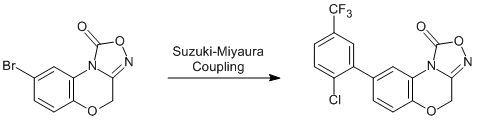
8-[2-Chloro-5-(trifluoromethyl)phenyl]-4H-[1,2,4]oxadiazolo-[3,4-c][1,4]benzoxazin-1-one
Abstract
:
Experimental Section
8-[2-Chloro-5-(trifluoromethyl)phenyl]-4H-[1,2,4]oxadiazolo[3,4-c][1,4]benzoxazin-1-one (4)
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgements
References and Notes
- Lee, Y.C.; Martin, E.; Murad, F. Human recombinant soluble guanylyl cyclase: Expression, purification, and regulation. Proc. Natl. Acad. Sci. USA 2000, 97, 10763–10768. [Google Scholar] [CrossRef] [PubMed]
- Wedel, B.; Humbert, P.; Harteneck, C.; Foerster, J.; Malkewitz, J.; Bӧhme, E.; Schultz, G.; Koesling, D. Mutation of His-105 in the beta(1)-subunit yields a nitric oxide-insensitive form of soluble guanylyl cyclase. Proc. Natl. Acad. Sci. USA 1994, 91, 2592–2596. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Schelvis, J.P.M.; Babcock, G.T.; Marletta, M.A. Identification of histidine 105 in the beta 1 subunit of soluble guanylate cyclase as the heme proximal ligand. Biochemistry 1998, 37, 4502–4509. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, T.C.; Garthwaite, J. The receptor-like properties of nitric oxide-activated soluble guanylyl cyclase in intact cells. Mol. Cell. Biochem. 2002, 230, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Ballou, D.P.; Zhao, Y.; Brandish, P.E.; Marletta, M.A. Revisiting the kinetics of nitric oxide (NO) binding to soluble guanylate cyclase: The simple NO-binding model is incorrect. Proc. Natl. Acad. Sci. USA 2002, 99, 12097–12101. [Google Scholar] [CrossRef] [PubMed]
- Kajiya, K.; Detmar, M. Method for inhibiting lymphangiogenesis and inflammation. U.S. Patent 2008,176,851, 24 July 2008. [Google Scholar]
- Ikeyama, K.; Fuziwara, S.; Denda, M. Method for accelerating cutaneous barrier recovery. U.S. Patent 2007,232,595, 4 October 2007. [Google Scholar]
- Dawson-Scully, K.; Sokolowski, M.; Kent, C.; Robertson, R.M.; Armstrong, G.A. Thermoprotective compositions of PKG pathway inhibitors and method of use thereof. U.S. Patent 2007,184,044, 9 August 2007. [Google Scholar]
- Baumann, M.; Bloch, W.; Korkmaz, Y. Modulating substances of the nitric oxid (no) - cyclic guanosine-3',5'-monophosphate (cgmp) signaling pathway for the treatment of dental disorders. WO Pat. 2005,046,660, 2005 26 May. [Google Scholar]
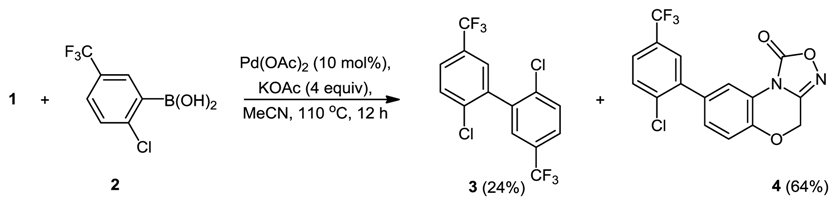
- Berezin, A.A.; Koutentis, P.A. Suzuki-Miyaura reactions of the soluble guanylate cyclase inhibitor NS2028: a non product specific route to C-8 substituted analogues. Tetrahedron 2011, 67, 4069–4078. [Google Scholar] [CrossRef]
- Drahushuk, A.T.; Choy, C.O.; Kumar, S.; McReynolds, J.H.; Olson, J.R. Modulation of cytochrome P450 by 5,5′-bis-trifluoromethyl-2,2′-dichlorobiphenyl, a unique environmental contaminant. Toxicology 1997, 120, 197–205. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Berezin, A.A.; Koutentis, P.A. 8-[2-Chloro-5-(trifluoromethyl)phenyl]-4H-[1,2,4]oxadiazolo-[3,4-c][1,4]benzoxazin-1-one. Molbank 2011, 2011, M728. https://doi.org/10.3390/M728
Berezin AA, Koutentis PA. 8-[2-Chloro-5-(trifluoromethyl)phenyl]-4H-[1,2,4]oxadiazolo-[3,4-c][1,4]benzoxazin-1-one. Molbank. 2011; 2011(2):M728. https://doi.org/10.3390/M728
Chicago/Turabian StyleBerezin, Andrey A., and Panayiotis A. Koutentis. 2011. "8-[2-Chloro-5-(trifluoromethyl)phenyl]-4H-[1,2,4]oxadiazolo-[3,4-c][1,4]benzoxazin-1-one" Molbank 2011, no. 2: M728. https://doi.org/10.3390/M728