Tumor Targeting with Methotrexate-Conjugated Zwitterionic Near-Infrared Fluorophore for Precise Photothermal Therapy
Abstract
:1. Introduction
2. Results
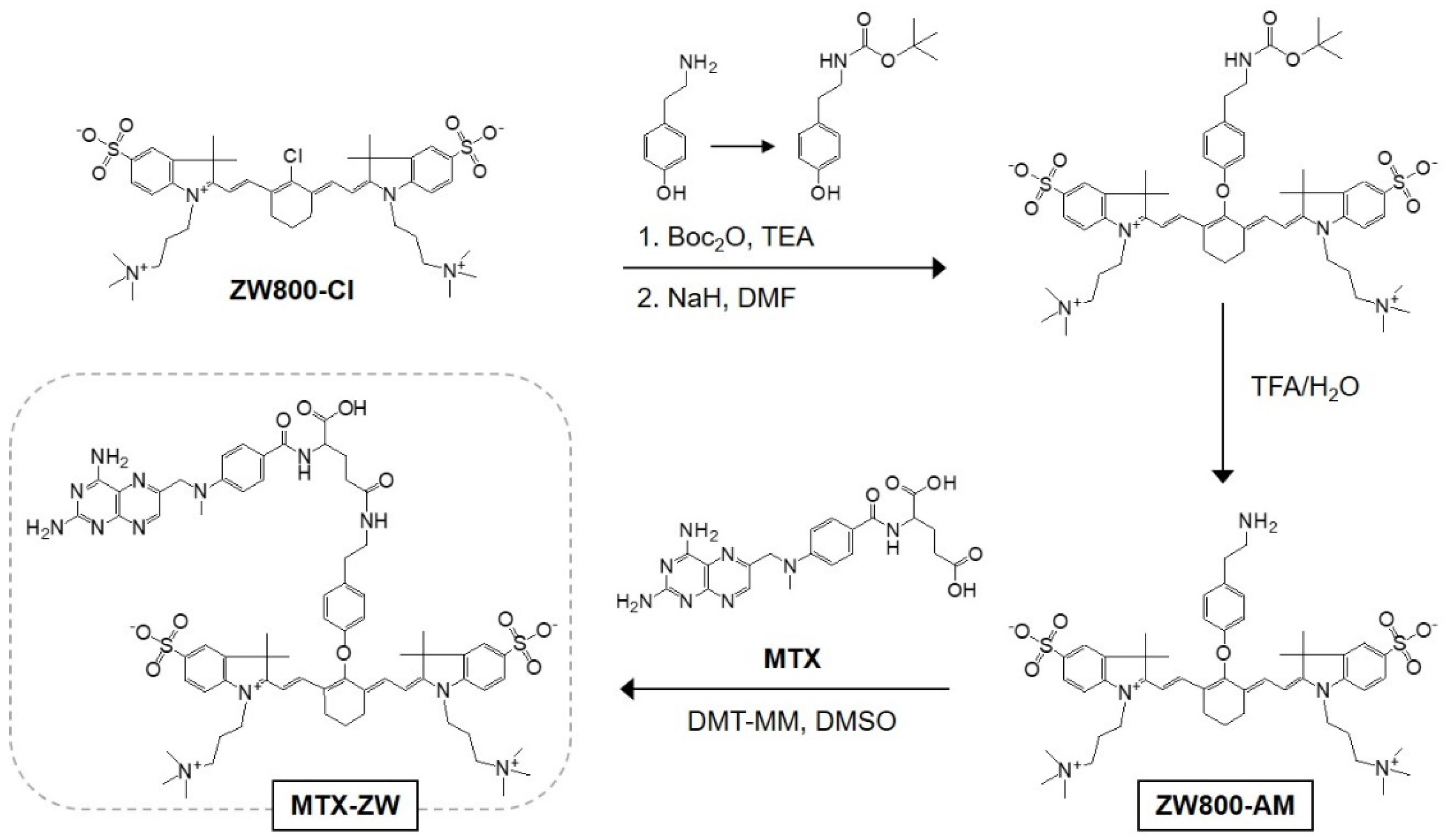
2.1. Synthesis and Characterization of MTX-ZW Conjugate
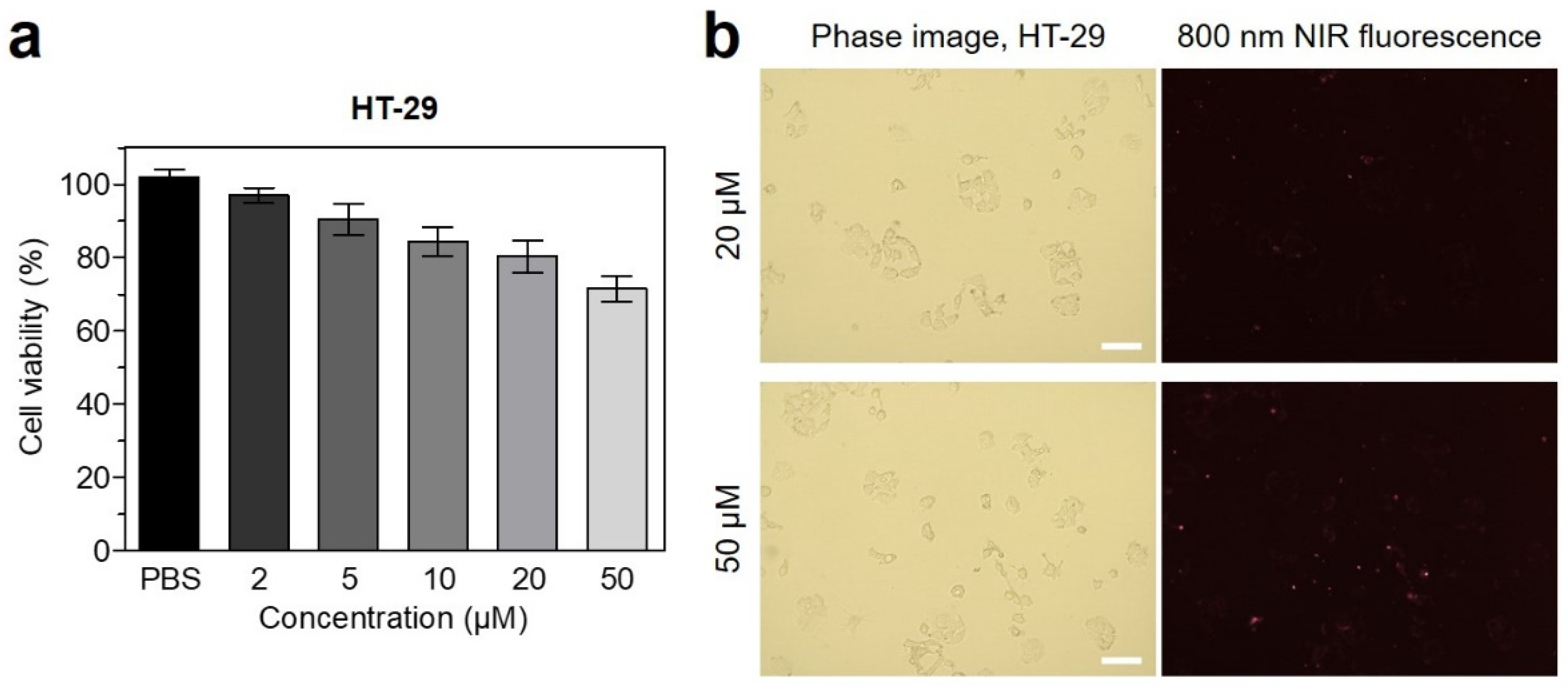
2.2. In Vitro Cytotoxicity and Cellular Uptake
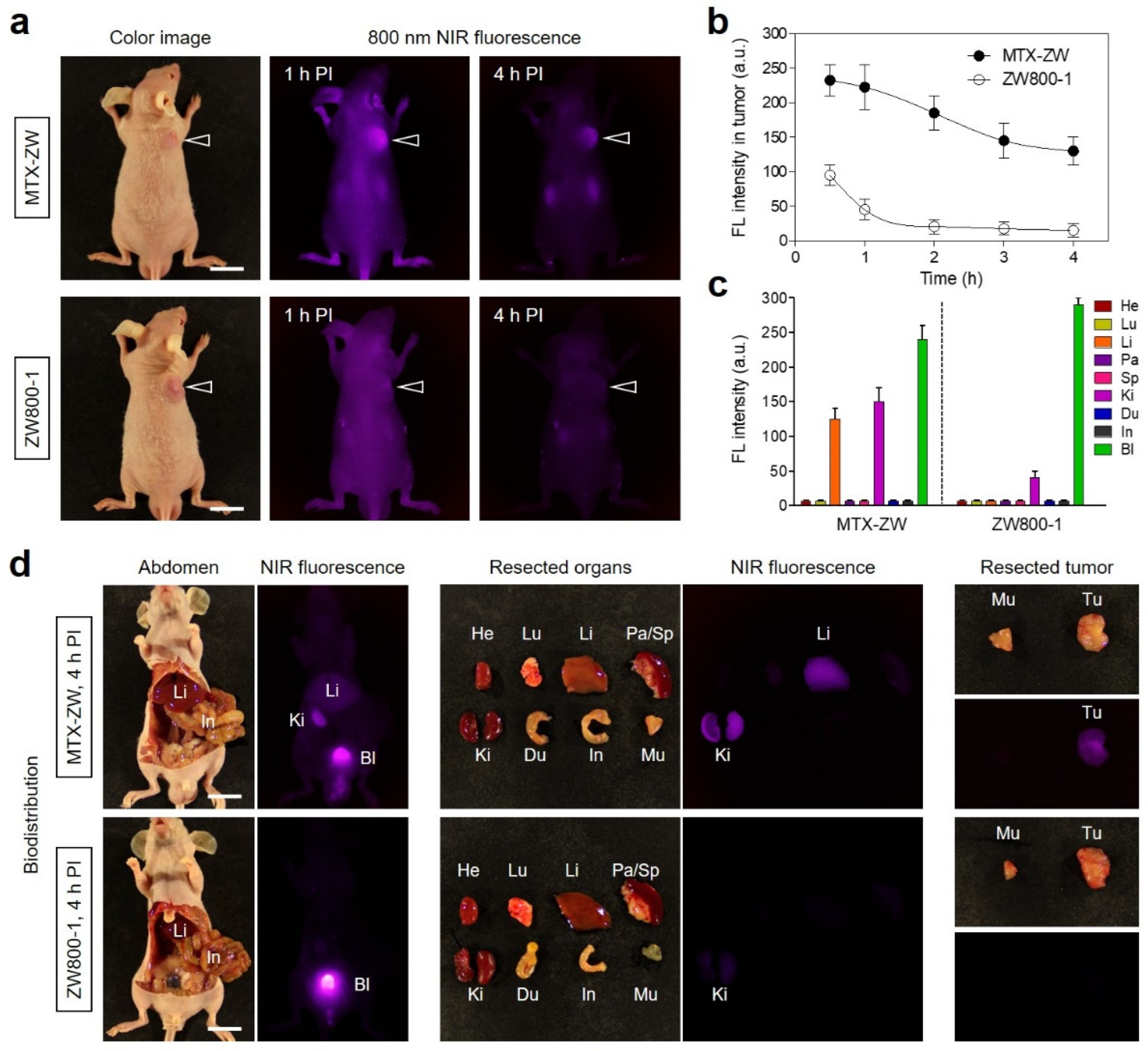
2.3. Time-Dependent In Vivo Tumor Imaging and Biodistribution
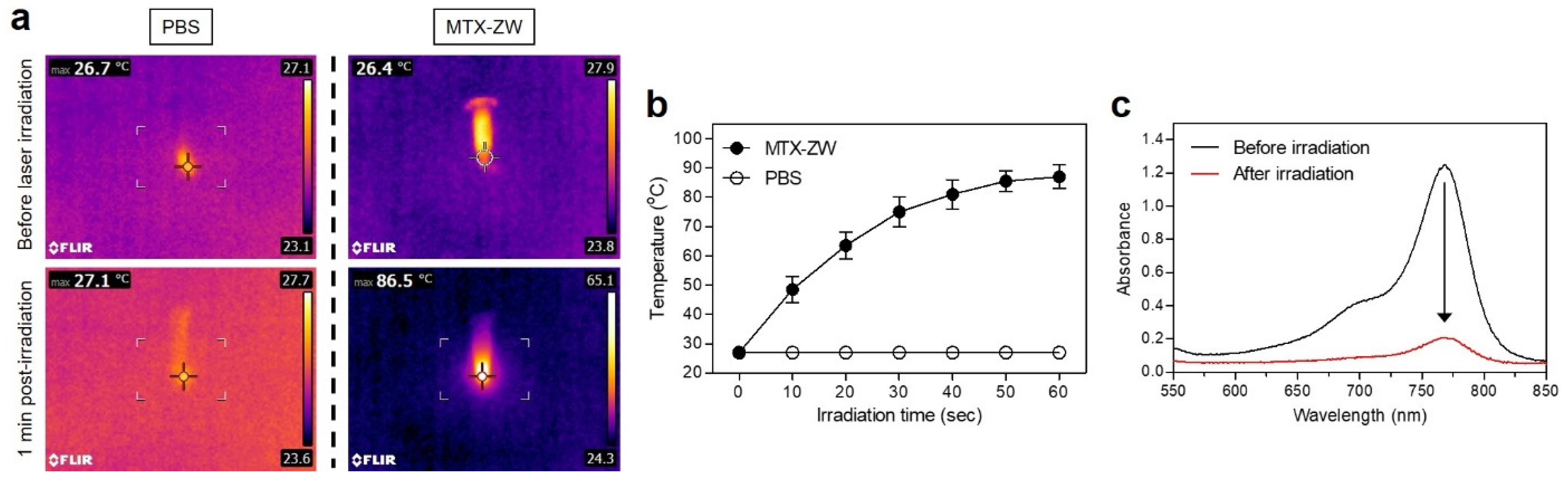
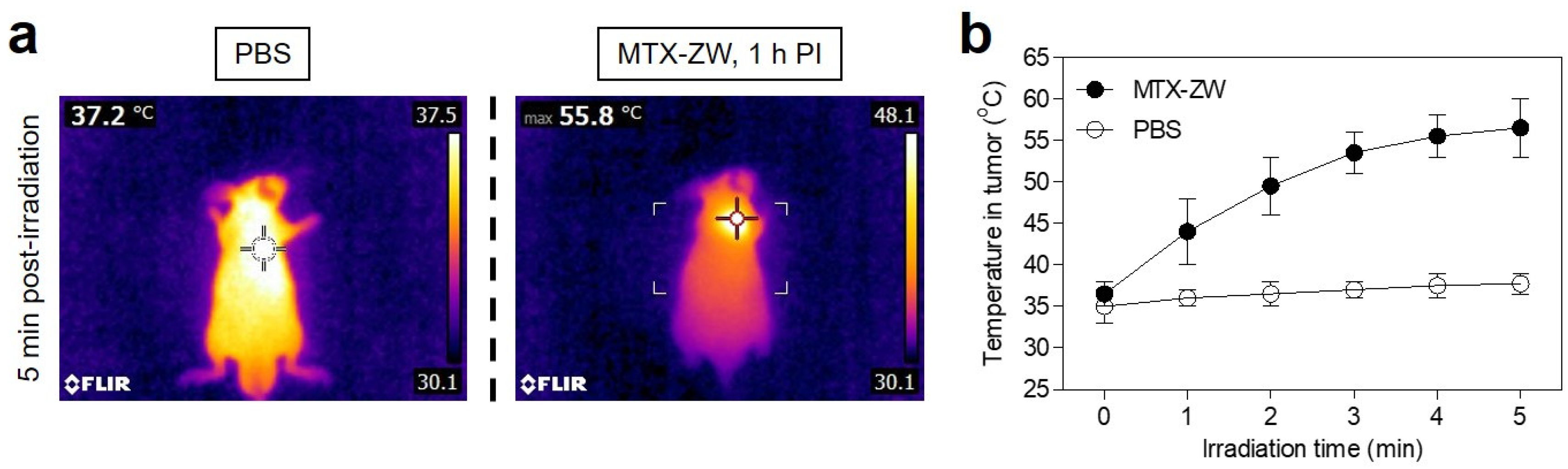
2.4. In Vitro and In Vivo Photothermal Effects
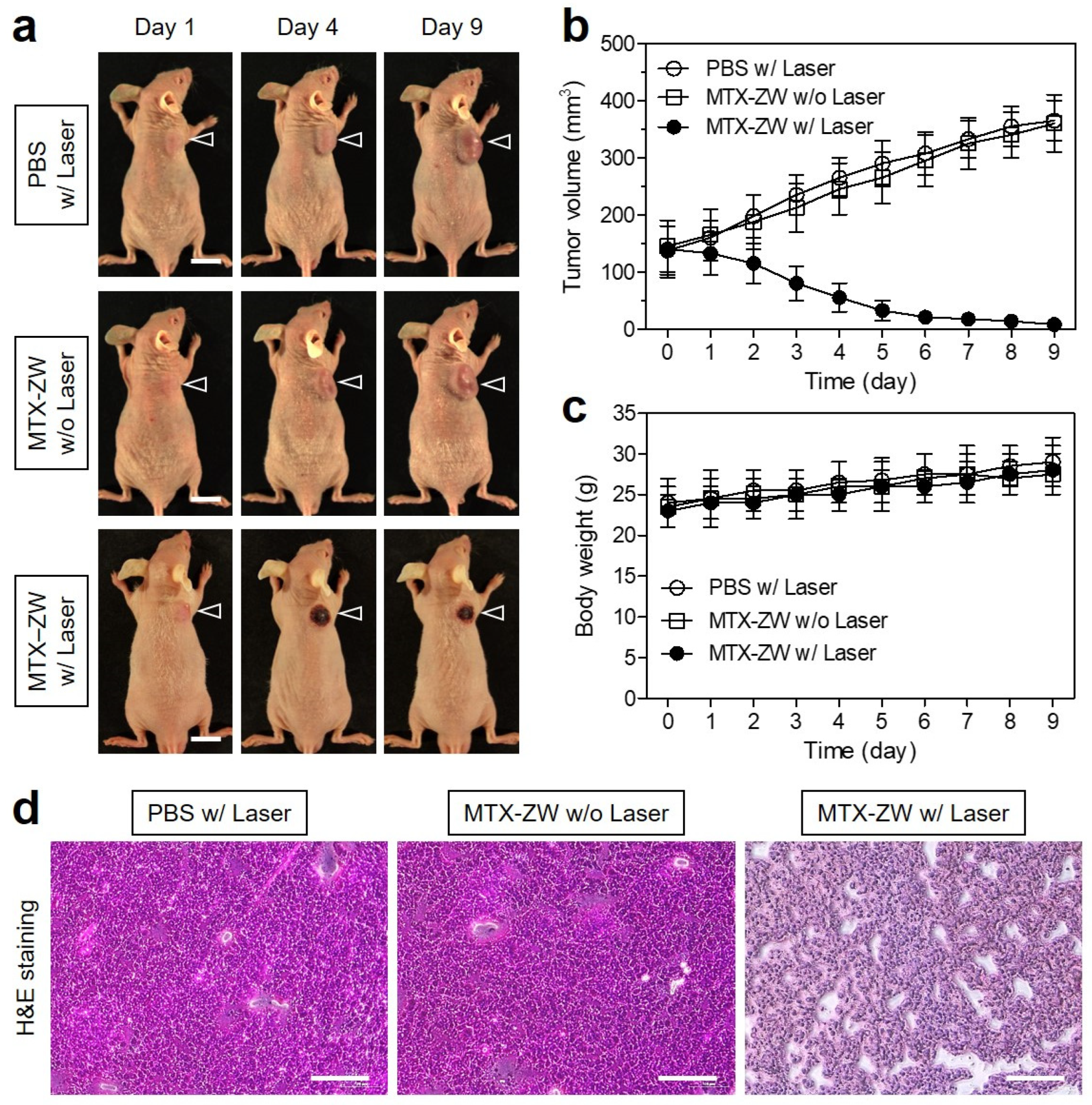
2.5. In Vivo Photothermal Therapeutic Efficacy
3. Discussion
4. Materials and Methods
4.1. Conjugation of Methotrexate to the Zwitterionic NIR Fluorophore (MTX-ZW)
4.2. Optical and Physicochemical Property Analyses
4.3. In Vitro Cell Binding and NIR Fluorescence Microscopy
4.4. In Vitro Cytotoxicity Assay
4.5. HT-29 Xenograft Mouse Model
4.6. In Vivo Biodistribution and Tumor Imaging
4.7. In Vivo Photothermal Therapeutic Efficacy
4.8. Statistical Analysis
4.9. Histological Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yin, X.; Cheng, Y.; Feng, Y.; Stiles, W.R.; Park, S.H.; Kang, H.; Choi, H.S. Phototheranostics for multifunctional treatment of cancer with fluorescence imaging. Adv. Drug. Deliv. Rev. 2022, 189, 114483. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.; Wu, J.B.; Pan, D. Review on near-infrared heptamethine cyanine dyes as theranostic agents for tumor imaging, tar-geting, and photodynamic therapy. J. Biomed. Opt. 2016, 21, 050901. [Google Scholar] [CrossRef] [PubMed]
- Owens, E.A.; Henary, M.; Fakhri, G.E.; Choi, H.S. Tissue-specific near-infrared fluorescence imaging. Acc. Chem. Res. 2016, 49, 1731–1740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, M.H.; Jo, G.; Lee, B.Y.; Kim, E.J.; Hyun, H. Rapid tumor targeting of renal-clearable ZW800-1 conjugate for efficient photothermal cancer therapy. Biomedicines 2021, 9, 1151. [Google Scholar] [CrossRef]
- Lee, S.; Lim, W.; Jo, D.; Jung, J.S.; Kim, S.; Jo, G.; Min, J.-J.; Choi, E.Y.; Hyun, H. Near-infrared fluorescent sorbitol probe for tumor diagnosis in vivo. J. Ind. Eng. Chem. 2018, 64, 80–84. [Google Scholar] [CrossRef]
- Lee, S.; Jung, J.S.; Jo, G.; Yang, D.H.; Koh, Y.S.; Hyun, H. Near-Infrared Fluorescent Sorbitol Probe for Targeted Photothermal Cancer Therapy. Cancers 2019, 11, 1286. [Google Scholar] [CrossRef] [Green Version]
- Choi, H.S.; Gibbs, S.L.; Lee, J.H.; Kim, S.H.; Ashitate, Y.; Liu, F.; Hyun, H.; Park, G.; Xie, Y.; Bae, S.; et al. Targeted zwitterionic near-infrared fluorophores for improved optical imaging. Nat. Biotechnol. 2013, 31, 148–153. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Ning, C.; Zhou, Z.; Yu, P.; Zhu, Y.; Tan, G.; Mao, C. Nanomaterials as photothermal therapeutic agents. Prog. Mater. Sci. 2019, 99, 1–26. [Google Scholar] [CrossRef]
- Doughty, A.C.V.; Hoover, A.R.; Layton, E.; Murray, C.K.; Howard, E.W.; Chen, W.R. Nanomaterial Applications in Photothermal Therapy for Cancer. Materials 2019, 12, 779. [Google Scholar] [CrossRef] [Green Version]
- Maturi, M.; Locatelli, E.; Sambri, L.; Tortorella, S.; Šturm, S.; Kostevšek, N.; Franchini, M.C. Synthesis of Ultrasmall Single-Crystal Gold-Silver Alloy Nanotriangles and Their Application in Photothermal Therapy. Nanomaterials 2021, 11, 912. [Google Scholar] [CrossRef]
- Choi, H.S.; Nasr, K.; Alyabyev, S.; Feith, D.; Lee, J.H.; Kim, S.H.; Ashitate, Y.; Hyun, H.; Patonay, G.; Strekowski, L.; et al. Synthesis and In Vivo Fate of Zwitterionic Near-Infrared Fluorophores. Angew. Chem. Int. Ed. 2011, 50, 6258–6263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, C.; Long, L.; Shi, C. Mitochondria-targeting IR-780 dye and its derivatives: Synthesis, mechanisms of action, and theranostic applications. Adv. Ther. 2018, 1, 1800069. [Google Scholar] [CrossRef]
- Hyun, H.; Bordo, M.W.; Nasr, K.; Feith, D.; Lee, J.H.; Kim, S.H.; Ashitate, Y.; Moffitt, L.A.; Rosenberg, M.; Henary, M.; et al. cGMP-compatible preparative scale synthesis of near-infrared fluorophores. Contrast Media Mol. Imaging 2012, 7, 516–524. [Google Scholar] [CrossRef] [PubMed]
- Khan, Z.A.; Tripathi, R.; Mishra, B. Methotrexate: A detailed review on drug delivery and clinical aspects. Expert Opin. Drug Deliv. 2012, 9, 151–169. [Google Scholar] [CrossRef]
- Woźniak, M.; Pastuch-Gawołek, G.; Makuch, S.; Wiśniewski, J.; Krenács, T.; Hamar, P.; Gamian, A.; Szeja, W.; Szkudlarek, D.; Krawczyk, M.; et al. In Vitro and In Vivo Efficacy of a Novel Glucose-Methotrexate Conjugate in Targeted Cancer Treatment. Int. J. Mol. Sci. 2021, 22, 1748. [Google Scholar] [CrossRef]
- Abolmaali, S.S.; Tamaddon, A.M.; Dinarvand, R. A review of therapeutic challenges and achievements of methotrexate delivery systems for treatment of cancer and rheumatoid arthritis. Cancer Chemother. Pharmacol. 2013, 71, 1115–1130. [Google Scholar] [CrossRef] [PubMed]
- Moshikur, R.M.; Chowdhury, M.R.; Wakabayashi, R.; Tahara, Y.; Moniruzzaman, M.; Goto, M. Ionic liquids with methotrexate moieties as a potential anticancer prodrug: Synthesis, characterization and solubility evaluation. J. Mol. Liq. 2019, 278, 226–233. [Google Scholar] [CrossRef]
- Chen, J.; Huang, L.; Lai, H.; Lu, C.; Fang, M.; Zhang, Q.; Luo, X. Methotrexate-loaded PEGylated chitosan nanoparticles: Synthesis, characterization, and in vitro and in vivo antitumoral activity. Mol. Pharm. 2014, 11, 2213–2223. [Google Scholar] [CrossRef]
- Li, S.; Sun, Z.; Meng, X.; Deng, G.; Zhang, J.; Zhou, K.; Li, W.; Zhou, L.; Gong, P.; Cai, L. Targeted Methotrexate Prodrug Conjugated with Heptamethine Cyanine Dye Improving Chemotherapy and Monitoring Itself Activating by Dual-Modal Imaging. Front. Mater. 2018, 5, 35. [Google Scholar] [CrossRef]
- Al-Natour, M.A.; Alazzo, A.; Ghaemmaghami, A.M.; Kim, D.H.; Alexander, C. LC-MS metabolomics comparisons of cancer cell and macrophage responses to methotrexate and polymer-encapsulated methotrexate. Int. J. Pharm. X 2019, 1, 100036. [Google Scholar] [CrossRef]
- Rostamizadeh, K.; Manafi, M.; Nosrati, H.; Manjilia, H.K.; Danafar, H. Methotrexate-conjugated mPEG–PCL copolymers: A novel approach for dual triggered drug delivery. New J. Chem. 2018, 42, 5937–5945. [Google Scholar] [CrossRef]
- Hou, M.; Gao, Y.E.; Shi, X.; Bai, S.; Ma, X.; Li, B.; Xiao, B.; Xue, P.; Kang, Y.; Xu, Z. Methotrexate-based amphiphilic prodrug nanoaggregates for co-administration of multiple therapeutics and synergistic cancer therapy. Acta Biomater. 2018, 77, 228–239. [Google Scholar] [CrossRef] [PubMed]
- Njiojob, C.N.; Owens, E.A.; Narayana, L.; Hyun, H.; Choi, H.S.; Henary, M. Tailored near-infrared contrast agents for image guided surgery. J. Med. Chem. 2015, 58, 2845–2854. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, D.; Teoh, C.L.; Park, S.J.; Kim, J.J.; Samanta, A.; Bi, R.; Dinish, U.S.; Olivo, M.; Piantino, M.; Louis, F.; et al. Seeing Elastin: A Near-Infrared Zwitterionic Fluorescent Probe for In Vivo Elastin Imaging. Chem 2018, 4, 1128–1138. [Google Scholar] [CrossRef] [Green Version]
- Park, M.H.; Jo, G.; Kim, E.J.; Hyun, H. Tumor-Targeted ZW800-1 Analog for Enhanced Tumor Imaging and Photothermal Therapy. Pharmaceutics 2021, 13, 1648. [Google Scholar] [CrossRef]
- Owens, E.A.; Hyun, H.; Kim, S.H.; Lee, J.H.; Park, G.; Ashitate, Y.; Choi, J.; Hong, G.H.; Alyabyev, S.; Lee, S.J. Highly charged cyanine fluorophores for trafficking scaffold degradation. Biomed. Mater. 2013, 8, 014109. [Google Scholar] [CrossRef]
- Zhu, H.; Cheng, P.; Chen, P.; Pu, K. Recent progress in the development of near-infrared organic photothermal and photodynamic nanotherapeutics. Biomater. Sci. 2018, 6, 746–765. [Google Scholar] [CrossRef]
- Kim, M.; Lee, J.H.; Nam, J.M. Plasmonic Photothermal Nanoparticles for Biomedical Applications. Adv. Sci. 2019, 6, 1900471. [Google Scholar] [CrossRef] [Green Version]
- Devarajan, P.V.; Patravale, V.B. Nanomedicine-prospects and challenges. Drug Deliv. Transl. Res. 2013, 3, 381. [Google Scholar] [CrossRef] [Green Version]
- Choi, H.S.; Liu, W.; Misra, P.; Tanaka, E.; Zimmer, J.P.; Ipe, B.I.; Bawendi, M.G.; Frangioni, J.V. Renal clearance of quantum dots. Nat. Biotechnol. 2007, 25, 1165–1170. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jo, G.; Kim, E.J.; Park, M.H.; Hyun, H. Tumor Targeting with Methotrexate-Conjugated Zwitterionic Near-Infrared Fluorophore for Precise Photothermal Therapy. Int. J. Mol. Sci. 2022, 23, 14127. https://doi.org/10.3390/ijms232214127
Jo G, Kim EJ, Park MH, Hyun H. Tumor Targeting with Methotrexate-Conjugated Zwitterionic Near-Infrared Fluorophore for Precise Photothermal Therapy. International Journal of Molecular Sciences. 2022; 23(22):14127. https://doi.org/10.3390/ijms232214127
Chicago/Turabian StyleJo, Gayoung, Eun Jeong Kim, Min Ho Park, and Hoon Hyun. 2022. "Tumor Targeting with Methotrexate-Conjugated Zwitterionic Near-Infrared Fluorophore for Precise Photothermal Therapy" International Journal of Molecular Sciences 23, no. 22: 14127. https://doi.org/10.3390/ijms232214127






