From Bipotent Neuromesodermal Progenitors to Neural-Mesodermal Interactions during Embryonic Development
Abstract
:1. Introduction
1.1. Paraxial Mesoderm
1.2. Neural Tube
1.3. The Neural Crest
2. Factors Underlying the Choice of NMPs to Generate Neural or Mesodermal Fates
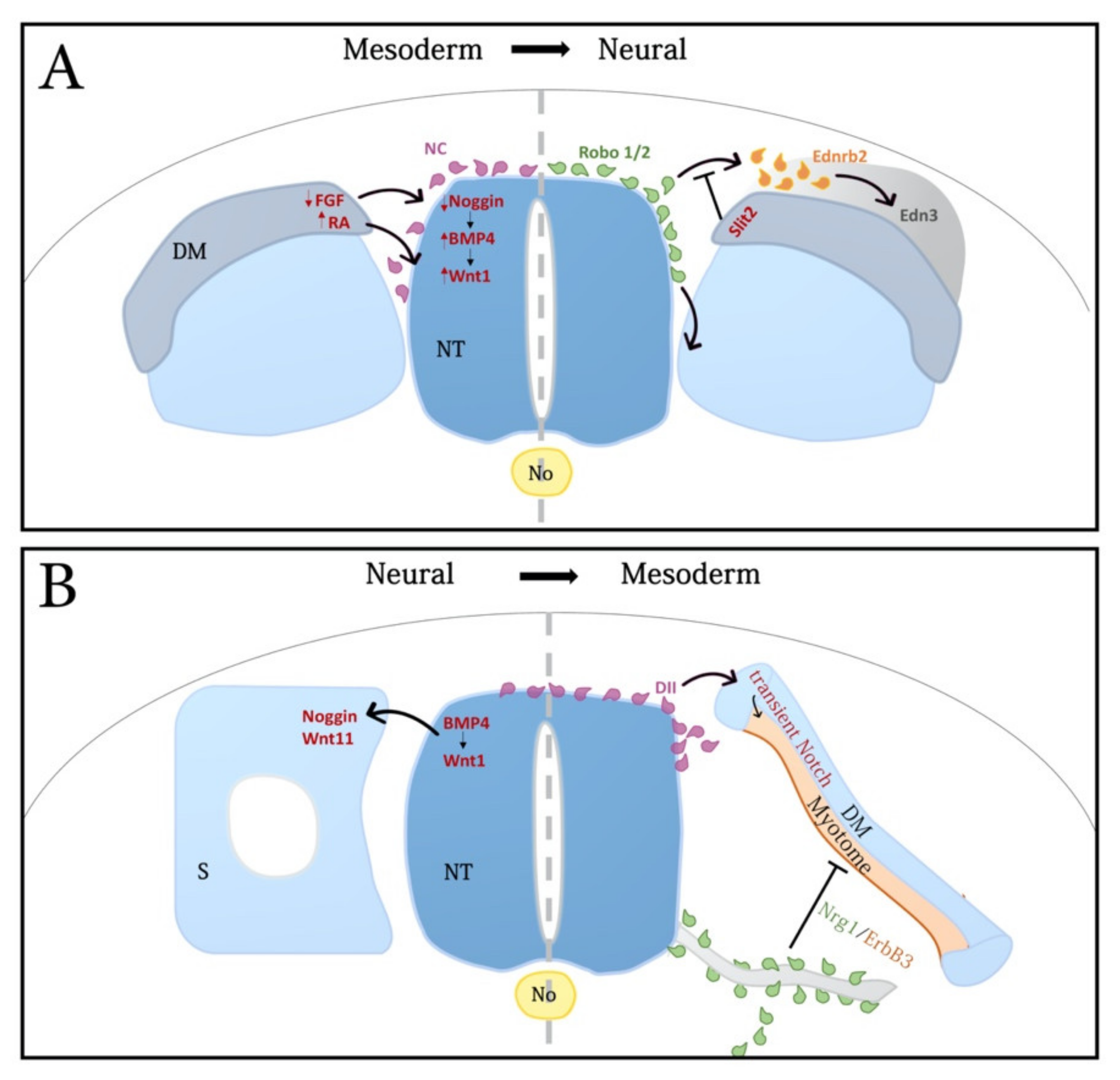
3. Mutual Interactions between Neural and Mesodermal Progenitors Shape the Body Plan
3.1. Mesoderm-Neural Tube Interactions
3.2. Interactions between NT and Somites Control Multiple Aspects of NC Development
3.2.1. NC Induction
3.2.2. The Timing of NC EMT
3.2.3. Segmental Migration of NC and Patterning of the PNS
3.2.4. Melanoblast Migration along the Dermomyotome-Derived Dermis
3.3. NC-Mesoderm Interactions in the Regulation of Myogenesis
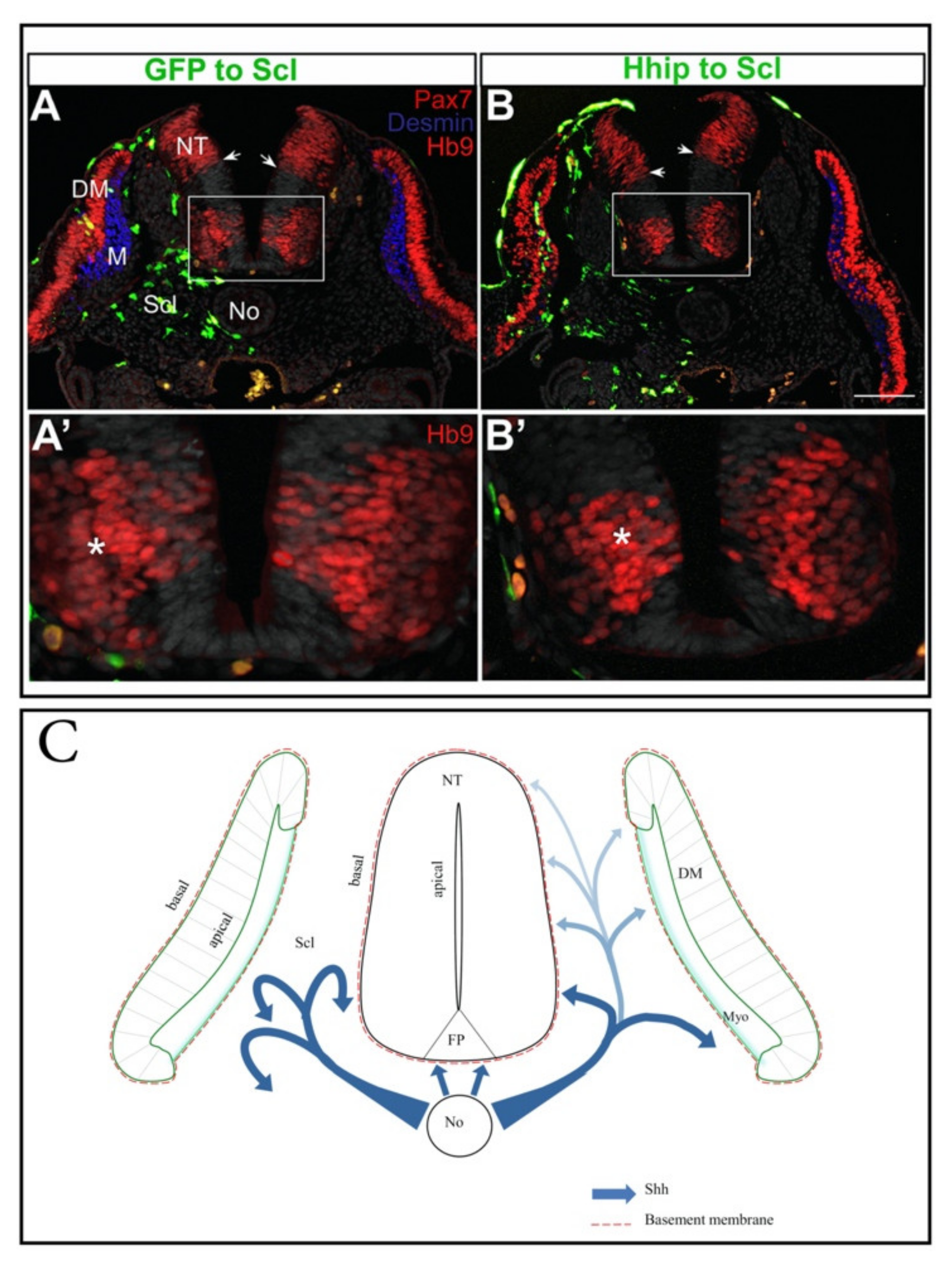
4. Shh, an Axial Midline Morphogen, Is Essential for Neural and Mesodermal Development
4.1. Shh in Muscle Development
4.2. Shh and Cartilage Development
4.3. Shh in the Coordination of Neural-Mesodermal Development
5. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Pourquie, O. Vertebrate somitogenesis. Annu. Dev.Biol. 2001, 17, 311–350. [Google Scholar] [CrossRef]
- Tzouanacou, E.; Wegener, A.; Wymeersch, F.J.; Wilson, V.; Nicolas, J.F. Redefining the Progression of Lineage Segregations during Mammalian Embryogenesis by Clonal Analysis. Dev. Cell 2009, 17, 365–376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henrique, D.; Abranches, E.; Verrier, L.; Storey, K.G. Neuromesodermal progenitors and the making of the spinal cord. Development 2015, 142, 2864–2875. [Google Scholar] [CrossRef] [Green Version]
- Steventon, B.; Martinez Arias, A. Evo-engineering and the cellular and molecular origins of the vertebrate spinal cord. Dev. Biol. 2017, 432, 3–13. [Google Scholar] [CrossRef]
- Frith, T.J.R.; Granata, I.; Wind, M.; Stout, E.; Thompson, O.; Neumann, K.; Stavish, D.; Heath, P.R.; Ortmann, D.; Hackland, J.O.S.; et al. Human axial progenitors generate trunk neural crest cells in vitro. eLife 2018, 7. [Google Scholar] [CrossRef]
- Hackland, J.O.S.; Shelar, P.B.; Sandhu, N.; Prasad, M.S.; Charney, R.M.; Gomez, G.A.; Frith, T.J.R.; García-Castro, M.I. FGF Modulates the Axial Identity of Trunk hPSC-Derived Neural Crest but Not the Cranial-Trunk Decision. Stem Cell Rep. 2019, 12, 920–933. [Google Scholar] [CrossRef] [Green Version]
- Gomez, G.A.; Prasad, M.S.; Wong, M.; Charney, R.M.; Shelar, P.B.; Sandhu, N.; Hackland, J.O.S.; Hernandez, J.C.; Leung, A.W.; Garcıá-Castro, M.I. WNT/β-catenin modulates the axial identity of embryonic stem cell-derived human neural crest. Development 2019, 146. [Google Scholar] [CrossRef] [Green Version]
- Lukoseviciute, M.; Mayes, S.; Sauka-Spengler, T. Neuromesodermal progenitor origin of trunk neural crest in vivo. BioRxiv 2021. [Google Scholar] [CrossRef]
- Brent, A.; Tabin, C. Developmental regulation of somite derivatives: Muscle, cartilage and tendon. Curr. Opin. Genet. Dev. 2002, 12, 548. [Google Scholar] [CrossRef]
- Christ, B.; Huang, R.; Scaal, M. Formation and differentiation of the avian sclerotome. Anat Embryol. 2004, 208, 333–350. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=15309628 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Scaal, M.; Christ, B. Formation and differentiation of the avian dermomyotome. Anat Embryol. 2004, 208, 411–424. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=15338303 (accessed on 2 July 2021). [CrossRef]
- Pourquie, O. Vertebrate somitogenesis: A novel paradigm for animal segmentation? Int. J. Dev. Biol. 2003, 47, 597–603. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=14756335 (accessed on 2 July 2021).
- Pourquie, O.; Fan, C.M.; Coltey, M.; Hirsinger, E.; Watanabe, Y.; Breant, C.; Francis-West, P.; Brickell, P.; Tessier-Lavigne, M.; Le Douarin, N.M. Lateral and axial signals involved in avian somite patterning: A role for BMP4. Cell 1996, 84, 461–471. [Google Scholar] [CrossRef] [Green Version]
- Brand-Saberi, B.; Christ, B. Evolution and development of distinct cell lineages derived from somites. Curr. Top. Dev. Biol. 2002, 48, 1–42. [Google Scholar]
- Kahane, N.; Cinnamon, Y.; Bachelet, I.; Kalcheim, C. The third wave of myotome colonization by mitotically competent progenitors: Regulating the balance between differentiation and proliferation during muscle development. Development 2001, 128, 2187–2198. [Google Scholar] [CrossRef] [PubMed]
- Cinnamon, Y.; Kahane, N.; Kalcheim, C. Characterization of the early development of specific hypaxial muscles from the ventrolateral myotome. Development 1999, 126, 4305–4315. [Google Scholar] [CrossRef]
- Gros, J.; Scaal, M.; Marcelle, C. A two-step mechanism for myotome formation in chick. Dev. Cell 2004, 6, 875–882. [Google Scholar] [CrossRef] [Green Version]
- Huang, R.; Christ, B. Origin of the epaxial and hypaxial myotome in avian embryos. Anat. Embryol. 2000, 202, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Kahane, N.; Cinnamon, Y.; Kalcheim, C. The cellular mechanism by which the dermomyotome contributes to the second wave of myotome development. Development 1998, 125, 4259–4271. [Google Scholar] [CrossRef]
- Ben-Yair, R.; Kalcheim, C. Lineage analysis of the avian dermomyotome sheet reveals the existence of single cells with both dermal and muscle progenitor fates. Development 2005, 132. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gros, J.; Manceau, M.; Thome, V.; Marcelle, C. A common somitic origin for embryonic muscle progenitors and satellite cells. Nature 2005, 435, 954–958. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=15843802 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Kassar-Duchosoy, L.; Giacone, E.; Gayraud-Morel, B.; Jory, A.; Gomes, D.; Tajbakhsh, S. Pax3/Pax7 mark a novel population of primitive myogenic cells during development. Genes Dev. 2005, 19, 1426–1431. [Google Scholar] [CrossRef] [Green Version]
- Buckingham, M.; Relaix, F. PAX3 and PAX7 as upstream regulators of myogenesis. Semin. Cell Dev. Biol. 2015. [Internet]. Available online: http://www.ncbi.nlm.nih.gov/pubmed/26424495 (accessed on 2 July 2021).
- Ben-Yair, R.; Kahane, N.; Kalcheim, C. Coherent development of dermomyotome and dermis from the entire mediolateral extent of the dorsal somite. Development 2003, 130. [Google Scholar] [CrossRef] [Green Version]
- Huang, R.J.; Zhi, Q.X.; Patel, K.; Wilting, J.; Christ, B. Dual origin and segmental organisation of the avian scapula. Development 2000, 127, 3789–3794. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=10934023 (accessed on 2 July 2021). [CrossRef]
- Cinnamon, Y.; Ben-Yair, R.; Kalcheim, C. Differential effects of N-cadherin-mediated adhesion on the development of myotomal waves. Development 2006, 133. [Google Scholar] [CrossRef] [Green Version]
- Kahane, N.; Kalcheim, C. Identification of early postmitotic cells in distinct embryonic sites and their possible roles in morphogenesis. Cell Tissue Res. 1998, 294, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Kahane, N.; Cinnamon, Y.; Kalcheim, C. The roles of cell migration and myofiber intercalation in patterning formation of the postmitotic myotome. Development 2002, 129, 2675–2687. [Google Scholar] [CrossRef] [PubMed]
- Halperin-Barlev, O.; Kalcheim, C. Sclerotome-derived slit1 drives directional migration and differentiation of robo2-expressing pioneer myoblasts. Development 2011, 138. [Google Scholar] [CrossRef] [Green Version]
- Kalcheim, C.; Kahane, N.; Cinnamon, Y.; Ben-Yair, R. Mechanisms of lineage segregation in the avian dermomyotome. Anat. Embryol. 2006, 211. [Google Scholar] [CrossRef]
- Saraga-Babic, M.; Krolo, M.; Sapunar, D.; Terzic, J.; Biocic, M. Differences in origin and fate between the cranial and caudal spinal cord during normal and disturbed human development. Acta Neuropathol. 1996, 91, 194–199. [Google Scholar] [CrossRef]
- Colas, J.F.; Schoenwolf, G.C. Towards a cellular and molecular understanding of neurulation. Dev. Dyn. 2001, 221, 117–145. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=11376482 (accessed on 2 July 2021). [CrossRef]
- Shimokita, E.; Takahashi, Y. Secondary neurulation: Fate-mapping and gene manipulation of the neural tube in tail bud. Dev. Growth Differ. 2011, 53, 401–410. [Google Scholar] [CrossRef]
- Gonzalez-Gobartt, E.; Blanco-Ameijeiras, J.; Usieto, S.; Allio, G.; Benazeraf, B.; Martí, E. Cell intercalation driven by SMAD3 underlies secondary neural tube formation. Dev. Cell 2021, 56, 1147.e6–1163.e6. [Google Scholar] [CrossRef] [PubMed]
- Andrews, M.G.; Kong, J.; Novitch, B.G.; Butler, S.J. New perspectives on the mechanisms establishing the dorsal-ventral axis of the spinal cord. Curr. Top. Dev. Biol. 2019, 132, 417–450. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30797516 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Helms, A.W.; Johnson, J.E. Specification of dorsal spinal cord interneurons. Curr. Opin. Neurobiol. 2003, 13, 42–49. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=12593981 (accessed on 2 July 2021). [CrossRef]
- Le Dréau, G.; Martí, E. The multiple activities of BMPs during spinal cord development. Cell. Mol. Life Sci. 2013, 70, 4293–4305. [Google Scholar] [CrossRef]
- Schoenwolf, G.C.; Smith, J.L. Mechanisms of neurulation: Traditional viewpoint and recent advances. Development 1990, 109, 243–270. [Google Scholar] [CrossRef] [PubMed]
- Yamada, T.; Placzek, M.; Tanaka, H.; Dodd, J.; Jessell, T.M. Control of cell pattern in the developing nervous system: Polarizing activity of the floor plate and notochord. Cell 1991, 64, 635–647. [Google Scholar] [CrossRef]
- Ward, L.; Pang, A.S.W.; Evans, S.E.; Stern, C.D. The role of the notochord in amniote vertebral column segmentation. Dev. Biol. 2018, 439, 3–18. [Google Scholar] [CrossRef]
- Cohen, M.; Briscoe, J.; Blassberg, R. Morphogen interpretation: The transcriptional logic of neural tube patterning. Curr Opin Genet Dev 2013, 23, 423–428. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23725799 (accessed on 2 July 2021). [CrossRef]
- Dessaud, E.; Yang, L.L.; Hill, K.; Cox, B.; Ulloa, F.; Ribeiro, A.; Mynett, A.; Novitch, B.G.; Briscoe, J. Interpretation of the sonic hedgehog morphogen gradient by a temporal adaptation mechanism. Nature 2007, 450, 717–720. Available online: http://www.ncbi.nlm.nih.gov/pubmed/18046410 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Ribes, V.; Balaskas, N.; Sasai, N.; Cruz, C.; Dessaud, E.; Cayuso, J.; Tozer, S.; Yang, L.L.; Novitch, B.; Marti, E.; et al. Distinct Sonic Hedgehog signaling dynamics specify floor plate and ventral neuronal progenitors in the vertebrate neural tube. Genes Dev. 2010, 24, 1186–1200. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=20516201 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Ericson, J.; Briscoe, J.; Rashbass, P.; Van Heyningen, V.; Jessell, T.M. Graded sonic hedgehog signaling and the specification of cell fate in the ventral neural tube. Cold Spring Harb. Symp. Quant. Biol. 1997, 62, 451–466. [Google Scholar] [PubMed]
- Gouti, M.; Metzis, V.; Briscoe, J. The route to spinal cord cell types: A tale of signals and switches. Trends Genet 2015, 31, 282–289. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25823696 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Jessell, T.M. Neuronal specification in the spinal cord: Inductive signals and transcriptional codes. Nat. Rev. Genet 2000, 1, 20–29. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=11262869 (accessed on 2 July 2021). [CrossRef]
- Briscoe, J.; Novitch, B.G. Regulatory pathways linking progenitor patterning, cell fates and neurogenesis in the ventral neural tube. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 57–70. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17282991 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Liem, K.F., Jr.; Tremml, G.; Jessell, T.M. A role for the roof plate and its resident TGFb-related proteins in neuronal patterning in the dorsal spinal cord. Cell 1997, 91, 127–138. [Google Scholar] [CrossRef] [Green Version]
- Le Dréau, G.; Garcia-Campmany, L.; Angeles Rabadán, M.; Ferronha, T.; Tozer, S.; Briscoe, J.; Martí, E. Canonical BMP7 activity is required for the generation of discrete neuronal populations in the dorsal spinal cord. Development 2012, 139, 259–268. [Google Scholar] [CrossRef] [Green Version]
- Hazen, V.M.; Phan, K.D.; Hudiburgh, S.; Butler, S.J. Inhibitory Smads differentially regulate cell fate specification and axon dynamics in the dorsal spinal cord. Dev. Biol. 2011, 356, 566–575. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21718693 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Augsburger, A.; Schuchardt, A.; Hoskins, S.; Dodd, J.; Butler, S. Bmps as mediators of roof plate repulsion of commissural neurons. Neuron 1999, 24, 127–141. [Google Scholar] [CrossRef] [Green Version]
- Andrews, M.G.; Del Castillo, L.M.; Ochoa-Bolton, E.; Yamauchi, K.; Smogorzewski, J.; Butler, S.J. BMPs direct sensory interneuron identity in the developing spinal cord using signal-specific not morphogenic activities. eLife 2017, 6. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28925352 (accessed on 2 July 2021). [CrossRef]
- Tanabe, Y.; Jessell, T.M. Diversity and pattern in the developing spinal cord. Science 1996, 274, 1115–1123. [Google Scholar] [CrossRef] [Green Version]
- Diez del Corral, R.; Olivera-Martinez, I.; Goriely, A.; Gale, E.; Maden, M.; Storey, K. Opposing FGF and retinoid pathways control ventral neural pattern, neuronal differentiation, and segmentation during body axis extension. Neuron 2003, 40, 65–79. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=14527434 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Lai, H.C.; Seal, R.P.; Johnson, J.E. Making sense out of spinal cord somatosensory development. Development 2016, 143, 3434–3448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Le Douarin, N.M.; Kalcheim, C. The Neural Crest, 2nd ed.; Cambridge University Press: New York, NY, USA, 1999. [Google Scholar]
- Piacentino, M.L.; Li, Y.; Bronner, M.E. Epithelial-to-mesenchymal transition and different migration strategies as viewed from the neural crest. Curr. Opin. Cell Biol. 2020, 66, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Nitzan, E.; Avraham, O.; Kahane, N.; Ofek, S.; Kumar, D.; Kalcheim, C. Dynamics of BMP and Hes1/Hairy1 signaling in the dorsal neural tube underlies the transition from neural crest to definitive roof plate. BMC Biol. 2016, 14, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ofek, S.; Wiszniak, S.; Kagan, S.; Tondl, M.; Schwarz, Q.; Kalcheim, C. Notch signaling is a critical initiator of roof plate formation as revealed by the use of RNA profiling of the dorsal neural tube. BMC Biol. 2021, 19. [Google Scholar] [CrossRef] [PubMed]
- Chizhikov, V.V.; Millen, K.J. Mechanisms of roof plate formation in the vertebrate CNS. Nat. Rev. Neurosci. 2004, 5, 808–812. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=15378040 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Guillot, C.; Michaut, A.; Rabe, B.; Pourquié, O. Dynamics of primitive streak regression controls the fate of neuro-mesodermal progenitors in the chicken embryo. eLife 2021, 1–36. [Google Scholar] [CrossRef]
- Goto, H.; Kimmey, S.C.; Row, R.H.; Matus, D.Q.; Martin, B.L. FGF and canonical Wnt signaling cooperate to induce paraxial mesoderm from tailbud neuromesodermal progenitors through regulation of a two-step epithelial to mesenchymal transition. Development 2017, 144, 1412–1421. [Google Scholar] [CrossRef] [Green Version]
- Kinney, B.A.; Al Anber, A.; Row, R.H.; Tseng, Y.J.; Weidmann, M.D.; Knaut, H.; Martin, B.L. Sox2 and Canonical Wnt Signaling Interact to Activate a Developmental Checkpoint Coordinating Morphogenesis with Mesoderm Fate Acquisition. Cell Rep. 2020, 33, 108311. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Antin, P.; Berx, G.; Blanpain, C.; Brabletz, T.; Bronner, M.; Campbell, K.; Cano, A.; Casanova, J.; Christofori, G.; et al. Guidelines and definitions for research on epithelial–mesenchymal transition. Nat. Rev. Mol. Cell Biol. 2020, 21, 341–352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chapman, D.L.; Papaioannou, V.E. Three neural tubes in mouse embryos with mutations in T-box gene Tbx6. Nature 1998, 391, 695–697. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, T.P.; Takada, S.; Yoshikawa, Y.; Wu, N.Y.; McMahon, A.P. T (Brachyury) is a direct target of Wnt3a during paraxial mesoderm specification. Genes Dev. 1999, 13, 3185–3190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoshikawa, Y.; Fujimori, T.; McMahon, A.P.; Takada, S. Evidence that absence of Wnt-3a signaling promotes neuralization instead of paraxial mesoderm development in the mouse. Dev.Biol. 1997, 183, 234–242. [Google Scholar] [CrossRef] [Green Version]
- Ciruna, B.G.; Schwartz, L.; Harpal, K.; Yamaguchi, T.P.; Rossant, J. Chimeric analysis of fibroblast growth factor receptor-1 (Fgfr1) function: A role for FGFR1 in morphogenetic movement through the primitive streak. Development 1997, 124, 2829–2841. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9226454 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Garriock, R.J.; Chalamalasetty, R.B.; Kennedy, M.W.; Canizales, L.C.; Lewandoski, M.; Yamaguchi, T.P. Lineage tracing of neuromesodermal progenitors reveals novel wnt-dependent roles in trunk progenitor cell maintenance and differentiation. Development 2015, 142, 1628–1638. [Google Scholar] [CrossRef] [Green Version]
- Wymeersch, F.J.; Huang, Y.; Blin, G.; Cambray, N.; Wilkie, R.; Wong, F.C.K.; Wilson, V. Position-dependent plasticity of distinct progenitor types in the primitive streak. eLife 2016, 5. [Google Scholar] [CrossRef]
- Cunningham, T.J.; Brade, T.; Sandell, L.L.; Lewandoski, M.; Trainor, P.A.; Colas, A.; Mercola, M.; Duester, G. Retinoic acid activity in undifferentiated neural progenitors is sufficient to fulfill its role in restricting Fgf8 expression for somitogenesis. PLoS ONE 2015, 10, 1–15. [Google Scholar] [CrossRef]
- Gouti, M.; Delile, J.; Stamataki, D.; Wymeersch, F.J.; Huang, Y.; Kleinjung, J.; Wilson, V.; Briscoe, J. A Gene Regulatory Network Balances Neural and Mesoderm Specification during Vertebrate Trunk Development. Dev. Cell 2017, 41, 243.e7–261.e7. [Google Scholar] [CrossRef]
- Appel, B.; Korzh, V.; Glasgow, E.; Thor, S.; Edlund, T.; Dawid, I.B.; Eisen, J.S. Motoneuron fate specification revealed by patterned LIM homeobox gene expression in embryonic zebrafish. Development 1995, 121, 4117–4125. [Google Scholar] [CrossRef]
- Eisen, J.S. Determination of primary motoneuron identity in developing zebrafish embryos. Science 1991, 252, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Bisgrove, B.W.; Raible, D.W.; Walter, V.; Eisen, J.S.; Grunwald, D.J. Expression of c-ret in the zebrafish embryo: Potential roles in motoneuronal development. J. Neurobiol. 1997, 33, 749–768. [Google Scholar] [CrossRef]
- Eisen, J.S.; Pike, S.H. The spt-1 mutation alters segmental arrangement and axonal development of identified neurons in the spinal cord of the embryonic zebrafish. Neuron 1991, 6, 767–776. [Google Scholar] [CrossRef]
- Lewis, K.E.; Eisen, J.S. Paraxial mesoderm specifies zebrafish primary motoneuron subtype identity. Development 2004, 131, 891–902. [Google Scholar] [CrossRef] [Green Version]
- Inoue, A.; Takahashi, M.; Hatta, K.; Hotta, Y.; Okamoto, H. Developmental regulation of Islet-1 mRNA expression during neuronal differentiation in embryonic zebrafish. Dev. Dyn. 1994, 199, 1–11. [Google Scholar] [CrossRef]
- Tokumoto, M.; Gong, Z.Y.; Tsubokawa, T.; Hew, C.L.; Uyemura, K.; Hotta, Y.; Okamoto, H. Molecular heterogeneity among primary motoneurons and within myotomes revealed by the differential mRNA expression of novel islet-1 homologs in embryonic zebrafish. Dev.Biol. 1995, 171, 578–589. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kimmel, C.B.; Miller, C.T.; Kruze, G.; Ullmann, B.; Bremiller, R.A.; Larison, K.D.; Snyder, H.C. The shaping of pharyngeal cartilages during early development of the zebrafish. Dev. Biol. 1998, 203, 245–263. [Google Scholar] [CrossRef] [Green Version]
- Roy, M.N.; Prince, V.E.; Ho, R.K. Heat shock produces periodic somitic disturbances in the zebrafish embryo. Mech. Dev. 1999, 85, 27–34. [Google Scholar] [CrossRef]
- Diez del Corral, R.; Breitkreuz, D.; Storey, K.G. Onset of neuronal differentiation is regulated by paraxial mesoderm and requires attenuation of FGF signalling. Development 2002, 129, 1681–1691. [Google Scholar] [CrossRef]
- Veenvliet, J.V.; Bolondi, A.; Kretzmer, H.; Haut, L.; Scholze-Wittler, M.; Schifferl, D.; Koch, F.; Guignard, L.; Kumar, A.S.; Pustet, M.; et al. Modeling mammalian trunk development in a dish. Science 2020, 370, eaba4937. Available online: https://pubmed.ncbi.nlm.nih.gov/33303587/ (accessed on 2 July 2021). [CrossRef] [PubMed]
- Sosic, D.; Brand-Saberi, B.; Schmidt, C.; Christ, B.; Olson, E.N. Regulation of paraxis expression and somite formation by ectoderm- and neural tube-derived signals. Dev.Biol. 1997, 185, 229–243. [Google Scholar] [CrossRef] [Green Version]
- Basch, M.L.; Bronner-Fraser, M. Neural crest inducing signals. Adv. Exp. Med. Biol. 2006, 589, 24–31. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=17076273 (accessed on 2 July 2021). [PubMed]
- Basch, M.L.; Bronner-Fraser, M.; Garcia-Castro, M.I. Specification of the neural crest occurs during gastrulation and requires Pax7. Nature 2006, 441, 218–222. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=16688176 (accessed on 2 July 2021). [CrossRef]
- Stuhlmiller, T.J.; Garcia-Castro, M.I. Current perspectives of the signaling pathways directing neural crest induction. Cell Mol Life Sci. 2012. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=22547091 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Steventon, B.; Araya, C.; Linker, C.; Kuriyama, S.; Mayor, R. Differential requirements of BMP and Wnt signalling during gastrulation and neurulation define two steps in neural crest induction. Development 2009, 136, 771–779. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=19176585 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Monsoro-Burq, A.H.; Fletcher, R.B.; Harland, R.M. Neural crest induction by paraxial mesoderm in Xenopus embryos requires FGF signals. Development 2003, 130, 3111–3124. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=12783784 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Bonstein, L.; Elias, S.; Frank, D. Paraxial-fated mesoderm is required for neural crest induction in Xenopus embryos. Dev. Biol. 1998, 193, 156–168. [Google Scholar] [CrossRef] [Green Version]
- Bonano, M.; Tribulo, C.; De Calisto, J.; Marchant, L.; Sanchez, S.S.; Mayor, R.; Aybar, M.J. A new role for the Endothelin-1/Endothelin-A receptor signaling during early neural crest specification. Dev. Biol. 2008, 323, 114–129. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=18775422 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Mayor, R.; Morgan, R.; Sargent, M.G. Induction of the prospective neural crest of xenopus. Development 1995, 121, 767–777. [Google Scholar] [CrossRef]
- Hong, C.S.; Park, B.Y.; Saint-Jeannet, J.P. Fgf8a induces neural crest indirectly through the activation of Wnt8 in the paraxial mesoderm. Development 2008, 135, 3903–3910. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=18997112 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Selleck, M.A.J.; Bronner-Fraser, M. Origins of the avian neural crest: The role of neural plate-epidermal interactions. Development 1995, 121, 525–538. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=7768190 (accessed on 2 July 2021). [CrossRef]
- Loring, J.F.; Erickson, C.A. Neural crest cell migratory pathways in the trunk of the chick embryo. Dev. Biol. 1987, 121, 220–236. [Google Scholar] [CrossRef]
- Teillet, M.-A.; Kalcheim, C.; Le Douarin, N.M. Formation of the dorsal root ganglia in the avian embryo: Segmental origin and migratory behavior of neural crest progenitor cells. Dev. Biol. 1987, 120. [Google Scholar] [CrossRef]
- Kalcheim, C.; Teillet, M.-A. Consequences of somite manipulation on the pattern of dorsal root ganglion development. Development 1989, 106, 85–93. [Google Scholar] [CrossRef]
- Sela-Donenfeld, D.; Kalcheim, C. Regulation of the onset of neural crest migration by coordinated activity of BMP4 and Noggin in the dorsal neural tube. Development 1999, 126, 4749–4762. [Google Scholar] [CrossRef] [PubMed]
- Burstyn-Cohen, T.; Stanleigh, J.; Sela-Donenfeld, D.; Kalcheim, C. Canonical Wnt activity regulates trunk neural crest delamination linking BMP/noggin signaling with G1/S transition. Development 2004, 131, 5327–5339. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sela-Donenfeld, D.; Kalcheim, C. Inhibition of noggin expression in the dorsal neural tube by somitogenesis: A mechanism for coordinating the timing of neural crest emigration. Development 2000, 127, 4845–4854. [Google Scholar] [CrossRef]
- Martínez-Morales, P.L.; del Corral, R.D.; Olivera-Martínez, I.; Quiroga, A.C.; Das, R.M.; Barbas, J.A.; Storey, K.G.; Morales, A.V. FGF and retinoic acid activity gradients control the timing of neural crest cell emigration in the trunk. J. Cell Biol. 2011, 194, 489–503. [Google Scholar] [CrossRef] [Green Version]
- Goldstein, R.S.; Kalcheim, C. Normal segmentation and size of the primary sympathetic ganglia depend upon the alternation of rostrocaudal properties of the somites. Development 1991, 112, 327–334. [Google Scholar] [CrossRef]
- Keynes, R.; Cook, G.; Davies, J.; Lumsden, A.; Norris, W.; Stern, C. Segmentation and the development of the vertebrate nervous system. J. Physiol. 1990, 84, 27–32. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=2193147 (accessed on 2 July 2021).
- Keynes, R.; Stern, C.D. Segmentation in the vertebrate nervous system. Nature 1984, 310, 786–789. [Google Scholar] [CrossRef]
- Kalcheim, C. Mechanisms of Early Neural crest Development: From Cell Specification to Migration; Elsevier: Amsterdam, The Netherlands, 2000; Volume 200. [Google Scholar]
- Goldstein, R.S.; Kalcheim, C. Determination of epithelial half-somites in skeletal morphogenesis. Development 1992, 116. [Google Scholar] [CrossRef]
- Bruggeman, B.J.; Maier, J.A.; Mohiuddin, Y.S.; Powers, R.; Lo, Y.; Guimaraes-Camboa, N.; Evans, S.M.; Harfe, B.D. Avian intervertebral disc arises from rostral sclerotome and lacks a nucleus pulposus: Implications for evolution of the vertebrate disc. Dev. Dyn. 2012, 241, 675–683. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=22354863 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Kuriyama, S.; Mayor, R. Molecular analysis of neural crest migration. Philos. Trans. R. Soc. L. B Biol. Sci. 2008, 363, 1349–1362. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=18198151 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Krull, C.E. Segmental organization of neural crest migration. Mech. Dev. 2001, 105, 37–45. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=11429280 (accessed on 2 July 2021). [CrossRef]
- Gammill, L.S.; Gonzalez, C.; Gu, C.; Bronner-Fraser, M. Guidance of trunk neural crest migration requires neuropilin 2/semaphorin 3F signaling. Development 2006, 133, 99–106. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=16319111 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Debby-Brafman, A.; Burstyn-Cohen, T.; Klar, A.; Kalcheim, C. F-spondin is expressed in somite regions avoided by neural crest cells and mediates the inhibition of distinct somitic domains to neural crest migration. Neuron 1999, 22, 475–488. [Google Scholar] [CrossRef] [Green Version]
- Roffers-Agarwal, J.; Gammill, L.S. Neuropilin receptors guide distinct phases of sensory and motor neuronal segmentation. Development 2009, 136, 1879–1888. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=19403658 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Schwarz, Q.; Maden, C.H.; Davidson, K.; Ruhrberg, C. Neuropilin-mediated neural crest cell guidance is essential to organise sensory neurons into segmented dorsal root ganglia. Development 2009, 136, 1785–1789. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=19386662 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Krull, C.E.; Lansford, R.; Gale, N.W.; Collazo, A.; Marcelle, C.; Yancopoulos, G.D.; Fraser, S.E.; Bronner Fraser, M.; Bronner-Fraser, M. Interactions of Eph-related receptors and ligands confer rostrocaudal pattern to trunk neural crest migration. Curr. Biol. 1997, 7, 571–580. [Google Scholar] [CrossRef] [Green Version]
- Erickson, C.A.; Duong, T.D.; Tosney, K.W. Descriptive and experimental analysis of the dispersion of neural crest cells along the dorsolateral path and their entry into ectoderm in the chick embryo. Dev. Biol. 1992, 151, 251–272. [Google Scholar] [CrossRef] [Green Version]
- Nitzan, E.; Pfaltzgraff, E.R.; Labosky, P.A.; Kalcheim, C. Neural crest and Schwann cell progenitor-derived melanocytes are two spatially segregated populations similarly regulated by Foxd3. Proc. Natl. Acad. Sci. USA 2013, 110. [Google Scholar] [CrossRef] [Green Version]
- Adameyko, I.; Lallemend, F.; Aquino, J.B.; Pereira, J.A.; Topilko, P.; Muller, T.; Fritz, N.; Beljajeva, A.; Mochii, M.; Liste, I.; et al. Schwann cell precursors from nerve innervation are a cellular origin of melanocytes in skin. Cell 2009, 139, 366–379. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=19837037 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Jia, L.; Cheng, L.; Raper, J. Slit/Robo signaling is necessary to confine early neural crest cells to the ventral migratory pathway in the trunk. Dev. Biol. 2005, 282, 411–421. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=15950606 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Oakley, R.A.; Lasky, C.J.; Erickson, C.A.; Tosney, K.W. Glycoconjugates mark a transient barrier to neural crest migration in the chicken embryo. Development 1994, 120, 103–114. [Google Scholar] [CrossRef]
- Oakley, R.A.; Tosney, K.W. Peanut agglutinin and chondroitin-6-sulfate are molecular markers for tissues that act as barriers to axon advance in the avian embryo. Dev. Biol. 1991, 147, 187–206. [Google Scholar] [CrossRef] [Green Version]
- Santiago, A.; Erickson, C.A. Ephrin-B ligands play a dual role in the control of neural crest cell migration. Development 2002, 129, 3621–3632. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.L.; Hall, R.; Erickson, C.A. Directing pathfinding along the dorsolateral path—The role of EDNRB2 and EphB2 in overcoming inhibition. Development 2008, 135, 4113–4122. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=19004859 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Lecoin, L.; Sakurai, T.; Ngo, M.T.; Abe, Y.; Yanagisawa, M.; Le Douarin, N.M. Cloning and characterization of a novel endothelin receptor subtype in the avian class. Proc. Natl. Acad. Sci. USA 1998, 95, 3024–3029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nataf, V.; Amemiya, A.; Yanagisawa, M.; Le Douarin, N.M. The expression pattern of endothelin 3 in the avian embryo. Mech. Dev. 1998, 73, 217–220. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9622636 (accessed on 2 July 2021). [CrossRef]
- Krispin, S.; Nitzan, E.; Kassem, Y.; Kalcheim, C. Evidence for a dynamic spatiotemporal fate map and early fate restrictions of premigratory avian neural crest. Development 2010, 137, 585–595. [Google Scholar] [CrossRef] [Green Version]
- Nagy, N.; Goldstein, A.M. Endothelin-3 regulates neural crest cell proliferation and differentiation in the hindgut enteric nervous system. Dev. Biol. 2006, 293, 203–217. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=16519884 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Noden, D.M.; Trainor, P.A. Relations and interactions between cranial mesoderm and neural crest populations. J. Anat. 2005, 207, 575–601. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=16313393 (accessed on 2 July 2021). [CrossRef]
- Couly, G.F.; Coltey, P.M.; Le Douarin, N.M. The developmental fate of the cephalic mesoderm in quail-chick chimeras. Development 1992, 114, 1–15. [Google Scholar] [CrossRef]
- Cerny, R.; Meulemans, D.; Berger, J.; Wilsch-Brauninger, M.; Kurth, T.; Bronner-Fraser, M.; Epperlein, H.H. Combined intrinsic and extrinsic influences pattern cranial neural crest migration and pharyngeal arch morphogenesis in axolotl. Dev. Biol. 2004, 266, 252–269. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=14738875 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Köntges, G.; Lumsden, A. Rhombencephalic neural crest segmentation is preserved throughout craniofacial ontogeny. Development 1996, 122, 3229–3242. [Google Scholar] [CrossRef]
- Noden, D.M. The embryonic origins of avian cephalic and cervical muscles and associated connective tissues. Am. J. Anat. 1983, 168, 257–276. [Google Scholar] [CrossRef]
- Grammatopoulos, G.A.; Bell, E.; Toole, L.; Lumsden, A.; Tucker, A.S. Homeotic transformation of branchial arch identity after Hoxa2 overexpression. Development 2000, 127, 5355–5365. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=11076757 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Schilling, T.F.; Kimmel, C.B. Musculoskeletal patterning in the pharyngeal segments of the zebrafish embryo. Development 1997, 124, 2945–2960. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9247337 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Grenier, J.; Teillet, M.A.; Grifone, R.; Kelly, R.G.; Duprez, D. Relationship between neural crest cells and cranial mesoderm during head muscle development. PLoS ONE 2009, 4, e4381. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=19198652 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Rinon, A.; Lazar, S.; Marshall, H.; Buchmann-Moller, S.; Neufeld, A.; Elhanany-Tamir, H.; Taketo, M.M.; Sommer, L.; Krumlauf, R.; Tzahor, E. Cranial neural crest cells regulate head muscle patterning and differentiation during vertebrate embryogenesis. Development 2007, 134, 3065–3075. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=17652354 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Sela-Donenfeld, D.; Kalcheim, C. Localized BMP4-noggin interactions generate the dynamic patterning of noggin expression in somites. Dev. Biol. 2002, 246. [Google Scholar] [CrossRef]
- Brill, G.; Kahane, N.; Carmeli, C.; Von Schack, D.; Barde, Y.-A.; Kalcheim, C. Epithelial-mesenchymal conversion of dermatome progenitors requires neural tube-derived signals: Characterization of the role of Neurotrophin-3. Development 1995, 121. [Google Scholar] [CrossRef]
- Spence, M.S.; Yip, J.; Erickson, C.A. The dorsal neural tube organizes the dermamyotome and induces axial myocytes in the avian embryo. Development 1996, 122, 231–241. [Google Scholar] [CrossRef]
- Olivera-Martinez, I.; Thelu, J.; Teillet, M.; Dhouailly, D. Dorsal dermis development depends on a signal from the dorsal neural tube, which can be substituted by Wnt-1. Mech. Dev. 2001, 100, 233–244. [Google Scholar] [CrossRef]
- Capdevila, J.; Tabin, C.; Johnson, R.L. Control of dorsoventral somite patterning by Wnt-1 and b-catenin. Dev. Biol. 1998, 193, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Ikeya, M.; Takada, S. Wnt signaling from the dorsal neural tube is required for the formation of the medial dermomyotome. Development 1998, 125, 4969–4976. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Tanaka, M.; Münsterberg, A. Expression of b-catenin in the developing chick myotome is regulated by myogenic signals. Development 2000, 127, 4105–4113. [Google Scholar] [CrossRef]
- Marcelle, C.; Stark, M.R.; Bronner-Fraser, M. Coordinate actions of BMPs, Wnts, Shh and noggin mediate patterning of the dorsal somite. Development 1997, 124, 3955–3963. [Google Scholar] [CrossRef] [PubMed]
- Rios, A.C.; Serralbo, O.; Salgado, D.; Marcelle, C. Neural crest regulates myogenesis through the transient activation of NOTCH. Nature 2011, 473, 532–535. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=21572437 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Van Ho, S.; Brohl, D.; Aurade, F.; Rattenbach, R.; Relaix, F. Neural crest cell lineage restricts skeletal muscle progenitor cell differentiation through Neuregulin-ErbB3 signaling. Dev. Cell 2011, 21, 273–287. [Google Scholar] [PubMed]
- Kalcheim, C. Regulation of Trunk Myogenesis by the Neural Crest: A New Facet of Neural Crest-Somite Interactions. Dev. Cell 2011, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borycki, A.G.; Mendham, L.; Emerson, C.P., Jr. Control of somite patterning by Sonic hedgehog and its downstream signal response genes. Development 1998, 125, 777–790. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9435297 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Briscoe, J. Making a grade: Sonic Hedgehog signalling and the control of neural cell fate. EMBO J. 2009, 28, 457–465. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=19197245 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Cairns, D.M.; Sato, M.E.; Lee, P.G.; Lassar, A.B.; Zeng, L. A gradient of Shh establishes mutually repressing somitic cell fates induced by Nkx3.2 and Pax3. Dev. Biol. 2008, 323, 152–165. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=18796301 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Ericson, J.; Rashbass, P.; Schedl, A.; Brenner-Morton, S.; Kawakami, A.; Van Heyningen, V.; Jessell, T.M.; Briscoe, J. Pax6 controls progenitor cell identity and neuronal fate in response to graded shh signaling. Cell 1997, 90, 169–180. [Google Scholar] [CrossRef] [Green Version]
- Gustafsson, M.K.; Pan, H.; Pinney, D.F.; Liu, Y.L.; Lewandowski, A.; Epstein, D.J.; Emerson, C.P., Jr. Myf5 is a direct target of long-range Shh signaling and Gli regulation for muscle specification. Genes Dev. 2002, 16, 114–126. [Google Scholar] [CrossRef] [Green Version]
- Goodrich, L.V.; Milenkovic, L.; Higgins, K.M.; Scott, M.P. Altered neural cell fates and medulloblastoma in mouse patched mutants. Science 1997, 277, 1109–1113. [Google Scholar] [CrossRef]
- Hidalgo, A.; Ingham, P. Cell patterning in the Drosophila segment: Spatial regulation of the segment polarity gene patched. Development 1990, 110, 291–301. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2081466 (accessed on 2 July 2021). [CrossRef]
- Van den Heuvel, M.; Ingham, P.W. smoothened encodes a receptor-like serpentine protein required for hedgehog signalling. Nature 1996, 382, 547–551. Available online: http://www.ncbi.nlm.nih.gov/pubmed/8700230 (accessed on 2 July 2021). [CrossRef]
- Caspary, T.; Larkins, C.E.; Anderson, K.V. The graded response to Sonic Hedgehog depends on cilia architecture. Dev. Cell 2007, 12, 767–778. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17488627 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Rohatgi, R.; Milenkovic, L.; Scott, M.P. Patched1 regulates hedgehog signaling at the primary cilium. Science 2007, 317, 372–376. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17641202 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Milenkovic, L.; Scott, M.P.; Rohatgi, R. Lateral transport of Smoothened from the plasma membrane to the membrane of the cilium. J. Cell Biol. 2009, 187, 365–374. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19948480 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Rohatgi, R.; Milenkovic, L.; Corcoran, R.B.; Scott, M.P. Hedgehog signal transduction by Smoothened: Pharmacologic evidence for a 2-step activation process. Proc. Natl. Acad. Sci. USA 2009, 106, 3196–3201. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19218434 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Briscoe, J.; Therond, P.P. The mechanisms of Hedgehog signalling and its roles in development and disease. Nat. Rev. Mol. Cell Biol. 2013, 14, 416–429. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23719536 (accessed on 2 July 2021). [CrossRef] [PubMed]
- Ribes, V.; Briscoe, J. Establishing and interpreting graded Sonic Hedgehog signaling during vertebrate neural tube patterning: The role of negative feedback. Cold Spring Harb. Perspect. Biol. 2009, 1, 1–16. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=20066087 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Chuang, P.T.; McMahon, A.P. Vertebrate hedgehog signalling modulated by induction of a hedgehog- binding protein. Nature 1999, 397, 617–621. [Google Scholar] [CrossRef]
- Ingham, P.W.; McMahon, A.P. Hedgehog signaling in animal development: Paradigms and principles. Genes Dev. 2001, 15, 3059–3087. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhoot, G.K.; Gustafsson, M.K.; Ai, X.; Sun, W.; Standiford, D.M.; Emerson, C.P., Jr. Regulation of Wnt signaling and embryo patterning by an extracellular sulfatase. Science 2001, 293, 1663–1666. [Google Scholar] [CrossRef]
- Izzi, L.; Levesque, M.; Morin, S.; Laniel, D.; Wilkes, B.C.; Mille, F.; Krauss, R.S.; McMahon, A.P.; Allen, B.L.; Charron, F. Boc and Gas1 each form distinct Shh receptor complexes with Ptch1 and are required for Shh-mediated cell proliferation. Dev. Cell 2011, 20, 788–801. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=21664577 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Allen, B.L.; Song, J.Y.; Izzi, L.; Althaus, I.W.; Kang, J.S.; Charron, F.; Krauss, R.S.; McMahon, A.P. Overlapping roles and collective requirement for the coreceptors GAS1, CDO, and BOC in SHH pathway function. Dev. Cell 2011, 20, 775–787. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=21664576 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Kahane, N.; Ribes, V.; Kicheva, A.; Briscoe, J.; Kalcheim, C. The transition from differentiation to growth during dermomyotome-derived myogenesis depends on temporally restricted hedgehog signaling. Development 2013, 140. [Google Scholar] [CrossRef] [Green Version]
- Briscoe, J.; Small, S. Morphogen rules: Design principles of gradient-mediated embryo patterning. Development 2015, 142, 3996–4009. Available online: http://www.ncbi.nlm.nih.gov/pubmed/26628090 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Stamataki, D.; Ulloa, F.; Tsoni, S.V.; Mynett, A.; Briscoe, J. A gradient of Gli activity mediates graded Sonic Hedgehog signaling in the neural tube. Genes Dev. 2005, 19, 626–641. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=15741323 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Cayuso, J.; Ulloa, F.; Cox, B.; Briscoe, J.; Marti, E. The Sonic hedgehog pathway independently controls the patterning, proliferation and survival of neuroepithelial cells by regulating Gli activity. Development 2006, 133, 517–528. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=16410413 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Charrier, J.B.; Lapointe, F.; Le Douarin, N.M.; Teillet, M.A. Anti-apoptotic role of Sonic hedgehog protein at the early stages of nervous system organogenesis. Development 2001, 128, 4011–4020. [Google Scholar] [CrossRef]
- Feng, X.; Adiarte, E.G.; Devoto, S.H. Hedgehog acts directly on the zebrafish dermomyotome to promote myogenic differentiation. Dev. Biol. 2006, 300, 736–746. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=17046741 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Hammond, C.L.; Hinits, Y.; Osborn, D.P.; Minchin, J.E.; Tettamanti, G.; Hughes, S.M. Signals and myogenic regulatory factors restrict pax3 and pax7 expression to dermomyotome-like tissue in zebrafish. Dev. Biol. 2007, 302, 504–521. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=17094960 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Maurya, A.K.; Tan, H.; Souren, M.; Wang, X.; Wittbrodt, J.; Ingham, P.W. Integration of Hedgehog and BMP signalling by the engrailed2a gene in the zebrafish myotome. Development 2011, 138, 755–765. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=21266411 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Wolff, C.; Roy, S.; Ingham, P.W. Multiple muscle cell identities induced by distinct levels and timing of hedgehog activity in the zebrafish embryo. Curr. Biol. 2003, 13, 1169–1181. [Google Scholar] [CrossRef] [Green Version]
- Teillet, M.A.; Watanabe, Y.; Jeffs, P.; Duprez, D.; Lapointe, F.; Le Douarin, N.M. Sonic hedgehog is required for survival of both myogenic and chondrogenic somitic lineages. Development 1998, 125, 2019–2030. [Google Scholar] [CrossRef]
- Borycki, A.G.; Brunk, B.; Tajbakhsh, S.; Buckingham, M.; Chiang, C.; Emerson, C.P., Jr. Sonic hedgehog controls epaxial muscle determination through Myf5 activation. Development 1999, 126, 4053–4063. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=10457014 (accessed on 2 July 2021). [CrossRef]
- Amthor, H.; Christ, B.; Patel, K. A molecular mechanism enabling continuous embryonic muscle growth—A balance between proliferation and differentiation. Development 1999, 126, 1041–1053. [Google Scholar] [CrossRef]
- Blagden, C.S.; Hughes, S.M. Extrinsic influences on limb muscle organisation. Cell Tissue Res. 1999, 296, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Du, S.J.; Devoto, S.H.; Westerfield, M.; Moon, R.T. Positive and negative regulation of muscle cell identity by members of the hedgehog and TGF-beta gene families. J. Cell Biol. 1997, 139, 145–156. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=9314535 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Kahane, N.; Cinnamon, Y.; Kalcheim, C. The origin and fate of pioneer myotomal cells in the avian embryo. Mech. Dev. 1998, 74, 59–73. [Google Scholar] [CrossRef]
- Kahane, N.; Ben-Yair, R.; Kalcheim, C. Medial pioneer fibers pattern the morphogenesis of early myoblasts derived from the lateral somite. Dev. Biol. 2007, 305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ben-Yair, R.; Kahane, N.; Kalcheim, C. LGN-dependent orientation of cell divisions in the dermomyotome controls lineage segregation into muscle and dermis. Development 2011, 138. [Google Scholar] [CrossRef] [Green Version]
- Buckingham, M.; Montarras, D. Skeletal muscle stem cells. Curr. Opin. Genet. Dev. 2008, 18, 330–336. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=18625314 (accessed on 2 July 2021). [CrossRef]
- Relaix, F.; Rocancourt, D.; Mansouri, A.; Buckingham, M. A Pax3/Pax7-dependent population of skeletal muscle progenitor cells. Nature 2005, 435, 948–953. [Google Scholar] [CrossRef] [Green Version]
- Chiang, C.; Litingtung, Y.; Lee, E.; Young, K.E.; Corden, J.L.; Westphal, H.; Beachy, P.A. Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature 1996, 383, 407–413. [Google Scholar] [CrossRef]
- Dockter, J.; Ordahl, C.P. Dorsoventral axis determination in the somite: A re-examination. Development 2000, 127, 2201–2206. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.-M.; Tessier-Lavigne, M. Patterning of mammalian somites by surface ectoderm and notochord: Evidence for sclerotome induction by a hedgehog homolog. Cell 1994, 79, 1175–1186. [Google Scholar] [CrossRef]
- Kahane, N.; Kalcheim, C. Neural tube development depends on notochord-derived sonic hedgehog released into the sclerotome. Development 2020, 147. [Google Scholar] [CrossRef]
- Stafford, D.A.; Brunet, L.J.; Khokha, M.K.; Economides, A.N.; Harland, R.M. Cooperative activity of noggin and gremlin 1 in axial skeleton development. Development 2011, 138, 1005–1014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, R.J.; Zhi, Q.X.; Schmidt, C.; Wilting, J.; Brand-Saberi, B.; Christ, B. Sclerotomal origin of the ribs. Development 2000, 527–532. [Google Scholar] [CrossRef]
- Kalcheim, C.; Ben-Yair, R. Cell rearrangements during development of the somite and its derivatives. Curr. Opin. Genet. Dev. 2005, 15. [Google Scholar] [CrossRef]
- Dubrulle, J.; Pourquie, O. Welcome to syndetome: A new somitic compartment. Dev. Cell 2003, 4, 611–612. Available online: http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?cmd=Retrieve&db=PubMed&dopt=Citation&list_uids=12737797 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Choi, K.S.; Harfe, B.D. Hedgehog signaling is required for formation of the notochord sheath and patterning of nuclei pulposi within the intervertebral discs. Proc. Natl. Acad. Sci. USA 2011, 108, 9484–9489. [Google Scholar] [CrossRef] [Green Version]
- Dietrich, S.; Gruss, P. undulated phenotypes suggest a role of Pax-1 for the development of vertebral and extravertebral structures. Dev.Biol. 1995, 167, 529–548. [Google Scholar] [CrossRef] [Green Version]
- Wood, W.M.; Otis, C.; Etemad, S.; Goldhamer, D.J. Development and patterning of rib primordia are dependent on associated musculature. Dev. Biol. 2020, 468, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Grifone, R.; Demignon, J.; Houbron, C.; Souil, E.; Niro, C.; Seller, M.J.; Hamard, G.; Maire, P. Six1 and Six4 homeoproteins are required for Pax3 and Mrf expression during myogenesis in the mouse embryo. Development 2005, 132, 2235–2249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elefteriou, F. Impact of the autonomic nervous system on the skeleton. Physiol. Rev. 2018, 98, 1083–1112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vyas, N.; Walvekar, A.; Tate, D.; Lakshmanan, V.; Bansal, D.; Lo Cicero, A.; Raposo, G.; Palakodeti, D.; Dhawan, J. Vertebrate Hedgehog is secreted on two types of extracellular vesicles with different signaling properties. Sci. Rep. 2014, 4, 7357. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25483805 (accessed on 2 July 2021). [CrossRef]
- Dierker, T.; Dreier, R.; Petersen, A.; Bordych, C.; Grobe, K. Heparan sulfate-modulated, metalloprotease-mediated sonic hedgehog release from producing cells. J. Biol. Chem. 2009, 284, 8013–8022. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19176481 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Chen, M.H.; Li, Y.J.; Kawakami, T.; Xu, S.M.; Chuang, P.T. Palmitoylation is required for the production of a soluble multimeric Hedgehog protein complex and long-range signaling in vertebrates. Genes Dev. 2004, 18, 641–659. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15075292 (accessed on 2 July 2021). [CrossRef] [PubMed] [Green Version]
- Parchure, A.; Vyas, N.; Mayor, S. Wnt and Hedgehog: Secretion of Lipid-Modified Morphogens. Trends Cell Biol. 2018, 28, 157–170. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29132729 (accessed on 2 July 2021). [CrossRef]
- Callejo, A.; Torroja, C.; Quijada, L.; Guerrero, I. Hedgehog lipid modifications are required for Hedgehog stabilization in the extracellular matrix. Development 2006, 133, 471–483. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16396909 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Etoc, F.; Metzger, J.; Ruzo, A.; Kirst, C.; Yoney, A.; Ozair, M.Z.; Brivanlou, A.H.; Siggia, E.D. A Balance between Secreted Inhibitors and Edge Sensing Controls Gastruloid Self-Organization. Dev. Cell 2016, 39, 302–315. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27746044 (accessed on 2 July 2021). [CrossRef] [Green Version]
- Phan-Everson, T.; Etoc, F.; Li, S.; Khodursky, S.; Yoney, A.; Brivanlou, A.H.; Siggia, E.D. Differential compartmentalization of BMP4/NOGGIN requires NOGGIN trans-epithelial transport. Dev. Cell 2021, 56, 1930.e5–1944.e5. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kahane, N.; Kalcheim, C. From Bipotent Neuromesodermal Progenitors to Neural-Mesodermal Interactions during Embryonic Development. Int. J. Mol. Sci. 2021, 22, 9141. https://doi.org/10.3390/ijms22179141
Kahane N, Kalcheim C. From Bipotent Neuromesodermal Progenitors to Neural-Mesodermal Interactions during Embryonic Development. International Journal of Molecular Sciences. 2021; 22(17):9141. https://doi.org/10.3390/ijms22179141
Chicago/Turabian StyleKahane, Nitza, and Chaya Kalcheim. 2021. "From Bipotent Neuromesodermal Progenitors to Neural-Mesodermal Interactions during Embryonic Development" International Journal of Molecular Sciences 22, no. 17: 9141. https://doi.org/10.3390/ijms22179141





