Imprinting Status of IGF2 in Cord Blood Cells of Han Chinese Newborns
Abstract
:1. Introduction
2. Materials and Methods
2.1 Collection of cord blood samples and clinical data
2.2 Nucleic acid preparation
2.3 Screening for informative samples
2.4 RNA reverse transcription and IGF2 imprinting assay
2.5 Statistical analysis
3. Results
3.1 IGF2 imprinting status in cord blood nucleated cells
3.2 Characteristics of parents of infants with LOI
3.3 Characteristics of infants with LOI
3.4 Inheritance assay of IGF2 LOI
4. Discussion
| Normal imprinting singletons (%) | LOI singletons (%) | p value | |
|---|---|---|---|
| Total | 216 | 57 | |
| Pre-pregnancy BMI (kg/m2, mean ± s.e.m) | 20.7 ± 2.54 | 20.8 ± 2.57 | 0.792 |
| Parity | |||
| 0 | 111(51.4) | 27(47.4) | |
| ≥ 1 | 105(48.6) | 30(52.6) | |
| Mean gestational age (week) (mean ± s.e.m.) | 39.5 ± 1.20 | 39.6 ± 0.91 | 0.111 |
| Maternal ages (years) | |||
| < 25 | 44(20.4) | 10(17.5) | |
| 25 – 29 | 121(56.0) | 29(50.9) | |
| 30 – 34 | 46(21.3) | 15(26.3) | |
| ≥ 35 | 5(2.3) | 3(5.3) | |
| Mean ± s.e.m. | 27.3 ± 3.55 | 27.9 ± 4.00 | 0.260 |
| Paternal ages (years) | |||
| < 25 | 11(5.1) | 4(7.0) | |
| 25 – 29 | 88(40.7) | 22(38.6) | |
| 30 – 34 | 79(36.6) | 14(24.6) | |
| ≥ 35 | 38(17.6) | 17(29.8) | 0.041a |
| Mean ± s.e.m. | 30.4 ± 4.47 | 31.5 ± 5.07 | 0.111 |
| Pregnancy complications | |||
| Hypertensive disorders | 21(9.7) | 3(5.3) | 0.290 |
| Diabetes mellitus | 7(3.2) | 1(1.8) | 0.554 |
| Oligoamnios | 11(5.1) | 1(1.8) | 0.270 |
| Polyhydramnios | 10(4.6) | 5(2.3) | 0.222 |
| Congenital defects | Normal imprinting | LOI | Notes |
|---|---|---|---|
| Inguinal/umbilical hernia | 2 | 1 | |
| Diaphragmatic hernia | 1 | 0 | Died in 3 days after birth |
| Congenital heart diseases | 1 | 0 | Ventricular septal defect 3mm |
| Sacrococcygeal teratoma | 1 | 0 | Benign |
| Congenital adrenal cortical hyperplasia | 0 | 1 | Familial |
| Items | Normal imprinting | LOI | p value |
|---|---|---|---|
| Total | 216 | 57 | |
| Sex | |||
| Male (%) | 99(45.8) | 32(56.1) | |
| Female (%) | 117(54.2) | 25(43.9) | |
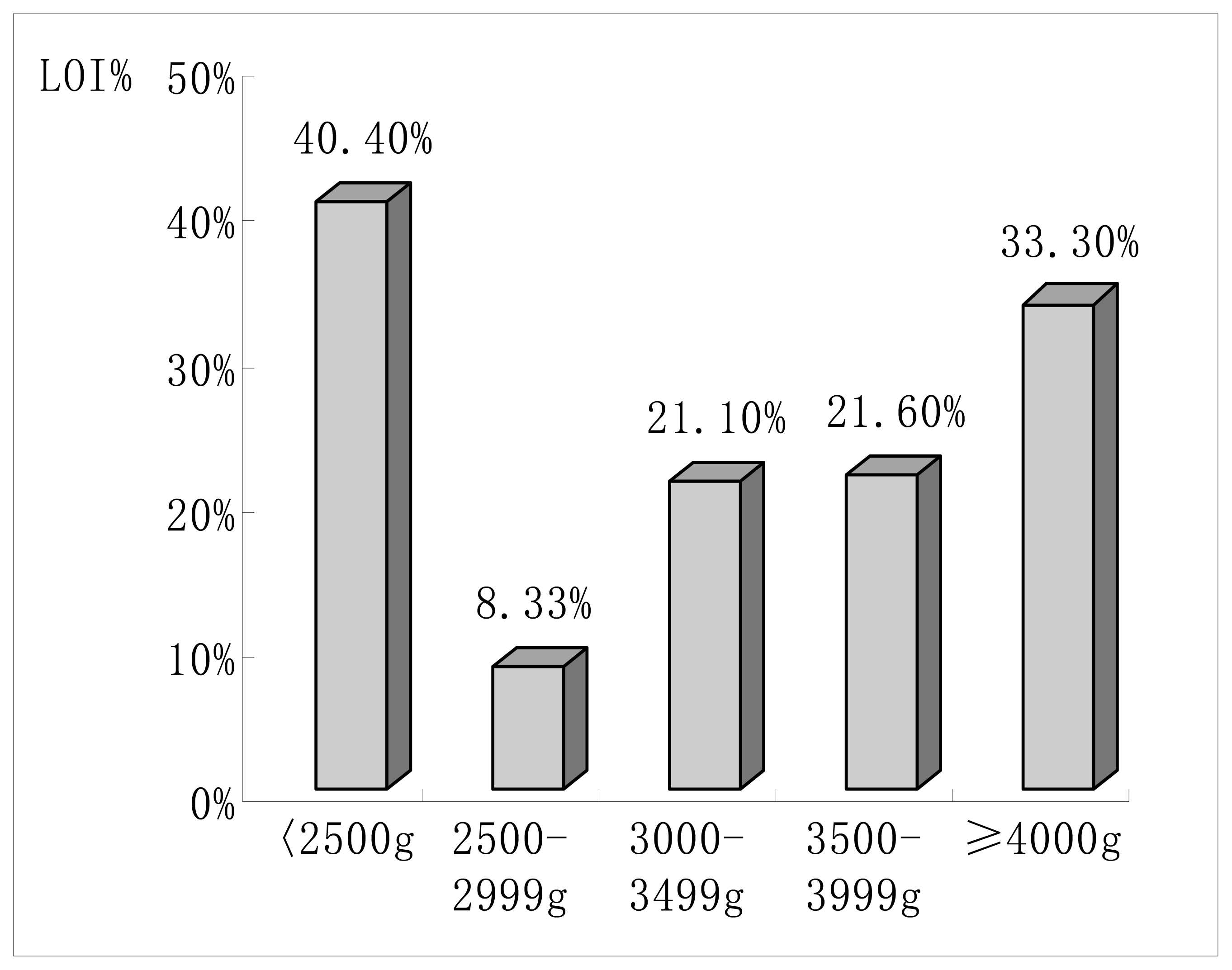
| Birth weight distribution (%) | |||
| < 2500g | 3(1.4) | 2(3.5) | 0.288 |
| 2500 – 2999g | 33(15.3) | 3(5.3) | 0.046a |
| 3000 – 3499g | 97(44.9) | 26(45.6) | 0.836 |
| 3500 – 3999g | 69(31.9) | 19(33.3) | 0.839 |
| ≥ 4000g | 14(6.5) | 7(12.3) | 0.159 |
| Physical constitution (mean ± s.e.m.) | |||
| Mean Birth weight | 3363.7 ± 427.7 | 3462.7 ± 460.2 | 0.127 |
| BMI (kg/m2) | 13.56 ± 1.34 | 13.61 ± 1.32 | 0.542 |
| Sitting height (cm) | 32.1 ± 1.93 | 32.2 ± 1.74 | 0.892 |
| Head circumference (cm) | 33.7 ± 1.42 | 33.7 ± 1.55 | 0.963 |
| Chest circumference (cm) | 32.8 ± 2.3 | 33.0 ± 1.63 | 0.450 |
| Shoulders radical lines (cm) | 12.4 ± 0.97 | 12.5 ± 0.96 | 0.608 |
| Abdominal perimeter (cm) | 33.2 ± 2.02 | 33.8 ± 2.27 | 0.053 |
| Other newborn diseases and defects (%) | |||
| Fetal distress | 27(47.4) | 9(15.8) | 0.514 |
| Asphyxia | 5(8.8) | 1(1.8) | 0.797 |
| Jaundice | 31(14.4) | 10(17.5) | 0.548 |
| Phenylketonuria | 0 | 0 | |
| Congenital hypothyrosis | 0 | 0 | |
Acknowledgement
References
- Feinberg, A.P.; Tycko, B. The history of cancer epigenetics. Nat. Rev. Cancer 2004, 4, 143–153. [Google Scholar]
- Hernandez, L.; Kozlov, S.; Piras, G.; Stewart, C.L. Paternal and maternal genomes confer opposite effects on proliferation, cell-cycle length, senescence, and tumor formation. Proc. Natl. Acad. Sci. USA 2003, 100, 13344–13349. [Google Scholar]
- Fukuzawa, R.; Breslow, N.E.; Morison, I.M.; Dwyer, P.; Kusafuka, T.; Kobayashi, Y.; Becroft, D.M.; Beckwith, J.B.; Perlman, E.J.; Reeve, A.E. Epigenetic differences between Wilms’ tumours in white and east-Asian children. Lancet 2004, 363, 446–451. [Google Scholar]
- Ravenel, J.D.; Broman, K.W.; Perlman, E.J.; Niemitz, E.L.; Jayawardena, T.M.; Bell, D.W.; Haber, D.A.; Uejima, H.; Feinberg, A.P. Loss of imprinting of insulin-like growth factor-II (IGF2) gene in distinguishing specific biologic subtypes of Wilms tumor. J.Natl. Cancer Inst 2001, 93, 1698–1703. [Google Scholar]
- Barton, S.C.; Surani, M.A.; Norris, M.L. Role of paternal and maternal genomes in mouse development. Nature 1984, 311, 374–376. [Google Scholar]
- Joyce, J.A.; Lam, W.K.; Catchpoole, D.J.; Jenks, P.; Reik, W.; Maher, E.R.; Schofield, P.N. Imprinting of IGF2 and H19: lack of reciprocity in sporadic Beckwith-Wiedemann syndrome. Hum. Mol. Genet 1997, 6, 1543–1548. [Google Scholar]
- Cui, H.; Horon, I.L.; Ohlsson, R.; Hamilton, S.R.; Feinberg, A.P. Loss of imprinting in normal tissue of colorectal cancer patients with microsatellite instability. Nat. Med 1998, 4, 1276–1280. [Google Scholar]
- Constancia, M.; Hemberger, M.; Hughes, J.; Dean, W.; Ferguson-Smith, A.; Fundele, R.; Stewart, F.; Kelsey, G.; Fowden, A.; Sibley, C.; Reik, W. Placental-specific IGF-II is a major modulator of placental and fetal growth. Nature 2002, 417, 945–948. [Google Scholar]
- Reik, W.; Constancia, M.; Fowden, A.; Anderson, N.; Dean, W.; Ferguson-Smith, A.; Tycko, B.; Sibley, C. Regulation of supply and demand for maternal nutrients in mammals by imprinted genes. J. Physiol 2003, 547, 35–44. [Google Scholar]
- Cooper, W.N.; Luharia, A.; Evans, G.A.; Raza, H.; Haire, A.C.; Grundy, R.; Bowdin, S.C.; Riccio, A.; Sebastio, G.; Bliek, J.; Schofield, P.N.; Reik, W.; Macdonald, F.; Maher, E.R. Molecular subtypes and phenotypic expression of Beckwith-Wiedemann syndrome. Eur. J. Hum. Genet 2005, 13, 1025–1032. [Google Scholar]
- Feinberg, A.P.; Cui, H.; Ohlsson, R. DNA methylation and genomic imprinting: insights from cancer into epigenetic mechanisms. Semin. Cancer Biol 2002, 12, 389–398. [Google Scholar]
- Cui, H.; Cruz-Correa, M.; Giardiello, F.M.; Hutcheon, D.F.; Kafonek, D.R.; Brandenburg, S.; Wu, Y.; He, X.; Powe, N.R.; Feinberg, A.P. Loss of IGF2 imprinting: a potential marker of colorectal cancer risk. Science 2003, 299, 1753–1755. [Google Scholar]
- Vorwerk, P.; Wex, H.; Bessert, C.; Hohmann, B.; Schmidt, U.; Mittler, U. Loss of imprinting of IGF-II gene in children with acute lymphoblastic leukemia. Leuk. Res 2003, 27, 807–812. [Google Scholar]
- Cruz-Correa, M.; Cui, H.; Giardiello, F.M.; Powe, N.R.; Hylind, L.; Robinson, A.; Hutcheon, D.F.; Kafonek, D.R.; Brandenburg, S.; Wu, Y.; He, X.; Feinberg, A.P. Loss of imprinting of insulin growth factor II gene: a potential heritable biomarker for colon neoplasia predisposition. Gastroenterology 2004, 126, 964–970. [Google Scholar]
- Woodson, K.; Flood, A.; Green, L.; Tangrea, J.A.; Hanson, J.; Cash, B.; Schatzkin, A.; Schoenfeld, P. Loss of insulin-like growth factor-II imprinting and the presence of screen-detected colorectal adenomas in women. J. Natl. Cancer Inst 2004, 96, 407–410. [Google Scholar]
- Hofmann, W.K.; Takeuchi, S.; Frantzen, M.A.; Hoelzer, D.; Koeffler, H.P. Loss of genomic imprinting of insulin-like growth factor 2 is strongly associated with cellular proliferation in normal hematopoietic cells. Exp. Hematol 2002, 30, 318–323. [Google Scholar]
- Sakatani, T.; Kaneda, A.; Iacobuzio-Donahue, C.A.; Carter, M.G.; de Boom Witzel, S.; Okano, H.; Ko, M.S.; Ohlsson, R.; Longo, D.L.; Feinberg, A.P. Loss of imprinting of Igf2 alters intestinal maturation and tumorigenesis in mice. Science 2005, 307, 1976–1978. [Google Scholar]
- Giacosa, A.; Franceschi, S.; La Vecchia, C.; Favero, A.; Andreatta, R. Energy intake, overweight, physical exercise and colorectal cancer risk. Eur. J. Cancer. Prev 1999, 8 Suppl 1, S53–60. [Google Scholar]
- Manousos, O.; Souglakos, J.; Bosetti, C.; Tzonou, A.; Chatzidakis, V.; Trichopoulos, D.; Adami, H.O.; Mantzoros, C. IGF-I and IGF-II in relation to colorectal cancer. Int. J. Cancer 1999, 83, 15–17. [Google Scholar]
- DeBaun, M.R.; King, A.A.; White, N. Hypoglycemia in Beckwith-Wiedemann syndrome. Semin. Perinatol 2000, 24, 164–171. [Google Scholar]
- Hussain, K.; Cosgrove, K.E.; Shepherd, R.M.; Luharia, A.; Smith, V.V.; Kassem, S.; Gregory, J.W.; Sivaprasadarao, A.; Christesen, H.T.; Jacobsen, B.B.; Brusgaard, K.; Glaser, B.; Maher, E.A.; Lindley, K.J.; Hindmarsh, P.; Dattani, M.; Dunne, M.J. Hyperinsulinemic hypoglycemia in Beckwith-Wiedemann syndrome due to defects in the function of pancreatic beta-cell adenosine triphosphate-sensitive potassium channels. J. Clin. Endocrinol. Metab 2005, 90, 4376–4382. [Google Scholar]
- Waterland, R.A.; Lin, J.R.; Smith, C.A.; Jirtle, R.L. Post-weaning diet affects genomic imprinting at the insulin-like growth factor 2 (Igf2) locus. Hum Mol Genet 2006, 15, 705–716. [Google Scholar]
- Sato, A.; Otsu, E.; Negishi, H.; Utsunomiya, T.; Arima, T. Aberrant DNA methylation of imprinted loci in superovulated oocytes. Hum. Reprod 2007, 22, 26–35. [Google Scholar]
- Miller, D.; Ostermeier, G.C. Spermatozoal RNA: Why is it there and what does it do? Gynecol. Obstet. Fertil 2006, 34, 840–846. [Google Scholar]
© 2007 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Dai, Y.; Wang, Z.; Li, J.; Gu, X.; Zheng, M.; Zhou, J.; Ye, X.; Yao, J.; Cui, I.; Hu, Y.; et al. Imprinting Status of IGF2 in Cord Blood Cells of Han Chinese Newborns. Int. J. Mol. Sci. 2007, 8, 273-283. https://doi.org/10.3390/i8040273
Dai Y, Wang Z, Li J, Gu X, Zheng M, Zhou J, Ye X, Yao J, Cui I, Hu Y, et al. Imprinting Status of IGF2 in Cord Blood Cells of Han Chinese Newborns. International Journal of Molecular Sciences. 2007; 8(4):273-283. https://doi.org/10.3390/i8040273
Chicago/Turabian StyleDai, Yimin, Zhiqun Wang, Jie Li, Xiangfang Gu, Mingming Zheng, Jianjun Zhou, Xiaodong Ye, Jincui Yao, Isabelle Cui, Yali Hu, and et al. 2007. "Imprinting Status of IGF2 in Cord Blood Cells of Han Chinese Newborns" International Journal of Molecular Sciences 8, no. 4: 273-283. https://doi.org/10.3390/i8040273




