Raiding the Coral Nurseries?
Abstract
: A recent shift in the pattern of commercial harvest in the Keppel Island region of the southern inshore Great Barrier Reef raises concern about the depletion of a number of relatively rare restricted range taxa. The shift appears to be driven by demand from the United States (US) for corals for domestic aquaria. Data from the annual status reports from the Queensland Coral Fishery were compared with export trade data to the US from the Convention on International Trade in Endangered Species (CITES). Evidence was found of recent increases in the harvest of species from the Mussidae family (Acanthastrea spp.) which appears to be largely driven by demand from the US. On present trends, the industry runs the risk of localized depletion of Blastomussa and Scolymia; evidenced by an increase in the harvest of small specimens and the trend of decreasing harvest despite a concurrent increase in demand. Considering their relatively high sediment tolerance compared to other reef-building species, and the current lack of information about their functional role in reef stability, the trend raises concerns about the impact of the harvest on local coral communities. The recent shift in harvest patterns could have impacts on slow-growing species by allowing harvest beyond the rate of population regeneration. In light of these factors, combined with the value of such species to local tourism, a commercial coral fishery based on uncommon but highly sought-after species may not be ecologically sustainable or economically viable in the Keppels.1. Introduction
Declining coral diversity on many of the world's coral reefs [1] and, in some cases, the regional depletion or extinctions of species has meant that many scleractinian corals are now listed as “critically endangered” (6 spp.), “endangered” (23 spp.) or “vulnerable” (199 spp.) by the International Union for Conservation of Nature [2]. Declining species diversity has been attributed to causes such as bleaching events [3-7], invasive species (e.g., Acantaster planci) [8-10], coral collecting [11-13] and land run-off [14] whereby regeneration following disturbance does not necessarily return the original species composition [15]. Changed species composition can then have implications for the rate of recovery from disturbance [16]. Trends of declining species diversity are expected to continue for the foreseeable future [17], shedding a new light on the growing world trade in reef corals for domestic aquaria.
The world marine aquarium trade is growing, driven mainly by demand for live specialty corals for domestic aquaria supplied primarily by stock collected from Indo-Pacific reefs. The trade has shifted over the past decade from supplying mainly fish, to supplying a wide range of invertebrates such as corals, mollusks and crustaceans. The number of specimens of live specialty corals being traded has increased from 10% of the total world coral trade in 1985 to 90% in 1997 [18,19]. In spite of this increase, the overall weight of traded live corals is now a quarter of the ∼4000 t traded in 1990. This is thought to have occurred because aquarists now covet smaller and lighter specimens of vibrantly colored stony corals with large polyps (“LPS”, e.g., Euphyllia, Acanthastrea, Scolymia, Goniopora, Catalaphyllia, Trachyphyllia and Heliofungia spp.) in contrast to the heavier Pocillopora, Porites, and Acropora specimens. Since 1990, all scleractinian corals have been declared “vulnerable to exploitation” by the Convention on the International Trade in Endangered Species (CITES). CITES is an agreement between signatory countries to implement specific controls on the trade of listed species. Because of sustainability concerns, collection and export of corals has now been banned by several signatories including the Philippines, Florida, Bali, Guam, Samoa, Puetro Rico and Hawaii. The United States (US) is also now considering banning the import of several species [20]. In spite of trade restrictions, the global increase in demand for specific corals appears to have arisen without adequate knowledge of critical aspects of the biology of target species including growth, reproduction or potential for regeneration. This situation has raised concern for the impact of the industry on reefs [21].
A commercial coral harvest industry has operated on the reefs along the Queensland coast in Australia since the 1980s. In 1983, the Queensland Coral Fishery (QCF) comprised 12 active collectors; two of which collected 65% of the total annual catch of 45 t from just four coral families. Seventy percent of the total catch comprised the ornamental P. damicornis [22]. By 2006, the fishery had grown to 59 collectors, sourcing 50% of their total annual catch of 38 t from 36 coral families of which 52 of these are now CITES-listed [23]. Past assessment of the fishery as ecologically sustainable was based on the historical focus of the industry on fast-growing acroporid and pocilloporid corals. Collection of 50 t y−1 was justified on the basis that the accretion at One Tree Island in the southern GBR was estimated to be ∼1875 t CaCO3 y−1 [24]. However, it is not logical to compare Holocene rates of growth with current rates as these changed over timescales of thousands of years. Harriott [25] also argues that on marginal, high latitude reefs where collection is typically focused, net reef accretion is low because carbonate production is matched by bioerosion losses. Even so, this method of determining sustainability may still be flawed because of the shift from fast-growing to slow-growing taxa and the current rate of decline of calcification as a result of atmospheric CO2 [26]. Moreover, such sustainability assessments merely inform managers about the total catch of corals whereas the industry now focuses on collecting specific taxa . In an attempt to address these issues, a risk-based regulatory framework has recently been introduced [27]. The arrangements specify management review reference points or “triggers”, based on past fisheries data and vulnerability and risk assessments for each taxa [28,29]. The methodology for assessing vulnerabity was based on that used by Ponder and Grayson [30]. Although this framework is an improvement, the historical data had limited species-specific resolution, many corals are still not reported to species level and the current two-year timeframe between actual catch and management review of the reference points is too long for meaningful intervention to occur. Moreover, most enforcement agents are not trained in the taxonomic identification of stony corals and fisheries data is assessed on reef-wide rather than local or regional scales, which makes species-specific triggers for localized depletion meaningless. Because the QCF is considered of low economic value compared to other marine wild-take industries, it is also left largely to self-regulate. However, self-regulation has a record of failing to protect fish stocks [31,32] and monitoring to ensure ecological sustainability is clearly not feasible considering the recent introduction of “roving” licenses [33].
The Keppels is a system of ∼12 km2 of coastal island fringing reefs near what is generally considered the southern-most extent of strong calcium carbonate accretion on the GBR. Compared to the main coral collection area near Cairns, which has extensive mid- and outer-shelf reefs, the area of reef targeted by collectors in the Keppels is small and isolated from the influence of other reef systems [34]. There are 59 licensed coral collectors in the GBR of which six are currently active in the Keppels. These six active licenses comprise ∼10% of the total of 59 fishery licenses in an area less than ∼1% of the ∼200,000 km2 of GBR reef available to the industry. A recent change from fixed area to roving licenses has meant that monitoring of fishery impacts is virtually impossible as collection is recorded in 36 nm2 grids and the typical geographic distribution of some of the targeted species can be as low as ∼0.003 nm2. The established long-term monitoring program of reefs in the Keppels by the Australian Institute of Marine Science [35] is unlikely to measure collection impacts because it monitors fixed transects at only six reefs. There is thus a risk that the current monitoring will not identify localized depletions of low abundance species.
In this study data from the annual QCF reports and CITES exports were reviewed to investigate evidence for localized depletion of targeted species that might also be related to changing market trends. The study focuses on the Keppels (23.1°S, 150.9°E, Figure 1) on the Eastern coast of Queensland which is close to a major air freight facility in Rockhampton, Australia. The adjacent coastal township of Yeppoon relies heavily on the reefs for tourism and recreation. Because of its location at the mouth of the second largest easterly flowing catchment in Australia, the waters of Keppel Bay are typically turbid and the highly sought-after sediment-tolerant specialist LPS [36,37], such as those from the Mussidae family are more prevalent than in other GBR regions [38]. There is concern that, because of the intense fishing effort in such a small area, there is a risk that recent shifts in harvest trends and current management arrangements may be allowing the collection of some species to exceed population regeneration rates.
2. Experimental Section
Information from the CITES trade database and from the annual status reports of the QCF were used to assess the market trends in the supply from the Keppels. The CITES trade database contains trade records that signatory Parties to the Convention compile annually on the trade of wild flora and fauna, including corals. The database can be a useful tool for assessing trends in trade volumes. The database is managed by the World Conservation Monitoring Centre, Cambridge, United Kingdom, on behalf of the CITES Secretariat [23]. Records pertaining to exports of live, wild harvested corals sold for commercial purposes to the US were extracted for 2006 to 2009. The data are referred to as the “CITES export data”.
Data on the collection of corals in the Keppels were sourced from the CFISH database and Annual Status Reports from the Queensland Coral Fishery (QCF) published by the Queensland's Department of Employment, Economic Development and Innovation [23,39]. The annual “coral” catch (including stony corals, sea anemones, soft corals and gorgonians) from 2006 to 2009 were reviewed to investigate the numbers of individual specimens (comprising whole or fragmented colonies) and total weight of coral taxa collected under the “specialty coral” category for the Keppels. Because of the low number of active harvest licenses in the study area and the competitive nature of the industry, higher resolution temporal or geographic data were not made available. At least for the aquarium fisheries, catch per unit of effort (CPUE) data have not proved to be a reliable measure, because searching time is usually not reported by collectors in logbooks [33]. In addition, a shift in the CPUE data from a more valuable to a less valuable target species might indicate that the population of the preferred species had declined, even if total catch remained high. Market and catch trends are more usefully assessed using taxa-specific catch numbers and weights.
Catch statistics sourced from the QCF for the Keppels were reviewed in the context of exports from Australia to the US which were sourced from CITES. Comparisons of catch and export data focused on four of the reported taxonomic groups for the quota years 2006/07, 2007/08 and 2008/09; Blastomussa, Scolymia, Mussidae and the solitary sea anemone (Heteractis crispa, order Actiniaria). All these are considered at negligible or low risk from QCF harvesting activities by Queensland Fisheries (Tables 1 and 2 in the Supplementary material) [28]. The risk analysis tool used in the Ecological Risk Assessment [29] for the fishery was based on consequence and likelihood assessments according to the AS/NZ Standard. A change of >±30% annual catch compared to the average for the previous two-year period triggers a review of the likely causes, and implications for current management arrangements in light of the sustainability of the fishery [40]. Estimates of annual catch weight of each taxa are derided by multiplying the numbers of specimens in each size category by a conversion factor to derive an estimate of the total catch weight.
3. Results and Discussion
Between 2006 and 2009 there were several significant changes in the collection trends in the Keppels. The harvest of specialty corals increased over the three years prior to 2009 from ∼20, 400 to ∼28, 400 pieces. The proportion of this catch that comprised LPS increased from 68% in 2006/07 to 77% in 2007/08 and 84% in 2008/09 (Figure 2). There was also a concurrent decrease in the CPUE from 122 kg d−1 in 2008 to 62 kg d−1 in 2009 and to 83 kg d−1 in 2010.
In 2009, the number of specimens harvested from approximately half of the 28 reported groups of specialty corals in the Keppels increased by >30% compared to the average for the previous two years and about half the reported groups decreased by >30% (Figure 3A). During the same period there were increases in the numbers of almost all coral taxa exported from Australia to the US (Figure 3B), suggesting that the US markets were driving this trend (Table 3 Supplementary Material).
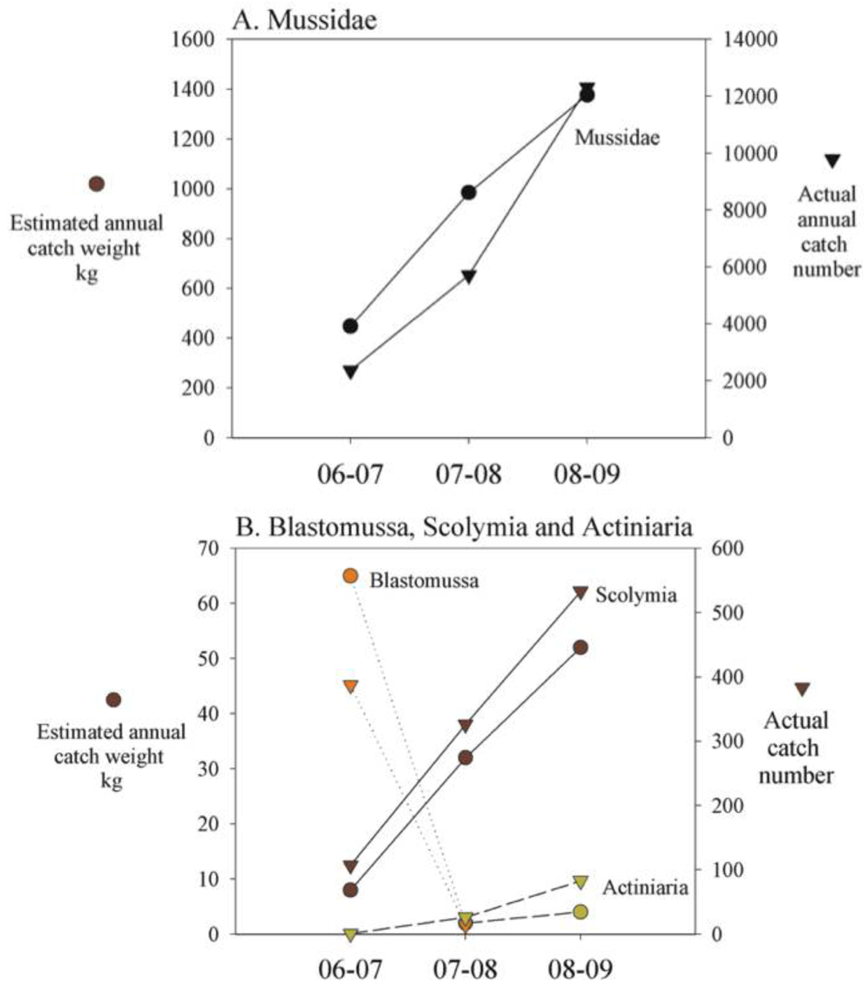
The catch of Mussidae increased by 205% to 12,298 specimens in 2008/09 compared to the average for the previous two-year period (4,036 specimens, Figure 3A). Australian exports to the US also increased 74% (Figure3B). The estimated weight of specimens increased 92% from 720 kg to 1376 kg (Figure 4A). Of the targeted species of Mussidae found in the southern GBR [41] only one (A. echinata Dana 1946) is listed by CITES as of least concern, three as vulnerable (A. bowerbankii Milne Edwards and Haime 1851, A. hemprechii Ehrenberg 1849, A. regularis Veron 2002) and a further three as near threatened (A. lordhowensis Veron & Pichon, 1982 A. hillae Wells 1955, A. rotundaflora Chevalier 1975) [2].
The catch of Scolymia increased by 146% to 533 specimens in 2008/09 compared to the average for the previous two-year period (Figure 3A). Australian exports to the US also increased 187% (Figure 3B). The estimated catch weight of Scolymia increased correspondingly by 160% from 320 kg to 520 kg (Figure 4B). Of the two species of Scolymia found on the Great Barrier Reef, S. australia Milne Edwards and Haime 1849 is listed by CITES as least concern and S. vitiensis Brüggemann, 1877 is listed as near threatened [2].
Between 2006/07 and 2007/08 the catch of Blastomussa decreased from 387 specimens to seven specimens. No specimens were reported as harvested in 2008/09 (Figure 3A). The estimated total catch weight decreased 188% from 65 kg in 2006/07 to 2 kg in 2007/08 (Figure 4B). Australian exports to the US increased 228% (Figure 3A). Of the two species of Blastomussa found in the southern GBR [41], one is listed as near threatened (B. wellsi Wijsman-Best, 1973) and the other is listed as of least concern (B. merleti Wells 1961) [2].
The catch of Actiniaria (H. crispa) more than doubled from 26 specimens in 2007/08 to 83 in 2008/09 (data not shown). The estimated catch weight increased 300% from 2 kg in 2007/08 to 4 kg in 2008/09 (Figure 4B). No export data were available for this species. None of the GBR sea anemones are listed on the IUCN Red list.
Over the past decade, the collection of corals by the QCF from the Keppel Bay Islands has changed from the collection of large specimens of mainly dead corals for ornamental purposes to small pieces of live, vibrantly colored LPS that are popular for domestic aquaria. In 1998, for instance, only 163 kg of Mussidae corals were collected in the Keppels but by 2006/07 this had reached 447 kg and by 2008/09 catch had exceeded 12, 000 pieces weighing more than 1,300 kg [33]. There appears to have also been a concurrent shift in catch to smaller coral pieces with ∼15% more individual 101 g–500 g pieces reported for 2007/08 and a similar trend for 2008/09 [39]. This shift combined with an increase in total catch numbers will have increased local collection impacts. There was also evidence for depletion of specimens from the genus Blastomussa (also Mussidae); whereas imports to the US of these species have increased, very few specimens were collected from 2006/07–2007/08 and no specimens were collected in the quota year 2008/09. It is possible that either long-term over-exploitation or harvest of immature specimens may have already depleted populations of Blastomussa species in this region. While US imports for several other taxa have increased, the numbers of the same taxa harvested from the Keppels decreased between 2006/07 and 2008/09. This could lead to local depletion of rare species; particularly if the trend continues. Of particular concern are species from the genus Scolymia and the sea anemone H. crispa (Actiniaria), which are particularly vulnerable to over-exploitation because they are solitary [42]. Additionally, as collectors target particular colors of LPS, there is concern about how the systematic removal of specimens with particular pigments may affect populations given that little is yet known about the functional significance of different color morphs to stress-tolerance. As all organisms play a functional role that contributes to the reef's capacity to remain in a stable state [43], the removal of even a single species or a subset of the population has the potential to have profound impacts on ecosystem stability and on the persistence of other species [44-46].
To be sustainable, collection of whole colonies or fragments of colonies must not exceed the rate at which the corals can regenerate by recruitment or regrowth. For example, for a growth rate of ∼3.0 cm yr−1 and natural mortality of −0.34 based on P. verrucosa [12,47] sustainable yield per recruit would be ∼72 g yr−1. If collected specimens are getting smaller or less abundant, then sustainable yield must also be reduced. Collectors may either be striving to maintain harvest value in the face of the sustained market demand for a diminishing resource, or they are selectively taking smaller portions of existing colonies to provide a source of replenishment. Without sustainable harvest yields, more of the harvested colonies may also be increasingly less mature, risking future recruitment rates. Alternatively, collectors may be fragmenting large, more mature colonies into smaller fragments to increase their profits (Figure 5). Although LPS have historically been collected as entire colonies [25], the latter would allow colonies to be taken that were previously considered too large for aquaria, potentially removing a previously unharvested source of recruitment to the local population. Either way, a change in the size of specimens would be concerning.
A coral fishery has existed in the Keppels for over 20 years, but little is known about harvest of species other than acroporid and pocilliporid corals prior to 2006. As recently as 2001, annual licenses only allowed collection from 200–400 m designated areas restricted to the reef edges [22]. Since 2006, the introduction of roving licenses, the relaxation of a 6 m depth limit and a shift in market demand to live specialty corals, combined with frequent disturbance may have had adverse impacts on some taxa as catch numbers appear to have become negligible in recent years despite increase in exports. For instance, sea anemones are now considered to be at extremely low abundances in the Keppels. While it is difficult to attribute exact cause, there is some evidence that their low numbers are due to past bleaching and over-exploitation [48,49]. Roving licenses were introduced in 2006 in response to the previous smaller fishery areas being fished out [25]. Their introduction was not favored by the Great Barrier Reef Marine Park Authority (GBRMPA) because of the obvious difficulty in monitoring impacts over wide, undesignated areas [33]. Roving could be considered a sign that fisherman are moving to other sites because of over-exploitation. Prior to their introduction, the industry was dominated by a small number of collectors and collection focused on a single ubiquitous species, P.damicornis, which made for comparatively easy monitoring and management. At that time, sustainability assessments were determined by the potential yield of P damicornis [22,33]. The GBRMPA assessed the damage caused by collecting P.damicornis as “minimal and restricted to small areas on a few reefs”. However, a ban on coral exports from the Philippines resulted in enquiries by large scale US and European importers. There was concern that overseas exports could lead to significant increases in the annual harvest in Australia. It appears that these concerns have been realized in the Keppels in that there has been an increase in the harvest of live specialty corals without knowledge of species-specific sustainable yields.
It is important to note that trends in global market demand for aquarium species may be changing so fast that managers cannot respond effectively. The time-frame for review of annual harvest is nearly two years. Log books are submitted at the end of the quota year but annual reports can take up to a year to complete. If the harvest data reveal that reference points have been triggered, a review of the management arrangements is conducted within three months [50]. Similarly, reviews occur following disturbance such as flood or bleaching. However, most impacts to coral health are immediate and, if severe, are often followed by months or even decades of recovery [51,52]. Surviving corals may be struggling to grow new skeletal material [53] produce lipids and reproduce [54] and have a higher risk of mortality [55] and disease [56]. For already sparse populations, continued harvest could affect the population regeneration before the next disturbance. Many coral species are already suffering from simultaneous stressors associated with anthropogenic activity and even minor harvesting could interact with these stressors and exacerbate negative effects on corals. It is likely that, over time, populations could fail to recover fully between events, particularly if small pockets of corals are virtually self-seeding. The populations of at least three of the targeted taxa, Blastomussa, Scolymia and H.crispa appear to be locally depleted. Regardless of the cause, the risk is that without timely and prolonged restrictions on harvest there could be a risk of further population declines. Since these species are already sparse and occur in small, isolated populations in the first place [35,57] there could be an “Allee” effect on the capacity of such a small population to regenerate [58,59].
Wild harvest fisheries are becoming increasingly more aware of the need to demonstrate sustainability in the context of the conservation and protection of biodiversity. In Australia, the industry recently introduced self-regulation initiatives [60]. For instance, a temporary moratorium on the collection of some species of sea anemones at some sites in the Keppels was implemented following a bleaching event in 2006. However the moratorium has clearly been ineffective in protecting sea anemones in the region because the commercial harvest of the most vulnerable of these species, the solitary H.crispa, increased 500% in the year after the initiation of the moratorium. Moreover, this occurred despite the scarcity of sea anemones in the region, their widely-known susceptibility to environmental stress [61] and over-exploitation [62] and despite two recommendations for closer management attention [48,49]. There also appears to be conflicting information in fishery management vulnerability assessments of sea anemones. For instance, in the 2009 risk assessment [29] the solitary sea anemone, H.crispa, is described as “spread widely throughout the Indo-Pacific” whereas in a 2008 CITES Non-detriment Finding [63] they are described as “relatively uncommon” and found in shallow(5–10 m) water and prone to bleaching”. In spite of this, H.crispa was assigned as only “moderately” susceptible to bleaching impacts and at negligible ecological risk [29] and was excluded from the moratorium. Such self-regulation initiatives may not be appropriate for slow-growing species and run the risk of being perceived as merely designed to improve the public perception of the industry. The current initiatives largely involve leaving bleached or damaged specimens alone following a disturbance but collecting only surviving healthy specimens which does not fit well with the principles of reef regeneration by seeding from surviving resilient areas of reef.
Compared to other Indo-Pacific countries such as Indonesia, Australia is resource-rich, has a lower population and less of an economic need for wild harvest of marine species. In 1985, the value of each license was $7600 and the industry employed the equivalent of 12 full-time employees [33]. By 2000, the gross value of production of the industry had increased to $427,000, or approximately $11,700 per license. Based on current domestic specimen retail prices, the 2009 value of the Queensland catch is ∼$0.3–$1.0 million while the average value of a license is ∼$17,000. There is no doubt that the retail value and profitability of corals has increased. For instance, in 1985, ornamental pieces sold for only ∼$1–$5 [22] whereas some rare live corals now sell for ∼$180. The increase in the number of licenses may have effectively absorbed any increase in overall profits. In a limited entry, quota-managed fishery latent effort is typically indicated by inactive permits and unfilled quota [64]. Generally, permits are left inactive only when licensees determine that the profits available in the fishery are too low to make it viable to purchase or activate them. Inactive fishery licenses can also become active at relatively short notice, drawing idle effort into the fishery, further limiting profits at a higher level of catch. Sparse populations could be fished down relatively quickly if holders of enough inactive licenses resume collecting. Moreover, the cost of managing fisheries in Queensland is estimated to be $1 b [64] whereas in 2007, domestic and international recreational snorkel and dive visitors to the GBR spent ∼$2.7 b (data from Tourism Queensland). There is also conflict between marine-based tourism and coral collectors [65] as they both target aesthetically pleasing and relatively accessible reefs. Resource management costs therefore appear to outweigh the economic value of the fishery, especially in light of their value to tourism.
Many of the coral species targeted by the QCF in the Keppels are found at very restricted locations within specific depths and environmental conditions and comprise <0.01% of the coral cover (based on data from the Australian Institute of Marine Science). It is possible that the targeted corals occur in higher abundance in small isolated pockets which are known only to collectors. However, if this were true then it is even further cause for alarm as there is a likelihood that such pockets are largely self-seeding. These factors make such species more vulnerable to over-exploitation as systematic harvest can result in the removal of almost all specimens in an area. Regeneration must necessarily therefore come from recruitment from unharvested areas that are within oceanographic range. These “refugia” are logically more likely to exist in deeper waters that are less likely to be affected by disturbance. In the Philippines, controls on the maximum depth limit for coral collection have meant that unharvested deepwater populations acted as reproductive reservoirs for shallow water recruitment and maintenance of sustainable harvest yields [12]. Unfortunately, in Australia, the relaxation of the 6m depth limit means that collectors can now access many areas of reef that were previously spared from disturbance. However a re-introduction of the 6m depth limit would be problematic because this would restrict collectors to those reef areas that are also relied upon by tourist operators. In any case, many of the species now targeted by collectors do not typically inhabit shallow reefs. The GBR is considered at high risk of species loss, not the least because inshore reefs are targeted by the QCF due to their accessibility to export facilities but also due to the fact that many of the harvested species are restricted to inshore reefs which are considered more vulnerable to chronic disturbance [66,67]. A recent report by the AIMS has highlighted the decline in coral reefs along the inshore GBR [16]. Accordingly, there is urgent need for more data on the distribution and population structure of restricted range species if managers are to accurately quantify their extinction risk.
Fundamental to the effective management of ecologically sustainable fisheries is having appropriate information available to make informed decisions as even moderate amounts of non-destructive aquarium extraction can negatively affect targeted populations [12,20,67]. To prevent the depletion of populations of highly sought-after species, particularly in areas close to transport like the Keppels, harvest quotas should be based on information on geographic distribution, growth, natural mortality and annual recruitment rates. The current total allowable catch quota (TAC) for the QCF, was originally based on acroporid and pocilloporid species for ornamental use, and now appears to be inappropriate. Quotas should be related to what is currently known about population dynamics and abundance of the species, with those species that are least abundant and the slowest growing having the lowest quotas. A reproductive “cushion” also needs to be maintained; whereby population replacement is assured by implementing a minimum collection size limit sufficiently greater than the size of colonies at reproductive maturity and the identification and protection of non-harvested population reserves. Alternatively, many of the taxa targeted by the international aquarium trade are listed as successful for aquaculture by Yates and Carlson [68]. Aquaculture is already feasible and, in many cases, corals that are reared in aquaria survive much longer than those harvested from the wild [69,70].
4. Conclusions
This study investigated a recent change in the commercial harvest of corals on inshore reefs in the Keppel Islands region of the southern Great Barrier Reef which appears to be driven by the US market demand for domestic aquaria. The results strongly suggest that the current management regime is no longer appropriate given the significant changes in market demand which is now focused on vibrantly colored stony corals with large polyps (LPS). These taxa are typically slow-growing and sparse compared to the fast growing and abundant acroporid and pocilloporid species which were the focus of the industry in the past. Without an effective, locally relevant monitoring and management regime, there is a risk that continued harvest levels may eventually cause local and even regional extinctions without triggering a management response. Even in the event of management intervention such as reduced species-specific quotas, for slow-growing species the loss may be long-term. Such extinctions could negatively impact reef stability by removing some of the key functional species groups. While these are not typically considered reef “builders”, they are stress-tolerant functional groups, the loss of which could tip the balance of these reefs from highly functionally diverse, stable systems to mono-specific stands of fast growing “weedy” species that are more susceptible to invasion by macro-algae, soft corals and unwanted “pest” species. To avoid the risk of local extinctions of these taxa, the fishery must now undergo a shift away from wild harvest to aquaculture if it is to be ecologically sustainable.
Supplementary Material
diversity-03-00466-s001.pdf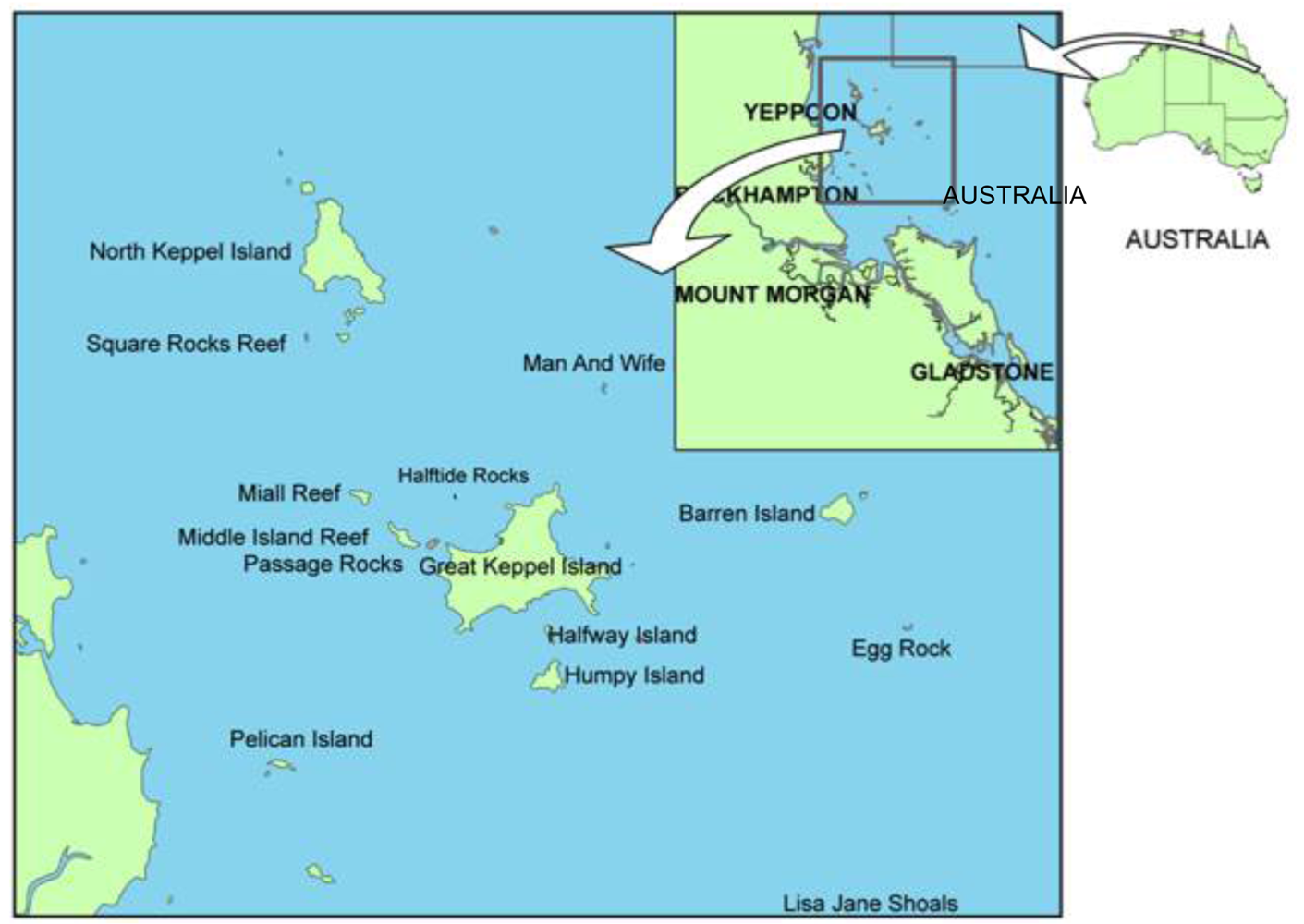



Acknowledgments
The author acknowledges the assistance of Queensland Fisheries for access to annual reports from the coral fishery and the Long Term Monitoring Team at the Australian Institute of Marine Science for providing information on the abundance of coral taxa.
References and Notes
- Pandolfi, J.M.; Bradbury, R.H.; Sala, E.; Hughes, T.P.; Bjorndal, K.A.; Cooke, R.G.; McArdle, D.; McClenachan, L.; Newman, M.J.H.; Paredes, G.; et al. Global trajectories of the long-term decline of coral reef ecosystems. Science 2003, 301, 955–958. [Google Scholar]
- IUCN Red list of threatened species. IUCN Red List Unit: Cambridge, UK, 2011. Available online: http://www.iucnredlist.org (accessed on 6 July 2011).
- Ostrander, G.K.; Armstrong, K.M.; Knobbe, E.T.; Gerace, D.; Scully, E.P. Rapid transition in the structure of a coral reef community: The effects of coral bleaching and physical disturbance. Proc. Natl. Acad. Sci. USA 2000, 97, 5297–5302. [Google Scholar]
- Smith, L.D.; Gilmour, J.P.; Heyward, A.J. Resilience of coral communities on an isolated system of reefs following catastrophic mass-bleaching. Coral Reefs 2008, 27, 197–205. [Google Scholar]
- Aronson, R.B.; Precht, W.F.; Toscano, M.A.; Koltes, K.H. The 1998 bleaching event and its aftermath on a coral reef in Belize. Mar. Biol. 2002, 141, 435–447. [Google Scholar]
- Loya, Y.; Sakai, K.; Yamazato, K.; Nakano, Y. Coral bleaching: the winners and the losers. Ecol. Lett. 2001, 4, 122–131. [Google Scholar]
- McClanahan, T.; Ateweberhan, M.; Omukoto, J. Long-term changes in coral colony size distributions on Kenyan reefs under different management regimes and across the 1998 bleaching event. Mar. Biol. 2008, 153, 755–768. [Google Scholar]
- Glynn, P.W. Some physical and biological determinants of coral community structure in the Eastern Pacific. Ecol. Monogr. 1976, 46, 431–456. [Google Scholar]
- Porter, J.W. Predation by Acanthaster and its effect on coral species diversity. Am. Nat. 1972, 106, 487–492. [Google Scholar]
- Porter, J.W. Community structure of coral reefs on opposite sides of the isthmus of Panama. Science 1974, 186, 543–545. [Google Scholar]
- Bentley, N. An overview of the exploitation, trade and management of corals in Indonesia. Traffic Bull. 1998, 17, 67–78. [Google Scholar]
- Ross, M.A. A quantitative study of the stony coral fishery in cebu, philippines. Mar. Ecol. 1984, 5, 75–91. [Google Scholar]
- Tsounis, G.; Rossi, S.; Grigg, R.; Santangelo, G.; Bramanti, L.; Gili, J.-M. The exploitation and conservation of precious corals. In Oceanography and Marine Biology: An Annual Review; Gibson, R.N., Atkinson, R.J.A., Gordon, J.D.M., Eds.; Taylor & Francis: London, UK, 2010. [Google Scholar]
- Nyström, M.; Moberg, F.; Tedengren, M. Natural and anthropogenic disturbance on reef corals in the inner gulf of thailand: Physiological effects of reduced salinity, copper and siltation. Proceedings of the 8th International Coral Reef Symposium, Panama City, Panama, 24–29 June 1996.
- Wakeford, M.; Done, T.J.; Johnson, C.R. Decadal trends in a coral community and evidence of changed disturbance regime. Coral Reefs 2008, 27, 1–13. [Google Scholar]
- Thompson, A.; Dolman, A. Coral bleaching: One disturbance too many for near-shore reefs of the Great Barrier Reef. Coral Reefs 2010, 29, 637–648. [Google Scholar]
- Carpenter, K.E.; Abrar, M.; Aeby, G.; Aronson, R.B.; Banks, S.; Bruckner, A.; Chiriboga, A.; Cortés, J.; Delbeek, J.C.; DeVantier, L.; et al. One-third of reef-building corals face elevated extinction risk from climate change and local impacts. Science 2008, 321, 560–563. [Google Scholar]
- Green, E.P.; Shirley, F. The Global Trade in Corals; WCMC-World Conservation Press: Cambridge, UK, 1999. [Google Scholar]
- Rhyne, A.; Rotjan, R.; Bruckner, A.; Tlusty, M. Crawling to collapse: Ecologically unsound ornamental invertebrate fisheries. PLoS One 2009, 4, e8413. [Google Scholar]
- Anonymous. International trade in coral and coral reef species: The role of the united states. In Report of the Trade Subgroup of the International Working Group to the U.S.; Coral Reef Task Force: Washington DC, USA; 2; March; 2000. [Google Scholar]
- Tissot, B.N.; Best, B.A.; Borneman, E.H.; Bruckner, A.W.; Cooper, C.H.; D'Agnes, H.; Fitzgerald, T.P.; Leland, A.; Lieberman, S.; Mathews Amos, A.; et al. How U.S. ocean policy and market power can reform the coral reef wildlife trade. Mar. Pol. 2010, 34, 1385–1388. [Google Scholar]
- Oliver, J.; McGinnity, P. Commercial coral collecting on the great barrier reef. Proceedings of the Fifth International Coral Reef Congress, Tahiti, French Polynesia, 27 May–1 June 1985.
- CITES Trade Statistics Derived from the CITES Trade Database; UNEP-World Conservation Monitoring Centre: Cambridge, UK. Available online: http://www.unep-wcmc.org/citestrade (accessed on 22 March 2011).
- Davies, P.J. Reef growth. In Perspectives in Coral Reefs; Barnes, D.J., Ed.; Brian Clouston: Manuka, Australia, 1983; pp. 69–106. [Google Scholar]
- Harriott, V.J. Can corals be harvested sustainably? AMBIO 2003, 32, 130–133. [Google Scholar]
- Cooper, T.F.; De'Ath, G.; Fabricius, K.E.; Lough, J.M. Declining coral calcification in massive porites in two nearshore regions of the northern great barrier reef. Glob. Change Biol. 2008, 14, 529–538. [Google Scholar]
- Anonymous. Policy for the Management of the Coral Fishery; Department of Primary Industries and Fisheries, Queensland Government: Queensland, Australia, 2009. [Google Scholar]
- Roelfs, A.J.; Silcock, R. A Vulnerability Assessment of Coral Taxa Harvested in the Queensland Coral Fishery; Department of Primary Industries and Fisheries: Queensland, Australia, 2009. [Google Scholar]
- Roelofs, A.J. Ecological Risk Assessment of the Queensland Coral Fishery; Department of Primary Industries and Fisheries: Queensland, Australia, 2008. [Google Scholar]
- Ponder, W.F.; Grayson, J.E. The Australian marine molluscs considered to be potentially vulnerable to the shell trade: A report / prepared for Environment Australia by Winston F. Ponder and Jillian E. Grayson; Australian Museum: Sydney, Australia, 1998. [Google Scholar]
- Schlager, E. Model Specification and Policy Analysis: The Governance of Coastal Fisheries. Ph.D. Thesis, Indiana University, Bloomington, IN, USA, 1990. [Google Scholar]
- Scott, A. Obstacles to fisheries self-government. Mar. Resour. Econ. 1993, 8, 187–199. [Google Scholar]
- Harriott, V.J. The Sustainability of Queensland's Coral Harvest Fishery: CRC Reef Research Centre Technical Report; CRC Reef Research Centre: Townsville, Australia, 2001. [Google Scholar]
- Luick, J.L.; Mason, L.; Hardy, T.; Furnas, M.J. circulation in the great barrier reef lagoon using numerical tracers and in situ data. Cont. Shelf Res. 2007, 27, 757–778. [Google Scholar]
- Thompson, A.; Davidson, J.; Schaffelke, B.; Sweatman, H. Reef Rescue Marine Monitoring Program Annual Report of AIMS Activities 2009/10 Project 3.7.1b Inshore Coral Reef Monitoring; Australian Institute of Marine Science (AIMS): Queensland, Australia, 2010. [Google Scholar]
- Sanders, D.; Baron-Szabo, R.C. Scleractinian assemblages under sediment input: Their characteristics and relation to the nutrient input concept. Palaeogeography, Palaeoclimatology, Palaeoecology 2005, 216, 139–181. [Google Scholar]
- Stafford-Smith, M. Sediment-rejection efficiency of 22 species of Australian scleractinian corals. Mar. Biol. 1993, 115, 229–243. [Google Scholar]
- Coles, S.L. Coral species diversity and environmental factors in the Arabian Gulf and the Gulf of Oman: A comparison to the Indo-Pacific region. Atoll Res. Bull. 1992, 507, 1–19. [Google Scholar]
- Anonymous. Annual status reports 2007 - 2009 Queensland Coral Fishery; Department of Employment Economic Development and Innovation: Queensland, Australia, 2009. [Google Scholar]
- Fletcher, W.; Chesson, J.; Fisher, M.; Sainsbury, K.; Hundloe, T.; Smith, A.; Whitworth, B. National ESD Reporting Framework for Australian Fisheries: The ‘How To’ Guide for Wild Capture Fisheries; Project 2000/145; Fisheries Research Development Corporation: Canberra, Australia, 2002. [Google Scholar]
- Veron, J.; Stafford-Smith, M.G. Coral ID Version 1.1; Australian Institute of Marine Science: Townsville, Australia, 2011. Available online: http://www.coralid.com (accessed on 26 July 2011).
- Shulman, M.; Robertson, D. Changes in the coral reefs of San Blas, Caribbean Panama: 1983 to 1990. Coral Reefs 1996, 15, 231–236. [Google Scholar]
- Carpenter, S.R.; Ives, A.R. Stability and diversity of ecosystems. Science 2007, 317, 58–62. [Google Scholar]
- Bush, S.L.; Precht, W.F.; Woodley, J.D.; Bruno, J.F. Indo-Pacific mushroom corals found on Jamaican reefs. Coral Reefs 2004, 23, 234–234. [Google Scholar]
- Paine, R.T. Ecological determinism in the competition for space: The Robert H. MacArthur award lecture. Ecology 1984, 65, 1339–1348. [Google Scholar]
- Bellwood, D.R.; Hoey, A.S.; Choat, J.H. Limited functional redundancy in high diversity systems: Resilience and ecosystem function on coral reefs. Ecol. Lett. 2003, 6, 281–285. [Google Scholar]
- Grigg, R.W. Resource management of precious corals. Mar. Ecol. 1984, 5, 57–74. [Google Scholar]
- Frisch, A.J.; Hobbs, J.-P.A. Rapid Assessment of Anemone and Anemonefish Populations at the Keppel Islands [Electronic Resource]: A Report to the Great Barrier Reef Marine Park Authority; Great Barrier Reef Marine Park Authority: Townsville, Australia, 2008. [Google Scholar]
- Jones, A.; Gardner, S.; Sinclair, B. Losing ‘nemo’: Bleaching and collection impacts inshore populations of anemonefish and their hosts the sea anemones. J. Fish Biol. 2008, 73, 753–761. [Google Scholar]
- Anonymous. Performance Measurement System: Queensland Coral Fishery; Department of Primary Industries and Fisheries: Queensland, Australia, 2009. [Google Scholar]
- Baker, A.C.; Glynn, P.W.; Riegl, B. Climate change and coral reef bleaching: An ecological assessment of long-term impacts, recovery trends and future outlook. Estuar. Coast. Shelf Sci. 2008, 80, 435–471. [Google Scholar]
- Guzman, H.; Cortés, J. Reef recovery 20 years after the 1982–1983 El Niño massive mortality. Mar. Biol. 2007, 151, 401–411. [Google Scholar]
- Jones, A.; Berkelmans, R. Potential costs of acclimatization to a warmer climate: Growth of a reef coral with heat tolerant vs. sensitive symbiont types. PLoS One 2010, 5, e10437. [Google Scholar]
- Jones, A.M.; Berkelmans, R. Tradeoffs to thermal acclimation: Energetics and reproduction of a reef coral with heat tolerant Symbiodinium type-D. J. Mar. Biol. 2011, 2011. [Google Scholar] [CrossRef]
- Anthony, K.R.N.; Connolly, S.R. Bleaching, energetics, and coral mortality risk: Effects of temperature, light and sediment regime. Limnol. Oceanogr. 2007, 52, 716–726. [Google Scholar]
- Maynard, J.A.; Anthony, K.R.N.; Harvell, C.D.; Burgman, M.A.; Beeden, R.H.; Sweatman, H.; Heron, S.F.; Lamb, J.B.; Willis, B.L. Predicting outbreaks of a climate-driven coral disease in the Great Barrier Reef. Coral Reefs 2010, 30, 485–495. [Google Scholar]
- Jones, A.M.; Berkelmans, R.; Houston, W. Species richness and community structure on a high latitude reef: Implications for conservation and management. Diversity 2011, 3, 329–355. [Google Scholar]
- Allee, W.C. Animal Aggregations: A Study in General Sociology; University of Chicago Press: Chicago, IL, USA, 1931. [Google Scholar]
- An ‘Allee’ effect applies to small populations where by increasing population density reduces the growth rate of the population.
- Anonymous. Coral Stress Response Plan for the Coral and Marine Aquarium Fish Fisheries; Department of Employment, Economic Development and Innovation: Queensland, Australia, 2009. [Google Scholar]
- Saenz-Agudelo, P.; Jones, G.P.; Thorrold, S.R.; Planes, S. Detrimental effects of host anemone bleaching on anemonefish populations. Coral Reefs 2011, 30, 497–506. [Google Scholar]
- Shuman, C.S.; Hodgson, G.; Ambrose, R. Population impacts of collecting sea anemones and anemonefish for the marine aquarium trade in the Philippines. Coral Reefs 2006, 24, 564–573. [Google Scholar]
- Atkinson, M.; Kerrigan, B.; Roelfs, A.; Smith, T. Non-Detriment Finding for CITES-Listed Corals in the Queensland Coral Fishery; WG9-Aquatic Invertebrates Case Study 4: Mexico, Queensland, Australia, 2008. [Google Scholar]
- Newton, P.; Wood, R.; Galeano, D.; Vieira, S.; Perry., R. Fishery Economic Status Report. Prepared for the Fisheries Resources Research Fund; Australian Bureau of Agricultural and Research Economics and Sciences: Canberra, Australia, 2007. [Google Scholar]
- Anonymous. Review of Recreational Take of Coral in Western Australia, A Discussion Paper; Western Australia Fisheries Management Authority: Perth, Australia, 2002. [Google Scholar]
- Carpenter, K.E.; Miclat, R.I.; Albaladejo, V.D.; Corpuz, V.T. The influence of the local abundance and diversity of Philippine reef fishes. Proceedings of the Fourth International Coral Reef Symposium, Manila, Philippines, May 1981.
- Roberts, C.M.; McClean, C.J.; Veron, J.E.N.; Hawkins, J.P.; Allen, G.R.; McAllister, D.E.; Mittermeier, C.G.; Schueler, F.W.; Spalding, M.; Wells, F.; et al. Marine biodiversity hotspots and conservation priorities for tropical reefs. Science 2002, 295, 1280–1284. [Google Scholar]
- Yates, K.; Carlson, B. Corals in aquariums: How to use selective collecting and innovative husbandry to promote reef conservation. Proceedings of the Seventh International Coral Reef Symposium, Guam, Micronesia, 22–27 June 1992.
- Borneman, E. Aquarium Corals: Selection, Husbandry, and Natural History; Microcosm Ltd: Neptune City, NJ, USA, 2001. [Google Scholar]
- Delbeek, J.C. Coral farming: Past, present and future trends. Aquarium Sci. Conserv. 2001, 3, 171–181. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jones, A.M. Raiding the Coral Nurseries? Diversity 2011, 3, 466-482. https://doi.org/10.3390/d3030466
Jones AM. Raiding the Coral Nurseries? Diversity. 2011; 3(3):466-482. https://doi.org/10.3390/d3030466
Chicago/Turabian StyleJones, Alison M. 2011. "Raiding the Coral Nurseries?" Diversity 3, no. 3: 466-482. https://doi.org/10.3390/d3030466
APA StyleJones, A. M. (2011). Raiding the Coral Nurseries? Diversity, 3(3), 466-482. https://doi.org/10.3390/d3030466



