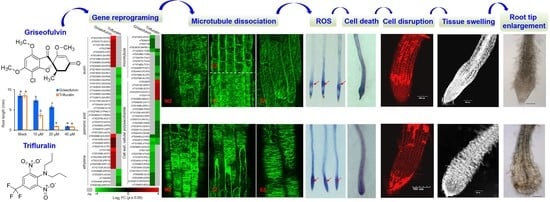
2.2. Abnormal Enlargement of the Root Tips Caused by Griseofulvin Is Different from Trifluralin
In early 1971, it was discovered that trifluralin, a successful commercial herbicide, inhibited root growth of corn and wheat and caused root swelling [
3]. To assess the exact effect of griseofulvin on
Arabidopsis roots, trifluralin was used as a positive control. While
Arabidopsis seedlings were cultured directly in 1/2 MS media containing various concentrations of griseofulvin or trifluralin at 22 °C with 100 μmol (photons) m
−2 s
−1 light intensity for 7 days (
Figure 2A), both griseofulvin and trifluralin significantly inhibited seedling growth under the concentrations of 10 to 40 μM. As the concentration increased, the inhibition of shoot, leaf, and root development became more pronounced. It is evident that griseofulvin drastically reduced root elongation as did trifluralin (
Figure 2B). Obviously, the effect of griseofulvin on root growth was a little weaker than that of trifluralin. As shown in
Figure 2C, the degree of inhibition of 40 μM griseofulvin was equivalent to that of 20 μM trifluralin. Thus, griseofulvin inhibits
Arabidopsis root growth at a higher effective dose than trifluralin.
To further observe the effect of griseofulvin and trifluralin on the tissue morphology of
Arabidopsis root tips, two-day-old
Arabidopsis seedlings grown on the conventional medium MS were transferred to new media containing different concentrations (0, 10, 20, and 40 μM) of griseofulvin or trifluralin (
Figure 2D). After 5 days of cultivation, root tips were observed under the microscope (
Figure 2E). Here, the abnormal enlargement of the root tips caused by griseofulvin and trifluralin is clearly visible. When treated with 10 μM or 20 μM griseofulvin, morphological changes in root tips are difficult to detect. However, 40 μM griseofulvin caused an abnormal enlargement of root tips that appeared to occur not in the apical meristematic zone (MZ) of trifluralin-treated roots, but mainly in its rear zones, which might be the transition zone (TZ) and elongation zone (EZ). In the following text we temporarily used TZ and EZ to state the action zones of griseofulvin and will provide further powerful evidence in the later observation of microtubules by confocal laser scanning microscope. The shape of the MZ remained normal after treatment with 40 μM griseofulvin. This result is in exact agreement with that in
Figure 1F. In the case of trifluralin, 20 μM or 40 μM trifluralin obviously caused an abnormal enlargement of the root tips, especially in the apical MZ. In this situation, the root tips treated with trifluralin lost their normal apical shape. The above results show that griseofulvin and trifluralin cause abnormal enlargement of
Arabidopsis root tips in a completely different zone.
In general, abnormal enlargement of plant tissues is associated with cell death. To verify whether griseofulvin causes cell death in different zones compared with trifluralin, cell death of the above samples was determined by trypan blue (TBD) staining. Dead cells were stained dark blue with TBD [
17]. In
Figure 2F, it can be seen that 10 μM griseofulvin caused slight cell death in the root TZ. As the concentration increased, the blue coloration in the root TZ became darker. At 40 μM griseofulvin, all cells already died not only in the TZ but also in the MZ of the root tips. With trifluralin, significant cell death was observed in the apical MZ after 10 μM trifluralin treatment. In the presence of 20 or 40 μM trifluralin, cell death in the apical MZ of root tips became more pronounced. These results indicate that the cell death caused by griseofulvin occurred first in the root TZ, but the initial cell death occurred in the root MZ of roots treated with trifluralin.
2.3. ROS Production Is an Early Event during Griseofulvin- and Trifluralin-Induced Cell Death of the Root Tips
ROS is considered a key element leading to cell death in both pathogen attack and herbicide treatment [
18]. To confirm the role of ROS in the process of cell death of
Arabidopsis root tips induced by griseofulvin or trifluralin, 3,3′-diaminobenzidine (DAB) and nitro-blue tetrazolium (NBT) staining were used to detect H
2O
2 and O
2·− production, respectively. In the presence of H
2O
2, DAB polymerizes and turns reddish-brown [
19]. Here, 2-day-old
Arabidopsis seedlings grown on the conventional medium were transferred to a new medium containing 40 μM griseofulvin or trifluralin and incubated for the indicated time before DAB or NBT staining (
Figure 3A). As a result, brown deposits were visible in root tips after 40 μM griseofulvin treatment for 6 h. With increasing time up to 9 and 12 h, the oxidized DAB deposits in the root tips became darker, indicating numerous H
2O
2 production (
Figure 3B). In plant cells, O
2·− can rapidly convert to H
2O
2 by superoxide dismutase (SOD) [
19] Intracellular O
2·− can react with NBT to generate the dark-blue insoluble formazan [
20]. The development of the NBT- O
2·− reaction products in the root tips revealed that O
2·− were detected as early as 3 h after 40 μM griseofulvin treatment, with the color continuing to darken with prolonged time to 9 h and 12 h (
Figure 3B). Higher magnification of NBT-stained root tips showed that 40 μM griseofulvin induced O
2·− production as early as 1 h, which occurred mainly in the TZ and the early EZ of the root tips (
Figure 3C). Data in
Figure 3D showed that 40 μM trifluralin significantly induced H
2O
2 and O
2·− production in the apical tissues of the root tips at 3 h to 12 h after treatment. In fact, large amounts of dark-blue deposits were produced in the apical MZ of trifluralin-treated root tips as early as 1 h (
Figure 3E). Obviously, the original location of ROS generation in the root tips was in good agreement with the initial occurrence of cell death (
Figure 2F) and morphological damage of the root tips (
Figure 2E) after griseofulvin and trifluralin treatment. Furthermore, ROS production was an early event in griseofulvin- and trifluralin-caused cell death in
Arabidopsis roots.
To demonstrate whether ROS was involved in cell death in
Arabidopsis roots after griseofulvin or trifluralin treatment, root tip viability was detected by fluorescein diacetate (FDA) staining. FDA fluorescence decreases as the dye leaks from dead cells, so a decrease in FDA fluorescence signal is used as an indicator of loss of cell viability [
21]. As shown in
Figure 4, 2-day-old
Arabidopsis seedlings grown on conventional medium were directly transferred and incubated for 1 to 3 days in new medium containing 40 μM griseofulvin or trifluralin (
Figure 4A–D), or pretreated with various ROS scavengers before incubation (
Figure 4E–H). It was evident that the FDA fluorescence intensity of root tips treated with griseofulvin and trifluralin decreased significantly with increasing duration (
Figure 4A–D). Remarkably, griseofulvin resulted in a visible decrease in FDA signal in the root TZ after 1 d and a strong decrease in fluorescence in the root TZ and EZ after 3 d and 5 d of treatment. However, the cells in the apical MZ of the root tips remained alive after 5 days of treatment with griseofulvin (
Figure 4A). With trifluralin, the FDA signals of almost all root tip cells decreased in a time-dependent manner after 1 to 3 days of treatment (
Figure 4C,D). Clearly, the mode of loss of cell viability was different in griseofulvin- and trifluralin-treated root tips. In addition, four general ROS scavengers, SOD, dimethyl thiourea (DMTU), diphenyleneiodonium (DPI), and N-acetyl-cysteine (NAC), were introduced to verify the role of ROS in cell death. SOD catalyzes O
2·− into O
2 and H
2O
2 [
18]. DMTU is a specific ROS scavenger [
22]. DPI is an inhibitor of NADPH oxidase, and NAC is the glutathione precursor [
23]. It was observed that pretreatment with SOD and DMTU significantly improved cell viability (
Figure 4E,F) and eliminated morphological damage (
Figure 4E) in roots treated with griseofulvin. The effects of DPI and NAC pretreatment on cell damage caused by griseofulvin were not visible (
Figure 4E,F). This suggests that the cell death caused by griseofulvin was due to the production of ROS, which may have originated from O
2. These four ROS scavengers were able to partially suppress the loss of cell viability and morphological damage caused by trifluralin in root tips (
Figure 4G,H). It was demonstrated that ROS plays an important role in trifluralin-induced cell death in root tips. Therefore, it was concluded that the formation of ROS as an early event is indeed involved in griseofulvin and trifluralin-induced cell death and abnormal enlargement of root tips, ultimately leading to root growth inhibition.
2.5. The Initial Zone of Microtubule Disruption in the Root Tip Cells Caused by Griseofulvin Is Different from Trifluralin
Microtubules play an important role in directional cell expansion, maintenance of cell morphology, and organization of the nucleus and organelles. Plant microtubules are composed of numerous heterodimers of α- and β-monomers that likely also contain a ring of γ-tubulin proteins [
25,
26]. MAPs represent all proteins involved in regulating microtubule polymerization, binding to microtubules, and stabilizing or promoting microtubule assembly [
1,
25]. Because griseofulvin could alter root tip cell morphology (
Figure 5C), it was expected that griseofulvin could affect microtubule dynamics, as well as trifluralin. Trifluralin could depolymerize microtubules and cause the mitotic spindle not to form, leading to misalignment and chromosome segregation during cell mitosis [
4]. Some studies suggest that griseofulvin targets tubulins and interferes with cell processing [
12,
13,
27], and it could also interfere with microtubule polymerization in HeLa cells [
9]. Nevertheless, its mode of action in plants is not yet clear.
To monitor the effect of griseofulvin on microtubule dynamics in root tips, specific
Arabidopsis plants expressing a microtubule reporter were used MBD-GFP [
28]. The root tip consists of three zones corresponding to the different states of the cells: meristematic zone (MZ), elongation zone (EZ), and differentiation zone (DZ). In addition, there is another transition zone (TZ) between the MZ and the EZ (
Figure 6A) [
29]. In the
Arabidopsis wild-type root, the orientation of the cortical microtubules is generally loosely longitudinal in the apical MZ cells and then shifts to transverse in the early TZ cells. In the EZ, it remains predominantly transverse and then shifts to the oblique direction in the DZ cells (
Figure 6A) [
26].
After 1 h of treatment with 40 μM griseofulvin, microtubules on the outer cell surfaces in four different zones of the root tips of the plant MBD-GFP maintained their normal dynamics and organization (
Figure 6B). After 3 h, griseofulvin caused massive dissociation of cortical microtubules in root TZ and early EZ cells and partially in fast EZ cells. Transverse microtubule alignment was barely observed, but numerous fluorescent protein particles were, indicating that the microtubules of root TZ and early EZ cells and partially of fast EZ cells were dramatically disrupted. With increasing time up to 6 and 9 h, griseofulvin caused greater dissociation of cortical microtubules in root TZ and EZ cells. It was also observed that some TZ and EZ cells began to deform (
Figure S1). However, during the first 9 h of griseofulvin treatment, no visible change in cortical microtubules was observed in root MZ and DZ cells (
Figure 6B and
Figure S1). After 12 h of griseofulvin treatment, cortical microtubules in zones other than the root DZ were severely damaged (
Figure 6B). Many cells in the apical TZ and early EZ almost lost their green fluorescence, indicating complete dissociation of cortical microtubules and cell death. Most cells in the rapid EZ exhibited randomly aligned microtubules and a twisted and swelling shape (as shown by the arrows). Some cells in the MZ also showed dissociation of cortical microtubules into small fragments that exhibited faint and diffuse green fluorescence (shown as arrows). After 24 h, root cells began to dissolve in earnest in all zones, especially in the MZ and TZ and in the early EZ cells (
Figure S1). Apparently, initial microtubule dissociations in the root TZ and early EZ cells already occurred when the roots were treated with griseofulvin within 3 h, which was a later event compared with the initial ROS production (
Figure 3B and
Figure 6B). This is likely to be the actual and direct reason for the root swelling caused by griseofulvin in the TZ. Dissociation of cortical microtubules in the apical MZ and EZ would certainly lead to inhibition of root growth.
However, in the presence of 40 μM trifluralin, the change in cortical microtubule alignment in
Arabidopsis root tip cells differed from that induced by griseofulvin (
Figure S2). The pronounced disintegration of cortical microtubules in four zones of root tips was observed 1 h after trifluralin treatment and became more severe with increasing time up to 9 and 12 h. Some cells with dissected microtubules showed a remarkable twisted and swollen shape and even rupture of the cell wall in different zones of roots treated with trifluralin for 12 h (
Figure S2). Such indiscriminate damage to all cells in whole root tips could be due to the destruction of α-tubulin, a central subunit of cell microtubules. Previous studies have shown that trifluralin destroys microtubules by binding to the α-tubulin subunit [
5,
30]. Undoubtedly, at the same concentration, trifluralin shows faster and more severe damage to cortical microtubules in root tips (
Figure 6B and
Figures S1 and S2). To clarify the detailed change process of cortical microtubules in different zones of root tips after trifluralin treatment,
Arabidopsis MBD-GFP roots were incubated with a lower concentration (20 μM) and shorter duration (10 to 60 min). Cortical microtubules in four zone cells maintained their normal orientation after 10 min of trifluralin treatment without visible damage (
Figure 6A and
Figure 7). After 20 min, dissociation of cortical microtubules in apical MZ cells had occurred. After 60 min, trifluralin caused substantial disintegration of cortical microtubules not only in the apical MZ but also in the other three zones of the root tips. Significant cell breakage was also observed in the MZ, TZ, and early EZ cells of trifluralin-treated roots (
Figure 7). These results indicate that trifluralin initially directs root tips to microtubules in apical MZ cells. Thus, the initial dissociation of cortical microtubules caused by griseofulvin and trifluralin occurred in the different zones, leading to cell fission and root tip swelling in the corresponding zones. All in all, our evidence suggests that the direct target of griseofulvin is not the α-tubulin protein, which is the primary target of trifluralin, because the two agents showed completely different behaviors in disrupting microtubule alignment in root tips (
Figure 6 and
Figure 7) [
9,
30]. Panda et al. [
9] suggested that the microtubule polymerization inhibited by griseofulvin in mammals was due to the disruption of the function of MAPs rather than the effect on αβγ-tubulins.
2.6. Transcriptome Analysis of Griseofulvin- and Trifluralin-Incubated Root Tips
To reveal the molecular mechanism of action of griseofulvin, large-scale transcriptome sequencing was performed on 5-day-old
Arabidopsis seedlings incubated with 40 μM griseofulvin or 1% DMSO (mock) for 1 h or with 20 μM trifluralin for 10 min before the first visible microtubule damage occurred. Comparative transcriptome analysis showed that 358 and 227 differentially expressed genes (DEGs) were upregulated at least twofold after griseofulvin and trifluralin, respectively (
p < 0.05) (
Figure 8A,
Table S1-1). Only 58 DEGs were present in both griseofulvin- and trifluralin-treated samples (
Figure 8A,
Table S1-2). Assignment of biological process terms from Gene Ontology (GO) for these 58 genes revealed 10 overrepresented groups (
p < 0.05). Among them, genes involved in stress, stimulus, oxidation–reduction, and cell-killing responses were significantly enriched (
Figure 8B). GO term enrichment analyses (
p < 0.05) for biological processes involving 300 genes specifically induced by griseofulvin showed that plant hormone- and ROS-related genes, especially ethylene-related genes, were significantly overrepresented (
Figure 8C,
Table S1-3). The 169 genes specifically upregulated by trifluralin had the most enriched terms in response to secondary metabolism, oxygenated compounds, salicylic acid, and so on (
Figure 8D,
Table S1-4). Obviously, griseofulvin mainly elicited a positive root response to ethylene, jasmonic acid, and abscisic acid. Trifluralin, on the other hand, increased the response to secondary metabolic processes of anthocyanins and flavonoids, as well as the response to salicylic acid and defense substances.
On the other hand, griseofulvin and trifluralin downregulated 563 and 333 DEGs, respectively, by at least twofold (
p < 0.05) (
Figure 8E,
Table S1-1). They had 53 common downregulated genes (
Figure 8E), which were significantly enriched in lipid metabolism-, catabolism-, and cell-wall-related responses (
Figure 8F,
Table S1-5). Based on the GO term enrichment (
p < 0.05) in biological processes, 510 genes specifically repressed by griseofulvin were mainly overrepresented in the single organism process, peptide transport, cell wall modification, and cell wall component of polysaccharides groups (
Figure 8G,
Table S1-6). However, 280 genes specifically repressed by trifluralin were significantly enriched in the hormone, especially auxin and abscisic acid, and developmental response groups (
Figure 8H,
Table S1-7). This is in direct contrast to griseofulvin, which remarkably induced hormone-dependent genes, particularly ethylene-, jasmonic-acid-, abscisic-acid-, and auxin-dependent signaling genes (
Figure 8C). Such differential responses to plant hormones may be one of the important molecular mechanisms of the microtubule disruption caused by griseofulvin and trifluralin in the different zone cells of root tips. Numerous studies have shown that plant hormones orchestrate root growth by controlling cell division, cell expansion, and cell differentiation in different zones of
Arabidopsis roots. Briefly, auxin and gibberellin mainly promote cell division, and abscisic acid inhibits cell proliferation in the root apical MZ. In the TZ, cytokinin is essentially required for cell differentiation, which was negatively affected by auxin and gibberellin. Ethylene, auxin, jasmonic acid, and abscisic acid suppress cell elongation in the EZ [
31,
32].
To investigate the detailed mechanism of microtubule disruption and abnormal root enlargement caused by griseofulvin and trifluralin, a series of auxin, jasmonic acid, ethylene, microtubule, cell wall, and cellular polysaccharide-responsive genes were selected according to previous reports (
Table S2) and shown in heat maps (
Figure 9A,C). Our results showed that the transcript levels of eight ethylene (ERS1, CTR1, AT1G71520, ACO1, ERF1B, ERF15, ERF114, ACO5), two jasmonic acid (bHLH041, ACX1), and nine auxin (HUP54, GH3.3, IAR3, AT2G37980, ILR1, SAUR5, SAUR51, SAUR59, SAUR76)-responsive genes were significantly upregulated by griseofulvin but not by trifluralin. In contrast, trifluralin significantly decreased the transcript levels of twelve auxin- (SAUR20, SAUR21, SAUR26, SAUR34, SAUR35, SAUR62, SAUR64, SAUR65, SAUR67, SAUR77, SHY2, ILL6), seven jasmonic-acid- (CYP94C1, CYP94B3, AOC1, AOC2, OPR3, JAR1, JAZ10) and five ethylene-responsive genes (ERF53, AT1G64380, ACS4, ERF34, ERF107) that were not significantly altered in griseofulvin-treated seedlings compared with sham treatment (
Figure 9A,
Table S2). This indicates that the effect of griseofulvin and trifluralin on hormone-responsive genes was completely different, which was clearly confirmed by the qRT-PCR results. As shown in
Figure 9B, the relative expression levels of five selected hormone-responsive genes, GH3.3, ILL6, CYP94C1, OPR3, and ERF53, were calculated by qRT-PCR using Actin2 as a control. Consistently, these five genes were significantly downregulated by trifluralin but upregulated by griseofulvin. Auxin is known to be important for stem cell maintenance, root meristem activity, and root zone patterning [
32]. Weak auxin signaling at the root tip would result in inhibiting cell division in the MZ and promoting cell elongation in the EZ [
31,
32]. The number of cells in the MZ is a critical determinant of root size and growth rate because it is controlled by auxin-dependent signaling and hormonal cross-talk [
33]. It is concluded that inhibition of cell division in the MZ due to low auxin-dependent gene expression may play a critical role in trifluralin-induced root growth inhibition. This may explain why trifluralin can cause the formation of ROS from root apical MZ cells, resulting in cell death and tissue swelling in the MZ during root growth inhibition (
Figure 2,
Figure 3D,E and
Figure 5D). For griseofulvin, significant enhancement of ethylene and auxin signaling should be the primary determinant of root growth inhibition and also the actual reason for the initial triggering of ROS production in the cells of the root TZ and early EZ and the subsequent triggering of cell death and swelling in the root TZ and early EZ (
Figure 2,
Figure 3B and
Figure 5C). Cell differentiation in the root TZ has been shown to be negatively affected by auxin [
31]. Ethylene is considered a potent hormonal inhibitor of cell expansion and elongation at the root tip and stimulates auxin synthesis and transport toward the root EZ during root growth and development, where it triggers a local auxin response that leads to suppression of cell elongation [
31,
32].
Among the thirteen microtubule-related genes, with the exception of the β-tubulin gene TUB5, which is repressed by both griseofulvin and trifluralin, the transcript levels of four MAP genes (IPGA1, AT5G66310, AT3G51150, MAP65-1) were significantly decreased by griseofulvin but not by trifluralin. Conversely, the transcript levels of three tubulin genes (TUB8, TUA1, TUA3) and five other MAP genes (TCS1, PCAP1, PCAP2, KLCR1, KLCR2) were significantly decreased by trifluralin but not by griseofulvin (
Figure 9C and
Figure S3,
Table S2). The synthesis of α- and β-tubulins as basic structural subunits of microtubules is regulated by the transcript levels of the genes that encode TUA and TUB [
34,
35]. In conjunction with the qPCR results showing that trifluralin significantly suppresses the expression level of the TUA3 gene but not that of the MAP65-1 gene, it is clear that trifluralin inhibits plant growth by binding to αβ-tubulins. This is consistent with previous evidence of αβ-tubulin mutant and molecular interactions between trifluralin and α-tubulin [
4,
5]. However, tubulin-directed damage cannot be invoked as an explanation for the initial occurrence of microtubule dissociation, ROS generation, cell death, and swelling in the root apical MZ in the presence of trifluralin, because all cells from different zones in the root tips share the common microtubule structures with αβ-tubulins. In contrast, griseofulvin significantly suppressed the expression of MAP genes such as MAP65-1, IPGA1, and AT3G1150 but not tubulin genes like TUA1, TUA3, or TUB8, suggesting that griseofulvin may disrupt microtubules by targeting MAPs (
Figure 9D and
Figure S3). This result is consistent with previous findings that griseofulvin is most likely to target MAP rather than the structural protein tubulin [
9]. Here, two MAPs encoded by AT5G66310 and AT3G51150 belong to the family of ATP-binding microtubule motor proteins. The IPGA1 protein is involved in the organization of cortical microtubules. The MAP65-1 protein, located on microtubule arrays, accumulates extensively in the root EZ and plays a critical role in root growth by promoting cell proliferation and axial expansion [
36]. MAPs have been shown to decorate cortical microtubules with different patterns, regulating microtubule dynamic instability, microtubule separation, and other array assembly processes [
25]. Some MAPs are important in promoting the formation of specific microtubule arrays that favor the continuation of cell division and also play a specific function in controlling auxin levels and the integration of hormone signaling [
33]. Recently, it was also discovered that carrier PIN-dependent auxin transport can exert feedback control over microtubule alignment and consequently affect cell wall properties and cell shape [
33]. Therefore, the disturbed balance of the complex auxin signaling network may be a reasonable explanation for the different localizations of initial ROS production, microtubule dissociation, cell death, and tissue swelling in root tips between griseofulvin and trifluralin.
In plant cells, the cell wall can also provide biophysical feedback to the orientation of microtubules in the cortex, so an undisturbed cellulose process is critical for maintaining normal microtubule orientation [
37]. To further investigate the relationship between cell wall and microtubule damage in root tips treated with griseofulvin or trifluralin, thirty-one genes related to cell wall and cellular polysaccharides were also shown in
Figure 9C. Twelve genes (GASA10, AT5G11420, XTH22, PGL3, ATHB-1, PHS2, CSLB4, CYCP2;1, SLK2, DGR2, ROPGAP3, BDG1) that showed no significant change in trifluralin-treated seedlings were significantly reduced in transcript levels by griseofulvin. In contrast, six genes were upregulated (AT2G20870, WAK1, ECS1, CDC20.1, AT3G01710, MADA1) and ten downregulated genes (LAC17, GRPL1, PSK2, NAC007, CRSP, IRX15-L, GAE6, CESA1, CESA3, CESA6) in trifluralin-treated seedlings showed no significant change in griseofulvin-treated samples compared with sham treatment. Interestingly, three common genes (C/VIF2, PRP4, XTH33) were downregulated by both griseofulvin and trifluralin. Further evidence for this was provided by the data of expression levels of WAK1, PSK2, GASA10, and CYCP2;1 examined in seedlings treated with griseofulvin or trifluralin by qRT-PCR compared with experimental conditions (
Figure 9D). Overall, griseofulvin shows a different pattern of influence on cell-wall- and cellular-polysaccharide-related genes than trifluralin. Previous studies have suggested that cellulose-dependent cell wall integrity is an important factor in controlling the polar distribution of auxin carriers of PIN proteins [
38]. Thus, this is likely a cause of the initial dissociation of microtubules in the different apical zones of roots, which is due to a differential response of auxin signaling between griseofulvin and trifluralin.