A Protons Exchanged Montmorillonite Clay as an Efficient Catalyst for the Reaction of Isobutylene Polymerization
Abstract
:Introduction
Experimental
A. Materials
- 1)
- n-C6H14 was refluxed with fuming sulfuric acid, washed neutral with distilled water, dried over molecular sieves, refluxed and distilled from CaH2 under a nitrogen atmosphere on the day of the experiment.
- 2)
- CH2Cl2 was dried over molecular sieves, distilled over CaH2, refluxed with triethylaluminium under nitrogen atmosphere overnight and freshly distilled on the day of the experiment.
- 3)
- “H-Maghnite xM”: The acid forms of “raw-Maghnite” is prepared by shaking the material raw (raw-Maghnite) with solution of sulfuric acid until saturation achieved over two days at room temperature, washing the mineral with water until sulfate-free and drying. The concentrations 0.05M, 0.10M, 0.15M, 0.20M, 0.25M, 0.30M and 0.35M of sulfuric acid treatment solutions were used to prepare “H-Maghnite0.05M”, “H-Maghnite0.10M”, “H-Maghnite0.15M” , “H-Maghnite0.20M”, “H-Maghnite0.25M”,”H-Maghnite0.30M” and “H-Maghnite0.35M” respectively.
- 4)
- Isobutylene (IB) was dried by passing the gas through a column packed with BaO, and condensing it under a nitrogen atmosphere.
- 5)
- Methanol was used as received
B. “Maghnite” and “H-Maghnite” characterization:
- 1)
- Samples for XRF analysis were prepared using the LiB4O7 fusion method. The resulting beads were analyzed on a Philips PW 2400XRF spectrometer in Laboratory of Inorganic Chemistry, Granada University, Spain
- 2)
- XRD profiles for pressed powder samples were recorded on a Philips PW 1710 diffractometer using Cu-Kα radiation (λ=1.5418A°)
- 3)
- IR absorption spectra were recorded on a ATI Matson FTIR N°9501165 spectrometer using the KBr pressed-disc technique, 0.5mg of sample was added to 300mg KBr and mixed for 3min in a vibratory grinder prior to pressing a 13mm disc.
- 4)
- High-resolution solid-state 29Si and 27Al MAS NMR spectra of untreated (raw-Maghnite) and acid treated (H-Maghnite0.25M) samples were recorded on a Brüker ASX 500 spectrometer at 59.6 and 130.3 MHz respectively. The sample spinning frequency was 4 KHz for 29Si and 11.5 KHz for 27Al.
C. Procedure and Polymer characterization
Results and Discussion
Catalyst Structure
| sample | Compositions wt% | IB conversion % | ||||||||||
| SiO2 | Al2O3 | Fe2O3 | CaO | MgO | Na2O | K2O | TiO2 | SO3 | As | PF* | ||
| Raw-Maghnite | 69.39 | 14.67 | 1.16 | 0.30 | 1.07 | 0.50 | 0.79 | 0.16 | 0.91 | 0.05 | 11 | 00 |
| H-Mag0.05M | 70.75 | 14.67 | 1.05 | 0.30 | 1.01 | 0.49 | 0.78 | 0.16 | 0.75 | 0.04 | 10 | 2 |
| H-Mag0.10M | 71.00 | 14.60 | 1.00 | 0.30 | 0.98 | 0.39 | 0.78 | 0.16 | 0.55 | 0.04 | 10.2 | 3.21 |
| H-Mag0.15M | 71.58 | 14.45 | 0.95 | 0.29 | 0.91 | 0.35 | 0.77 | 0.15 | 0.42 | 0.03 | 10.1 | 10 |
| H-Mag0.20M | 71.65 | 14.20 | 0.80 | 0.28 | 0.85 | 0.30 | 0.77 | 0.15 | 0.39 | 0.01 | 10.6 | 13.7 |
| H-Mag0.25M | 71.70 | 14.03 | 0.71 | 0.28 | 0.80 | 0.21 | 0.77 | 0.15 | 0.34 | 0.01 | 11 | 65 |
| H-Mag0.30M | 73.20 | 13.85 | 0.70 | 0.27 | 0.78 | 0.20 | 0.76 | 0.13 | 0.31 | 0.02 | 9.78 | 30 |
| H-Mag0.35M | 75.31 | 13.52 | 0.71 | 0.26 | 0.78 | 0.18 | 0.75 | 0.13 | 0.32 | 0.01 | 8.03 | 27 |
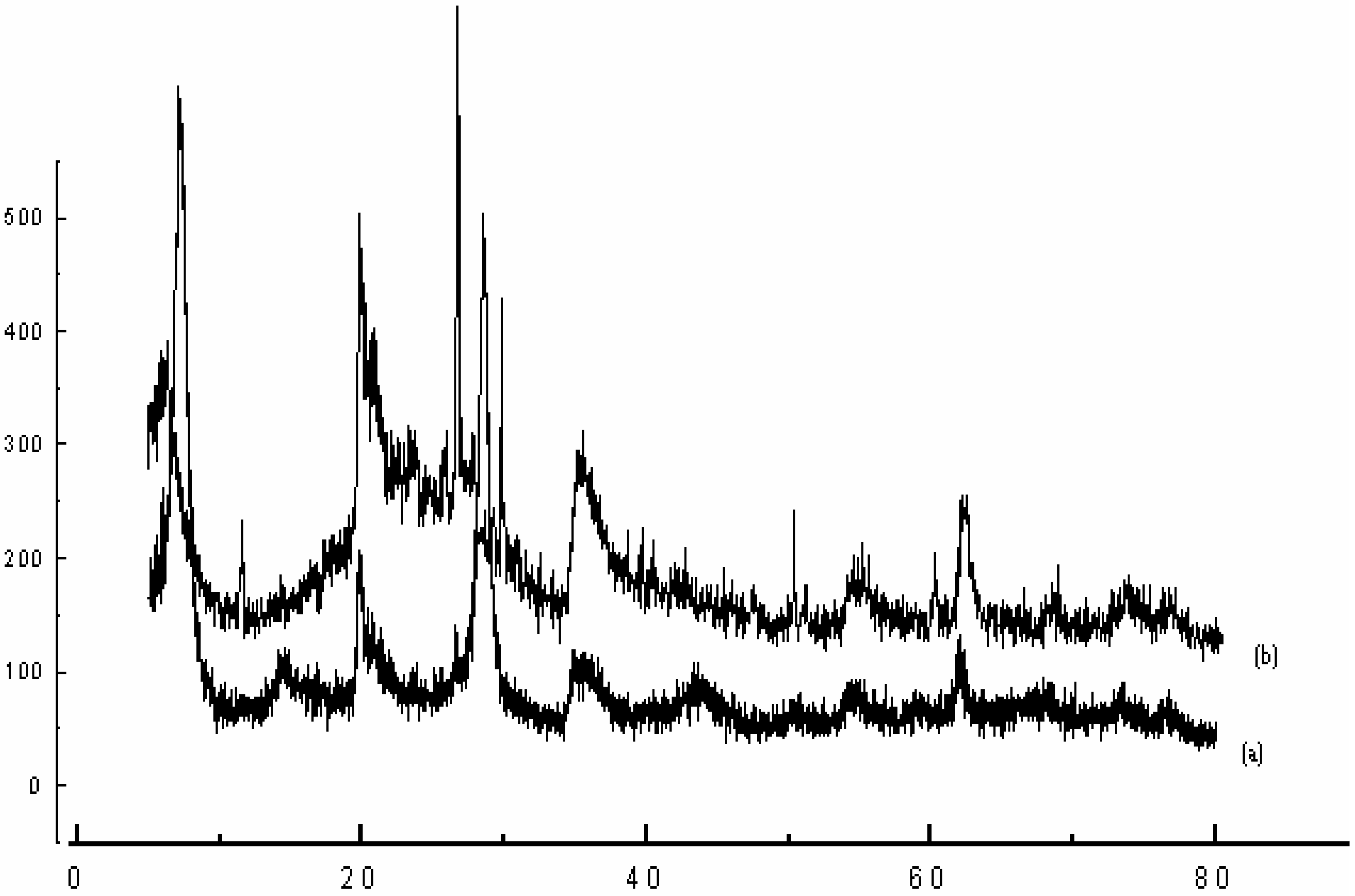
| samples | dhkl (Å) | hkl | Nature of sample |
| Raw-Maghnite | 12.50 | 001 | Montmorillonite |
| 4.47 | 110 | Montmorillonite | |
| 4.16 | ,, | Quartz | |
| 3.35 | ,, | Quartz | |
| 3.21 | ,, | Feldspath | |
| 3.03 | ,, | Calcite | |
| 2.55 | 200 | Montmorillonite | |
| 1.68 | 009 | Montmorillonite | |
| 1.49 | 060 | Montmorillonite | |
| H-Maghnite0.25M | 15.02 | 001 | Montmorillonite |
| 4.47 | 110 | Montmorillonite | |
| 4.16 | ,, | Quartz | |
| 3.35 | ,, | Quartz | |
| 3.21 | ,, | Feldspath | |
| 3.03 | ,, | Calcite | |
| 2.55 | 200 | Montmorillonite | |
| 1.68 | 009 | Montmorillonite | |
| 1.49 | 060 | Montmorillonite |



Polymerization and Products characterization
| Experiment | Solvent | IB g | [IB] mol/l | “H-Maghnite0.25M”(g) | Yield % | Mn | Mw | Mw/Mn |
|---|---|---|---|---|---|---|---|---|
| 1 | Bulk | 10 | - | 0.5 | 47 | 728 | 3269 | 4.49 |
| 2 | Bulk | 10 | - | 1.0 | 65 | 713 | 3020 | 4.24 |
| 3 | Bulk | 10 | - | 1.5 | 72 | 520 | 2410 | 4.63 |
| 4 | CH2Cl2 | 10 | 2 | 1.0 | 37 | 3270 | 3839 | 1.17 |
| 5 | C6H14 | 10 | 2 | 1.0 | 50 | 3690 | 4291 | 1.16 |
Effect of “H-Maghnite0.25M” proportion
| Time (minutes) | 0 | 30 | 60 | 90 | 120 | 150 | 180 | 240 | 300 | |
| Yield% | a | 0 | 15 | 25 | 35 | 38 | 43 | 47 | 54 | 56 |
| b | 0 | 30 | 40 | 47 | 55 | 60 | 65 | 75 | 83 | |
| c | 0 | 35 | 48 | 55 | 63 | 68 | 72 | 80 | 81 | |
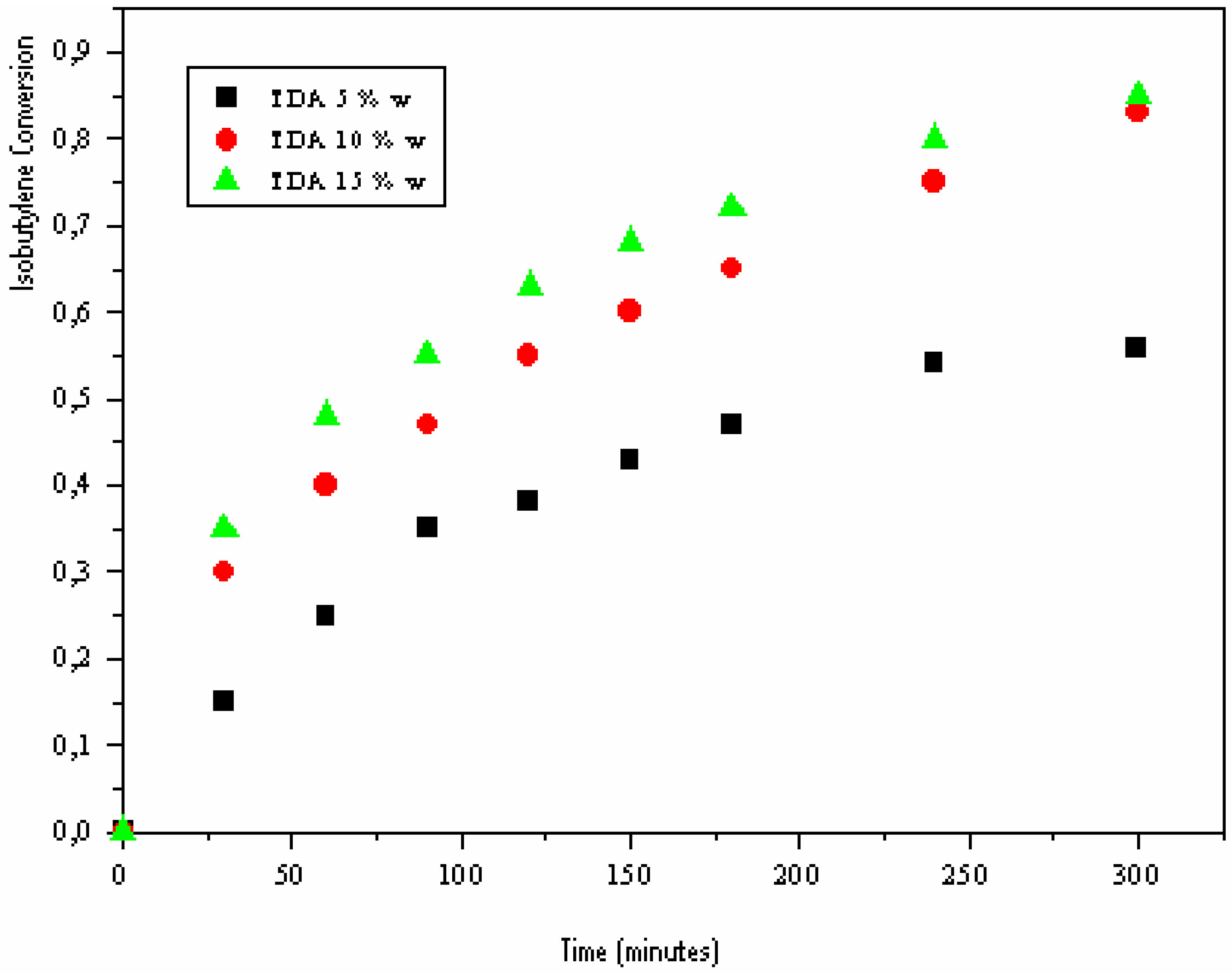
Effect of solvent:
Characterization of products
| Proton type | (a) | (b) | (c) | (d) | (e) | (f) |
| δ in ppm | 0.99 | 1.11 | 1.41 | 4.64 | 4.84 | 5.14 |
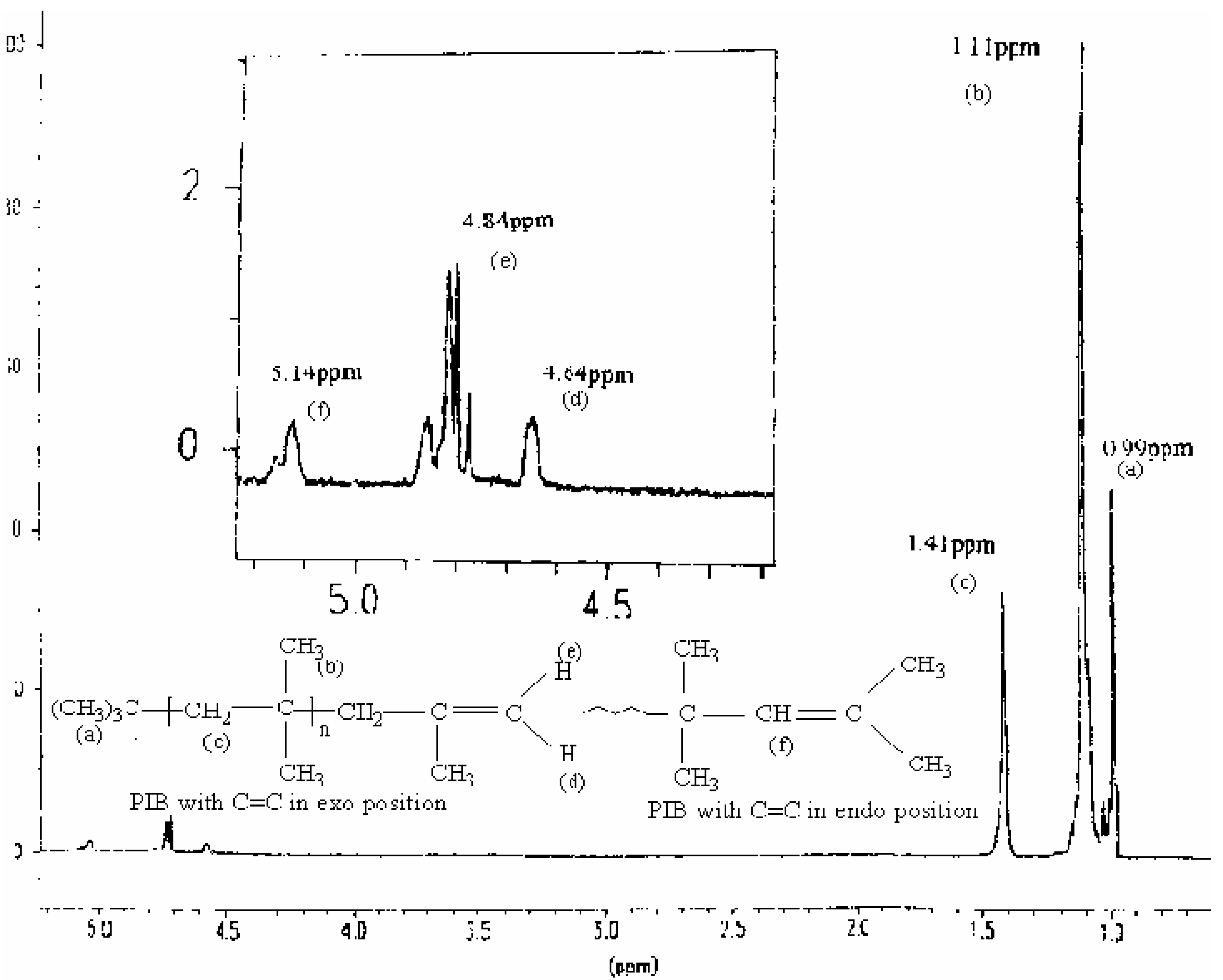

Mechanism of the reaction


- Transfer to monomer:
- Transfer to initiator:
Conclusion
References
- Thomas, C.L.; Hickey, J.; Stecker, G. Ind. Eng. Chem. 1950, 42, 866. [CrossRef]
- Kaplan, H. U.S.Patent. 3287422, 1966.
- Hojabri, F. J. Appl. Chem. Biotechnol. 1971, 21, 87. [CrossRef]
- Ballantine, J.A.; Davies, M.; Purnell, H. Chemical conversions using sheet silicates: novel interlamellar dehydrations of alcohols to ethers and polymers. J. C. S. Chem. Comm. 1981, 427–428. [Google Scholar] [CrossRef]
- Odian, G. La Polymerisation: principes et Applications; Ed.Technica: New York, 1994; pp. 222–226. [Google Scholar]
- Breen, C.; Madejovà, J.; Komadel, P. J.Mater.Chem 1995, 5(3), 496–474.
- Farmer, V.C. Infrared Spectra of Minerals; Farmer, V.C., Ed.; Mineralogical Society: London, 1974; p. 331. [Google Scholar]
- Moeke, H.H.W. Infrared Spectra of Minerals; Farmer, V.C., Ed.; Mineralogical Society: London, 1974; p. 365. [Google Scholar]
- Madejovà, J.; Bednànikovà, E.; Komadel, P.; Cicel, B. Proc.11th Conf. Chem. Miner. Petrol. Ceske Budéjovica 1990; Konta, J., Ed.; Charles University: Prague, 1993; p. 267. [Google Scholar]
- Komarneni, S.; Fyfe, C.A.; Kennedy, G. J.; Strobhl, H. J. Am. Ceram. Soc. 1986, 69, 645.
- Samajovà, E.; Kraus, I.; Lajcàkovà, A. Geol. Carpath. Ser. Clays 1992, 42, 21.
- Thompson, J.G. Clay Miner 1984, 19, 169.
- Tkàc, I.; Komadel, P.; Müle, D. Clay Miner 1994, 29, 11.
- Francis, S.A.; Archer, E.D. Anal.Chem. 1963, 35, 1363–1369. [CrossRef]
- Stanley, L. M.; Ingham, J. D.; Miler, J. A. J. Org. Magn. Reson. 1977, 10, 198. [CrossRef]
© 2002 by MDPI (http://www.mdpi.org).
Share and Cite
Harrane, A.; Meghabar, R.; Belbachir, M. A Protons Exchanged Montmorillonite Clay as an Efficient Catalyst for the Reaction of Isobutylene Polymerization. Int. J. Mol. Sci. 2002, 3, 790-800. https://doi.org/10.3390/i3070790
Harrane A, Meghabar R, Belbachir M. A Protons Exchanged Montmorillonite Clay as an Efficient Catalyst for the Reaction of Isobutylene Polymerization. International Journal of Molecular Sciences. 2002; 3(7):790-800. https://doi.org/10.3390/i3070790
Chicago/Turabian StyleHarrane, Amine, Rachid Meghabar, and Mohammed Belbachir. 2002. "A Protons Exchanged Montmorillonite Clay as an Efficient Catalyst for the Reaction of Isobutylene Polymerization" International Journal of Molecular Sciences 3, no. 7: 790-800. https://doi.org/10.3390/i3070790







