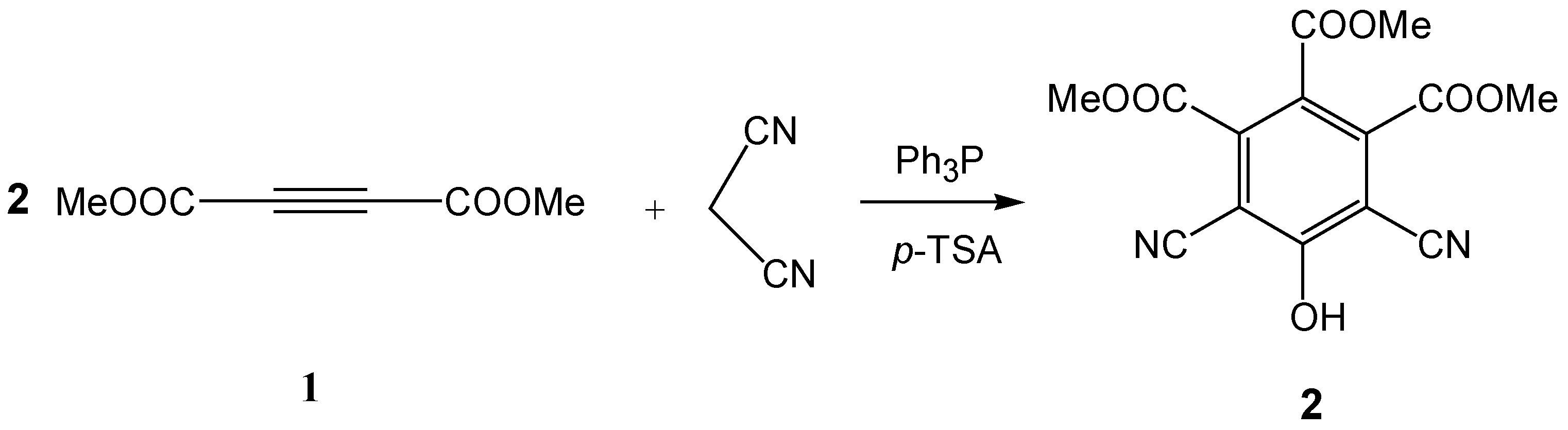
Trimethyl 4,6-Dicyano-5-hydroxybenzene-1,2,3-tricarboxylate
Abstract
:Experimental
Structural Characterization
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgements
References and Notes
- Lakowicz, J.R. Principle of Fluorescence Spectroscopy, 3rd ed.; Springer: New York, NY, USA, 2006. [Google Scholar]
- Hussain, H.H.; Babic, G.; Durst, T.; Wright, J.S.; Flueraru, M.; Chichirau, A.; Chepelev, L.L. Development of novel antioxidants: Design, synthesis, and reactivity. J. Org. Chem. 2003, 68, 7023–7032. [Google Scholar] [CrossRef] [PubMed]
- Vosooghi, M.; Rajabalian, S.; Sorkhi, M.; Badinloo, M.; Nakhjiri, M.; Negahbani, A.S.; Asadipour, A.; Mahdavi, M.; Shafiee, A.; Foroumadi, A. Synthesis and cytotoxic activity of some 2-amino-4-aryl-3-cyano-7-(dimethylamino)-4H-chromenes. Res. Pharm. Sci. 2010, 5, 9–14. [Google Scholar] [PubMed]
- Wright, J.S.; Johnson, E.R.; DiLiabio, G.A. Predicting the activity of phenolic antioxidants: Theoretical method, analysis of substituent effects, and application to major families of antioxidants. J. Am. Chem. Soc. 2001, 123, 1173–1183. [Google Scholar] [CrossRef] [PubMed]
- Samshuddin, S.; Narayana, B.; Sarojini, B.K. Ethyl 4,4''-difluoro-5'-hydroxy-1,1':3',1''-terphenyl-4'-carboxylate. Molbank 2011, 2011, M745. [Google Scholar] [CrossRef]
- Crystallographic data for compound 2 reported in this paper have been deposited with the Cambridge Crystallographie Data Center as supplementary publication CCDC No. 842814. These data can be obtained free of charge via www.ccdc.com.ac.uk/data_request/cif.



| CH3OH | CH3CN | CHCl3 | ||||
| Comp. | λAbs. | λFlu. | λAbs. | λFlu. | λAbs. | λFlu. |
| 2 | 237 | 469 | 226 | 459 | 335 | 455 |
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zonouzi, A.; Mirzazadeh, R.; Ng, S.W. Trimethyl 4,6-Dicyano-5-hydroxybenzene-1,2,3-tricarboxylate. Molbank 2012, 2012, M775. https://doi.org/10.3390/M775
Zonouzi A, Mirzazadeh R, Ng SW. Trimethyl 4,6-Dicyano-5-hydroxybenzene-1,2,3-tricarboxylate. Molbank. 2012; 2012(3):M775. https://doi.org/10.3390/M775
Chicago/Turabian StyleZonouzi, Afsaneh, Roghieh Mirzazadeh, and Seik Weng Ng. 2012. "Trimethyl 4,6-Dicyano-5-hydroxybenzene-1,2,3-tricarboxylate" Molbank 2012, no. 3: M775. https://doi.org/10.3390/M775
APA StyleZonouzi, A., Mirzazadeh, R., & Ng, S. W. (2012). Trimethyl 4,6-Dicyano-5-hydroxybenzene-1,2,3-tricarboxylate. Molbank, 2012(3), M775. https://doi.org/10.3390/M775





