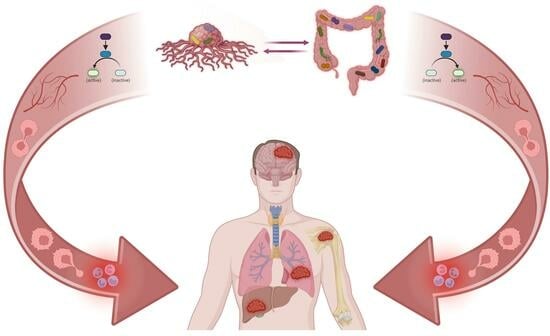
Exploring the Role of the Gut and Intratumoral Microbiomes in Tumor Progression and Metastasis
Abstract
:1. Introduction
2. The Mechanisms of Tumor Progression and Metastasis
3. The Relationship between Microbiome and Cancer Progression-Related Processes
3.1. Microbiome in Epithelial-to-Mesenchymal Transition
3.2. Microbiome and Angiogenesis
4. The Studies of Microbiome Composition in Metastatic Disease
4.1. Gastrointestinal Malignancies
4.2. Lung Cancer
4.3. Breast Cancer
4.4. Head and Neck Cancers
4.5. Genitourinary Cancers
4.6. Metastatic Melanoma
5. Microbiota Modulation and Cancer Progression
6. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sevcikova, A.; Izoldova, N.; Stevurkova, V.; Kasperova, B.; Chovanec, M.; Ciernikova, S.; Mego, M. The Impact of the Microbiome on Resistance to Cancer Treatment with Chemotherapeutic Agents and Immunotherapy. Int. J. Mol. Sci. 2022, 23, 488. [Google Scholar] [CrossRef] [PubMed]
- Cullin, N.; Azevedo Antunes, C.; Straussman, R.; Stein-Thoeringer, C.K.; Elinav, E. Microbiome and cancer. Cancer Cell 2021, 39, 1317–1341. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D. Hallmarks of Cancer: New Dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef] [PubMed]
- Massague, J.; Ganesh, K. Metastasis-Initiating Cells and Ecosystems. Cancer Discov. 2021, 11, 971–994. [Google Scholar] [CrossRef] [PubMed]
- Gerstberger, S.; Jiang, Q.; Ganesh, K. Metastasis. Cell 2023, 186, 1564–1579. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Yu, B.; Rao, B.; Sun, Y.; Yu, J.; Wang, D.; Cui, G.; Ren, Z. The effect of the intratumoral microbiome on tumor occurrence, progression, prognosis and treatment. Front. Immunol. 2022, 13, 1051987. [Google Scholar] [CrossRef] [PubMed]
- Galeano Nino, J.L.; Wu, H.; LaCourse, K.D.; Kempchinsky, A.G.; Baryiames, A.; Barber, B.; Futran, N.; Houlton, J.; Sather, C.; Sicinska, E.; et al. Effect of the intratumoral microbiota on spatial and cellular heterogeneity in cancer. Nature 2022, 611, 810–817. [Google Scholar] [CrossRef]
- Liu, J.; Luo, F.; Wen, L.; Zhao, Z.; Sun, H. Current Understanding of Microbiomes in Cancer Metastasis. Cancers 2023, 15, 1893. [Google Scholar] [CrossRef]
- Hofman, P.; Vouret-Craviari, V. Microbes-induced EMT at the crossroad of inflammation and cancer. Gut Microbes 2012, 3, 176–185. [Google Scholar] [CrossRef]
- Caven, L.T.; Brinkworth, A.J.; Carabeo, R.A. Chlamydia trachomatis induces the transcriptional activity of host YAP in a Hippo-independent fashion. Front. Cell Infect. Microbiol. 2023, 13, 1098420. [Google Scholar] [CrossRef]
- Fu, A.; Yao, B.; Dong, T.; Chen, Y.; Yao, J.; Liu, Y.; Li, H.; Bai, H.; Liu, X.; Zhang, Y.; et al. Tumor-resident intracellular microbiota promotes metastatic colonization in breast cancer. Cell 2022, 185, 1356–1372.e1326. [Google Scholar] [CrossRef] [PubMed]
- Brandt, S.; Kwok, T.; Hartig, R.; Konig, W.; Backert, S. NF-κB activation and potentiation of proinflammatory responses by the Helicobacter pylori CagA protein. Proc. Natl. Acad. Sci. USA 2005, 102, 9300–9305. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Grabowska, A.M.; Clarke, P.A.; Whelband, E.; Robinson, K.; Argent, R.H.; Tobias, A.; Kumari, R.; Atherton, J.C.; Watson, S.A. Helicobacter pylori potentiates epithelial:mesenchymal transition in gastric cancer: Links to soluble HB-EGF, gastrin and matrix metalloproteinase-7. Gut 2010, 59, 1037–1045. [Google Scholar] [CrossRef] [PubMed]
- Fares, J.; Fares, M.Y.; Khachfe, H.H.; Salhab, H.A.; Fares, Y. Molecular principles of metastasis: A hallmark of cancer revisited. Signal Transduct. Target. Ther. 2020, 5, 28. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P. Epithelial-mesenchymal transitions in tumour progression. Nat. Rev. Cancer 2002, 2, 442–454. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Antin, P.; Berx, G.; Blanpain, C.; Brabletz, T.; Bronner, M.; Campbell, K.; Cano, A.; Casanova, J.; Christofori, G.; et al. Guidelines and definitions for research on epithelial-mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2020, 21, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, M.; Giordano, A.; Jackson, S.; Hess, K.R.; De Giorgi, U.; Mego, M.; Handy, B.C.; Ueno, N.T.; Alvarez, R.H.; De Laurentiis, M.; et al. Circulating tumor cells as prognostic and predictive markers in metastatic breast cancer patients receiving first-line systemic treatment. Breast Cancer Res. 2011, 13, R67. [Google Scholar] [CrossRef]
- Mego, M.; Karaba, M.; Minarik, G.; Benca, J.; Silvia, J.; Sedlackova, T.; Manasova, D.; Kalavska, K.; Pindak, D.; Cristofanilli, M.; et al. Circulating Tumor Cells with Epithelial-to-mesenchymal Transition Phenotypes Associated with Inferior Outcomes in Primary Breast Cancer. Anticancer Res. 2019, 39, 1829–1837. [Google Scholar] [CrossRef]
- Fridrichova, I.; Kalinkova, L.; Ciernikova, S. Clinical Relevancy of Circulating Tumor Cells in Breast Cancer: Epithelial or Mesenchymal Characteristics, Single Cells or Clusters? Int. J. Mol. Sci. 2022, 23, 12141. [Google Scholar] [CrossRef]
- Kim, M.Y.; Oskarsson, T.; Acharyya, S.; Nguyen, D.X.; Zhang, X.H.; Norton, L.; Massague, J. Tumor self-seeding by circulating cancer cells. Cell 2009, 139, 1315–1326. [Google Scholar] [CrossRef]
- Comen, E.; Norton, L. Self-seeding in cancer. Recent Results Cancer Res. 2012, 195, 13–23. [Google Scholar] [CrossRef]
- Park, J.; Wysocki, R.W.; Amoozgar, Z.; Maiorino, L.; Fein, M.R.; Jorns, J.; Schott, A.F.; Kinugasa-Katayama, Y.; Lee, Y.; Won, N.H.; et al. Cancer cells induce metastasis-supporting neutrophil extracellular DNA traps. Sci. Transl. Med. 2016, 8, 361ra138. [Google Scholar] [CrossRef] [PubMed]
- Cools-Lartigue, J.; Spicer, J.; McDonald, B.; Gowing, S.; Chow, S.; Giannias, B.; Bourdeau, F.; Kubes, P.; Ferri, L. Neutrophil extracellular traps sequester circulating tumor cells and promote metastasis. J. Clin. Investig. 2013, 123, 3446–3458. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Wang, Q.; Shen, X.; Du, J. The emerging role of neutrophil extracellular traps in cancer: From lab to ward. Front. Oncol. 2023, 13, 1163802. [Google Scholar] [CrossRef] [PubMed]
- Adrover, J.M.; McDowell, S.A.C.; He, X.Y.; Quail, D.F.; Egeblad, M. NETworking with cancer: The bidirectional interplay between cancer and neutrophil extracellular traps. Cancer Cell 2023, 41, 505–526. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.; Lee, S.M.L.; Bazhin, A.V.; Guba, M.; Werner, J.; Niess, H. Neutrophil extracellular traps facilitate cancer metastasis: Cellular mechanisms and therapeutic strategies. J. Cancer Res. Clin. Oncol. 2023, 149, 2191–2210. [Google Scholar] [CrossRef]
- Nepali, P.R.; Kyprianou, N. Anoikis in phenotypic reprogramming of the prostate tumor microenvironment. Front. Endocrinol. 2023, 14, 1160267. [Google Scholar] [CrossRef] [PubMed]
- Paoli, P.; Giannoni, E.; Chiarugi, P. Anoikis molecular pathways and its role in cancer progression. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2013, 1833, 3481–3498. [Google Scholar] [CrossRef]
- Pretzsch, E.; Bosch, F.; Neumann, J.; Ganschow, P.; Bazhin, A.; Guba, M.; Werner, J.; Angele, M. Mechanisms of Metastasis in Colorectal Cancer and Metastatic Organotropism: Hematogenous versus Peritoneal Spread. J. Oncol. 2019, 2019, 7407190. [Google Scholar] [CrossRef]
- Peinado, H.; Zhang, H.; Matei, I.R.; Costa-Silva, B.; Hoshino, A.; Rodrigues, G.; Psaila, B.; Kaplan, R.N.; Bromberg, J.F.; Kang, Y.; et al. Pre-metastatic niches: Organ-specific homes for metastases. Nat. Rev. Cancer 2017, 17, 302–317. [Google Scholar] [CrossRef]
- Kaplan, R.N.; Riba, R.D.; Zacharoulis, S.; Bramley, A.H.; Vincent, L.; Costa, C.; MacDonald, D.D.; Jin, D.K.; Shido, K.; Kerns, S.A.; et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 2005, 438, 820–827. [Google Scholar] [CrossRef] [PubMed]
- Huang, H. Matrix Metalloproteinase-9 (MMP-9) as a Cancer Biomarker and MMP-9 Biosensors: Recent Advances. Sensors 2018, 18, 3249. [Google Scholar] [CrossRef] [PubMed]
- Kryczek, I.; Wei, S.; Keller, E.; Liu, R.; Zou, W. Stroma-derived factor (SDF-1/CXCL12) and human tumor pathogenesis. Am. J. Physiol. Cell Physiol. 2007, 292, C987–C995. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Zhao, H.; Chen, H.; Yao, Q. CXCR4 in breast cancer: Oncogenic role and therapeutic targeting. Drug Des. Dev. Ther. 2015, 9, 4953–4964. [Google Scholar] [CrossRef]
- Muller, A.; Homey, B.; Soto, H.; Ge, N.; Catron, D.; Buchanan, M.E.; McClanahan, T.; Murphy, E.; Yuan, W.; Wagner, S.N.; et al. Involvement of chemokine receptors in breast cancer metastasis. Nature 2001, 410, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, R.N.; Rafii, S.; Lyden, D. Preparing the “soil”: The premetastatic niche. Cancer Res. 2006, 66, 11089–11093. [Google Scholar] [CrossRef] [PubMed]
- De Palma, M.; Biziato, D.; Petrova, T.V. Microenvironmental regulation of tumour angiogenesis. Nat. Rev. Cancer 2017, 17, 457–474. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Ji, Y.R.; Lee, Y.M. Crosstalk between angiogenesis and immune regulation in the tumor microenvironment. Arch. Pharm. Res. 2022, 45, 401–416. [Google Scholar] [CrossRef]
- Zirlik, K.; Duyster, J. Anti-Angiogenics: Current Situation and Future Perspectives. Oncol. Res. Treat. 2018, 41, 166–171. [Google Scholar] [CrossRef]
- Ornitz, D.M.; Itoh, N. The Fibroblast Growth Factor signaling pathway. Wiley Interdiscip. Rev. Dev. Biol. 2015, 4, 215–266. [Google Scholar] [CrossRef]
- Harry, J.A.; Ormiston, M.L. Novel Pathways for Targeting Tumor Angiogenesis in Metastatic Breast Cancer. Front. Oncol. 2021, 11, 772305. [Google Scholar] [CrossRef] [PubMed]
- Olejarz, W.; Kubiak-Tomaszewska, G.; Chrzanowska, A.; Lorenc, T. Exosomes in Angiogenesis and Anti-Angiogenic Therapy in Cancers. Int. J. Mol. Sci. 2020, 21, 5840. [Google Scholar] [CrossRef] [PubMed]
- Roche, J. The Epithelial-to-Mesenchymal Transition in Cancer. Cancers 2018, 10, 52. [Google Scholar] [CrossRef] [PubMed]
- Umar, S. Enteric pathogens and cellular transformation: Bridging the gaps. Oncotarget 2014, 5, 6573–6575. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Acloque, H.; Huang, R.Y.; Nieto, M.A. Epithelial-mesenchymal transitions in development and disease. Cell 2009, 139, 871–890. [Google Scholar] [CrossRef] [PubMed]
- Chandrakesan, P.; Roy, B.; Jakkula, L.U.; Ahmed, I.; Ramamoorthy, P.; Tawfik, O.; Papineni, R.; Houchen, C.; Anant, S.; Umar, S. Utility of a bacterial infection model to study epithelial-mesenchymal transition, mesenchymal-epithelial transition or tumorigenesis. Oncogene 2014, 33, 2639–2654. [Google Scholar] [CrossRef] [PubMed]
- Basu, M.; Philipp, L.M.; Baines, J.F.; Sebens, S. The Microbiome Tumor Axis: How the Microbiome Could Contribute to Clonal Heterogeneity and Disease Outcome in Pancreatic Cancer. Front. Oncol. 2021, 11, 740606. [Google Scholar] [CrossRef]
- Wan, G.; Xie, M.; Yu, H.; Chen, H. Intestinal dysbacteriosis activates tumor-associated macrophages to promote epithelial-mesenchymal transition of colorectal cancer. Innate Immun. 2018, 24, 480–489. [Google Scholar] [CrossRef]
- Henstra, C.; van Praagh, J.; Olinga, P.; Nagelkerke, A. The gastrointestinal microbiota in colorectal cancer cell migration and invasion. Clin. Exp. Metastasis 2021, 38, 495–510. [Google Scholar] [CrossRef]
- Wang, Q.; Yu, C.; Yue, C.; Liu, X. Fusobacterium nucleatum produces cancer stem cell characteristics via EMT-resembling variations. Int. J. Clin. Exp. Pathol. 2020, 13, 1819–1828. [Google Scholar]
- Baud, J.; Varon, C.; Chabas, S.; Chambonnier, L.; Darfeuille, F.; Staedel, C. Helicobacter pylori initiates a mesenchymal transition through ZEB1 in gastric epithelial cells. PLoS ONE 2013, 8, e60315. [Google Scholar] [CrossRef] [PubMed]
- Marques, M.S.; Melo, J.; Cavadas, B.; Mendes, N.; Pereira, L.; Carneiro, F.; Figueiredo, C.; Leite, M. Afadin Downregulation by Helicobacter pylori Induces Epithelial to Mesenchymal Transition in Gastric Cells. Front. Microbiol. 2018, 9, 2712. [Google Scholar] [CrossRef] [PubMed]
- Li, W.T.; Iyangar, A.S.; Reddy, R.; Chakladar, J.; Bhargava, V.; Sakamoto, K.; Ongkeko, W.M.; Rajasekaran, M. The Bladder Microbiome Is Associated with Epithelial-Mesenchymal Transition in Muscle Invasive Urothelial Bladder Carcinoma. Cancers 2021, 13, 3649. [Google Scholar] [CrossRef] [PubMed]
- Abd-El-Raouf, R.; Ouf, S.A.; Gabr, M.M.; Zakaria, M.M.; El-Yasergy, K.F.; Ali-El-Dein, B. Escherichia coli foster bladder cancer cell line progression via epithelial mesenchymal transition, stemness and metabolic reprogramming. Sci. Rep. 2020, 10, 18024. [Google Scholar] [CrossRef] [PubMed]
- Katz, J.; Onate, M.D.; Pauley, K.M.; Bhattacharyya, I.; Cha, S. Presence of Porphyromonas gingivalis in gingival squamous cell carcinoma. Int. J. Oral. Sci. 2011, 3, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Abdulkareem, A.A.; Shelton, R.M.; Landini, G.; Cooper, P.R.; Milward, M.R. Periodontal pathogens promote epithelial-mesenchymal transition in oral squamous carcinoma cells in vitro. Cell Adhes. Migr. 2018, 12, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Ha, N.H.; Woo, B.H.; Kim, D.J.; Ha, E.S.; Choi, J.I.; Kim, S.J.; Park, B.S.; Lee, J.H.; Park, H.R. Prolonged and repetitive exposure to Porphyromonas gingivalis increases aggressiveness of oral cancer cells by promoting acquisition of cancer stem cell properties. Tumour Biol. 2015, 36, 9947–9960. [Google Scholar] [CrossRef] [PubMed]
- Sztukowska, M.N.; Ojo, A.; Ahmed, S.; Carenbauer, A.L.; Wang, Q.; Shumway, B.; Jenkinson, H.F.; Wang, H.; Darling, D.S.; Lamont, R.J. Porphyromonas gingivalis initiates a mesenchymal-like transition through ZEB1 in gingival epithelial cells. Cell Microbiol. 2016, 18, 844–858. [Google Scholar] [CrossRef]
- Peng, Z.; Wan, P.; Deng, Y.; Shen, W.; Liu, R. Lipopolysaccharide exacerbates to the migration, invasion, and epithelial-mesenchymal transition of esophageal cancer cells by TLR4/NF-κB axis. Environ. Toxicol. 2023, 38, 1090–1099. [Google Scholar] [CrossRef]
- Nikolaieva, N.; Sevcikova, A.; Omelka, R.; Martiniakova, M.; Mego, M.; Ciernikova, S. Gut Microbiota-MicroRNA Interactions in Intestinal Homeostasis and Cancer Development. Microorganisms 2022, 11, 107. [Google Scholar] [CrossRef]
- Zhu, Z.; Huang, J.; Li, X.; Xing, J.; Chen, Q.; Liu, R.; Hua, F.; Qiu, Z.; Song, Y.; Bai, C.; et al. Gut microbiota regulate tumor metastasis via circRNA/miRNA networks. Gut Microbes 2020, 12, 1788891. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Liu, L.; Li, H.; Qin, H.; Sun, Z. Clinical significance of Fusobacterium nucleatum, epithelial-mesenchymal transition, and cancer stem cell markers in stage III/IV colorectal cancer patients. OncoTargets Ther. 2017, 10, 5031–5046. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Weng, W.; Peng, J.; Hong, L.; Yang, L.; Toiyama, Y.; Gao, R.; Liu, M.; Yin, M.; Pan, C.; et al. Fusobacterium nucleatum Increases Proliferation of Colorectal Cancer Cells and Tumor Development in Mice by Activating Toll-Like Receptor 4 Signaling to Nuclear Factor-κB, and Up-regulating Expression of MicroRNA-21. Gastroenterology 2017, 152, 851–866.e824. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, T.; Miko, E.; Ujlaki, G.; Yousef, H.; Csontos, V.; Uray, K.; Bai, P. The involvement of oncobiosis and bacterial metabolite signaling in metastasis formation in breast cancer. Cancer Metastasis Rev. 2021, 40, 1223–1249. [Google Scholar] [CrossRef] [PubMed]
- Ujlaki, G.; Kovacs, T.; Vida, A.; Kokai, E.; Rauch, B.; Schwarcz, S.; Miko, E.; Janka, E.; Sipos, A.; Hegedus, C.; et al. Identification of Bacterial Metabolites Modulating Breast Cancer Cell Proliferation and Epithelial-Mesenchymal Transition. Molecules 2023, 28, 5898. [Google Scholar] [CrossRef] [PubMed]
- Sari, Z.; Miko, E.; Kovacs, T.; Janko, L.; Csonka, T.; Lente, G.; Sebo, E.; Toth, J.; Toth, D.; Arkosy, P.; et al. Indolepropionic Acid, a Metabolite of the Microbiome, Has Cytostatic Properties in Breast Cancer by Activating AHR and PXR Receptors and Inducing Oxidative Stress. Cancers 2020, 12, 2411. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Cheng, J.; Zhang, J.; Zhou, F.; He, X.; Shi, Y.; Tao, Y. The Role of Respiratory Microbiota in Lung Cancer. Int. J. Biol. Sci. 2021, 17, 3646–3658. [Google Scholar] [CrossRef] [PubMed]
- Castro, P.R.; Bittencourt, L.F.F.; Larochelle, S.; Andrade, S.P.; Mackay, C.R.; Slevin, M.; Moulin, V.J.; Barcelos, L.S. GPR43 regulates sodium butyrate-induced angiogenesis and matrix remodeling. Am. J. Physiol. Heart Circ. Physiol. 2021, 320, H1066–H1079. [Google Scholar] [CrossRef]
- Nguyen, T.T.; Lian, S.; Ung, T.T.; Xia, Y.; Han, J.Y.; Jung, Y.D. Lithocholic Acid Stimulates IL-8 Expression in Human Colorectal Cancer Cells via Activation of Erk1/2 MAPK and Suppression of STAT3 Activity. J. Cell. Biochem. 2017, 118, 2958–2967. [Google Scholar] [CrossRef]
- Saha, S.; Chowdhury, P.; Pal, A.; Chakrabarti, M.K. Downregulation of human colon carcinoma cell (COLO-205) proliferation through PKG-MAP kinase mediated signaling cascade by E. coli heat stable enterotoxin (STa), a potent anti-angiogenic and anti-metastatic molecule. J. Appl. Toxicol. 2008, 28, 475–483. [Google Scholar] [CrossRef]
- Yang, S.; Dai, H.; Lu, Y.; Li, R.; Gao, C.; Pan, S. Trimethylamine N-Oxide Promotes Cell Proliferation and Angiogenesis in Colorectal Cancer. J. Immunol. Res. 2022, 2022, 7043856. [Google Scholar] [CrossRef] [PubMed]
- Salahshouri, P.; Emadi-Baygi, M.; Jalili, M.; Khan, F.M.; Wolkenhauer, O.; Salehzadeh-Yazdi, A. A Metabolic Model of Intestinal Secretions: The Link between Human Microbiota and Colorectal Cancer Progression. Metabolites 2021, 11, 456. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Li, H.; Xie, H.; Wu, X.; Lan, P. The malignant role of exosomes in the communication among colorectal cancer cell, macrophage and microbiome. Carcinogenesis 2019, 40, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.H.; Zhao, L.; Zhang, X.; Nakatsu, G.; Han, J.; Xu, W.; Xiao, X.; Kwong, T.N.Y.; Tsoi, H.; Wu, W.K.K.; et al. Gavage of Fecal Samples from Patients with Colorectal Cancer Promotes Intestinal Carcinogenesis in Germ-Free and Conventional Mice. Gastroenterology 2017, 153, 1621–1633.e1626. [Google Scholar] [CrossRef] [PubMed]
- De Spiegeleer, B.; Verbeke, F.; D’Hondt, M.; Hendrix, A.; Van De Wiele, C.; Burvenich, C.; Peremans, K.; De Wever, O.; Bracke, M.; Wynendaele, E. The quorum sensing peptides PhrG, CSP and EDF promote angiogenesis and invasion of breast cancer cells in vitro. PLoS ONE 2015, 10, e0119471. [Google Scholar] [CrossRef] [PubMed]
- Wynendaele, E.; Verbeke, F.; D’Hondt, M.; Hendrix, A.; Van De Wiele, C.; Burvenich, C.; Peremans, K.; De Wever, O.; Bracke, M.; De Spiegeleer, B. Crosstalk between the microbiome and cancer cells by quorum sensing peptides. Peptides 2015, 64, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Hilmi, M.; Kamal, M.; Vacher, S.; Dupain, C.; Ibadioune, S.; Halladjian, M.; Sablin, M.P.; Marret, G.; Ajgal, Z.C.; Nijnikoff, M.; et al. Intratumoral microbiome is driven by metastatic site and associated with immune histopathological parameters: An ancillary study of the SHIVA clinical trial. Eur. J. Cancer 2023, 183, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Hajjar, J.; Mendoza, T.; Zhang, L.; Fu, S.; Piha-Paul, S.A.; Hong, D.S.; Janku, F.; Karp, D.D.; Ballhausen, A.; Gong, J.; et al. Associations between the gut microbiome and fatigue in cancer patients. Sci. Rep. 2021, 11, 5847. [Google Scholar] [CrossRef]
- Sethi, V.; Kurtom, S.; Tarique, M.; Lavania, S.; Malchiodi, Z.; Hellmund, L.; Zhang, L.; Sharma, U.; Giri, B.; Garg, B.; et al. Gut Microbiota Promotes Tumor Growth in Mice by Modulating Immune Response. Gastroenterology 2018, 155, 33–37.e36. [Google Scholar] [CrossRef]
- Spakowicz, D.; Hoyd, R.; Muniak, M.; Husain, M.; Bassett, J.S.; Wang, L.; Tinoco, G.; Patel, S.H.; Burkart, J.; Miah, A.; et al. Inferring the role of the microbiome on survival in patients treated with immune checkpoint inhibitors: Causal modeling, timing, and classes of concomitant medications. BMC Cancer 2020, 20, 383. [Google Scholar] [CrossRef]
- Dzutsev, A.; Goldszmid, R.S.; Viaud, S.; Zitvogel, L.; Trinchieri, G. The role of the microbiota in inflammation, carcinogenesis, and cancer therapy. Eur. J. Immunol. 2015, 45, 17–31. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Cai, G.; Qiu, Y.; Fei, N.; Zhang, M.; Pang, X.; Jia, W.; Cai, S.; Zhao, L. Structural segregation of gut microbiota between colorectal cancer patients and healthy volunteers. ISME J. 2012, 6, 320–329. [Google Scholar] [CrossRef] [PubMed]
- Viljoen, K.S.; Dakshinamurthy, A.; Goldberg, P.; Blackburn, J.M. Quantitative profiling of colorectal cancer-associated bacteria reveals associations between Fusobacterium spp., enterotoxigenic Bacteroides fragilis (ETBF) and clinicopathological features of colorectal cancer. PLoS ONE 2015, 10, e0119462. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Fan, L.; Lin, Y.; Shen, W.; Qi, Y.; Zhang, Y.; Chen, Z.; Wang, L.; Long, Y.; Hou, T.; et al. Fusobacterium nucleatum promotes colorectal cancer metastasis through miR-1322/CCL20 axis and M2 polarization. Gut Microbes 2021, 13, 1980347. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Su, T.; Zhang, Y.; Lee, A.; He, J.; Ge, Q.; Wang, L.; Si, J.; Zhuo, W.; Wang, L. Fusobacterium nucleatum promotes colorectal cancer metastasis by modulating KRT7-AS/KRT7. Gut Microbes 2020, 11, 511–525. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhang, L.; Li, M.; Zhang, Y.; Sun, M.; Wang, L.; Lin, J.; Cui, Y.; Chen, Q.; Jin, C.; et al. Fusobacterium nucleatum reduces METTL3-mediated m6A modification and contributes to colorectal cancer metastasis. Nat. Commun. 2022, 13, 1248. [Google Scholar] [CrossRef] [PubMed]
- Bullman, S.; Pedamallu, C.S.; Sicinska, E.; Clancy, T.E.; Zhang, X.; Cai, D.; Neuberg, D.; Huang, K.; Guevara, F.; Nelson, T.; et al. Analysis of Fusobacterium persistence and antibiotic response in colorectal cancer. Science 2017, 358, 1443–1448. [Google Scholar] [CrossRef]
- Wang, F.; He, M.M.; Yao, Y.C.; Zhao, X.; Wang, Z.Q.; Jin, Y.; Luo, H.Y.; Li, J.B.; Wang, F.H.; Qiu, M.Z.; et al. Regorafenib plus toripalimab in patients with metastatic colorectal cancer: A phase Ib/II clinical trial and gut microbiome analysis. Cell Rep. Med. 2021, 2, 100383. [Google Scholar] [CrossRef]
- Ambalam, P.; Raman, M.; Purama, R.K.; Doble, M. Probiotics, prebiotics and colorectal cancer prevention. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 119–131. [Google Scholar] [CrossRef]
- Dejea, C.M.; Fathi, P.; Craig, J.M.; Boleij, A.; Taddese, R.; Geis, A.L.; Wu, X.; DeStefano Shields, C.E.; Hechenbleikner, E.M.; Huso, D.L.; et al. Patients with familial adenomatous polyposis harbor colonic biofilms containing tumorigenic bacteria. Science 2018, 359, 592–597. [Google Scholar] [CrossRef]
- Stakelum, A.; Zaborowski, A.; Collins, D.; Winter, D.C. The influence of the gastrointestinal microbiome on colorectal metastasis: A narrative review. Colorectal Dis. 2020, 22, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Wachsmannova, L.; Stevurkova, V.; Ciernikova, S. Changes in SNAI1 and VIM gene expression in Caco2 cells after cocultivation with bacteria from colorectal cancer biopsies. Neoplasma 2019, 66, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Yan, Y.; Chen, D.; Yang, Y. Quxie Capsule Modulating Gut Microbiome and Its Association with T cell Regulation in Patients with Metastatic Colorectal Cancer: Result From a Randomized Controlled Clinical Trial. Integr. Cancer Ther. 2020, 19, 1534735420969820. [Google Scholar] [CrossRef] [PubMed]
- Li, J.J.; Zhu, M.; Kashyap, P.C.; Chia, N.; Tran, N.H.; McWilliams, R.R.; Bekaii-Saab, T.S.; Ma, W.W. The role of microbiome in pancreatic cancer. Cancer Metastasis Rev. 2021, 40, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.Y.; Kim, T.B.; Kim, J.; Choi, H.W.; Kim, E.J.; Yoo, H.J.; Lee, S.; Jun, H.R.; Yoo, W.; Kim, S.; et al. Diversity in the Extracellular Vesicle-Derived Microbiome of Tissues according to Tumor Progression in Pancreatic Cancer. Cancers 2020, 12, 2346. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.; Chen, X.; Hong, L.; Zhao, X.; Cui, G.; Li, A.; Liu, Y.; Zhou, L.; Sun, R.; Shen, S.; et al. Nanoparticle Conjugation of Ginsenoside Rg3 Inhibits Hepatocellular Carcinoma Development and Metastasis. Small 2020, 16, e1905233. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.; Su, X.; Yuan, M.; Zhang, S.; He, J.; Deng, Q.; Qiu, W.; Dong, H.; Cai, S. The characterization of lung microbiome in lung cancer patients with different clinicopathology. Am. J. Cancer Res. 2019, 9, 2047–2063. [Google Scholar]
- Yu, G.; Gail, M.H.; Consonni, D.; Carugno, M.; Humphrys, M.; Pesatori, A.C.; Caporaso, N.E.; Goedert, J.J.; Ravel, J.; Landi, M.T. Characterizing human lung tissue microbiota and its relationship to epidemiological and clinical features. Genome Biol. 2016, 17, 163. [Google Scholar] [CrossRef]
- Gowing, S.D.; Chow, S.C.; Cools-Lartigue, J.J.; Chen, C.B.; Najmeh, S.; Jiang, H.Y.; Bourdeau, F.; Beauchamp, A.; Mancini, U.; Angers, I.; et al. Gram-positive pneumonia augments non-small cell lung cancer metastasis via host toll-like receptor 2 activation. Int. J. Cancer 2017, 141, 561–571. [Google Scholar] [CrossRef]
- Jungnickel, C.; Wonnenberg, B.; Karabiber, O.; Wolf, A.; Voss, M.; Wolf, L.; Honecker, A.; Kamyschnikow, A.; Herr, C.; Bals, R.; et al. Cigarette smoke-induced disruption of pulmonary barrier and bacterial translocation drive tumor-associated inflammation and growth. Am. J. Physiol. Lung Cell Mol. Physiol. 2015, 309, L605–L613. [Google Scholar] [CrossRef]
- Hadzega, D.; Minarik, G.; Karaba, M.; Kalavska, K.; Benca, J.; Ciernikova, S.; Sedlackova, T.; Nemcova, P.; Bohac, M.; Pindak, D.; et al. Uncovering Microbial Composition in Human Breast Cancer Primary Tumour Tissue Using Transcriptomic RNA-seq. Int. J. Mol. Sci. 2021, 22, 9058. [Google Scholar] [CrossRef] [PubMed]
- Buchta Rosean, C.; Bostic, R.R.; Ferey, J.C.M.; Feng, T.Y.; Azar, F.N.; Tung, K.S.; Dozmorov, M.G.; Smirnova, E.; Bos, P.D.; Rutkowski, M.R. Preexisting Commensal Dysbiosis Is a Host-Intrinsic Regulator of Tissue Inflammation and Tumor Cell Dissemination in Hormone Receptor-Positive Breast Cancer. Cancer Res. 2019, 79, 3662–3675. [Google Scholar] [CrossRef] [PubMed]
- Wenhui, Y.; Zhongyu, X.; Kai, C.; Zhaopeng, C.; Jinteng, L.; Mengjun, M.; Zepeng, S.; Yunshu, C.; Peng, W.; Yanfeng, W.; et al. Variations in the Gut Microbiota in Breast Cancer Occurrence and Bone Metastasis. Front. Microbiol. 2022, 13, 894283. [Google Scholar] [CrossRef] [PubMed]
- Parhi, L.; Alon-Maimon, T.; Sol, A.; Nejman, D.; Shhadeh, A.; Fainsod-Levi, T.; Yajuk, O.; Isaacson, B.; Abed, J.; Maalouf, N.; et al. Breast cancer colonization by Fusobacterium nucleatum accelerates tumor growth and metastatic progression. Nat. Commun. 2020, 11, 3259. [Google Scholar] [CrossRef] [PubMed]
- Chiba, A.; Bawaneh, A.; Velazquez, C.; Clear, K.Y.J.; Wilson, A.S.; Howard-McNatt, M.; Levine, E.A.; Levi-Polyachenko, N.; Yates-Alston, S.A.; Diggle, S.P.; et al. Neoadjuvant Chemotherapy Shifts Breast Tumor Microbiota Populations to Regulate Drug Responsiveness and the Development of Metastasis. Mol. Cancer Res. 2020, 18, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Eun, Y.G.; Lee, J.W.; Kim, S.W.; Hyun, D.W.; Bae, J.W.; Lee, Y.C. Oral microbiome associated with lymph node metastasis in oral squamous cell carcinoma. Sci. Rep. 2021, 11, 23176. [Google Scholar] [CrossRef] [PubMed]
- Qiao, H.; Tan, X.R.; Li, H.; Li, J.Y.; Chen, X.Z.; Li, Y.Q.; Li, W.F.; Tang, L.L.; Zhou, G.Q.; Zhang, Y.; et al. Association of Intratumoral Microbiota with Prognosis in Patients with Nasopharyngeal Carcinoma from 2 Hospitals in China. JAMA Oncol. 2022, 8, 1301–1309. [Google Scholar] [CrossRef]
- Yuan, L.; Yang, P.; Wei, G.; Hu, X.; Chen, S.; Lu, J.; Yang, L.; He, X.; Bao, G. Tumor microbiome diversity influences papillary thyroid cancer invasion. Commun. Biol. 2022, 5, 864. [Google Scholar] [CrossRef]
- Peiffer, L.B.; White, J.R.; Jones, C.B.; Slottke, R.E.; Ernst, S.E.; Moran, A.E.; Graff, J.N.; Sfanos, K.S. Composition of gastrointestinal microbiota in association with treatment response in individuals with metastatic castrate resistant prostate cancer progressing on enzalutamide and initiating treatment with anti-PD-1 (pembrolizumab). Neoplasia 2022, 32, 100822. [Google Scholar] [CrossRef]
- Ma, J.; Gnanasekar, A.; Lee, A.; Li, W.T.; Haas, M.; Wang-Rodriguez, J.; Chang, E.Y.; Rajasekaran, M.; Ongkeko, W.M. Influence of Intratumor Microbiome on Clinical Outcome and Immune Processes in Prostate Cancer. Cancers 2020, 12, 2524. [Google Scholar] [CrossRef]
- Salgia, N.J.; Bergerot, P.G.; Maia, M.C.; Dizman, N.; Hsu, J.; Gillece, J.D.; Folkerts, M.; Reining, L.; Trent, J.; Highlander, S.K.; et al. Stool Microbiome Profiling of Patients with Metastatic Renal Cell Carcinoma Receiving Anti-PD-1 Immune Checkpoint Inhibitors. Eur. Urol. 2020, 78, 498–502. [Google Scholar] [CrossRef]
- Agarwal, A.; Modliszewski, J.; Davey, L.; Reyes-Martinez, M.; Runyambo, D.; Corcoran, D.; Dressman, H.; George, D.J.; Valdivia, R.H.; Armstrong, A.J.; et al. Investigating the role of the gastrointestinal microbiome in response to immune checkpoint inhibitors (ICIs) among patients (pts) with metastatic renal cell carcinoma (mRCC). J. Clin. Oncol. 2020, 38, 730. [Google Scholar] [CrossRef]
- Dizman, N.; Hsu, J.; Bergerot, P.G.; Gillece, J.D.; Folkerts, M.; Reining, L.; Trent, J.; Highlander, S.K.; Pal, S.K. Randomized trial assessing impact of probiotic supplementation on gut microbiome and clinical outcome from targeted therapy in metastatic renal cell carcinoma. Cancer Med. 2021, 10, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Hahn, A.W.; Froerer, C.; VanAlstine, S.; Rathi, N.; Bailey, E.B.; Stenehjem, D.D.; Agarwal, N. Targeting Bacteroides in Stool Microbiome and Response to Treatment with First-Line VEGF Tyrosine Kinase Inhibitors in Metastatic Renal-Cell Carcinoma. Clin. Genitourin. Cancer 2018, 16, 365–368. [Google Scholar] [CrossRef] [PubMed]
- Gopalakrishnan, V.; Spencer, C.N.; Nezi, L.; Reuben, A.; Andrews, M.C.; Karpinets, T.V.; Prieto, P.A.; Vicente, D.; Hoffman, K.; Wei, S.C.; et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 2018, 359, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Mizuhashi, S.; Kajihara, I.; Sawamura, S.; Kanemaru, H.; Makino, K.; Aoi, J.; Makino, T.; Masuguchi, S.; Fukushima, S.; Ihn, H. Skin microbiome in acral melanoma: Corynebacterium is associated with advanced melanoma. J. Dermatol. 2021, 48, e15–e16. [Google Scholar] [CrossRef] [PubMed]
- Matson, V.; Fessler, J.; Bao, R.; Chongsuwat, T.; Zha, Y.; Alegre, M.L.; Luke, J.J.; Gajewski, T.F. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science 2018, 359, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Wargo, J.A.; Gopalakrishnan, V.; Spencer, C.; Karpinets, T.; Reuben, A.; Andrews, M.C.; Tetzlaff, M.T.; Lazar, A.; Hwu, P.; Hwu, W.J.; et al. Association of the diversity and composition of the gut microbiome with responses and survival (PFS) in metastatic melanoma (MM) patients (pts) on anti-PD-1 therapy. J. Clin. Oncol. 2017, 15, 3008. [Google Scholar] [CrossRef]
- Vitali, F.; Colucci, R.; Di Paola, M.; Pindo, M.; De Filippo, C.; Moretti, S.; Cavalieri, D. Early melanoma invasivity correlates with gut fungal and bacterial profiles. Br. J. Dermatol. 2022, 186, 106–116. [Google Scholar] [CrossRef]
- Ciernikova, S.; Mego, M.; Hainova, K.; Adamcikova, Z.; Stevurkova, V.; Zajac, V. Modification of microflora imbalance: Future directions for prevention and treatment of colorectal cancer? Neoplasma 2015, 62, 345–352. [Google Scholar] [CrossRef]
- Wang, Z.; Li, L.; Wang, S.; Wei, J.; Qu, L.; Pan, L.; Xu, K. The role of the gut microbiota and probiotics associated with microbial metabolisms in cancer prevention and therapy. Front. Pharmacol. 2022, 13, 1025860. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Hu, M.; Xu, F.; Wu, X.; Dong, D.; Wang, W. Preventive effect of Lactobacillus reuteri on melanoma. Biomed. Pharmacother. 2020, 126, 109929. [Google Scholar] [CrossRef] [PubMed]
- Pereira, F.V.; Melo, A.C.L.; Silva, M.B.; de Melo, F.M.; Terra, F.F.; Castro, I.A.; Perandini, L.A.; Miyagi, M.T.; Sato, F.T.; Origassa, C.S.T.; et al. Interleukin-6 and the Gut Microbiota Influence Melanoma Progression in Obese Mice. Nutr. Cancer 2021, 73, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhou, X.; Wang, Y.; Wang, D.; Ke, Y.; Zeng, X. Propionate and Butyrate Produced by Gut Microbiota after Probiotic Supplementation Attenuate Lung Metastasis of Melanoma Cells in Mice. Mol. Nutr. Food Res. 2021, 65, e2100096. [Google Scholar] [CrossRef] [PubMed]
- Le Noci, V.; Guglielmetti, S.; Arioli, S.; Camisaschi, C.; Bianchi, F.; Sommariva, M.; Storti, C.; Triulzi, T.; Castelli, C.; Balsari, A.; et al. Modulation of Pulmonary Microbiota by Antibiotic or Probiotic Aerosol Therapy: A Strategy to Promote Immunosurveillance against Lung Metastases. Cell Rep. 2018, 24, 3528–3538. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sung, C.Y.; Lee, N.; Ni, Y.; Pihlajamaki, J.; Panagiotou, G.; El-Nezami, H. Probiotics modulated gut microbiota suppresses hepatocellular carcinoma growth in mice. Proc. Natl. Acad. Sci. USA 2016, 113, E1306–E1315. [Google Scholar] [CrossRef] [PubMed]
- Dizman, N.; Meza, L.; Bergerot, P.; Alcantara, M.; Dorff, T.; Lyou, Y.; Frankel, P.; Cui, Y.; Mira, V.; Llamas, M.; et al. Nivolumab plus ipilimumab with or without live bacterial supplementation in metastatic renal cell carcinoma: A randomized phase 1 trial. Nat. Med. 2022, 28, 704–712. [Google Scholar] [CrossRef]
- Jakubauskas, M.; Jakubauskiene, L.; Leber, B.; Horvath, A.; Strupas, K.; Stiegler, P.; Schemmer, P. Probiotic Supplementation Attenuates Chemotherapy-Induced Intestinal Mucositis in an Experimental Colorectal Cancer Liver Metastasis Rat Model. Nutrients 2023, 15, 1117. [Google Scholar] [CrossRef]
- Shang, F.; Jiang, X.; Wang, H.; Chen, S.; Wang, X.; Liu, Y.; Guo, S.; Li, D.; Yu, W.; Zhao, Z.; et al. The inhibitory effects of probiotics on colon cancer cells: In vitro and in vivo studies. J. Gastrointest. Oncol. 2020, 11, 1224–1232. [Google Scholar] [CrossRef]
- Baruch, E.N.; Youngster, I.; Ben-Betzalel, G.; Ortenberg, R.; Lahat, A.; Katz, L.; Adler, K.; Dick-Necula, D.; Raskin, S.; Bloch, N.; et al. Fecal microbiota transplant promotes response in immunotherapy-refractory melanoma patients. Science 2021, 371, 602–609. [Google Scholar] [CrossRef]


| Study | Study Design | Disease | Purpose | Patients (n) | Intervention | Study Status |
|---|---|---|---|---|---|---|
| NCT03941080 | An observational prospective study | Metastatic CRC | To confirm the microbial taxa associated with treatment response and side effects in metastatic or irresectable disease | 300 adults/ older adults | Enrolled patients will be newly diagnosed with an indication for standard palliative systemic treatment. | Recruiting |
| NCT04579484 | An observational prospective study | Metastatic breast cancer | To determine the gut microbiome in fecal samples of patients with ER+ HER2− breast cancer and assess the relationship between dietary factors and microbiome | 20 adults/ older adults | Patients will receive endocrine therapy with an aromatase inhibitor combined with an inhibitor of cyclin-dependent kinases 4 and 6. | Recruiting |
| NCT04804956 | An observational prospective study | Metastatic rectal cancer | To identify the profile of the mesorectal microbiome and correlation with poor prognosis prediction | 100 adults | Participants will receive neoadjuvant treatment. | Recruiting |
| NCT04579978 | An observational prospective study | Metastatic solid cancer | To study changes in the gut microbial community after ICI and evaluate bacterial species associated with treatment efficacy | 60 adults/ older adults | Patients will be enrolled in the study for planned standard-of-care ICI. | Recruiting |
| NCT05878977 | An interventional open-label study | Metastatic melanoma | To define novel markers for the prediction of therapy response | 150 adults/ older adults | Immunotherapy will consist of PD-1 and CTLA-4 inhibitors. | Recruiting |
| NCT05635149 | An observational prospective study | Metastatic CRC | To assess the composition of the gut microbiome and its association with treatment efficacy | 100 adults/ older adults | Patients will be treated with Fruquintinib, ICI plus RT, or Fruquintinib and ICI alone. | Recruiting |
| NCT05753839 | An interventional randomized open-label study with parallel assignment | Metastatic clear cell renal cell carcinoma/kidney cancer | To correlate the gut and urine microbiome compositions with OS, PFS, and ORR | 40 adults/ older adults | Patients will receive ICI followed by maintenance therapy with ICI or cytoreductive nephrectomy ± metastasectomy after ICI. | Recruiting |
| NCT04090710 | An interventional randomized study with parallel assignment | Metastatic renal cell carcinoma | To investigate the changes in the gut microbiome via analysis of stool samples | 78 children/ adults/ older adults | Patients will undergo cytoreductive stereotactic body RT with a combination of ICIs vs. one ICI alone. | Recruiting |
| NCT04243720 | An observational prospective study | Metastatic solid cancer | To determine changes in the gut microbiome associated with resistance to immunotherapy | 100 adults/ older adults | Only participants who progressed on immunotherapy will be enrolled in this study. | Recruiting |
| NCT04148378 | An observational case-only prospective study | CRC neoplasms/ metastatic CRC/ colorectal sarcoma/ adenocarcinoma | To correlate microbiome composition with type of disease | 100 children/ adults/ older adults | There is no intervention for the study. | Unknown |
| NCT04516135 | An interventional randomized open-label study with parallel assignment | Metastatic gynecologic cancers | To describe overall diversity, richness, and specific microbial dynamics in the gut and vaginal microbiomes | 108 adults/ older adults | Females will be treated with 3D conformal RT/intensity-modulated RT/volume-modulated arc therapy at the physician’s discretion for 1 fraction in the absence of RT-induced toxicities or progression. | Recruiting |
| NCT04214015 | An observational case-only prospective study | Metastatic mesothelioma | To analyze the relative abundance of bacterial members in the gut microbiome | 100 children/ adults/ older adults | There is no intervention for the study. | Unknown |
| NCT03818061 | An interventional non-randomized study with parallel assignment | Metastatic HNSCC | To characterize the gut microbiome in immunotherapy using whole-metagenome sequencing | 33 adults/ older adults | Patients with/without human papillomavirus will receive atezolizumab combined with bevacizumab. | Active, but not recruiting |
| NCT03698461 | An interventional open-label study with single-group assignment | Metastatic neoplasms/ colorectal neoplasms/ colonic neoplasms/ rectal neoplasms | To determine fecal microbial profile in different time frames | 20 adults/ older adults | Anti-cancer treatment will consist of atezolizumab with bevacizumab, levoleucovorin, oxaliplatin, and 5-fluorouracil. | Active, but not recruiting |
| NCT03977571 | An interventional randomized open-label study with parallel assignment | Metastatic renal cell carcinoma/ kidney cancer/ synchronous neoplasm | To correlate the gut microbiome with OS, PFS, and ORR | 400 adults/ older adults | Patients will receive deferred cytoreductive nephrectomy/no surgery following nivolumab with ipilimumab or tyrosine kinase inhibitors. | Recruiting |
| NCT04636775 | An observational prospective study | Metastatic non-small-cell lung cancer | To assess the correlations between gut microbiome composition and adverse effects and differences between responders and non-responders | 46 adults/ older adults | Patients will be treated with immunotherapy using ICI. | Recruiting |
| NCT04219137 | An observational prospective study | Metastatic EGA | To study the microbiome in feces and rectal swab samples | 120 adults/ older adults | Participants will undergo platinum-based chemotherapy. | Unknown |
| NCT03161756 | An interventional non-randomized study parallel assignment | Metastatic melanoma | To explore associations between the gut microbiome and therapy response | 72 adults/ older adults | Nivolumab alone or in combination with ipilimumab will be administered intravenously plus denosumab subcutaneously. | Active, but not recruiting |
| NCT04720768 | An interventional open-label study with sequential assignment | Metastatic melanoma | To identify fecal biomarkers associated with therapy response/resistance | 78 adults/ older adults | Patients will receive combined treatment with encorafenib, binimetinib, and palbociclib. | Recruiting |
| NCT03340129 | An interventional randomized open-label study with parallel assignment | Metastatic melanoma | To observe the diversity and composition of the gut microbiome and to determine the correlation between mucosal integrity and microbes | 218 adults/ older adults | Treatment will include ipilimumab and nivolumab with concurrent intracranial stereotactic RT or ipilimumab and nivolumab alone. | Recruiting |
| Malignancy | Study Type Preclinical/Clinical | Intervention | Changes in Microbial Composition | Major Findings | Ref. |
|---|---|---|---|---|---|
| CRC with liver/lung metastases | Patients | Regorafenib plus toripalimab | Fusobacterium, Alistipes, Bilophila, and Acidaminococcus | A higher level of specific bacteria was observed in non-responders. Shorter PFS correlated with a higher amount of Fusobacterium. | [88] |
| FAP | Patients/ mice | No intervention provided | E. coli and ETBF | Both bacterial taxa were biofilm members in FAP tissues from patients. Colonization with E. coli and ETBF increased DNA damage and IL-17 production in carcinogen-treated mice. | [90] |
| CRC with liver/lung metastases | Patients | Quxie capsules | Actinobacteria, Oscillibacter, Eubacterium, and Lachnospiraceae | Capsules increased butyrate-producing, immunity-stimulating, and anti-cancer bacterial taxa and enhanced Th cells, both CD4 and CD8 cells. | [93] |
| PDAC with lymph node metastases | Patients | No intervention | Leuconostoc, Sutterella, Comamonas, and Turicibacter | Lower levels of Leuconostoc and Sutterella were documented in tumors with a size ≥3 cm. An increase in lymph node metastases correlated with a higher abundance of Comamonas and Turicibacter. On the contrary, Streptococcus dominated recurrence-free tumors. | [95] |
| Hepatocellular carcinoma | Mice | NpRg3 | Bacteroidetes, Verrucomicrobia, and Firmicutes | Developed NpRg3 remodeled gut microbiome via reduced Firmicutes and increased Bacteroidetes and Verrucomicrobia in stool samples. Moreover, NpRg3 attenuated tumor development and lung metastatic formation in dimethylnitrosamine-induced spontaneous murine carcinoma. | [96] |
| Lung cancer | Patients | Systemic therapy/surgical resection | Legionella and Thermus | Thermus was abundant in the lung microbiome in patients with advanced cancer stages, while Legionella was enriched in patients with developed metastases. Alpha diversity in tumor tissues was lower than in non-malignant lung tissue samples. | [98] |
| Hormone receptor-positive breast cancer | Mice | Antibiotic cocktail (vancomycin, ampicillin, metronidazole, neomycin, and gentamicin) | Blautia, Alistipes, Blautia, Escherichia/Shigella, and Bilophila | Orally gavaged antibiotics caused commensal dysbiosis with a higher abundance of specific genera in poorly metastatic mice. Antibiotics promoted tumor cell dissemination to the lungs/peripheral blood/and lymph nodes. | [102] |
| Breast cancer | Patients | No intervention | Streptococcus, Campylobacter, Moraxellaceae, Lactobacillales, Bacilli, Epsilonproteobacteria, Veillonella, Acinetobacter, Pseudomonadales, Megamonas, and Akkermansia | Listed bacteria, except for Megamonas and Akkermansia, were increased in stool samples of patients with bone metastases. However, the results showed lowered levels of Megamonas and Akkermansia. Bacterial diversity was reduced in the order of normal controls, patients without metastases, and patients with bone metastases. | [103] |
| Breast cancer | Patients | Neoadjuvant chemotherapy | Streptococcus, Pseudomonas, Brevundimonas, and Staphylococcus | Chemotherapy decreased intratumoral Streptococcus and increased Pseudomonas. The development of distant metastases correlated with a higher presence of Brevundimonas and Staphylococcus in primary breast tumors. | [105] |
| Oral squamous cell carcinoma | Patients | Therapeutic neck dissection due to positive lymph node metastases | Tannerella, Fusobacterium, Prevotella, Stomatobaculum, Bifidobacterium, Finegoldia Peptostreptococcaceae, and Shuttleworthia | Two taxa—Tannerella and Fusobacterium—were enriched in the oral microbiome of patients without metastases. Other genera from the listed panel increased in patients with developed lymph node metastases. Differences in alpha diversity between the oral microbiome of 2 analyzed groups were not significant. | [106] |
| Castrate-resistant prostate cancer | Patients | Immune checkpoint inhibitor (pembrolizumab) | A. muciniphila, B. thetaiotaomicron, B. fragilis, and R. unassigned | A. muciniphila was depleted in pembrolizumab responders, while other listed microbes were higher in responding patients. | [109] |
| Renal cell carcinoma | Patients | Immune checkpoint inhibitor (nivolumab or nivolumab plus ipilimumab | A. muciniphila, B. adolescentis, B. intestinihominis, Odoribacter splanchnicus, Bacteroides ovatus, and Eggerthella lenta | A. muciniphila, B. adolescentis, B. intestinihominis, and O. splanchnicus correlated with clinical benefit in metastatic patients, while B. ovatus and E. lenta were associated with no clinical benefit from immunotherapy. | [111] |
| Renal cell carcinoma | Patients | Immune checkpoint inhibitor | Akkermansia | The presence of Akkermansia was documented in both responding and non-responding patients to immunotherapy. Therefore, host-specific or tumor factors might affect therapy response. | [112] |
| Melanoma | Patients | Immune checkpoint inhibitor | Lactobacillales, Clostridiales/Ruminococcaceae, Faecalibacterium, Bacteroidales, B. thetaiotaomicron, E. coli, and Anaerotruncus colihominis | Lactobacillales dominated the oral microbiome of all metastatic patients. Clostridiales/Ruminococcaceae, Faecalibacterium, and alpha diversity were greater in responders, while Bacteroidales, B. thetaiotaomicron, E. coli, and A. colihominis were abundant in non-responders. | [115] |
| Melanoma | Patients | Immune checkpoint inhibitor | B. longum, C. aerofaciens, E. faecium, R. obeum, and R. intestinalis | The authors observed a higher abundance of R. obeum and R. intestinalis in non-responders, while the other 3 species were enriched significantly in the responder gut microbiome. | [117] |
| Melanoma | Patients | Immune checkpoint inhibitor | Clostridiales and Bacteroidales | Higher bacterial diversity with the prevalence of Clostridiales was observed in the gut microbiome of responding patients. However, the dominance of Bacteroidales within the gut microbiome characterized non-responders. | [118] |
| Melanoma | Patients | No intervention | Corynebacterium | In swab samples, Corynebacterium was the most detected taxa in advanced-stage patients. However, the authors did not detect associations between cutaneous microbiome and cancer stage. | [116] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sevcikova, A.; Mladosievicova, B.; Mego, M.; Ciernikova, S. Exploring the Role of the Gut and Intratumoral Microbiomes in Tumor Progression and Metastasis. Int. J. Mol. Sci. 2023, 24, 17199. https://doi.org/10.3390/ijms242417199
Sevcikova A, Mladosievicova B, Mego M, Ciernikova S. Exploring the Role of the Gut and Intratumoral Microbiomes in Tumor Progression and Metastasis. International Journal of Molecular Sciences. 2023; 24(24):17199. https://doi.org/10.3390/ijms242417199
Chicago/Turabian StyleSevcikova, Aneta, Beata Mladosievicova, Michal Mego, and Sona Ciernikova. 2023. "Exploring the Role of the Gut and Intratumoral Microbiomes in Tumor Progression and Metastasis" International Journal of Molecular Sciences 24, no. 24: 17199. https://doi.org/10.3390/ijms242417199