Rheological Behaviors of Polyacrylonitrile/1-Butyl-3- Methylimidazolium Chloride Concentrated Solutions
Abstract
:1. Introduction
2. Experimental
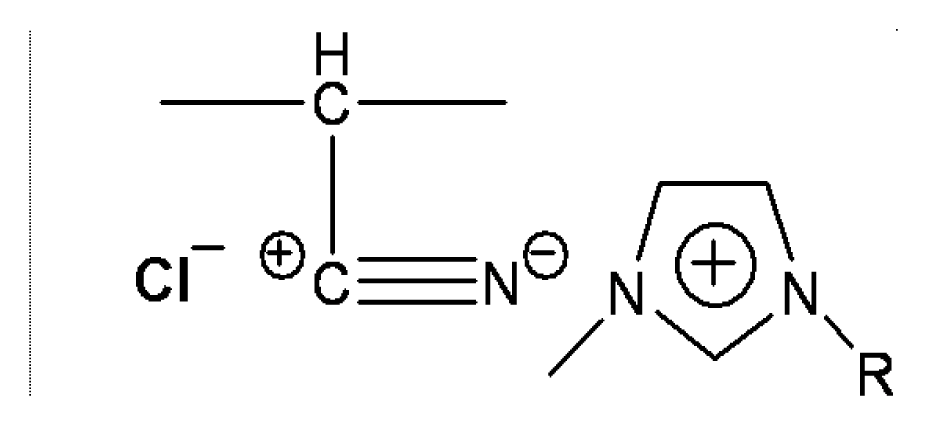
2.1 Materials
2.2 Preparation of concentrated solutions
2.3 Rheometry
2.4 Results and discussion
Conclusions

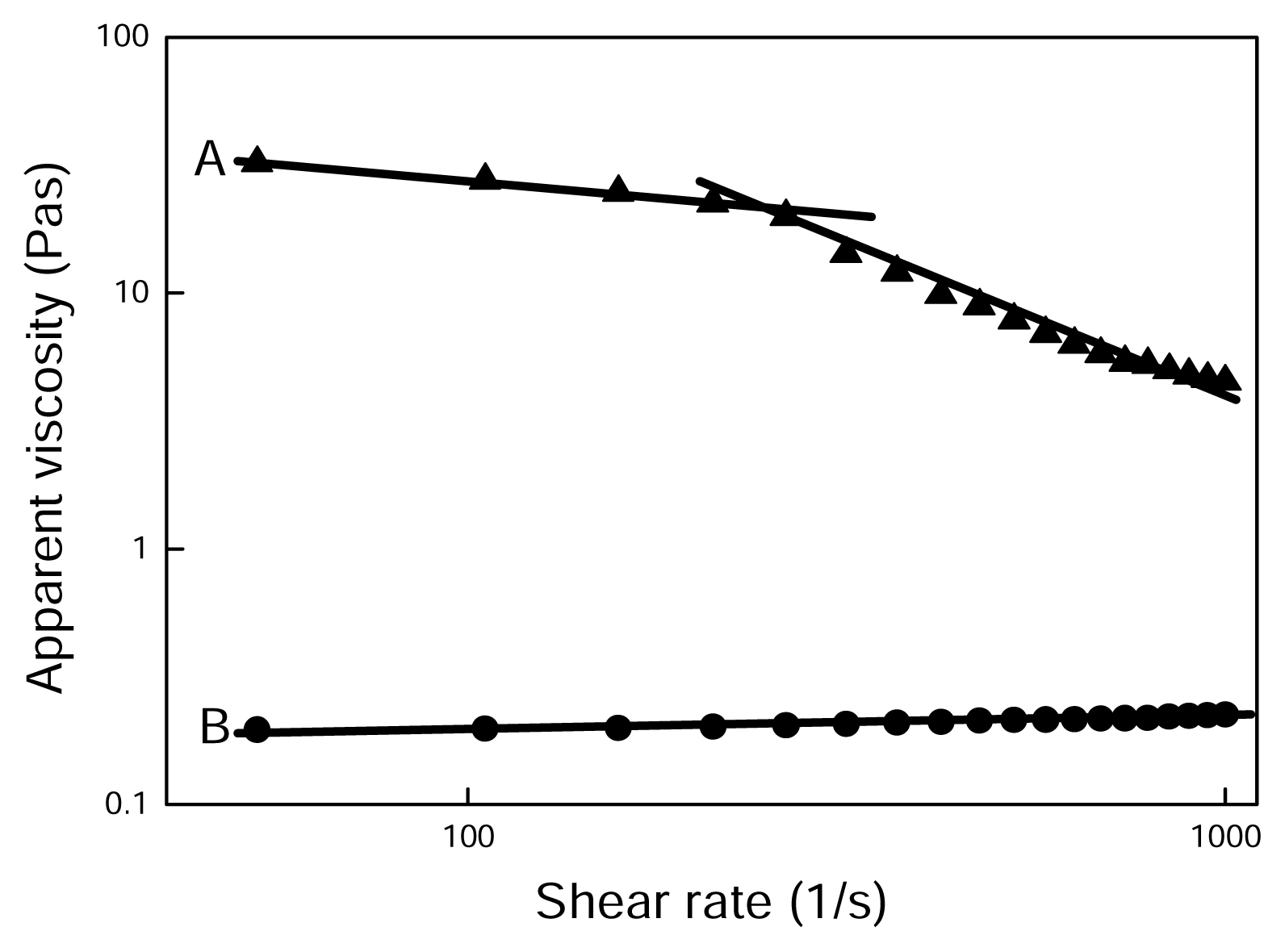
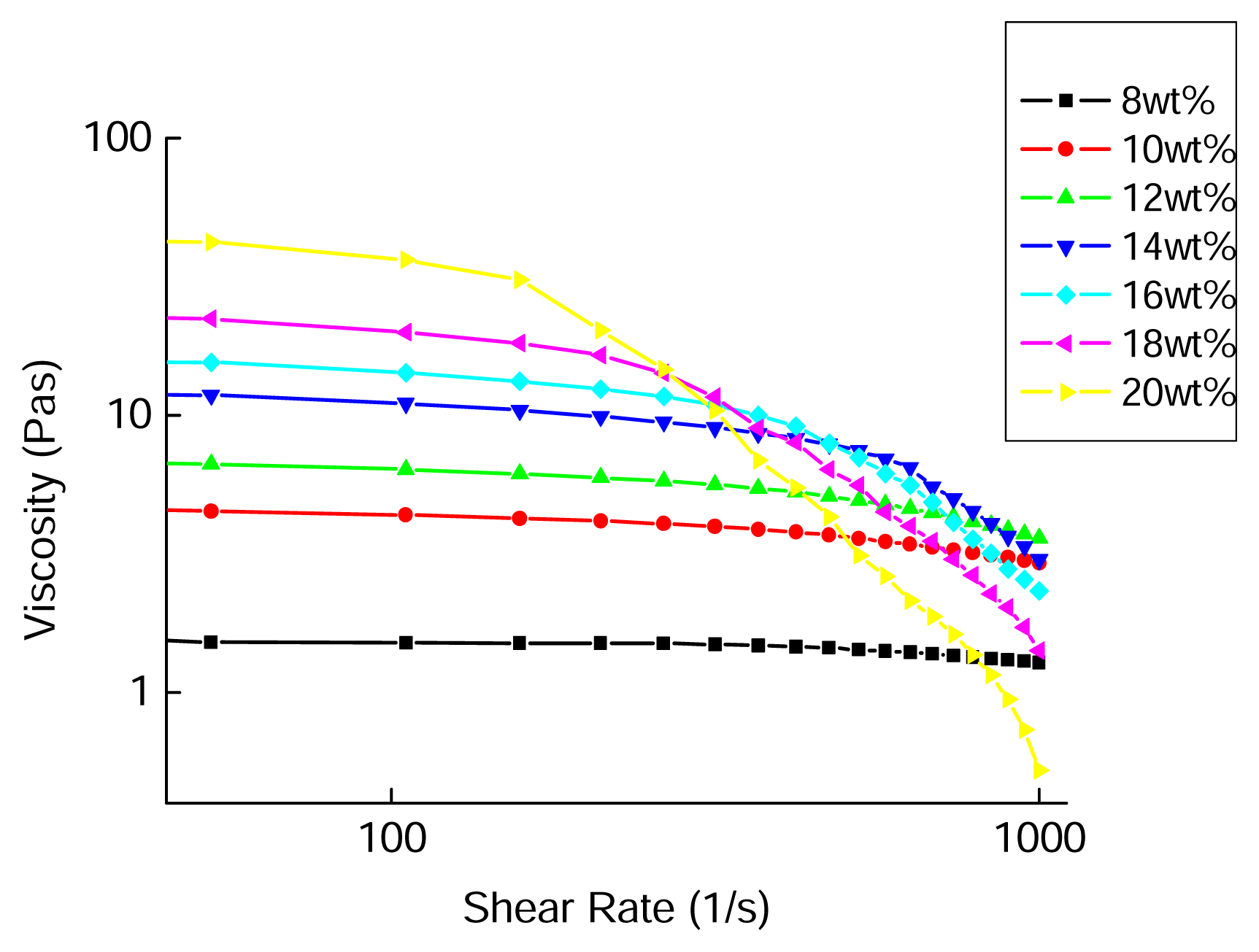
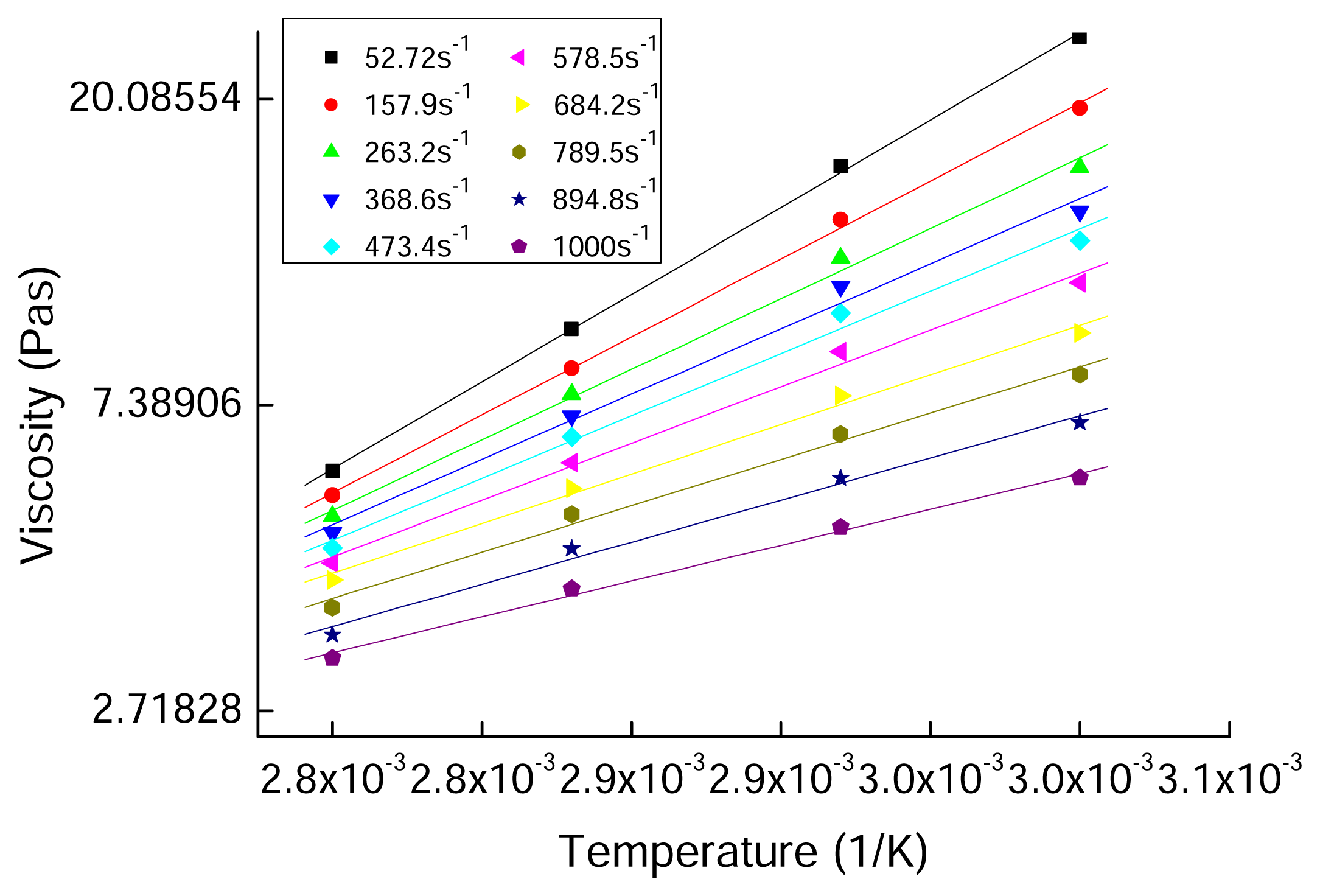
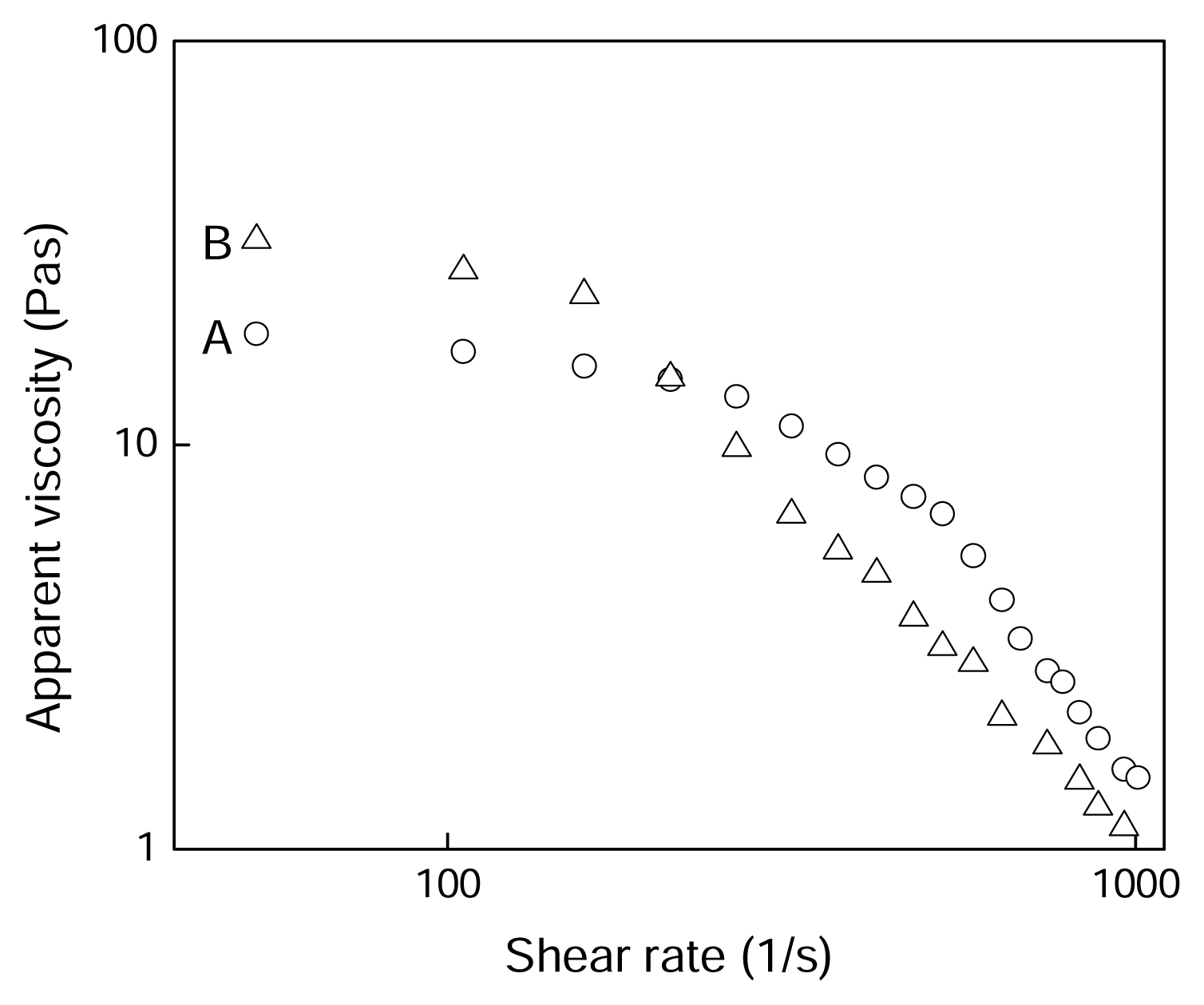


| PAN/[BMIM]Cl | n | R2 |
|---|---|---|
| Stage1 | 0.74504 | 0.993 |
| Stage2 | −0.08613 | 0.981 |
Acknowledgements
References and Notes
- Minagawa, M.; Takasu, T.; Morita, T.; Shirai, H.; Fujikura, Y.; Kameda, Y. The steric effect of solvent molecules in the dissolution of polyacrylonitrile from five different N,N-dimethylformamide derivatives as studied using Raman spectroscopy. Polymer 1996, 37, 463–467. [Google Scholar]
- Rogers, R.D.; Sedden, K.R. Ionic Liquids-Solvents of the Future. Science 2003, 302, 792–793. [Google Scholar]
- Hurley, F.H.; Wier, T.P. Electrodeposition of Metals from Fused Quaternary Ammonium Salts. J. Electrochem. SOC 1951, 98, 203–206. [Google Scholar]
- Welton, T. Room-Temperature Ionic Liquids. Solvents for Synthesis and Catalysis. Chem. Rev 1999, 99, 2071–2082. [Google Scholar]
- Chiappe, C.; Pieraccini, D. Ionic Liquids: Solvent Properties and Organic Reactivity. J .Phys .Org. Chem 2005, 18, 275–297. [Google Scholar]
- Kubisa, P. Application of Ionic Liquids as Solvents for Polymerization Processes. Prog. Polym. Sci 2004, 29, 3–12. [Google Scholar]
- Carlin, R.T.; Osteryoung, R.A.; Wilkes, J.S.; Rovang, J. Studies of Titanium(IV) Chloride in a Strongly Lewis Acidic Molten Salt: Electrochemistry and Titanium NMR and Electronic Spectroscopy. Inorg. Chem 1990, 29, 3003–3009. [Google Scholar]
- Huddleston, J.G.; Willauer, H.D.; Swatloski, R.P.; Visser, A.E.; Rogers, R.D. Room Temperature Ionic Liquids as Novel Media for ‘Clean’ Liquid-Liquid Extraction. Chem. Commun 1998, 16, 1765–1766. [Google Scholar]
- Najdanovic-Visak, V.; Esperanca, J.M.; Rebelo, L.P.; Nunes da Ponte, M.; Guedes, H.J.; Seddon, K.R.; Szydlowski, J. Phase Behaviour of Room Temperature Ionic Liquid Solutions: An Unusually Large Co-Solvent Effect in (Water + Ethanol). Phys. Chem. Chem. Phys 2002, 4, 1701–1703. [Google Scholar]
- Tanaka, H.; Gomez, M.A.; Tonelli, A.E.; Chichester-Hicks, S.V.; Haddon, R.C.; Bell, T.; Bell, A.T. Studies of the phase transitions in poly[bis(4-ethylphenoxy)phosphazene] and poly[bis(4-tertbutylphenoxy)phosphazene]. 1. Phosphorus-31 NMR study. Macromolecules 1989, 22(3), 1031–1036. [Google Scholar]
- Marwanta, E.; Mizumo, T.; Nakamura, N.; Ohno, H. Improved ionic conductivity of nitrile rubber/ionic liquid composites. Polymer 2005, 46, 3795–3800. [Google Scholar]
- Ferry, A.; Edman, L.; Forsyth, M.; MacFarlane, D.R.; Sun, J. NMR and Raman studies of a novel fast-ion-conducting polymer-in-salt electrolyte based on LiCF3SO3 and PAN. Electrochim. Acta 2000, 45, 1237–1242. [Google Scholar]
© 2007 by MDPI Reproduction is permitted for noncommercial purposes.
Share and Cite
Liu, W.; Cheng, L.; Zhang, H.; Zhang, Y.; Wang, H.; Yu, M. Rheological Behaviors of Polyacrylonitrile/1-Butyl-3- Methylimidazolium Chloride Concentrated Solutions. Int. J. Mol. Sci. 2007, 8, 180-188. https://doi.org/10.3390/i8030180
Liu W, Cheng L, Zhang H, Zhang Y, Wang H, Yu M. Rheological Behaviors of Polyacrylonitrile/1-Butyl-3- Methylimidazolium Chloride Concentrated Solutions. International Journal of Molecular Sciences. 2007; 8(3):180-188. https://doi.org/10.3390/i8030180
Chicago/Turabian StyleLiu, Weiwei, Lingyan Cheng, Hongyan Zhang, Yumei Zhang, Huaping Wang, and Mingfang Yu. 2007. "Rheological Behaviors of Polyacrylonitrile/1-Butyl-3- Methylimidazolium Chloride Concentrated Solutions" International Journal of Molecular Sciences 8, no. 3: 180-188. https://doi.org/10.3390/i8030180




