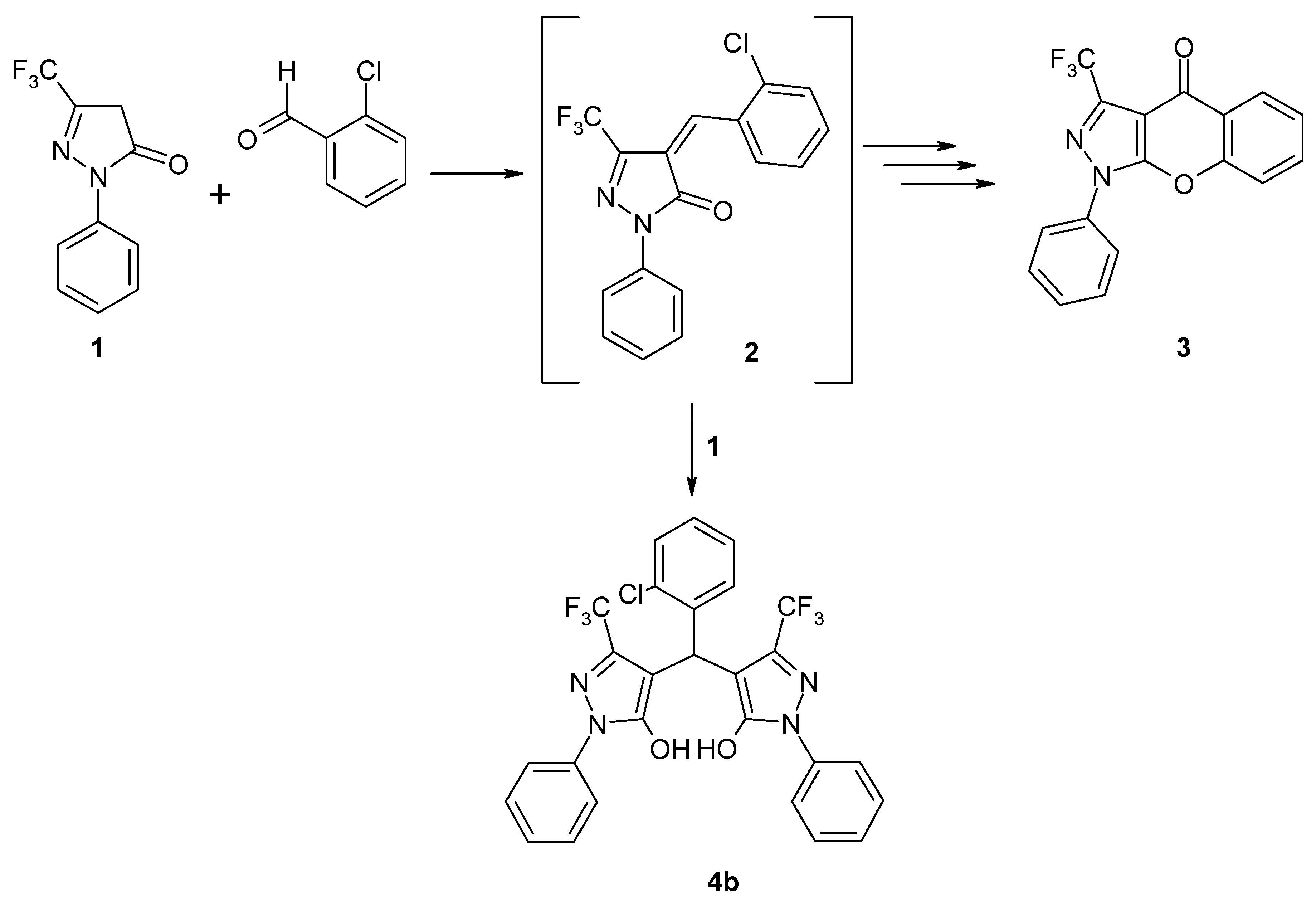
4,4’-[(2-Chlorophenyl)methylene]bis[1-phenyl-3-(trifluoromethyl)-1H-pyrazol-5-ol]
Abstract
:Experimental
4,4’-[(2-Chlorophenyl)methylene]bis[1-phenyl-3-(trifluoromethyl)-1H-pyrazol-5-ol] (4b)
Supplementary materials
Supplementary File 1Supplementary File 2Supplementary File 3Acknowledgements
References and Notes
- Bieringer, S.; Holzer, W. 4-Acyl-5-hydroxy-1-phenyl-3-trifluoromethylpyrazoles: Synthesis and NMR Spectral Investigations. Heterocycles 2006, 68, 1825–1836. [Google Scholar]
- Sobahi, T.R. Thermal condensation of 3-trifluoromethyl- and 3-amino-1-phenyl-2-pyrazolin-5-ones with aromatic aldehydes: Synthesis of 4-arylidenepyrazolones and pyrazolopyranopyrazoles. Indian J. Chem, Sect. B 2006, 45, 1315–1318. [Google Scholar] [CrossRef]
- Zohdi, H.F.; Elghandour, A.H.H.; Rateb, N.M.; Sallam, M.M.M. Reactions with 5-trifluoromethyl-2,4-dihydropyrazol-3-one derivatives: a new route for the synthesis of fluorinated polyfunctionally substituted pyrazole and pyrano[2,3-c]pyrazole derivatives. J. Chem. Res. Synop. 1992, 396–397. [Google Scholar] [CrossRef]
- Yao, C.S.; Yu, C.X.; Tu, S.J.; Shi, D.Q.; Wang, X.S.; Zhu, Y.Q.; Yang, H.Z. The synthesis of 4,4’-arylmethylene-bis(3-trifluoromethyl)-1-phenyl-1H-pyrazol-5-ol) in aqueous media without catalyst. J. Fluorine Chem. 2007, 128, 105–109. [Google Scholar] [CrossRef]
- Braun, S.; Kalinowski, H.O.; Berger, S. 150 and More Basic NMR Experiments, 2nd expanded ed.; Wiley-VCH: Weinheim, New York, 1998. [Google Scholar]
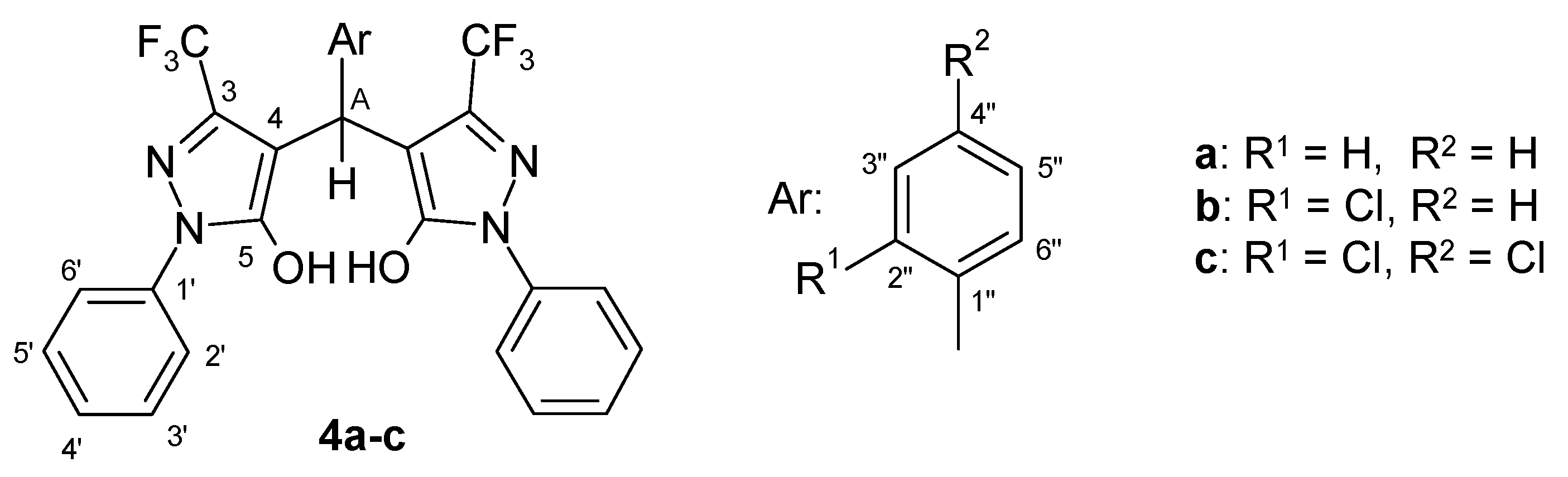
| comp. | OH | HA | H of N-phenyl | H of aryl | 19F | ||||||
| 2’,6’ | 3’,5’ | 4’ | 2’’ | 3’’ | 4’’ | 5’’ | 6’’ | C F3 | |||
| 4a | 8.33a | 5.22 | 7.87 | 7.41 | 7.23 | 7.26 | 7.26 | 7.12 | 7.26 | 7.26 | –60.2 |
| 4b | 6.88a | 5.68 | 7.82 | 7.42 | 7.25 | – | 7.32 | 7.18 | 7.25 | 7.72 | –60.0 |
| 4c | 6.42a | 5.63 | 7.81 | 7.42 | 7.25 | – | 7.47b | – | 7.36c | 7.68d | –60.1 |
| comp. | CA | CF3 | C of pyrazole | |||||||||||||
| C-3 | C-4 | C-5 | ||||||||||||||
| 4a | 32.8
1J = 125.0 | 122.4
1J = 270.0 | 138.8
2J(C3,F) = 34.5 3J(C3,HA) = 4.0 | 100.4
2J(C4,HA) = 8.8 | 157.8
3J(C5,HA) = 7.9 | |||||||||||
| 4b | 30.9
1J = 127.5 | 122.1
1J = 270.1 | 137.5
2J(C3,F) = 35.4 3J(C3,HA) = 4.0 | 100.2
2J(C4,HA) = 8.6 | 156.9
3J(C5,HA) = 7.3 | |||||||||||
| 4c | 30.7
1J = 128.0 | 122.1
1J = 270.1 | 137.5
2J(C3,F) = 35.7 3J(C3,HA) = 4.0 | 99.8
2J(C4,HA) = 8.7 | 156.8
3J(C5,HA) = 7.0 | |||||||||||
| comp. | C of N-phenyl | C of aryl | ||||||||||||||
| 1’ | 2’,6’ | 3’,5’ | 4’ | 1’’ | 2’’ | 3’’ | 4’’ | 5’’ | 6’’ | |||||||
| 4a | 139.2 | 120.9 | 128.6 | 125.5 | 143.5 | 127.0 | 128.0 | 125.6 | 128.0 | 127.0 | ||||||
| 4b | 138.9 | 121.1 | 128.7 | 125.8 | 139.7 | 131.7 | 129.2 | 127.7 | 126.3 | 130.6 | ||||||
| 4c | 138.8 | 121.2 | 128.7 | 125.9 | 138.7 | 132.6 | 128.6 | 131.4 | 126.5 | 131.9 | ||||||
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Guo, C.; Holzer, W. 4,4’-[(2-Chlorophenyl)methylene]bis[1-phenyl-3-(trifluoromethyl)-1H-pyrazol-5-ol]. Molbank 2009, 2009, M605. https://doi.org/10.3390/M605
Guo C, Holzer W. 4,4’-[(2-Chlorophenyl)methylene]bis[1-phenyl-3-(trifluoromethyl)-1H-pyrazol-5-ol]. Molbank. 2009; 2009(3):M605. https://doi.org/10.3390/M605
Chicago/Turabian StyleGuo, Changbin, and Wolfgang Holzer. 2009. "4,4’-[(2-Chlorophenyl)methylene]bis[1-phenyl-3-(trifluoromethyl)-1H-pyrazol-5-ol]" Molbank 2009, no. 3: M605. https://doi.org/10.3390/M605




