animals descending from the original breed.
18. On the History and Future of Diversity
- (I)
- Domestication and subsequent interaction with wild populations;
- (II)
- Migrations followed by natural adaptation to agricultural habitats in diverse environments and during the subsequent periods of human history; and
- (III)
- A relatively recent systematic breed-oriented selection.
- A sampling of a part of the diversity of the ancestor species, but is usually followed by introgression of wild animals during the dispersal of the domesticates [226]. The taurine domestication was estimated to involve only 80 females [17], but later introgression of aurochs males on different continents probably introduced additional diversity. For African taurine cattle this is now accepted [33].
- Zebu and taurine cattle are the domestic forms of two well-diverged but cross-fertile aurochs subspecies from Southwest Asia [79] and the Indus Valley [34]. These were adapted to different environments, which with the many intermediate crossbreds ensured a potential adaptation of the domestic animals to climates ranging from temperate to tropical.
- In addition to taurine and zebu cattle Asia harbors also domestic cattle descending from other bovine species with many combinations of mixed-species origin.
- Europe remained completely taurine with mere traces of putative zebu introgression, while Asia, Africa, America and Australia harbor both taurine, zebu and taurindicine breeds in different climactic zones.
- In Africa, the diversity pattern has been determined by consecutive immigrations of short-horned taurine, long-horned taurines and zebus, by introgression of African aurochs, by disease resistance (trypanotolerance, resistance to rinderpest), by the wide range of management systems (sedentary, transhumance, nomadic pastoralism).
- The modulation of horn development illustrates an early and flexible adaptation to local requirements or preferences, short-horned or hornless animals being convenient for stabling.
- The decrease in size is a domestic adaptation, but may also reflect the capacity of feeding cattle during the winter period. During antiquity the large Epirote cattle and its Roman descendants created a difference with the more common small cattle, but this may not have survived during the Middle Ages [54]. European cattle started to regain their size since the 15th century.
- In the Roman era, production purposes were multiple as evidenced by the preponderance of draught cattle in Italy and of dairy cattle in central and northern Europe [90].
- Because the migration of the Germanic tribes were the last major movements of European people, it is plausible that from the Middle Ages differentiation of European cattle was mainly due to isolation by distance. This process was only partially undercut by trading of cattle causing gene flow between neighboring regions and is still reflected by the genetic clustering of the present breeds [8].
- In the tropical zones, diversity patterns were decided largely by the tropical adaptation and resistance to rinderpest. This has led to widespread incrossing of zebu in African and American taurine populations.
- Cattle were introduced in America only after 1492 with the import of Iberian and Northwest-European cattle, followed in the 19th century by English, Dutch and zebu breeds and in the 20th century by European continental beef breeds.
- After the Middle Ages, cultural and technical progress and the growing demand for food rationalized the European cattle husbandry. Starting in the 18th century this led to an organized management of regional breeds: genetically isolated groups of phenotypically homogeneous animals. This took place all over Europe, where cattle exhibitions soon catalyzed the merging of early breeds from neighboring regions. This improved the productivity of European breeds, changed appreciably their appearance and emphasized the differences between breeds.
- Since the 19th century several highly productive breeds spread to other countries and continents (Table S2), where separate herd books were kept. Thus, several groups of closely related breeds were formed differing mainly in nationality.
- A less productive and often abortive development was the introduction of highly productive European breeds in tropical countries where the intensive management required for these cattle cannot be ensured.
- In the New World, a creative attitude to breeding led to a number of taurine or taurindicine synthetic breeds, the result of crossing cattle from different origins. Several of these are highly successful (Table S3).
- A focus on productive breeds diminished the population sizes of local breeds, several of which disappeared by crossing with sires from productive breeds. This is being counteracted by successful conservation efforts.
Acknowledgments
Author Contributions
Supplementary Materials
Conflicts of Interest
References and Notes
- Diamond, J. Guns, Germs, and Steel; Norton Company: New York, NY, USA, 1997. [Google Scholar]
- Conolly, J.; Manning, K.; Colledge, S.; Dobney, K.; Shennan, S. Species distribution modelling of ancient cattle from early Neolithic sites in SW Asia and Europe. Holocene 2012, 24, 3–14. [Google Scholar]
- Price, T.D. Europe’s First Farmers: An Introduction; Cambridge University Press: Cambridge, UK, 2000. [Google Scholar]
- Brass, M. Reconsidering the emergence of social complexity in early Saharan pastoral societies, 5000–2500 BC. Sahara 2007, 18, 1–16. [Google Scholar]
- Ajmone-Marsan, P.; Garcia, J.F.; Lenstra, J.A. On the origin of cattle: how aurochs became cattle and colonized the world. Evol. Anthropol. 2010, 19, 148–157. [Google Scholar] [CrossRef]
- Rifkin, B. Beyond Beef: The Rise and Fall of the Cattle Culture; Dutton: London, UK, 1992. [Google Scholar]
- Felius, M. Cattle Breeds, an Encyclopedia; Misset Uitgeverij: Doetinchem, The Netherlands, 1995. [Google Scholar]
- Felius, M.; Koolmees, P.A.; Theunissen, B.; European Cattle Genetic Diversity Consortium; Lenstra, J.A. On the breeds of cattle—Historic and current classifications. Diversity 2011, 3, 660–692. [Google Scholar] [CrossRef]
- Felius, M.; Theunissen, B.; Lenstra, J.A. On the conservation of cattle—the role of breeds. J. Agric. Sci. 2014, in press. [Google Scholar]
- Lenstra, J.A.; Bradley, D.G. Systematics and phylogeny of cattle. In The Genetics of Cattle; Fries, R., Ruvinsky, A., Eds.; CABI Publishing: Oxon, UK; New York, NY, USA, 1999; pp. 1–14. [Google Scholar]
- Lenstra, J.A.; Theunissen, B.; Felius, M. Domestic cattle and buffaloes. In Ecology, Evolution and Behaviour of Wild Cattle: Implications for Conservation; Melletti, M., Burton, J., Eds.; Cambridge University Press: Cambridge, UK, 2014. [Google Scholar]
- Ho, S.Y.; Larson, G.; Edwards, C.J.; Heupink, T.H.; Lakin, K.E.; Holland, P.W.; Shapiro, B. Correlating Bayesian date estimates with climatic events and domestication using a bovine case study. Biol. Lett. 2008, 4, 370–374. [Google Scholar] [CrossRef] [PubMed]
- Achilli, A.; Bonfiglio, S.; Olivieri, A.; Malusa, A.; Pala, M.; Hooshiar Kashani, B.; Perego, U.A.; Ajmone-Marsan, P.; Liotta, L.; Semino, O.; et al. The multifaceted origin of taurine cattle reflected by the mitochondrial genome. PLoS One 2009, 4, e5753. [Google Scholar] [CrossRef]
- Hassanin, A.; An, J.; Ropiquet, A.; Nguyen, T.T.; Couloux, A. Combining multiple autosomal introns for studying shallow phylogeny and taxonomy of Laurasiatherian mammals: Application to the tribe Bovini (Cetartiodactyla, Bovidae). Mol. Phylogenet. Evol. 2013, 66, 766–775. [Google Scholar] [CrossRef] [PubMed]
- Bollongino, R.; Burger, J.; Powell, A.; Mashkour, M.; Vigne, J.D.; Thomas, M.G. Modern taurine cattle descended from small number of Near-Eastern founders. Mol. Biol. Evol. 2012, 29, 2101–2104. [Google Scholar] [CrossRef] [PubMed]
- Gautier, Y.; Gautier, C. L’Art du Sahara; Seuil: Paris, France, 1996. [Google Scholar]
- Van Vuure, C. Retracing the Aurochs; Universität Paderborn: Höxter, Germany, 2001. [Google Scholar]
- Helmer, D.; Gourichon, L.; Monchot, H.; Peters, J.; Saña Segui, M. Identifying early domestic cattle from prepottery Neolithic sites on the middle Euphrates using sexual dimorphism. In The First Steps of Animal Domestication; Vigne, J.D., Peters, J., Helmer, D., Eds.; Oxbow Books: Oxford, UK, 2005; pp. 86–95. [Google Scholar]
- Vigne, J.D. The origins of animal domestication and husbandry: A major change in the history of humanity and the biosphere. C. R. Biol. 2011, 334, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Barker, G. Prehistoric Farming in Europe; Cambridge University Press: Cambridge, UK, 1985. [Google Scholar]
- Zeder, M.A.; Emshwiller, E.; Smith, B.D.; Bradley, D.G. Documenting domestication: The intersection of genetics and archaeology. Trends Genet. 2006, 22, 139–155. [Google Scholar] [CrossRef] [PubMed]
- Evershed, R.P.; Payne, S.; Sherratt, A.G.; Copley, M.S.; Coolidge, J.; Urem-Kotsu, D.; Kotsakis, K.; Ozdogan, M.; Ozdogan, A.E.; Nieuwenhuyse, O.; et al. Earliest date for milk use in the Near East and southeastern Europe linked to cattle herding. Nature 2008, 455, 528–531. [Google Scholar] [CrossRef] [PubMed]
- Bonfiglio, S.; Achilli, A.; Olivieri, A.; Negrini, R.; Colli, L.; Liotta, L.; Ajmone-Marsan, P.; Torroni, A.; Ferretti, L. The enigmatic origin of bovine mtDNA haplogroup R: Sporadic interbreeding or an independent event of Bos primigenius domestication in Italy? PLoS One 2010, 5, e15760. [Google Scholar] [CrossRef]
- Lenstra, J.A.; Ajmone-Marsan, P.; Beja-Pereira, A.; Bollongino, R.; Bradley, D.G.; Colli, L.; de Gaetano, A.; Edwards, C.J.; Felius, M.; Ferretti, L.; et al. Meta-analysis of mitochondrial DNA reveals several population bottlenecks during worldwide migrations of cattle. Diversity 2014, 6, 178–187. [Google Scholar] [CrossRef]
- Colledge, S.; Conolly, J.; Dobney, K.; Manning, K.; Shennan, S. Origins and Spread of Domestic Animals in Southwest Asia and Europe; Left Coast Press: Wallnut Creek, CA, USA, 2013. [Google Scholar]
- Jarrige, J.F. Mehrgarh Neolithic. In Proceedings of the International Seminar on the “First Farmers in Global Perspective”, Lucknow, India, 18–20 January 2006; pp. 135–154.
- Arbuckle, B.S.; Makarewicz, C.A. The early management of cattle (Bos taurus) in Neolithic central Anatolia. Antiquity 2009, 83, 669–686. [Google Scholar]
- Epstein, H.; Mason, I.L. The Origin of the Domestic Animals of Africa; African Publishing Corporation: New York, NY, USA; London, UK; Munich, Germany, 1971. [Google Scholar]
- McInerney, J. The Cattle of the Sun. Cows and Culture in the World of the Ancient Greeks; Princeton University Press: Princeton, NJ, USA, 2010. [Google Scholar]
- Brass, M. Revisiting a hoary chestnut: The nature of early cattle domestication in North-East Africa. Sahara 2012, 24, 7–12. [Google Scholar]
- Linseele, V.; van Neer, W.; Thys, S.; Phillipps, R.; Cappers, R.; Wendrich, W.; Holdaway, S. New archaeozoological data from the Fayum “Neolithic” with a critical assessment of the evidence for early stock keeping in Egypt. PLoS One 2014, 9, e108517. [Google Scholar] [CrossRef]
- Gifford-Gonzalez, D.; Hanotte, O. Domesticating Animals in Africa: Implications of Genetic and Archaeological Findings. J. World Prehist. 2011, 24, 1–23. [Google Scholar] [CrossRef]
- Stock, F.; Gifford-Gonzalez, D. Genetics and African cattle domestication. Afr. Archaeol. Rev. 2013, 30, 51–72. [Google Scholar] [CrossRef]
- Chen, S.; Lin, B.Z.; Baig, M.; Mitra, B.; Lopes, R.J.; Santos, A.M.; Magee, D.A.; Azevedo, M.; Tarroso, P.; Sasazaki, S.; et al. Zebu cattle are an exclusive legacy of the South Asia Neolithic. Mol. Biol. Evol. 2010, 27, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Paijmans, J.L.; Chang, F.; Wu, X.; Chen, G.; Lei, C.; Yang, X.; Wei, Z.; Bradley, D.G.; Orlando, L.; et al. Morphological and genetic evidence for early Holocene cattle management in northeastern China. Nat. Commun. 2013. [Google Scholar] [CrossRef]
- Gautier, A. Animal Domestication in North Africa. In Proceedings of the ACACIA Conference Aridity, Change and Conflict in Africa; Königswinter, Germany, 1–3 October 2003, Bollig, M., Bubenzer, I., Vogelsang, R., Wotzka, H., Eds.; Heinrich-Barth-Institut: Cologne, Germany; pp. 75–89.
- Beja-Pereira, A.; Caramelli, D.; Lalueza-Fox, C.; Vernesi, C.; Ferrand, N.; Casoli, A.; Goyache, F.; Royo, L.J.; Conti, S.; Lari, M.; et al. The origin of European cattle: Evidence from modern and ancient DNA. Proc. Natl. Acad. Sci. USA 2006, 103, 8113–8118. [Google Scholar] [CrossRef] [PubMed]
- Achilli, A.; Olivieri, A.; Pellecchia, M.; Uboldi, C.; Colli, L.; Al-Zahery, N.; Accetturo, M.; Pala, M.; Hooshiar Kashani, B.; Perego, U.A.; et al. Mitochondrial genomes of extinct aurochs survive in domestic cattle. Curr. Biol. 2008, 18, R157–R158. [Google Scholar] [CrossRef]
- Qiu, Q.; Zhang, G.; Ma, T.; Qian, W.; Wang, J.; Ye, Z.; Cao, C.; Hu, Q.; Kim, J.; Larkin, D.M.; et al. The yak genome and adaptation to life at high altitude. Nat. Genet. 2012, 44, 946–949. [Google Scholar] [CrossRef] [PubMed]
- Payne, W.J.A.; Hodges, J. Tropical Cattle: Origin, Breeding and Breeding Policies; Blackwell Science: Oxford, UK, 1997. [Google Scholar]
- Rasteiro, R.; Chikhi, L. Female and male perspectives on the Neolithic transition in Europe: Clues from ancient and modern genetic data. PLoS One 2013, 8, e60944. [Google Scholar] [CrossRef]
- Perez-Pardal, L.; Royo, L.J.; Beja-Pereira, A.; Curik, I.; Traore, A.; Fernandez, I.; Solkner, J.; Alonso, J.; Alvarez, I.; Bozzi, R.; et al. Y-specific microsatellites reveal an African subfamily in taurine (Bos taurus) cattle. Anim. Genet. 2009, 41, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Edwards, C.J.; Ginja, C.; Kantanen, J.; Pérez-Pardal, L.; Tresset, A.; Stock, F.; Gama, L.T.; Penedo, M.C.T.; Bradley, D.G.; Lenstra, J.A.; et al. Dual origins of dairy cattle farming—Evidence from a comprehensive survey of European Y-chromosomal variation. PLoS One 2011, 6, e15922. [Google Scholar] [CrossRef]
- Hongo, H.; Pearson, J.Ö.B.; Ilgezdi, G. The process of ungulate domestication at Çayönü, southeastern Turkey: A multidisciplinary approach focusing on Bos sp. and Cervus elaphus. Anthropozool 2009, 44, 63–78. [Google Scholar] [CrossRef]
- Kantanen, J.; Edwards, C.J.; Bradley, D.G.; Viinalass, H.; Thessler, S.; Ivanova, Z.; Kiselyova, T.; Cinkulov, M.; Popov, R.; Stojanovic, S.; et al. Maternal and paternal genealogy of Eurasian taurine cattle (Bos taurus). Heredity 2009, 103, 404–415. [Google Scholar] [CrossRef] [PubMed]
- Decker, J.E.; McKay, S.D.; Rolf, M.M.; Kim, J.; Molina Alcala, A.; Sonstegard, T.S.; Hanotte, O.; Gotherstrom, A.; Seabury, C.M.; Praharani, L.; et al. Worldwide patterns of ancestry, divergence, and admixture in domesticated cattle. PLoS Genet. 2014, 10, e1004254. [Google Scholar] [CrossRef] [PubMed]
- Van Neer, W. Egyptian Longhorn Cattle from the Elite Cemetery at HK6: Not Just a Load of Old Bull. Nekhen News 2010, 22, 8–9. [Google Scholar]
- Gautier, A.; van Neer, W. Animal remains from Mahal Teglinos (Kassala, Sudan) and the arrival of pastoralism in the southern Atbai. J. Afr. Archaeol. 2006, 4, 223–233. [Google Scholar] [CrossRef]
- Holmes, J.A. Ecology. How the Sahara became dry. Science 2008, 320, 752–753. [Google Scholar] [CrossRef] [PubMed]
- Dunne, J.; Evershed, R.P.; Salque, M.; Cramp, L.; Bruni, S.; Ryan, K.; Biagetti, S.; di Lernia, S. First dairying in green Saharan Africa in the fifth millennium BC. Nature 2012, 486, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Bonfiglio, S.; Ginja, C.; de Gaetano, A.; Achilli, A.; Olivieri, A.; Colli, L.; Tesfaye, K.; Agha, S.H.; Gama, L.T.; Cattonaro, F.; et al. Origin and spread of Bos taurus: New clues from mitochondrial genomes belonging to haplogroup T1. PLoS One 2012, 7, e38601. [Google Scholar] [CrossRef]
- Anderung, C.; Bouwman, A.; Persson, P.; Carretero, J.M.; Ortega, A.I.; Elburg, R.; Smith, C.; Arsuaga, J.L.; Ellegren, H.; Gotherstrom, A.; et al. Prehistoric contacts over the Straits of Gibraltar indicated by genetic analysis of Iberian Bronze Age cattle. Proc. Natl. Acad. Sci. USA 2005, 102, 8431–8435. [Google Scholar] [CrossRef] [PubMed]
- Perez-Pardal, L.; Royo, L.J.; Beja-Pereira, A.; Chen, S.; Cantet, R.J.; Traore, A.; Curik, I.; Solkner, J.; Bozzi, R.; Fernandez, I.; et al. Multiple paternal origins of domestic cattle revealed by Y-specific interspersed multilocus microsatellites. Heredity 2010, 105, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Bökönyi, S. History of Domestic Mammals in Central and Eastern Europe; Akadémiai Kiadó: Budapest, Hungary, 1974. [Google Scholar]
- Guilaine, J. De la Vague à la Tombe. La Conquête Néolithique le la Méditerranée; Le Seuil: Paris, France, 2003. [Google Scholar]
- Gkiasta, M.; Russell, T.; Shennan, S.; Steele, J. Neolithic transition in Europe: The radiocarbon record revisited. Antiquity 2003, 77, 45–62. [Google Scholar]
- Cymbron, T.; Freeman, A.; Malheiro, M.I.; Vigne, J.D.; Bradley, D. Microsatellite diversity suggests different histories for Mediterranean and Northern European cattle populations. Proc. R. Soc. B: Biol. Sci. 2005, 272, 1837–1843. [Google Scholar] [CrossRef]
- Tresset, A.; Vigne, J.D. Substitution of species, techniques and symbols at the Mesolithic-Neolithic transition in Western Europe. Proc. Br. Acad. 2007, 144, 189–210. [Google Scholar]
- Benecke, N. Der Mensch Und Seine Haustiere; Theiss: Stuttgart, Germany, 1994. [Google Scholar]
- Tresset, A. French connections II: Of cows and men. In Neolithic Settlement in Ireland and Western Britain; Armit, I., Murphy, E., Nelis, E., Simpson, D., Eds.; Oxbow Books: Oxford, UK, 2003; pp. 18–30. [Google Scholar]
- Salque, M.; Bogucki, P.I.; Pyzel, J.; Sobkowiak-Tabaka, I.; Grygiel, R.; Szmyt, M.; Evershed, R.P. Earliest evidence for cheese making in the sixth millennium BC in northern Europe. Nature 2013, 493, 522–525. [Google Scholar] [CrossRef] [PubMed]
- Copley, M.S.; Berstan, R.; Dudd, S.N.; Docherty, G.; Mukherjee, A.J.; Straker, V.; Payne, S.; Evershed, R.P. Direct chemical evidence for widespread dairying in prehistoric Britain. Proc. Natl. Acad. Sci. USA 2003, 100, 1524–1529. [Google Scholar] [CrossRef] [PubMed]
- Balasse, M.; Tresset, A. Early weaning of Neolithic domestic cattle (Bercy, France) revealed by intra-tooth variation in nitrogen isotope ratios. J. Archeol. Sci. 2002, 29, 853–859. [Google Scholar] [CrossRef]
- Itan, Y.; Powell, A.; Beaumont, M.A.; Burger, J.; Thomas, M.G. The origins of lactase persistence in Europe. PLoS Comput. Biol. 2009, 5, e1000491. [Google Scholar] [CrossRef]
- Curry, A. The milk revolution. Nature 2013, 500, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Stock, F.; Edwards, C.J.; Bollongino, R.; Finlay, E.K.; Burger, J.; Bradley, D.G. Cytochrome b sequences of ancient cattle and wild ox support phylogenetic complexity in the ancient and modern bovine populations. Anim. Genet. 2009, 40, 694–700. [Google Scholar] [CrossRef] [PubMed]
- Wint, W.; Robinson, T. Gridded Livestock of the World 2007; FAO: Rome, Italy, 2007. [Google Scholar]
- Allchin, F.R. Early domestic animals in India and Pakistan. In The Domestication and Exploitation of Plants and Animals; Ucko, P.J., Dimbelby, G.W., Eds.; Gerald Duckworth: London, UK, 1969; pp. 317–322. [Google Scholar]
- Allchin, B.; Allchin, R.A. The Birth of Indian Civilization: India and Pakistan before 500 BC; Penguin: London, UK, 1974. [Google Scholar]
- Naik, S.N. Origin and domestication of zebu cattle (Bos indicus). J. Hum. Evol. 1978, 7, 23–30. [Google Scholar] [CrossRef]
- Cai, D.; Sun, Y.; Tang, Z.; Hud, S.; Li, W.; Zhao, X.; Xiang, H.; Zhou, H. The origins of Chinese domestic cattle as revealed by ancient DNA analysis. J. Archaeol. Sci. 2014, 41, 423–434. [Google Scholar] [CrossRef]
- Li, R.; Zhang, X.M.; Campana, M.G.; Huang, J.P.; Chang, Z.H.; Qi, X.B.; Shi, H.; Su, B.; Zhang, R.F.; Lan, X.Y.; et al. Paternal origins of Chinese cattle. Anim. Genet. 2013, 44, 446–449. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, K.; Olsson, M.; van Tol, H.T.; Mikko, S.; Vlamings, B.H.; Andersson, G.; Rodriguez-Martinez, H.; Purwantara, B.; Paling, R.W.; Colenbrander, B.; et al. On the origin of Indonesian cattle. PLoS One 2009, 4, e5490. [Google Scholar] [CrossRef]
- Cullen, H.M.; deMenocal, P.B.; Hemming, S.; Hemming, G.; Brown, F.H.; Guilderson, T.; Sirocko, F. Climate change and the collapse of the Akkadian empire: Evidence from the deep sea. Geology 2000, 28, 379–382. [Google Scholar] [CrossRef]
- Edwards, C.J.; Baird, J.F.; MacHugh, D.E. Taurine and zebu admixture in Near Eastern cattle: A comparison of mitochondrial, autosomal and Y-chromosomal data. Anim. Genet. 2007, 38, 520–524. [Google Scholar] [CrossRef] [PubMed]
- Pieragostini, E.; Scaloni, A.; Rullo, R.; di Luccia, A. Identical marker alleles in Podolic cattle (Bos taurus) and Indian zebu (Bos indicus). Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2000, 127, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hanotte, O.; Bradley, D.G.; Ochieng, J.W.; Verjee, Y.; Hill, E.W.; Rege, J.E.O. African pastoralism: Genetic imprints of origins and migrations. Science 2002, 296, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Brandt, S.A.; Carder, N. Pastoral rock art in the horn of Africa: Making sense of udder chaos. World Archaeol. 1987, 19, 195–213. [Google Scholar] [CrossRef]
- Bradley, D.G.; Loftus, R.T.; Cunningham, C.; MacHugh, D.E. Genetics and domestic cattle origin. Evol. Anthopol. 1998, 6, 79–86. [Google Scholar] [CrossRef]
- Epstein, H.; Mason, I.L. Cattle. In Evolution of Domesticated Animals; Mason, I.L., Ed.; Longman Group: Essex, UK, 1984. [Google Scholar]
- Clason, A.T. Huisdieren in voor- en vroeghistorische tijden [Domestic animal in prehistory and early history]. Boerderij 1977, 61, 12–17. [Google Scholar]
- Clason, A.T. What’s new in the Bronze Age? In Discussion with the Past; Van Es, W.A., Ed.; Foundation for Promoting Archaeology: Zwolle, The Netherlands, 1999; pp. 35–40. [Google Scholar]
- Zeuner, F.E. A History of Domesticated Animals; Harper & Row: New York, NY, USA, 1963. [Google Scholar]
- Kron, G. Roman livestock farming in southern Italy: The case against environmental determinism. In Espaces Integrés et Ressources Naturelles dans L’Empire Romain; Clavel-Lévêque, M., Hermon, E., Eds.; Presses Universitaires de Franche-Comté: Besançon, France, 2004; pp. 119–134. [Google Scholar]
- Tacitus. In Germania IV; 98 AD.
- Aristoteles. In Historia Animalium; ca. 350 BC.
- Kron, G. Archaeozoological evidence for the productivity of Roman livestock farming. Münstersche Beiträge zur Antiken Handelsgeschichte 2002, 21, 53–73. [Google Scholar]
- Archimedes. The Cattle Problem in English Verse by S.J.P. Hillion & H.W. Lenstra Jr.; Mercator: Santpoort, The Netherlands, 1999. [Google Scholar]
- Fussel, G.E. The Classical Tradition in West European Farming; Newton Abbot: Devon, UK, 1972. [Google Scholar]
- MacKinnon, M. Cattle “breed” variation and improvement in Roman Italy: Connecting the zooarchaeological and ancient textual evidence. World Archaeol. 2010, 42, 55–73. [Google Scholar] [CrossRef]
- Engeler, W. Das Schweizerische Braunvieh; Huber & Co: Frauenfeld, Germany, 1947. [Google Scholar]
- Jewell, P.A. Changes in size and type of cattle from prehistoric to mediaeval times in Britain. J. Anim. Breed. Genet. 1962, 77, 159–167. [Google Scholar]
- Armitage, P.L. Developments in British cattle husbandry. Ark 1982, 9, 50–54. [Google Scholar]
- Newfield, T.P. A great Carolingian panzootic: The probable extent, diagnosis and impact of an early ninth-century cattle pestilence. Argos 2012, 46, 200–210. [Google Scholar]
- Davis, S.J.M. The Archaeology of Animals; Batsford: London, UK, 1987. [Google Scholar]
- Lenstra, J.A.; Felius, M. Genetic aspects of domestication. In The Genetics of Cattle, 2nd ed.; Garrick, D.J., Ruvinsky, A, Eds.; CABI: Wallingford, UK, 2014. [Google Scholar]
- Kompan, D.; Cividini, A. (Eds.) Brachycerous Cattle in Balkan Countries and Their Perspectives; Biotechnical Faculty: Domzale, Slovenia, 2008.
- Mane, P. Fort comme un boeuf. L’image des bovins dans l’agriculture médiévale. In Aurochs, le Retour. Aurochs, Vaches & Autres Bovins de la Préhistoire à Nos Jours; Center Jurassien du Patrimoine: Lons-le-Saunier, France, 1994; pp. 166–177. [Google Scholar]
- Hemming, J. Bos primigenius in Britain: Or, why do fairy cows have red ears? Folklore 2002, 113, 71–82. [Google Scholar] [CrossRef]
- Clason, A.T. Zeldzame Huisdierrassen; Thieme: Zutphen, The Netherlands, 1980. [Google Scholar]
- White, L., Jr. Medieval Technology and Social Change; Oxford University Press: Oxford, UK, 1964. [Google Scholar]
- Langdon, J. Horses, Oxen and Technological Innovation: The Use of Draught Animals in English Farming from 1066–1500; Cambridge University Press: Cambridge, UK, 1986. [Google Scholar]
- Astill, G.; Langdon, J. Medieval Farming and Technology: The Impact of Agricultural Change in Northwest Europe; Brill: Leiden, The Netherlands, 1997. [Google Scholar]
- Armitage, P.L. A preliminary description of British cattle from the late twelfth to the early sixteenth century. Ark 1980, 7, 405–413. [Google Scholar]
- Aberth, J. An Environmental History of the Middle Ages: The Crucible of Nature; Taylor and Francis Books: Oxon, UK, 2013. [Google Scholar]
- Pickl, O. Der Viehandel von Ungarn nach Oberitalien vom 14. bis zum 17 Jahrhundert. In Internationaler Ochsenhandel (1350–1750). Beiträge zur Wirtshaftsgeschichte Band 9; Westermann, E., Ed.; Klett-Cotta: Stuttgart, Germany, 1979; pp. 125–136. [Google Scholar]
- Westermann, E. Internationaler Ochsenhandel (1350–1750). In Beiträge zur Wirtshaftsgeschichte Band 9; Klett-Cotta: Stuttgart, Germany, 1979. [Google Scholar]
- Hughes, P.G. Wales and the Drovers; Golden Grove Book Company: Barry, UK, 1988. [Google Scholar]
- Petersen, L. The Danish cattle trade during the sixteenth and seventeenth centuries. Scand. Econ. Hist. Rev. 1970, 18, 69–85. [Google Scholar] [CrossRef]
- Gijsbers, W. Kapitale Ossen. In De Internationale Handel in Slachtvee in Noordwest-Europa (1300–1750); Verloren: Hilversum, The Netherlands, 1999. [Google Scholar]
- Wenger, H. Glück Im Stall; Buchverlag Verbandsdruckerei: Bern, Switzerland, 1954. [Google Scholar]
- Bodó, I.; Gera, I.; Koppány, G. The Hungarian Grey Cattle Breed, 2nd ed.; Association of the Hungarian Grey Cattle Breeders: Budapest, Hungary, 2004. [Google Scholar]
- Bartosiewicz, L. Hungarian Grey cattle: A traditional European breed. Anim. Genet. Resour. 1997, 21, 49–60. [Google Scholar] [CrossRef]
- Engel, P. The Realm of St Stephen: A History of Medieval Hungary, 895–1526; I.B.Tauris: London, UK; New York, NY, USA, 2001. [Google Scholar]
- Appuhn, K. Ecologies of Beef: Eighteenth-Century Epizootics and the Environmental History of Early Modern Europe. Environ. Hist. 2010, 15, 268–287. [Google Scholar] [CrossRef]
- Ciani, F.; Matassino, D. Il bovino grigio allevato in Italia: Origine ed evoluzione.Nota 2: Il bovino macrocero [Grey cattle reared in Italy: Origin and evolution. Note 1: Long-horned cattle]. Taurus Speciale 2001, 13, 89–99. [Google Scholar]
- Bishko, C.J. The peninsular background of Latin American cattle ranching. Hisp. Am. Hist. Rev. 1953, 32, 491–515. [Google Scholar] [CrossRef]
- Rouse, J.E. The Criollo, Spanish Cattle in the Americas; University of Oklahoma Press: Norman, France, 1977. [Google Scholar]
- Bieleman, J. Techniek in Nederland in De Twintigste Eeuw III [Technique in the Netherlands in the Twentieth Century III]; Walburg Pers: Zutphen, The Netherlands, 2000. [Google Scholar]
- Felius, M. Schoone Koeien [Beautiful cows]. In Vorstelijk Vee [Lordly Livestock]; Erkelens, W., Frankenhuis, M., Zandering, R., Eds.; Thoth: Bussum, The Netherlands, 2002; pp. 18–35. [Google Scholar]
- Etienne, C.; Liébault, J. L’Agriculture et Maison Rustique; 1529. [Google Scholar]
- De Serres, O. Le Theatre D’agriculture et Mesnage. Des Champs; J. Mettayer: Paris, France, 1600. [Google Scholar]
- Etienne, C.; Stevens, K.; Libout, J. De Velt.-Bouw of De Landtwinninghe; Michiel Colijn: Amsterdam, The Netherlands, 1622. [Google Scholar]
- Rozier, A. Le Cour Complet d’Agriculture; Rue et Hotel Serpente: Paris, France, 1782. [Google Scholar]
- De Francourt, M.; Spindler, F.; Quemene, Y. Élements d’Histoire des Races Rovines et Ovines en France; Société d’Ethnozootechnique: Clermont-Ferrand, France, 2002. [Google Scholar]
- Markham, G. Cheap and Good Husbandry; London, UK, 1614. [Google Scholar]
- Russell, N. Like Engend’Ring Like; Cambrige University Press: Cambridge, UK, 1986. [Google Scholar]
- Westermann, E. (Ed.) Internationaler Ochsenhandel 1350–1710; Klett-Cotta: Stuttgart, Germany, 1979.
- Toulson, S. The Drovers; Shire Publications: Bucks, UK, 1980. [Google Scholar]
- Haldane, A.R.B. The Drove Roads of Scotland; Nelson: London, UK, 1952. [Google Scholar]
- Carter, F.W. Trade and Urban Development in Poland: An Economic Geography of Cracow, from its Origins to 1795; Cambridge University Press: Cambridge, UK, 1994. [Google Scholar]
- Broad, J. Cattle plague in eighteenth-century England. Agric. Hist. Rev. 1983, 32, 104–115. [Google Scholar]
- Hengeveld, G.J. Het Rundvee, Zijne Verschillende Soorten, Rassen En Veredeling [Cattle, Different Types, Breeds and Improvement]; De Erven Loosjes: Haarlem, The Netherlands, 1865. [Google Scholar]
- Koolmees, P.A. Epizootic Diseases in the Netherlands, 1713–2002: Veterinary Science, Agricultural Policy, and Public Response. In Healing the Herds: Disease, Livestock Economies, and the Globalization of Veterinary Medicine; Brown, K., Gilfoyle, D., Eds.; Ohio University Press: Athens, Greece, 2010; pp. 19–41. [Google Scholar]
- Theunissen, B. Darwin and his pigeons. The analogy between artificial and natural selection revisited. J. Hist. Biol. 2012, 45, 179–212. [Google Scholar] [CrossRef] [PubMed]
- Thomas, R. Zooarchaeology, Improvement and the British Agricultural Revolution. Int. J. Hist. Archaeol. 2005, 9, 71–88. [Google Scholar] [CrossRef]
- Stanley, P. Robert Bakewell and the Longhorn Breed of Cattle; Farming Press: Ipswich, UK, 1995. [Google Scholar]
- Hall, S.J.G.; Clutton-Brock, J. Two Hundred Years of British Farm Livestock; British Museum: London, UK, 1989. [Google Scholar]
- Youatt, W. Cattle, Their Breeds, Management and Diseases; Baldwin and Cradock: London, UK, 1834. [Google Scholar]
- Gade, D.W. Cattle. In The Cambridge World History of Food; Kiple, K.F., Ed.; Cambridge University Press: Cambridge, UK, 2012; pp. 489–496. [Google Scholar]
- Bewick, T. A General History of Quadrupeds; Hodgson, Beilby and Bewick: Newcastle upon Tyne, UK, 1790. [Google Scholar]
- Concours Régionaux d’Animaux Reproducteurs Tenus a Besançon, Grenoble, Périgeues, Rennes, Arra, Bourges, Clermont et Rouen et Concours Universel de Paris en 1855; Paris, France, 1856.
- Gunn, W.D. Cattle of Southern India; Volume III, Bulletin No. 60; Department of Agriculture: Madras, India, 1909. [Google Scholar]
- Porter, V. (Ed.) A Mason’s World Dictionary of Livestock Breeds, Types and Varieties, 5th ed.; CABI Publishing: Oxon, UK, 2002.
- Intergovernmental Technical Working Group on Animal Genetic Resources, Eight Session, Rome, 26–28 November 2014. Available online: dad.fao.org (accessed on 6 November 2014).
- Béranger, C.; Vissac, B. An holistic approach to livestock farming systems: Theoretical and methodological aspects. In The Study of Livestock Farming Systems in a Research and Development Framework. EAAP Publication 63; Gibon, A., Flamant, J.C., Eds.; Wageningen University Press: Wageningen, The Netherlands, 1994; pp. 5–17. [Google Scholar]
- Landwirtschaftliches Tieralbum; Paul Paray: Berlin, Germany, 1899.
- United States Consular Reports Cattle and Dairy Farming Part I; House Executive Document, No. 51; Government Printing Office: Washington, DC, USA, 1888.
- Haring, F.; Hammond, J.; Johannson, I. Handbuch Der Tierzüchtung; Paul Parey: Hamburg/Berlin, Germany, 1961. [Google Scholar]
- Averdunk, G.; Krogmeier, D. Minor and dual-purpose Bos taurus breeds. In Encyclopedia of Dairy Sciences, 2nd ed.; Fuquay, J.W., Ed.; Elsevier: Oxford, UK, 2011; pp. 293–299. [Google Scholar]
- Decker, J.E.; Pires, J.C.; Conant, G.C.; McKay, S.D.; Heaton, M.P.; Chen, K.; Cooper, A.; Vilkki, J.; Seabury, C.M.; Caetano, A.R.; et al. Resolving the evolution of extant and extinct ruminants with high-throughput phylogenomics. Proc. Natl. Acad. Sci. USA 2009, 106, 18644–18649. [Google Scholar] [CrossRef] [PubMed]
- Medugorac, I.; Medugorac, A.; Russ, I.; Veit-Kensch, C.E.; Taberlet, P.; Luntz, B.; Mix, H.M.; Forster, M. Genetic diversity of European cattle breeds highlights the conservation value of traditional unselected breeds with high effective population size. Mol. Ecol. 2009, 18, 3394–3410. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, O.; Akin, O.; Metin Yener, S.; Ertugrul, M.; Wilson, R.T. The domestic livestock resources of Turkey: Cattle local breeds and types and their conservation status. Anim. Genet. Resour. 2012, 50, 65–73. [Google Scholar] [CrossRef]
- Israel has a worldwide reputation for agricultural accomplishments. Available online: http://www.israeldairy.com (accessed on 6 November 2014).
- Van Engelen, A.; Livestock, Livelihood and Rural Development Freelance Consultancy, Almaty, Kazakhstan. Personal communication, 2013.
- Granberg, L.; Kantanen, J.; Soini, K. Sakha Ynaga Cattle of the Yakuts; Finnish Academy of Science and Letters: Helsinki, Finland, 2009. [Google Scholar]
- Matsuura, T. The Livestock Farmers of Japan; Japan Intermational Agriculture Council: Tokyo, Japan, 1984. [Google Scholar]
- Uesaka, S. Beef Cattle in Japan; Kyoto University: Kyoto, Japan, 1970. [Google Scholar]
- Premium Wagyu Beef from Kobe Beef Stor e.com! Available online: http://www.kobebeefstore.org (accessed on 6 November 2014).
- Shahai, R.; Vijh, R.K. Domestic Animal Diversity; SI Publications: Karnal, India, 2000. [Google Scholar]
- Wallace, R. India in 1887; Oliver and Boyd: Edinburgh, UK, 1888. [Google Scholar]
- Vivekanandan, P. Breed Keepers and Best Practices. Linking Breeds, Natural Resources, Indigenous Knowledge and Institutions; SEVA: Madurai, India, 2012. [Google Scholar]
- Saleem, M.; Rahim, I.; Jalali, S.; Rueff, H.; Khan, M.; Maselli, D.; Wiesman, U.; Muhammad, S. Morphological characterization of Achai cattle in sedentary and transhumant systems in Pakistan. Anim. Genet. Resour. 2013, 52, 83–90. [Google Scholar] [CrossRef]
- Iype, S. Vechur cattle—From extinction to sustainability. Anim. Genet. Resour. 2013, 52, 105–110. [Google Scholar] [CrossRef]
- Mittal, J.P.; Prasad, S.; Phillips, C.J.C. Rathi—A new breed of cattle from the Indian desert. New Techn. Cattle Prod. 1989, 2, 244–246. [Google Scholar]
- Farooq, U.; Samad, H.A.; Sher, F.; Asim, M.; Khan, M.A. Cholistan and Cholistani breed of cattle. Pak. Vet. J. 2010, 30, 126–130. [Google Scholar]
- Sharma, R.; Maitra, A.; Pandey, A.K.; Mishra, B.P. Differentiation of four Indian aboriginal cattle populations revealed by STR markers. Genetika 2012, 48, 719–725. [Google Scholar] [PubMed]
- Sharma, R.; Maitra, A.; Singh, P.K.; Tantia, M.S. Genetic diversity and relationship of cattle populations of East India: Distinguishing lesser known cattle populations and established breeds based on STR markers. Springerplus 2013. [Google Scholar] [CrossRef] [Green Version]
- Cockrill, W.R. The water buffalo: A review. Br. Vet. J. 1981, 137, 8–17. [Google Scholar] [PubMed]
- Mondal, M.; Baruah, K.K.; Rajkhowa, C. Mithun: An Animal of Indian Pride. Livest. Res. Rural Dev. 2014, 26. article #6. [Google Scholar]
- Gou, X.; Wang, Y.; Yang, S.; Deng, W.; Mao, H. Genetic diversity and origin of Gayal and cattle in Yunnan revealed by mtDNA control region and SRY gene sequence variation. J. Anim. Breed. Genet. 2010, 127, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Doutresoulle, G. L’Élevage en Afrique Occidentale Française; Éditions Larose: Paris, France, 1947. [Google Scholar]
- Barré, N.; Garris, G.; Camus, E. Propagation of the tick Amblyomma variegatum in the Carribean. Rev. Sci. Tech. Off. Int. Epizoot. 1995, 14, 841–855. [Google Scholar]
- The Eradication of Rinderpest from Africa: A Great Milestone; African Union Interafrican Bureau for Animal Resources: Nairobi, Kenya, 2011.
- Rowe, J.; Hødnebø, K. Rinderpest in the Sudan. 1988–1980 The Mystery of the Missing Panzootic. Sudan. Afr. 1994, 5, 149–178. [Google Scholar]
- Spinage, C.A. Cattle Plague: A History; Kluwer Academic: New York, NY, USA, 2003. [Google Scholar]
- Hanotte, O.; Tawah, C.L.; Bradley, D.G.; Okomo, M.; Verjee, Y.; Ochieng, J.; Rege, J.E. Geographic distribution and frequency of a taurine Bos taurus and an indicine Bos indicus Y specific allele amongst sub-saharan African cattle breeds. Mol. Ecol. 2000, 9, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Flori, L.; Gonzatti, M.I.; Thevenon, S.; Chantal, I.; Pinto, J.; Berthier, D.; Aso, P.M.; Gautier, M. A quasi-exclusive European ancestry in the Senepol tropical cattle breed highlights the importance of the slick locus in tropical adaptation. PLoS One 2012, 7, e36133. [Google Scholar] [CrossRef]
- Ginja, C.; Telo Da Gama, L.; Penedo, M.C. Analysis of STR markers reveals high genetic structure in Portuguese native cattle. J. Hered. 2010, 101, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Ginja, C.; Penedo, M.C.; Melucci, L.; Quiroz, J.; Martinez Lopez, O.R.; Revidatti, M.A.; Martinez-Martinez, A.; Delgado, J.V.; Gama, L.T. Origins and genetic diversity of New World Creole cattle: Inferences from mitochondrial and Y chromosome polymorphisms. Anim. Genet. 2010, 41, 128–141. [Google Scholar] [CrossRef] [PubMed]
- Rouse, J.E. World Cattle III Cattle of North America; University of Oklahoma Press: Norman, France, 1973. [Google Scholar]
- Bowling, G.A. The Introduction of Cattle into Colonial North America. J. Dairy Sci. 1942, 25, 129–154. [Google Scholar] [CrossRef]
- Dechambre, P. Les Bovins; Ch Amat Éditeur: Paris, France, 1913; p. 26. [Google Scholar]
- Warwick, E.J. Fifty years of progress in breeding beef cattle. J. Anim. Sci. 1958, 17, 922–943. [Google Scholar]
- Borges Lopes, M.A.; Marquez de Rezende, E.M. ABCZ História E Histórias; Câmara Brasileira do Livro: Sao Paulo, Brazil, 2001. [Google Scholar]
- Dos Santos, R. Os Cruzamentos Na Pecuária. Tropical Crossing in Tropical Livestock; Editora Agropecuária Tropical Ltda: Uberaba, Brazil, 1999. [Google Scholar]
- Martinez, A.M.; Gama, L.T.; Canon, J.; Ginja, C.; Delgado, J.V.; Dunner, S.; Landi, V.; Martin-Burriel, I.; Penedo, M.C.; Rodellar, C.; et al. Genetic footprints of Iberian cattle in America 500 years after the arrival of Columbus. PLoS One 2012, 7, e49066. [Google Scholar] [PubMed]
- Freyer, G.; Konig, S.; Fischer, B.; Bergfeld, U.; Cassell, B.G. Invited review: Crossbreeding in dairy cattle from a German perspective of the past and today. J. Dairy Sci. 2008, 91, 3725–3743. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, M.K.; Norberg, E.; Pedersen, J.; Christensen, L.G. Invited review: Crossbreeding in dairy cattle: A Danish perspective. J. Dairy Sci. 2008, 91, 4116–4128. [Google Scholar] [CrossRef] [PubMed]
- Field, T.G.; Taylor, R.E. Scientific Farm Animal Production, 9th ed.; MacMillan: London, UK, 2007. [Google Scholar]
- Handbook of Australian Livestock; Australian Meat & Livestock Corporation: North Sydney, Australia, 1984.
- Trotter, M.; McCullock, B. Rare Breeds of Heritage Livestock in New Zealand; David Bateman: Albany, Auckland, New Zealand, 2010. [Google Scholar]
- Buchanan, D.S.; Lenstra, J.A. Breeds of cattle. In The Genetics of Cattle, 2nd ed.; Garrick, D.J., Ruvinsky, A., Eds.; CABI: Wallingford, UK, 2014. [Google Scholar]
- Hansen, P.J. Current and future assisted reproductive technologies for mammalian farm animals. Adv. Exp. Med. Biol. 2014, 752, 1–22. [Google Scholar] [PubMed]
- Cundiff, L.V.; Gregory, K.E.; Koch, R.M.; Dickerson, G.E. Genetic Diversity among Cattle Breeds and its Use to Increase Beef Production Efficiency in a Temperate Environment. In 3rd World Congress on Genetics Applied to Livestock Production; University of Nebraska: Lincoln, NE, USA, 1986; pp. 271–282. [Google Scholar]
- Cundiff, L.V. Beef cattle: Breeds and Genetics. In Bell Encyclopedia of Animal Science; Pond, W.G., Bell, A.W., Eds.; CRC Press: Boca Raton, FL, USA, 2004. [Google Scholar]
- Stock, K.F.; Reents, R. Genomic selection: Status in different species and challenges for breeding. Reprod. Domest. Anim. 2013, 48, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Hayes, B.J.; Lewin, H.A.; Goddard, M.E. The future of livestock breeding: Genomic selection for efficiency, reduced emissions intensity, and adaptation. Trends Genet. 2013, 29, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Taberlet, P.; Valentini, A.; Rezaei, H.R.; Naderi, S.; Pompanon, F.; Negrini, R.; Ajmone-Marsan, P. Are cattle, sheep, and goats endangered species? Mol. Ecol. 2008, 17, 275–284. [Google Scholar]
- FAO. Molecular Genetic Characterization of Animal Genetic Resources. In FAO Animal Production and Health Guidlines No. 9; FAO: Rome, Italy, 2011. [Google Scholar]
- Sahiwal Cow (Zaibi) World Record Holder in Sahiwal Breed. Available online: http://www.rccsc.com.pk (accessed on 6 November 2014).
- Roschinsky, R.; Kluszczynska, M.; Sölkner, J.; Puskur, R.; Wurzinger, M. Smallholder experiences with dairy cattle crossbreeding in the tropics: from introduction to impact. Animal 2014, in press. [Google Scholar]
- FAO. The State of the World’s Animal Genetic Resources for Food and Agriculture; FAO: Rome, Italy, 2007. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org (accessed on 11 November 2014).
- Quittet, E.; Denis, B. Races Bovines Françaises; La Maison Rustique: Paris, France, 1963. [Google Scholar]
- Denis, B.; Avon, L. Races Bovines, Histoire, Aptitudes, Situation Actuelle; Éditions Castor & Pollux: Chaumont, France, 2010; p. 324. [Google Scholar]
- Sanchez Belda, A. Razas Bovinas Españoles; Minosterio de Agricultura Pesca y Alimentación: Madrid, Spain, 1984. [Google Scholar]
- Fernández Rodriquez, M.; Gómez Fernández, M.; Delgado Bermejo, J.V.; Adán Belmonte, S.; Jiménez Cabras, M. Cuiá de Campo de las Razas Autóctonas Españolas; Ministerio de Medio Ambiente y Medio Rural y Marino: Madrid, Spain, 2010. [Google Scholar]
- French, M.H.; Johansson, I.; Joshi, N.R.; McLaughlin, R.A. European Breeds of Cattle; FAO: Rome, Italy, 1966. [Google Scholar]
- SAVE. Rare Breeds and Varieties of Greece; SAVE: Konstanz, Germany, 2010. [Google Scholar]
- Rege, J.E.O.; Tawah, C.L. The state of African cattle genetic resources II, Geographical distribution, characteristics and uses of present-day breeds and strains. Anim. Genet. Resour. Inf. 1999, 26, 1–25. [Google Scholar] [CrossRef]
- Wuletaw, Z. Survey of Indigenous Cattle Genetic Resources, Husbandry Practices and Cattle Breeding Objectives in North Gondar, Ethiopia. Master’s Thesis, Alemaya University, Dire Dawa, Ethiopia, 2004. [Google Scholar]
- Zerabruk, M.; Vangen, O. The Abergelle and Irob cattle breeds of North Ethiopia: Description and on-farm characterization. Anim. Genet. Resour. 2005, 36, 7–20. [Google Scholar]
- Tadesse, D. On Farm Phenotypic Characterization of Cattle Genetic Resources and their Production Systems in South and North Wollo Zones of Amhara Region North Eastern Ethiopia. Master’s Thesis, Alemaya University, Dire Dawa, Ethiopia, 2005. [Google Scholar]
- Getachew, F. On-Farm Characterization of Cattle Genetic Resources and their Production Systems in Awi, East and West Gojjam Zones of Amhara Region, Ethiopia. Master’s Thesis, Alemaya University, Dire Dawa, Ethiopia, 2006. [Google Scholar]
- Chebo, C.; Ayalew, W.; Wuletaw, Z. On-farm phenotypical characterization of indigenous cattle populations of Gamo Gaffa zone, Southern Ethiopia. Anim. Genet. Resour. 2013, 52, 71–82. [Google Scholar] [CrossRef]
- Mwambene, P.L.; Katule, A.M.; Chenyambuga, S.W.; Plante, Y.; Mwakilembe, P.A.A. Fipa cattle in the southwestern highlands of Tanzania: Molecular characterization. Anim. Genet. Resour. 2012, 51, 31–43. [Google Scholar] [CrossRef]
- Sponenberg, D.P. Florida Cracker and Pineywoods: Unique American Cattle. ALBC News 1993, 10, 1–5. [Google Scholar]
- Aguirre Riofrio, L.; Apoloa, G.; Chalcoa, L.; Martínez, A. Caracterización genética de la población bovina criolla de la Región Sur del Ecuador y su relación genética con otras razas bovinas. Anim. Genet. Resour. Inf. 2014, 54, 93–101. [Google Scholar] [CrossRef]
- Hyndham, D.L.; Littlejohn, R.P.; Williams, J.L.; Crawford, A.M. Enderby Island cattle: What breed are they derived from? Proc. Assoc. Adv. Anim. Breed. Genet. 2001, 14, 329–331. [Google Scholar]
- Christman, C.; American Livestock Breeds Conservancy, Pittsboro, NC, USA. 2005; Unpublished work.
- USDA. National Animal Germplasm Program (NAGP). Available online: http://nrrc.ars.usda.gov/A-GRIN/main_webpage/ars?record_source=US (accessed on 27 October 2014).
- Cryoconservation of cattle breeds in The Netherlands. Available online: http://edepot.wur.nl/5602 (accessed on 27 October 2014).
- Groeneveld, L.F.; Lenstra, J.A.; Eding, H.; Toro, M.A.; Scherf, B.; Pilling, D.; Negrini, R.; Finlay, E.K.; Jianlin, H.; Groeneveld, E.; et al. Genetic diversity in farm animals: A review. Anim. Genet. 2010, 41, 6–31. [Google Scholar] [CrossRef] [PubMed]
- Daetwyler, H.D.; Capitan, A.; Pausch, H.; Stothard, P.; van Binsbergen, R.; Brondum, R.F.; Liao, X.; Djari, A.; Rodriguez, S.C.; Grohs, C.; et al. Whole-genome sequencing of 234 bulls facilitates mapping of monogenic and complex traits in cattle. Nat. Genet. 2014, 46, 858–865. [Google Scholar] [CrossRef] [PubMed]
- Larson, G.; Burger, J. A population genetics view of animal domestication. Trends Genet. 2013, 29, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Kaupe, B.; Winter, A.; Fries, R.; Erhardt, G. DGAT1 polymorphism in Bos indicus and Bos taurus cattle breeds. J. Dairy Res. 2004, 71, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Medugorac, I.; Seichter, D.; Graf, A.; Russ, I.; Blum, H.; Gopel, K.H.; Rothammer, S.; Forster, M.; Krebs, S. Bovine polledness—An autosomal dominant trait with allelic heterogeneity. PLoS One 2012, 7, e39477. [Google Scholar] [CrossRef] [PubMed]
- O’Rourke, B.A.; Greenwood, P.L.; Arthur, P.F.; Goddard, M.E. Inferring the recent ancestry of myostatin alleles affecting muscle mass in cattle. Anim. Genet. 2013, 44, 86–90. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, A.; Pruvost, M.; Reissmann, M.; Benecke, N.; Brockmann, G.A.; Castanos, P.; Cieslak, M.; Lippold, S.; Llorente, L.; Malaspinas, A.S.; et al. Coat color variation at the beginning of horse domestication. Science 2009. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Felius, M.; Beerling, M.-L.; Buchanan, D.S.; Theunissen, B.; Koolmees, P.A.; Lenstra, J.A. On the History of Cattle Genetic Resources. Diversity 2014, 6, 705-750. https://doi.org/10.3390/d6040705
Felius M, Beerling M-L, Buchanan DS, Theunissen B, Koolmees PA, Lenstra JA. On the History of Cattle Genetic Resources. Diversity. 2014; 6(4):705-750. https://doi.org/10.3390/d6040705
Chicago/Turabian StyleFelius, Marleen, Marie-Louise Beerling, David S. Buchanan, Bert Theunissen, Peter A. Koolmees, and Johannes A. Lenstra. 2014. "On the History of Cattle Genetic Resources" Diversity 6, no. 4: 705-750. https://doi.org/10.3390/d6040705
APA StyleFelius, M., Beerling, M.-L., Buchanan, D. S., Theunissen, B., Koolmees, P. A., & Lenstra, J. A. (2014). On the History of Cattle Genetic Resources. Diversity, 6(4), 705-750. https://doi.org/10.3390/d6040705