Self-Organizing Molecular Field Analysis on a New Series of COX-2 Selective Inhibitors: 1,5-Diarylimidazoles
Abstract
:1. Introduction
2. Computational Methods
2.1. Data sets and biological activities
2.2. Molecular modeling and docking
| compda | R | -Log(IC50) | R | compda | -Log(IC50) |
|---|---|---|---|---|---|
| ZA1 | 4-F | 7.8539 | ZA16 | 4-OMe-2-F | 7.8239 |
| ZA2 | 3-F | 7.1871 | ZA17 | 3,4-diCl | 8.2218 |
| ZA3 | 2-F | 7.5528 | ZA18 | 4-OMe-3-F | 8.3979 |
| ZA4 | H | 6.9101 | ZA19 | 4-Me-3-F | 8.2218 |
| ZA5 | 4-Cl | 7.7447 | ZA20 | 4-OMe-3-Me | 7.8861 |
| ZA6 | 4-Me | 7.7959 | ZA21 | 4-Me-3-OMe | 7.8239 |
| ZA7 | 4-OMe | 7.9586 | ZA22 | 4-Cl-3-Me | 7.9586 |
| ZA8 | 4-OEt | 8.3979 | ZA23 | 4-NMe-3-Cl | 7.5686 |
| ZA9 | 4-Pr | 7.0000 | ZA24 | 4-OMe-3-Cl | 8.0969 |
| ZA10 | 4-Pr' | 7.4089 | ZA25 | 4-OEt-3-Cl | 8.1549 |
| ZA11 | 4-SMe | 6.9586 | ZA26 | 4-OEt-3-F | 7.602 |
| ZA12 | 4-SEt | 7.2757 | ZA27 | 4-F-3-OMe | 7.7959 |
| ZA13 | 4-NH2 | 6.7282 | ZA28 | 4-OMe-3,5-diCl | 8.1549 |
| ZA14 | 4-NEt2 | 7.0969 | ZA29 | 3,5-diF | 7.1249 |
| ZA15 | 2,4-diF | 8.1549 |

2.3. SOMFA 3D-QSAR models
| Model No | Alignment | Charge | Resolution of grid (Å) |
|---|---|---|---|
| 1 | a | AM1 | 0.5 |
| 2 | a | AM1 | 1 |
| 3 | a | MNDO | 0.5 |
| 4 | a | MNDO | 1 |
| 5 | a | PM3 | 0.5 |
| 6 | a | PM3 | 1 |
| 7 | b | AM1 | 0.5 |
| 8 | b | AM1 | 1 |
| 9 | b | MNDO | 0.5 |
| 10 | b | MNDO | 1 |
| 11 | b | PM3 | 0.5 |
| 12 | b | PM3 | 1 |
| Model | c1 | r2 | s | F | rCV2 |
|---|---|---|---|---|---|
| 1 | 0.685 | 0.478 | 0.351 | 24.711 | 0.455 |
| 2 | 0.679 | 0.464 | 0.356 | 23.342 | 0.439 |
| 3 | 0.765 | 0.458 | 0.358 | 22.858 | 0.434 |
| 4 | 0.752 | 0.446 | 0.361 | 21.736 | 0.421 |
| 5 | 0.633 | 0.503 | 0.342 | 27.316 | 0,473 |
| 6 | 0.658 | 0.479 | 0.351 | 24.835 | 0.451 |
| 7 | 0.500 | 0.525 | 0.335 | 29.820 | 0.486 |
| 8 | 0.514 | 0.532 | 0.333 | 30.680 | 0.500 |
| 9 | 0.512 | 0.546 | 0.327 | 32.515 | 0.507 |
| 10 | 0.506 | 0.544 | 0.328 | 32.149 | 0.500 |
| 11 | 0.644 | 0.498 | 0.344 | 26.826 | 0.458 |
| 12 | 0.645 | 0.497 | 0.344 | 26.716 | 0.464 |
| Compd | Observed | Predicted | Residuala | Compd | Observed | Predicted | Residuala |
|---|---|---|---|---|---|---|---|
| 1 | 7.85 | 7.46 | 0.40 | 12 | 7.28 | 7.43 | -0.16 |
| 2 | 7.19 | 7.57 | -0.38 | 13 | 6.73 | 7.42 | -0.70 |
| 3 | 7.55 | 7.70 | -0.15 | 14 | 7.10 | 7.12 | -0.02 |
| 4 | 6.91 | 7.11 | -0.20 | 15 | 8.15 | 7.94 | 0.21 |
| 5 | 7.75 | 8.00 | -0.25 | 16 | 7.82 | 7.70 | 0.13 |
| 6 | 7.80 | 7.57 | 0.23 | 17 | 8.22 | 7.79 | 0.43 |
| 7 | 7.96 | 7.69 | 0.27 | 18 | 8.40 | 8.15 | 0.25 |
| 8 | 8.40 | 7.76 | 0.64 | 19 | 8.22 | 8.21 | 0.01 |
| 9 | 7.00 | 7.18 | -0.18 | 20 | 7.89 | 7.73 | 0.16 |
| 10 | 7.41 | 7.32 | 0.09 | 21 | 7.82 | 7.58 | 0.24 |
| 11 | 6.96 | 7.41 | -0.45 | 22 | 7.96 | 8.02 | -0.06 |
| Compd | Observed | Predicted | Residuala | Compd | Observed | Predicted | Residuala |
|---|---|---|---|---|---|---|---|
| 1 | 7.59 | 8.02 | -0.43 | 5 | 7.80 | 7.92 | -0.13 |
| 2 | 8.10 | 8.11 | -0.01 | 6 | 8.15 | 7.66 | 0.50 |
| 3 | 8.15 | 7.82 | 0.34 | 7 | 7.13 | 7.66 | -0.53 |
| 4 | 7.60 | 7.83 | -0.23 |
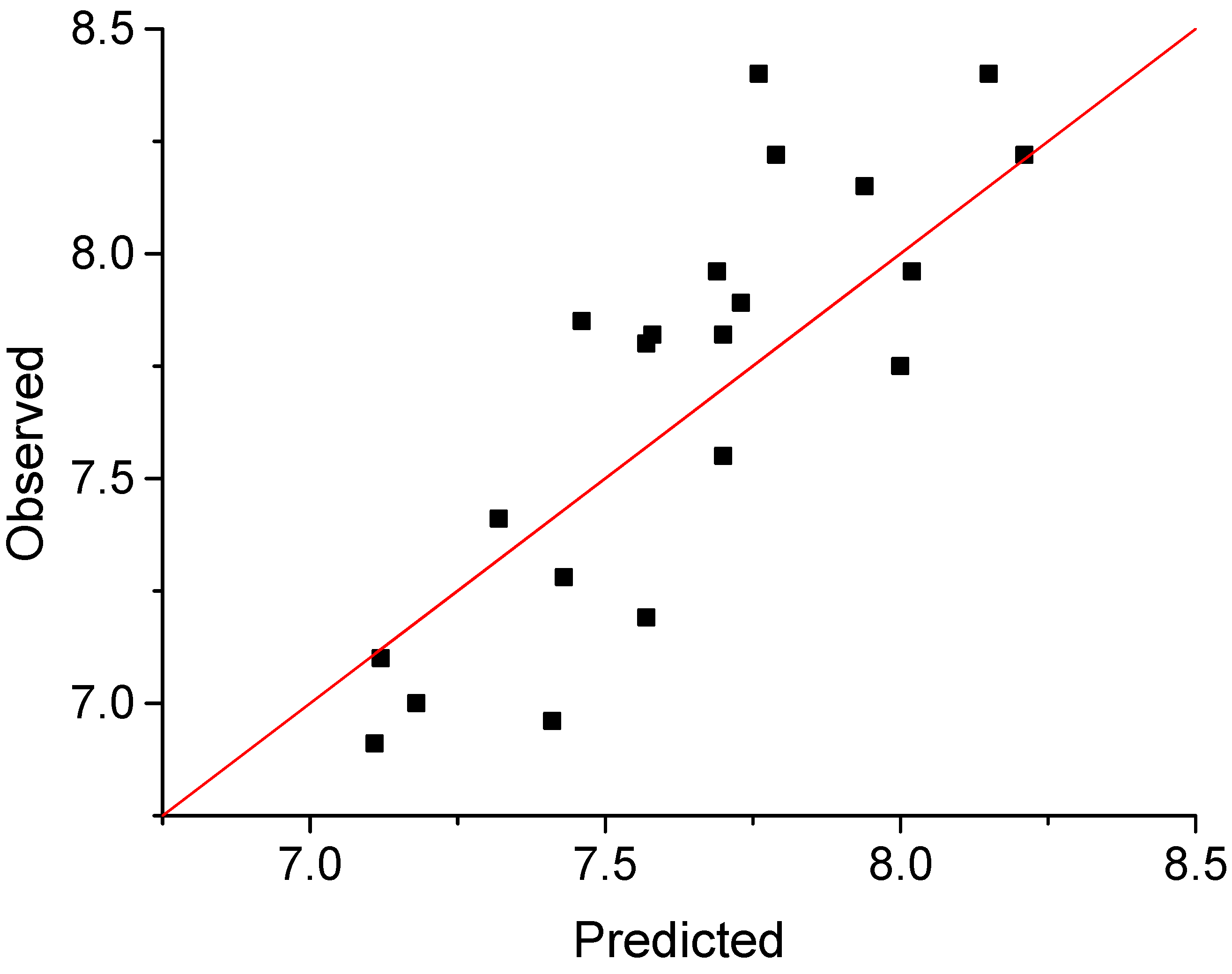
3. Results and Discussion
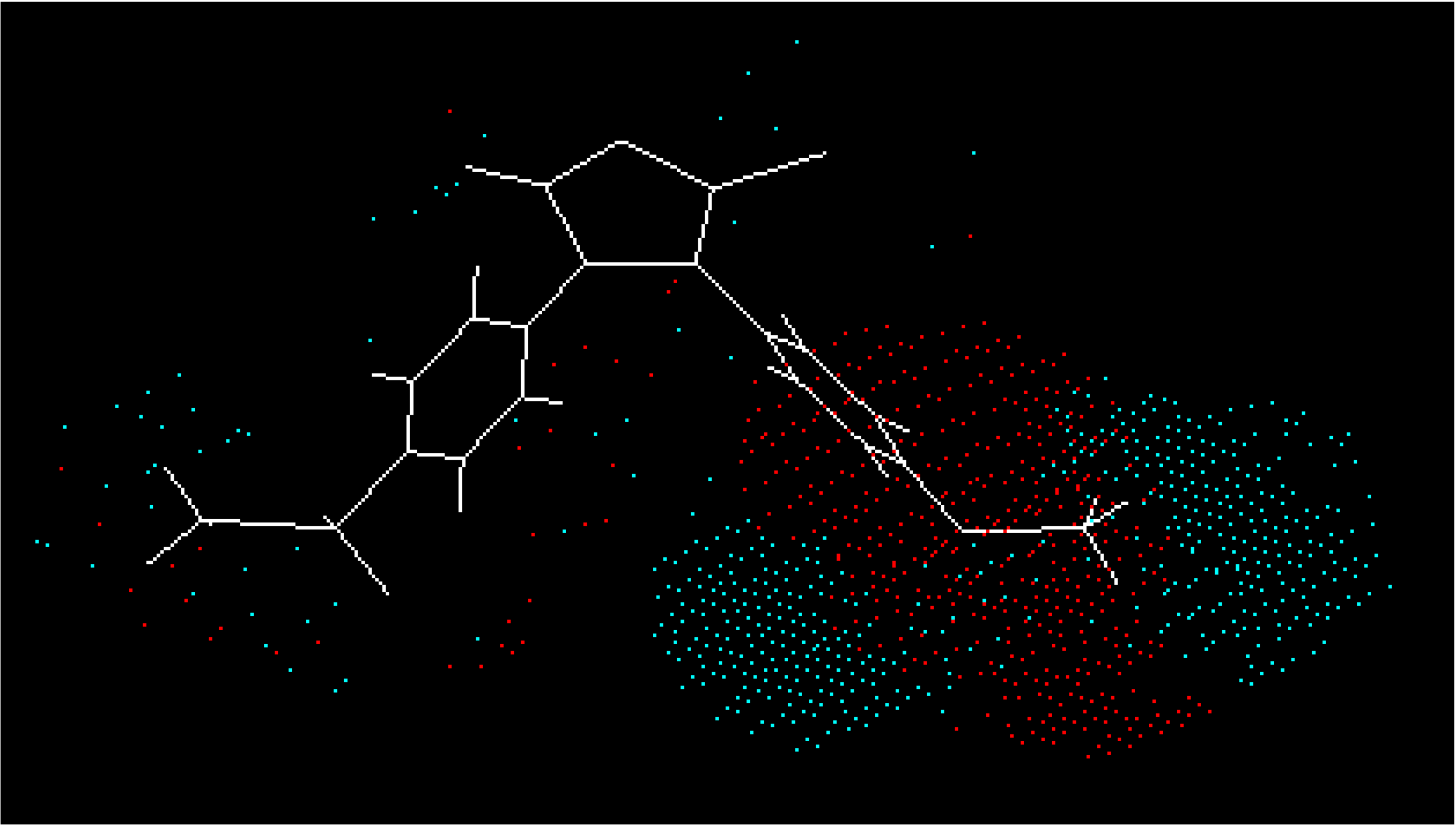
4.Conclusion
Acknowledgements
References and notes
- Vane, J. R.; Botting, R. M. Mechanism of action of anti-inflammatory drugs. Scand. J. Rheumatol. Suppl. 1996, 102, 9–21. [Google Scholar] [CrossRef]
- Dannhardt, G.; Kiefer, W. Cyclooxygenase inhibitors–current status and future prospects. Eur. J. Med. Chem. 2001, 36(2), 109–126. [Google Scholar] [CrossRef]
- Picot, D.; Loll, P. J.; Garavito, R. M. The X-ray crystal structure of the membrane protein prostaglandin H2 synthase-1. Nature 1994, 367, 243–249. [Google Scholar] [CrossRef]
- Crofford, L. J. COX-1 and COX-2 Tissue Expression: Implications and Predictions. J. Rheumatol. 1997, 24 (Suppl 49), 15–19. [Google Scholar]
- Marnett, L. J. Cyclooxygenase mechanisms. Curr. Opin. Chem. Biol. 2000, 4(5), 545–552. [Google Scholar] [CrossRef]
- Laneuville, O.; Breuer, D. K.; Dewitt, D. L.; Hla, T.; Funk, C. D.; Smith, W. L. Differential inhibition of human prostaglandin endoperoxide H synthases-1 and -2 by nonsteroidal anti-inflammatory drugs. J. Pharmacol. Exp. Ther. 1994, 271(2), 927–934. [Google Scholar]
- Marnett, L. J.; Kalgutkar, A. S. Design of selective inhibitors of cyclooxygenase-2 as nonulcerogenic anti-inflammatory agents. Curr. Opin. Chem. Biol. 1998, 2(4), 482–490. [Google Scholar] [CrossRef]
- Carmen, A.; Jose, A.; Alberto, F.; et al. Synthesis and structure-activity relationship of a new series of COX-2 selective inhibitors:1,5-diarylimidazoles. J. Med. Chem. 2003, 46(11), 3463–3475. [Google Scholar] [CrossRef]
- Robinson, D. D.; Winn, P. J.; Lyne, P. D.; Richards, W. G. Self-Organizing Molecular Field Analysis: A Tool for Structure-Activity Studies. J. Med. Chem. 1999, 42(4), 573–583. [Google Scholar](b) SOMFA2 v2.0.0, a novel suite of programs which can carry out an advanced 3-D QSAR analysis of a series of molecules, can be downloaded from author’s homepage: http://bellatrix.pcl.ox.ac.uk. (c)Robinson, D. D. SOMFA2 Instruction Manual; New Chemistry Laboratory: Oxford University, UK, 1999. [Google Scholar]
- Cramer, R. D., III; Patteerson, D. E.; Bunce, J. D. Comparative molecular field analysis (CoMFA). 1. Effect of shape on binding of steroids to carrier proteins. J. Am. Chem. Soc. 1988, 110(18), 5959–5967. [Google Scholar] [CrossRef]
- CAChe 6.1.1. Evaluation. CAChe Group, Fujitsu Limited: Beaverton, USA, 2005.
- Pedretti, A.; Villa, L.; Vistoli, G. VEGA: A Versatile Program to Convert, Handle and Visualize Molecular Structures on Windows-based PCs. J. Mol. Graph. Model. 2002, 21(1), 47–49. [Google Scholar] (b) VEGA ZZ Release 2.0.5.52, a versatile program to convert, handle and visualize molecular structure on Windows-based PCs; its executable and the source code can be free downloaded from: http://www.ddl.unimi.it/vega/index2.htm.
- NoSA V2.30 Build 2003.02.11, a statistics software with simple interface, which can be downloaded from its homepage: http://nosa.diy.myrice.com/ as a shareware.
© 2006 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Li, S.; Zheng, Y. Self-Organizing Molecular Field Analysis on a New Series of COX-2 Selective Inhibitors: 1,5-Diarylimidazoles. Int. J. Mol. Sci. 2006, 7, 220-229. https://doi.org/10.3390/i7070220
Li S, Zheng Y. Self-Organizing Molecular Field Analysis on a New Series of COX-2 Selective Inhibitors: 1,5-Diarylimidazoles. International Journal of Molecular Sciences. 2006; 7(7):220-229. https://doi.org/10.3390/i7070220
Chicago/Turabian StyleLi, Shunlai, and Yan Zheng. 2006. "Self-Organizing Molecular Field Analysis on a New Series of COX-2 Selective Inhibitors: 1,5-Diarylimidazoles" International Journal of Molecular Sciences 7, no. 7: 220-229. https://doi.org/10.3390/i7070220





