GPCR-Based Dopamine Sensors—A Detailed Guide to Inform Sensor Choice for In Vivo Imaging
Abstract
1. Introduction
1.1. Measuring Neuromodulator Release During Behavior
1.2. Heterogeneity of Brain Dopamine Systems
1.3. Measuring Dopamine Across Brain Regions
1.4. Sensor Choice Depends on Brain Region Dopamine Levels and Other Experimental Modalities
2. Currently Available Methods to Measure Dopamine Release
2.1. Analytical Chemistry: Microdialysis (In Vivo) and FSCV (Ex Vivo and In Vivo)
2.2. False Fluorescent Neurotransmitters (Ex Vivo)
2.3. Carbon Nanotubes (Ex Vivo)
2.4. GPCR FRET-Based Sensors: CNiFERs (Ex Vivo and In Vivo)
2.5. GPCR Signal Transduction Sensors: iTango2 and SPARK (Ex Vivo and In Vivo)
2.6. Genetically Encoded Calcium Sensors (Ex Vivo and In Vivo)
3. Catalogue of GPCR Biosensors for Dopamine
3.1. The GPCR Dopamine Sensor Toolbox (Ex Vivo and In Vivo)
3.2. Advantages of GPCR Dopamine Biosensors
3.3. Limitations of GPCR Dopamine Biosensors
4. Regional Heterogeneity of Dopamine Systems Across the Brain
4.1. Mapping Dopamine Average Content Using Biochemistry
4.2. Mapping Basal Dopamine Levels Using In Vivo Microdialysis
4.3. Insights into Phasic Dopamine Levels Using FSCV
5. Practical Considerations for Sensor Choice: One Sensor Does Not Fit All
5.1. Matching Brain Region Dopamine Levels to Sensor Affinity and Dynamic Range
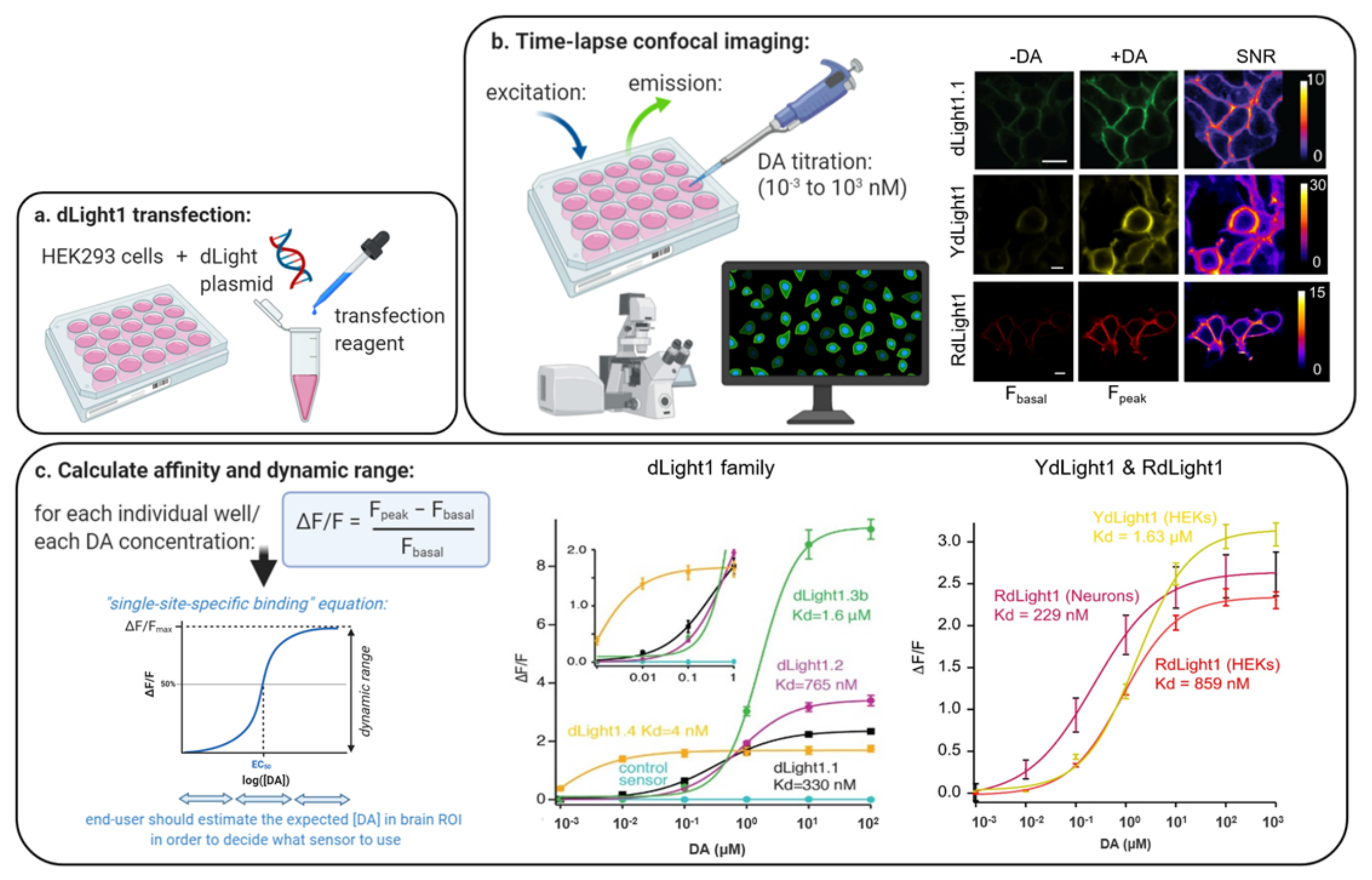
5.1.1. Affinity
5.1.2. Dynamic Range
5.1.3. Sensor Choice Based on Affinity and Dynamic Range
5.1.4. Published In vivo Validations of DA Sensors in Brain Regions with Dense vs. Sparse Innervation
5.2. Sensor Molecular Specificity Matters in Brain Regions with Dual Dopamine/Norepinephrine Innervation
5.3. Maximizing Sensor Kinetics Allows to Sparse out Individual Transients in Response to Closely Related Stimuli
5.4. Future Developments in Sensor Brightness/Subcellular Expression will Improve 1 and 2-Photon Imaging
5.5. Sensor Color Spectra Permits Dual Imaging and Multiplexing with Optogenetics and Photopharmacology
5.6. Molecular Scaffold as a Double-Edged Sword for Pharmacology and Drug Discovery
6. Piloting Sensor Use in the Laboratory
6.1. Sensor Validation at the Neuroanatomical Level
6.2. Sensor Validation In Vivo Using Optogenetic Stimulation
6.3. Sensor Validation In Vivo Using Pharmacology
6.4. Sensor Validation In Vivo Using Behavioral Stimulation
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAV | Adeno-associated virus |
| Amy | Amygdala |
| AP | Action potential |
| Au1 | Primary auditory cortex |
| BNST | Bed nucleus of the stria terminalis |
| CAG | Human cytomegalovirus promoter |
| cAMP | Cyclic adenosine monophosphate |
| CNiFERs | Cell-based neurotransmitter fluorescent engineered reporters |
| CNO | Clozapine-n-oxide |
| cpFP | Circularly-permuted fluorescent protein |
| cpGFP | Circularly-permuted green fluorescent protein |
| cpmApple | Circularly-permuted mApple (fluorescent protein) |
| CPu | Caudate-Putamen (= striatum) |
| CRB | Cerebellum |
| DA | Dopamine |
| dFF | Normalized fluorescent response (ΔF/F) |
| DR | Dorsal raphe nuclei |
| DRD1-5 | Genes encoding D1-D5 dopamine receptors |
| EC50 | Half maximal effective concentration |
| Emis | Emission wavelength |
| ENT | Enthorhinal cortex |
| Exc | Excitation wavelength |
| FFNs | False fluorescent neurotransmitters |
| FRET | Förster resonance energy transfer |
| FSCV | Fast-scan cyclic voltammetry |
| GECIs | Genetically-encoded calcium indicators |
| GFAP | Glial fibrillary acidic protein |
| GPCR | G protein-coupled receptor |
| GPe | Globus pallidus externus |
| GRAB | GPCR-activation based sensors |
| GTP | Guanosine triphosphate |
| HEK-293 | Human embryonic kidney cell line |
| HPC | Hippocampal formation |
| HPLC | High-performance liquid chromatography |
| hSyn | Human synapsin promoter |
| Hyp | Hypothalamus |
| Kd | Dissociation constant |
| LS | Lateral septum |
| M1 | Primary motor cortex |
| M2 | Secondary motor cortex |
| mPFC | Medial prefrontal cortex |
| MS | Medial septum |
| NAc | Nucleus accumbens |
| ND | Not determined |
| NE | Norepinephrine |
| OT | Olfactory tubercle |
| PBP | Periplasmic Binding Protein |
| PVT | Paraventricular thalamus |
| ROI | Region of Interest |
| SN | Substantia nigra |
| SNc | Subtantia nigra pars compacta |
| SNR | Signal-to-noise ratio |
| SWCNT | Single-walled carbon nanotubes |
| TeA | Temporal association cortex |
| TEV | Tobacco Etch Virus peptide sequence |
| TIRF | Total internal reflection microscopy |
| TRE | Tetracycline response element |
| tTA | Tetracycline-controlled transactivator |
| VP | Ventral pallidum |
| VMAT2 | Vesicular monoamine transporter 2 |
| VTA | Ventral Tegmental Area |
References
- Bargmann, C.I. Beyond the connectome: How neuromodulators shape neural circuits. BioEssays 2012, 34, 458–465. [Google Scholar] [CrossRef]
- Marder, E. Neuromodulation of neuronal circuits: Back to the future. Neuron 2012, 76, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Picciotto, M.R.; Higley, M.J.; Mineur, Y.S. Acetylcholine as a Neuromodulator: Cholinergic Signaling Shapes Nervous System Function and Behavior. Neuron 2012, 76, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Tritsch, N.X.; Sabatini, B.L. Dopaminergic modulation of synaptic transmission in cortex and striatum. Neuron 2012, 76, 33–50. [Google Scholar] [CrossRef] [PubMed]
- Miyawaki, A.; Llopis, J.; Heim, R.; McCaffery, J.M.; Adams, J.A.; Ikura, M.; Tsien, R.Y. Fluorescent indicators for Ca2+ based on green fluorescent proteins and calmodulin. Nature 1997, 388, 882–887. [Google Scholar] [CrossRef] [PubMed]
- Akerboom, J.; Chen, T.-W.; Wardill, T.J.; Tian, L.; Marvin, J.S.; Mutlu, S.; Calderon, N.C.; Esposti, F.; Borghuis, B.G.; Sun, X.R.; et al. Optimization of a GCaMP Calcium Indicator for Neural Activity Imaging. J. Neurosci. 2012, 32, 13819–13840. [Google Scholar] [CrossRef]
- Chen, T.-W.; Wardill, T.J.; Sun, Y.; Pulver, S.R.; Renninger, S.L.; Baohan, A.; Schreiter, E.R.; Kerr, R.A.; Orger, M.B.; Jayaraman, V.; et al. Ultrasensitive fluorescent proteins for imaging neuronal activity. Nature 2013, 499, 295–300. [Google Scholar] [CrossRef]
- Dana, H.; Sun, Y.; Mohar, B.; Hulse, B.K.; Kerlin, A.M.; Hasseman, J.P.; Tsegaye, G.; Tsang, A.; Wong, A.; Patel, R.; et al. High-performance calcium sensors for imaging activity in neuronal populations and microcompartments. Nat. Methods 2019, 16, 649–657. [Google Scholar] [CrossRef]
- Tian, L.; Hires, S.A.; Mao, T.; Huber, D.; Chiappe, M.E.; Chalasani, S.H.; Petreanu, L.; Akerboom, J.; McKinney, S.A.; Schreiter, E.R.; et al. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat. Methods 2009, 6, 875–881. [Google Scholar] [CrossRef]
- Colgan, L.A.; Cavolo, S.L.; Commons, K.G.; Levitan, E.S. Action Potential-Independent and Pharmacologically Unique Vesicular Serotonin Release from Dendrites. J. Neurosci. 2012, 32, 15737–15746. [Google Scholar] [CrossRef]
- Sulzer, D.; Cragg, S.J.; Rice, M.E. Striatal dopamine neurotransmission: Regulation of release and uptake. Basal Ganglia 2016, 6, 123–148. [Google Scholar] [CrossRef] [PubMed]
- Nadim, F.; Bucher, D. Neuromodulation of Neurons and Synapses. Curr. Opin. Neurobiol. 2014, 29, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Rusakov, D.A.; Savtchenko, L.P.; Zheng, K.; Henley, J.M. Shaping the synaptic signal: Molecular mobility inside and outside the cleft. Trends Neurosci. 2011, 34, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Gerfen, C.R.; Surmeier, D.J. Modulation of striatal projection systems by dopamine. Annu. Rev. Neurosci. 2011, 34, 441–466. [Google Scholar] [CrossRef]
- Berridge, K.C. The debate over dopamine’s role in reward: The case for incentive salience. Psychopharmacology 2007, 191, 391–431. [Google Scholar] [CrossRef]
- Salamone, J.D.; Correa, M.; Yang, J.-H.; Rotolo, R.; Presby, R. Dopamine, Effort-Based Choice, and Behavioral Economics: Basic and Translational Research. Front. Behav. Neurosci. 2018, 12, 52. [Google Scholar] [CrossRef]
- Bromberg-Martin, E.S.; Matsumoto, M.; Hikosaka, O. Dopamine in motivational control: Rewarding, aversive, and alerting. Neuron 2010, 68, 815–834. [Google Scholar] [CrossRef]
- Berke, J.D. What does dopamine mean? Nat. Neurosci. 2018, 21, 787–793. [Google Scholar] [CrossRef]
- Costa, R.M. Plastic corticostriatal circuits for action learning: What’s dopamine got to do with it? Ann. N. Y. Acad. Sci. 2007, 1104, 172–191. [Google Scholar] [CrossRef]
- Klaus, A.; Alves da Silva, J.; Costa, R.M. What, If, and When to Move: Basal Ganglia Circuits and Self-Paced Action Initiation. Annu. Rev. Neurosci. 2019, 42, 459–483. [Google Scholar] [CrossRef]
- Goldman-Rakic, P.S. The cortical dopamine system: Role in memory and cognition. Adv. Pharmacol. 1998, 42, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Ott, T.; Nieder, A. Dopamine and Cognitive Control in Prefrontal Cortex. Trends Cogn. Sci. (Regul. Ed.) 2019, 23, 213–234. [Google Scholar] [CrossRef]
- Vander Weele, C.M.; Siciliano, C.A.; Matthews, G.A.; Namburi, P.; Izadmehr, E.M.; Espinel, I.C.; Nieh, E.H.; Schut, E.H.S.; Padilla-Coreano, N.; Burgos-Robles, A.; et al. Dopamine enhances signal-to-noise ratio in cortical-brainstem encoding of aversive stimuli. Nature 2018, 563, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Matthews, G.A.; Nieh, E.H.; Vander Weele, C.M.; Halbert, S.A.; Pradhan, R.V.; Yosafat, A.S.; Glober, G.F.; Izadmehr, E.M.; Thomas, R.E.; Lacy, G.D.; et al. Dorsal Raphe Dopamine Neurons Represent the Experience of Social Isolation. Cell 2016, 164, 617–631. [Google Scholar] [CrossRef]
- Kenny, P.J.; Voren, G.; Johnson, P.M. Dopamine D2 receptors and striatopallidal transmission in addiction and obesity. Curr. Opin. Neurobiol. 2013, 23, 535–538. [Google Scholar] [CrossRef]
- Han, W.; Tellez, L.A.; Perkins, M.H.; Perez, I.O.; Qu, T.; Ferreira, J.; Ferreira, T.L.; Quinn, D.; Liu, Z.-W.; Gao, X.-B.; et al. A Neural Circuit for Gut-Induced Reward. Cell 2018, 175, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Berland, C.; Montalban, E.; Perrin, E.; Di Miceli, M.; Nakamura, Y.; Martinat, M.; Sullivan, M.; Davis, X.S.; Shenasa, M.A.; Martin, C.; et al. Circulating Triglycerides Gate Dopamine-Associated Behaviors through DRD2-Expressing Neurons. Cell Metab. 2020, 31, 773–790. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.B.; Alves da Silva, J.; Almeida, J.; Cui, G.; Gerfen, C.R.; Costa, R.M.; Oliveira-Maia, A.J. Postingestive Modulation of Food Seeking Depends on Vagus-Mediated Dopamine Neuron Activity. Neuron 2020. [Google Scholar] [CrossRef] [PubMed]
- Beeler, J.A.; Faust, R.P.; Turkson, S.; Ye, H.; Zhuang, X. Low Dopamine D2 Receptor Increases Vulnerability to Obesity Via Reduced Physical Activity, Not Increased Appetitive Motivation. Biol. Psychiatry 2016, 79, 887–897. [Google Scholar] [CrossRef] [PubMed]
- Friend, D.M.; Devarakonda, K.; O’Neal, T.J.; Skirzewski, M.; Papazoglou, I.; Kaplan, A.R.; Liow, J.-S.; Guo, J.; Rane, S.G.; Rubinstein, M.; et al. Basal Ganglia Dysfunction Contributes to Physical Inactivity in Obesity. Cell Metab. 2017, 25, 312–321. [Google Scholar] [CrossRef]
- Welch, A.C.; Zhang, J.; Lyu, J.; McMurray, M.S.; Javitch, J.A.; Kellendonk, C.; Dulawa, S.C. Dopamine D2 receptor overexpression in the nucleus accumbens core induces robust weight loss during scheduled fasting selectively in female mice. Mol. Psychiatry 2019. [Google Scholar] [CrossRef] [PubMed]
- Labouesse, M.A.; Sartori, A.M.; Weinmann, O.; Simpson, E.H.; Kellendonk, C.; Weber-Stadlbauer, U. Striatal dopamine 2 receptor upregulation during development predisposes to diet-induced obesity by reducing energy output in mice. Proc. Natl. Acad. Sci. USA 2018, 115, 10493–10498. [Google Scholar] [CrossRef]
- Folgueira, C.; Beiroa, D.; Porteiro, B.; Duquenne, M.; Puighermanal, E.; Fondevila, M.F.; Barja-Fernández, S.; Gallego, R.; Hernández-Bautista, R.; Castelao, C.; et al. Hypothalamic dopamine signalling regulates brown fat thermogenesis. Nat. Metab. 2019, 1, 811–829. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, P.; Dinan, T.G. Prolactin and dopamine: What is the connection? A review article. J. Psychopharmacol. (Oxf.) 2008, 22, 12–19. [Google Scholar] [CrossRef]
- Björklund, A.; Dunnett, S.B. Dopamine neuron systems in the brain: An update. Trends Neurosci. 2007, 30, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Dahlstroem, A.; Fuxe, K. Evidence for the existence of monoamine-containing neurons in the central nervous system. I. Demonstration of monoamines in the cell bodies of brain stem neurons. Acta Physiol. Scand. Suppl. 1964, 62 (Suppl. 232), 1–55. [Google Scholar]
- Lyons, D.J.; Hellysaz, A.; Broberger, C. Prolactin Regulates Tuberoinfundibular Dopamine Neuron Discharge Pattern: Novel Feedback Control Mechanisms in the Lactotrophic Axis. J. Neurosci. 2012, 32, 8074–8083. [Google Scholar] [CrossRef]
- Lin, R.; Liang, J.; Wang, R.; Yan, T.; Zhou, Y.; Liu, Y.; Feng, Q.; Sun, F.; Li, Y.; Li, A.; et al. The Raphe Dopamine System Controls the Expression of Incentive Memory. Neuron 2020, 106, 498–514. [Google Scholar] [CrossRef]
- Beas, B.S.; Wright, B.J.; Skirzewski, M.; Leng, Y.; Hyun, J.H.; Koita, O.; Ringelberg, N.; Kwon, H.-B.; Buonanno, A.; Penzo, M.A. The locus coeruleus drives disinhibition in the midline thalamus via a dopaminergic mechanism. Nat. Neurosci. 2018, 21, 963–973. [Google Scholar] [CrossRef]
- Kempadoo, K.A.; Mosharov, E.V.; Choi, S.J.; Sulzer, D.; Kandel, E.R. Dopamine release from the locus coeruleus to the dorsal hippocampus promotes spatial learning and memory. Proc. Natl. Acad. Sci. USA 2016, 113, 14835–14840. [Google Scholar] [CrossRef]
- Rice, M.E.; Patel, J.C. Somatodendritic dopamine release: Recent mechanistic insights. Philos Trans. R. Soc. Lond. B. Biol. Sci. 2015, 370. [Google Scholar] [CrossRef] [PubMed]
- Adams, B.W.; Moghaddam, B. Tactile stimulation activates dopamine release in the lateral septum. Brain Res. 2000, 858, 177–180. [Google Scholar] [CrossRef]
- Clark, M.; Bracci, E. Dichotomous Dopaminergic Control of Ventral Pallidum Neurons. Front. Cell Neurosci. 2018, 12. [Google Scholar] [CrossRef] [PubMed]
- Meszaros, J.; Cheung, T.; Erler, M.M.; Kang, U.J.; Sames, D.; Kellendonk, C.; Sulzer, D. Evoked transients of pH-sensitive fluorescent false neurotransmitter reveal dopamine hot spots in the globus pallidus. eLife 2018, 7, e42383. [Google Scholar] [CrossRef] [PubMed]
- Aransay, A.; RodrÃguez-LÃ3pez, C.; GarcÃa-Amado, M.; Clascá, F.; Prensa, L. Long-range projection neurons of the mouse ventral tegmental area: A single-cell axon tracing analysis. Front. Neuroanat. 2015, 9. [Google Scholar] [CrossRef]
- Lindvall, O.; Björklund, A. The glyoxylic acid fluorescence histochemical method: A detailed account of the methodology for the visualization of central catecholamine neurons. Histochemistry 1974, 39, 97–127. [Google Scholar] [CrossRef]
- Jones, S.R.; Mickelson, G.E.; Collins, L.B.; Kawagoe, K.T.; Wightman, R.M. Interference by pH and Ca2+ ions during measurements of catecholamine release in slices of rat amygdala with fast-scan cyclic voltammetry. J. Neurosci. Methods 1994, 52, 1–10. [Google Scholar] [CrossRef]
- Koch, S.; Perry, K.W.; Nelson, D.L.; Conway, R.G.; Threlkeld, P.G.; Bymaster, F.P. R-fluoxetine Increases Extracellular DA, NE, As Well As 5-HT in Rat Prefrontal Cortex and Hypothalamus: An in vivo Microdialysis and Receptor Binding Study. Neuropsychopharmacology 2002, 27, 949–959. [Google Scholar] [CrossRef]
- Muller, A.; Joseph, V.; Slesinger, P.A.; Kleinfeld, D. Cell-based reporters reveal in vivo dynamics of dopamine and norepinephrine release in murine cortex. Nat. Methods 2014, 11, 1245–1252. [Google Scholar] [CrossRef]
- Kim, M.W.; Wang, W.; Sanchez, M.I.; Coukos, R.; von Zastrow, M.; Ting, A.Y. Time-gated detection of protein-protein interactions with transcriptional readout. Elife 2017, 6. [Google Scholar] [CrossRef]
- Lee, D.; Creed, M.; Jung, K.; Stefanelli, T.; Wendler, D.J.; Oh, W.C.; Mignocchi, N.L.; Lüscher, C.; Kwon, H.-B. Temporally precise labeling and control of neuromodulatory circuits in the mammalian brain. Nat. Methods 2017, 14, 495–503. [Google Scholar] [CrossRef]
- Broussard, G.J.; Liang, Y.; Fridman, M.; Unger, E.K.; Meng, G.; Xiao, X.; Ji, N.; Petreanu, L.; Tian, L. In vivo measurement of afferent activity with axon-specific calcium imaging. Nat. Neurosci. 2018, 21, 1272–1280. [Google Scholar] [CrossRef] [PubMed]
- Mahn, M.; Prigge, M.; Ron, S.; Levy, R.; Yizhar, O. Biophysical constraints of optogenetic inhibition at presynaptic terminals. Nat. Neurosci. 2016, 19, 554–556. [Google Scholar] [CrossRef]
- Howe, M.W.; Dombeck, D.A. Rapid signalling in distinct dopaminergic axons during locomotion and reward. Nature 2016, 535, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Parker, N.F.; Cameron, C.M.; Taliaferro, J.P.; Lee, J.; Choi, J.Y.; Davidson, T.J.; Daw, N.D.; Witten, I.B. Reward and choice encoding in terminals of midbrain dopamine neurons depends on striatal target. Nat. Neurosci. 2016, 19, 845–854. [Google Scholar] [CrossRef] [PubMed]
- Gubernator, N.G.; Zhang, H.; Staal, R.G.W.; Mosharov, E.V.; Pereira, D.B.; Yue, M.; Balsanek, V.; Vadola, P.A.; Mukherjee, B.; Edwards, R.H.; et al. Fluorescent False Neurotransmitters Visualize Dopamine Release from Individual Presynaptic Terminals. Science 2009, 324, 1441–1444. [Google Scholar] [CrossRef] [PubMed]
- Beyene, A.G.; Delevich, K.; Del Bonis-O’Donnell, J.T.; Piekarski, D.J.; Lin, W.C.; Thomas, A.W.; Yang, S.J.; Kosillo, P.; Yang, D.; Prounis, G.S.; et al. Imaging striatal dopamine release using a nongenetically encoded near infrared fluorescent catecholamine nanosensor. Sci. Adv. 2019, 5, eaaw3108. [Google Scholar] [CrossRef]
- Patriarchi, T.; Cho, J.R.; Merten, K.; Howe, M.W.; Marley, A.; Xiong, W.-H.; Folk, R.W.; Broussard, G.J.; Liang, R.; Jang, M.J.; et al. Ultrafast neuronal imaging of dopamine dynamics with designed genetically encoded sensors. Science 2018, 360, eaat4422. [Google Scholar] [CrossRef]
- Patriarchi, T.; Mohebi, A.; Sun, J.; Marley, A.; Liang, R.; Dong, C.; Puhger, K.; Mizuno, G.O.; Davis, C.M.; Wiltgen, B.; et al. An expanded palette of dopamine sensors for multiplex imaging in vivo. Nat. Methods 2020, 1–9. [Google Scholar] [CrossRef]
- Sun, F.; Zeng, J.; Jing, M.; Zhou, J.; Feng, J.; Owen, S.F.; Luo, Y.; Li, F.; Wang, H.; Yamaguchi, T.; et al. A Genetically Encoded Fluorescent Sensor Enables Rapid and Specific Detection of Dopamine in Flies, Fish, and Mice. Cell 2018, 174, 481–496. [Google Scholar] [CrossRef]
- Sun, F.; Zhou, J.; Dai, B.; Qian, T.; Zeng, J.; Li, X.; Zhuo, Y.; Zhang, Y.; Tan, K.; Feng, J.; et al. New and improved GRAB fluorescent sensors for monitoring dopaminergic activity in vivo. bioRxiv 2020. [Google Scholar] [CrossRef]
- Siciliano, C.A.; Tye, K.M. Leveraging calcium imaging to illuminate circuit dysfunction in addiction. Alcohol 2019, 74, 47–63. [Google Scholar] [CrossRef]
- Gunaydin, L.A.; Grosenick, L.; Finkelstein, J.C.; Kauvar, I.V.; Fenno, L.E.; Adhikari, A.; Lammel, S.; Mirzabekov, J.J.; Airan, R.D.; Zalocusky, K.A.; et al. Natural Neural Projection Dynamics Underlying Social Behavior. Cell 2014, 157, 1535–1551. [Google Scholar] [CrossRef]
- Kim, C.K.; Yang, S.J.; Pichamoorthy, N.; Young, N.P.; Kauvar, I.; Jennings, J.H.; Lerner, T.N.; Berndt, A.; Lee, S.Y.; Ramakrishnan, C.; et al. Simultaneous fast measurement of circuit dynamics at multiple sites across the mammalian brain. Nat. Methods 2016, 13, 325–328. [Google Scholar] [CrossRef]
- Sych, Y.; Chernysheva, M.; Sumanovski, L.T.; Helmchen, F. High-density multi-fiber photometry for studying large-scale brain circuit dynamics. Nat. Methods 2019, 16, 553–560. [Google Scholar] [CrossRef] [PubMed]
- Helmchen, F.; Fee, M.S.; Tank, D.W.; Denk, W. A Miniature Head-Mounted Two-Photon Microscope: High-Resolution Brain Imaging in Freely Moving Animals. Neuron 2001, 31, 903–912. [Google Scholar] [CrossRef]
- Flusberg, B.A.; Nimmerjahn, A.; Cocker, E.D.; Mukamel, E.A.; Barretto, R.P.J.; Ko, T.H.; Burns, L.D.; Jung, J.C.; Schnitzer, M.J. High-speed, miniaturized fluorescence microscopy in freely moving mice. Nat. Methods 2008, 5, 935–938. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Zhang, Y.; Lecoq, J.; Jung, J.C.; Li, J.; Zeng, H.; Niell, C.M.; Schnitzer, M.J. Long-Term Optical Access to an Estimated One Million Neurons in the Live Mouse Cortex. Cell Rep. 2016, 17, 3385–3394. [Google Scholar] [CrossRef]
- Sofroniew, N.J.; Flickinger, D.; King, J.; Svoboda, K. A large field of view two-photon mesoscope with subcellular resolution for in vivo imaging. Elife 2016, 5. [Google Scholar] [CrossRef] [PubMed]
- Beyene, A.G.; Yang, S.J.; Landry, M.P. Review Article: Tools and trends for probing brain neurochemistry. J. Vac. Sci. Technol. A 2019, 37, 040802. [Google Scholar] [CrossRef] [PubMed]
- Oe, Y.; Wang, X.; Patriarchi, T.; Konno, A.; Ozawa, K.; Yahagi, K.; Hirai, H.; Tian, L.; McHugh, T.J.; Hirase, H. Distinct temporal integration of noradrenaline signaling by astrocytic second messengers during vigilance. Nat. Commun. 2020, 11, 471. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Zhang, C.; Lischinsky, J.E.; Jing, M.; Zhou, J.; Wang, H.; Zhang, Y.; Dong, A.; Wu, Z.; Wu, H.; et al. A Genetically Encoded Fluorescent Sensor for Rapid and Specific In Vivo Detection of Norepinephrine. Neuron 2019, 102, 745–761. [Google Scholar] [CrossRef] [PubMed]
- Jing, M.; Zhang, P.; Wang, G.; Feng, J.; Mesik, L.; Zeng, J.; Jiang, H.; Wang, S.; Looby, J.C.; Guagliardo, N.A.; et al. A genetically encoded fluorescent acetylcholine indicator for in vitro and in vivo studies. Nat. Biotechnol. 2018, 36, 726–737. [Google Scholar] [CrossRef] [PubMed]
- Jing, M.; Li, Y.; Zeng, J.; Huang, P.; Skirzewski, M.; Kljakic, O.; Peng, W.; Qian, T.; Tan, K.; Zou, J.; et al. An optimized acetylcholine sensor for monitoring in vivo cholinergic activity. Nat. Methods 2020. [Google Scholar] [CrossRef]
- Wan, J.; Peng, W.; Li, X.; Qian, T.; Song, K.; Zeng, J.; Deng, F.; Hao, S.; Feng, J.; Zhang, P.; et al. A genetically encoded GRAB sensor for measuring serotonin dynamics in vivo. bioRxiv 2020. [Google Scholar] [CrossRef]
- Peng, W.; Wu, Z.; Song, K.; Zhang, S.; Li, Y.; Xu, M. Regulation of sleep homeostasis mediator adenosine by basal forebrain glutamatergic neurons. Science 2020, 369. [Google Scholar] [CrossRef]
- Dana, H.; Mohar, B.; Sun, Y.; Narayan, S.; Gordus, A.; Hasseman, J.P.; Tsegaye, G.; Holt, G.T.; Hu, A.; Walpita, D.; et al. Sensitive red protein calcium indicators for imaging neural activity. eLife 2016, 5, e12727. [Google Scholar] [CrossRef]
- Justice, J.B. The Evolution of in Vivo Voltammetry and Microdialysis. In Catecholamine Research: From Molecular Insights to Clinical Medicine; Nagatsu, T., Nabeshima, T., McCarty, R., Goldstein, D.S., Eds.; Advances in Behavioral Biology; Springer: Boston, MA, USA, 2002; pp. 301–304. ISBN 978-1-4757-3538-3. [Google Scholar]
- Budygin, E.A.; Kilpatrick, M.R.; Gainetdinov, R.R.; Wightman, R.M. Correlation between behavior and extracellular dopamine levels in rat striatum: Comparison of microdialysis and fast-scan cyclic voltammetry. Neurosci. Lett. 2000, 281, 9–12. [Google Scholar] [CrossRef]
- Darvesh, A.S.; Carroll, R.T.; Geldenhuys, W.J.; Gudelsky, G.A.; Klein, J.; Meshul, C.K.; Van der Schyf, C.J. In vivo brain microdialysis: Advances in neuropsychopharmacology and drug discovery. Expert Opin. Drug Discov. 2011, 6, 109–127. [Google Scholar] [CrossRef]
- Fuchs, H.; Hauber, W. Dopaminergic innervation of the rat globus pallidus characterized by microdialysis and immunohistochemistry. Exp. Brain Res. 2004, 154, 66–75. [Google Scholar] [CrossRef]
- Wightman, R.M. Detection technologies. Probing cellular chemistry in biological systems with microelectrodes. Science 2006, 311, 1570–1574. [Google Scholar] [CrossRef] [PubMed]
- Zachek, M.K.; Park, J.; Takmakov, P.; Wightman, R.M.; McCarty, G.S. Microfabricated FSCV-compatible microelectrode array for real-time monitoring of heterogeneous dopamine release. Analyst 2010, 135, 1556–1563. [Google Scholar] [CrossRef]
- Schwerdt, H.N.; Kim, M.J.; Amemori, S.; Homma, D.; Yoshida, T.; Shimazu, H.; Yerramreddy, H.; Karasan, E.; Langer, R.; Graybiel, A.M.; et al. Subcellular probes for neurochemical recording from multiple brain sites. Lab Chip 2017, 17, 1104–1115. [Google Scholar] [CrossRef] [PubMed]
- Briand, L.A.; Gritton, H.; Howe, W.M.; Young, D.A.; Sarter, M. Modulators in concert for cognition: Modulator interactions in the prefrontal cortex. Prog. Neurobiol. 2007, 83, 69–91. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.B.; Schmitz, Y.; Mészáros, J.; Merchant, P.; Hu, G.; Li, S.; Henke, A.; Lizardi-Ortiz, J.E.; Karpowicz, R.J.; Morgenstern, T.J.; et al. Fluorescent false neurotransmitter reveals functionally silent dopamine vesicle clusters in the striatum. Nat. Neurosci. 2016, 19, 578–586. [Google Scholar] [CrossRef]
- Dunn, M.; Henke, A.; Clark, S.; Kovalyova, Y.; Kempadoo, K.A.; Karpowicz, R.J.; Kandel, E.R.; Sulzer, D.; Sames, D. Designing a norepinephrine optical tracer for imaging individual noradrenergic synapses and their activity in vivo. Nat. Comm. 2018, 9, 2838. [Google Scholar] [CrossRef] [PubMed]
- Kruss, S.; Landry, M.P.; Vander Ende, E.; Lima, B.M.A.; Reuel, N.F.; Zhang, J.; Nelson, J.; Mu, B.; Hilmer, A.; Strano, M. Neurotransmitter Detection Using Corona Phase Molecular Recognition on Fluorescent Single-Walled Carbon Nanotube Sensors. J. Am. Chem. Soc. 2014, 136, 713–724. [Google Scholar] [CrossRef]
- Kruss, S.; Salem, D.P.; Vuković, L.; Lima, B.; Ende, E.V.; Boyden, E.S.; Strano, M.S. High-resolution imaging of cellular dopamine efflux using a fluorescent nanosensor array. Proc. Natl. Acad. Sci. USA 2017, 114, 1789–1794. [Google Scholar] [CrossRef]
- Dinarvand, M.; Elizarova, S.; Daniel, J.; Kruss, S. Imaging of Monoamine Neurotransmitters with Fluorescent Nanoscale Sensors. ChemPlusChem 2020, 85, 1465–1480. [Google Scholar] [CrossRef]
- Nguyen, Q.-T.; Schroeder, L.F.; Mank, M.; Muller, A.; Taylor, P.; Griesbeck, O.; Kleinfeld, D. An in vivo biosensor for neurotransmitter release and in situ receptor activity. Nat. Neurosci. 2010, 13, 127–132. [Google Scholar] [CrossRef]
- Barnea, G.; Strapps, W.; Herrada, G.; Berman, Y.; Ong, J.; Kloss, B.; Axel, R.; Lee, K.J. The genetic design of signaling cascades to record receptor activation. Proc. Natl. Acad. Sci. USA 2008, 105, 64–69. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, H.K.; Ben-Tabou de-Leon, S.; Wong, A.M.; Jagadish, S.; Ishimoto, H.; Barnea, G.; Kitamoto, T.; Axel, R.; Anderson, D.J. Visualizing Neuromodulation In Vivo: TANGO-Mapping of Dopamine Signaling Reveals Appetite Control of Sugar Sensing. Cell 2012, 148, 583–595. [Google Scholar] [CrossRef] [PubMed]
- Howe, M.; Ridouh, I.; Allegra Mascaro, A.L.; Larios, A.; Azcorra, M.; Dombeck, D.A. Coordination of rapid cholinergic and dopaminergic signaling in striatum during spontaneous movement. eLife 2019, 8, e44903. [Google Scholar] [CrossRef]
- Liu, C.; Kaeser, P.S. Mechanisms and regulation of dopamine release. Curr. Opin. Neurobiol. 2019, 57, 46–53. [Google Scholar] [CrossRef]
- Ford, C.P. The role of D2-autoreceptors in regulating dopamine neuron activity and transmission. Neuroscience 2014, 282, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Sturgill, J.; Hegedus, P.; Li, S.; Chevy, Q.; Siebels, A.; Jing, M.; Li, Y.; Hangya, B.; Kepecs, A. Basal forebrain-derived acetylcholine encodes valence-free reinforcement prediction error. bioRxiv 2020. [Google Scholar] [CrossRef]
- Wu, M.; Minkowicz, S.; Dumrongprechachan, V.; Hamilton, P.; Xiao, L.; Kozorovitskiy, Y. Ketamine restores escape behavior by re-engaging dopamine systems to drive cortical spinogenesis. bioRxiv 2020. [Google Scholar] [CrossRef]
- Patriarchi, T.; Cho, J.R.; Merten, K.; Marley, A.; Broussard, G.J.; Liang, R.; Williams, J.; Nimmerjahn, A.; von Zastrow, M.; Gradinaru, V.; et al. Imaging neuromodulators with high spatiotemporal resolution using genetically encoded indicators. Nat. Protoc. 2019, 14, 3471–3505. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, C.; Goto, Y.; Tomizawa, Y.; Fukata, Y.; Fukata, M.; Harpsøe, K.; Gloriam, D.E.; Aoki, K.; Takeuchi, T. A genetically encoded red fluorescence dopamine biosensor enables dual imaging of dopamine and norepinephrine. bioRxiv 2020. [Google Scholar] [CrossRef]
- Mohebi, A.; Pettibone, J.R.; Hamid, A.A.; Wong, J.-M.T.; Vinson, L.T.; Patriarchi, T.; Tian, L.; Kennedy, R.T.; Berke, J.D. Dissociable dopamine dynamics for learning and motivation. Nature 2019, 570, 65–70. [Google Scholar] [CrossRef]
- Yuan, L.; Dou, Y.-N.; Sun, Y.-G. Topography of Reward and Aversion Encoding in the Mesolimbic Dopaminergic System. J. Neurosci. 2019, 39, 6472–6481. [Google Scholar] [CrossRef]
- Lutas, A.; Kucukdereli, H.; Alturkistani, O.; Carty, C.; Sugden, A.U.; Fernando, K.; Diaz, V.; Flores-Maldonado, V.; Andermann, M.L. State-specific gating of salient cues by midbrain dopaminergic input to basal amygdala. Nat. Neurosci. 2019, 22, 1820–1833. [Google Scholar] [CrossRef] [PubMed]
- Iino, Y.; Sawada, T.; Yamaguchi, K.; Tajiri, M.; Ishii, S.; Kasai, H.; Yagishita, S. Dopamine D2 receptors in discrimination learning and spine enlargement. Nature 2020, 579, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Malik, A.N.; Mikhael, J.G.; Bech, P.; Tsutsui-Kimura, I.; Sun, F.; Zhang, Y.; Li, Y.; Watabe-Uchida, M.; Gershman, S.J.; et al. A unified framework for dopamine signals across timescales. bioRxiv 2019, 803437. [Google Scholar] [CrossRef]
- Lee, S.J.; Lodder, B.; Chen, Y.; Patriarchi, T.; Tian, L.; Sabatini, B.L. Cell-type specific asynchronous modulation of PKA by dopamine during reward based learning. bioRxiv 2019, 839035. [Google Scholar] [CrossRef]
- Gallo, E.F.; Greenwald, J.; Teboul, E.; Martyniuk, K.M.; Li, Y.; Javitch, J.A.; Balsam, P.D.; Kellendonk, C. Dopamine D2 receptors modulate the cholinergic pause and inhibitory learning. bioRxiv 2020. [Google Scholar] [CrossRef]
- Augustine, V.; Ebisu, H.; Zhao, Y.; Lee, S.; Ho, B.; Mizuno, G.O.; Tian, L.; Oka, Y. Temporally and Spatially Distinct Thirst Satiation Signals. Neuron 2019, 103, 242–249. [Google Scholar] [CrossRef]
- Alhadeff, A.L.; Goldstein, N.; Park, O.; Klima, M.L.; Vargas, A.; Betley, J.N. Natural and Drug Rewards Engage Distinct Pathways that Converge on Coordinated Hypothalamic and Reward Circuits. Neuron 2019, 103, 891–908. [Google Scholar] [CrossRef]
- Corre, J.; van Zessen, R.; Loureiro, M.; Patriarchi, T.; Tian, L.; Pascoli, V.; Lüscher, C. Dopamine neurons projecting to medial shell of the nucleus accumbens drive heroin reinforcement. eLife 2018, 7, e39945. [Google Scholar] [CrossRef]
- Liu, Y.; Jean-Richard-dit-Bressel, P.; Yau, J.O.-Y.; Willing, A.; Prasad, A.A.; Power, J.M.; Killcross, S.; Clifford, C.W.G.; McNally, G.P. The mesolimbic dopamine signatures of relapse to alcohol-seeking. bioRxiv 2020. [Google Scholar] [CrossRef]
- de Jong, J.W.; Afjei, S.A.; Pollak Dorocic, I.; Peck, J.R.; Liu, C.; Kim, C.K.; Tian, L.; Deisseroth, K.; Lammel, S. A Neural Circuit Mechanism for Encoding Aversive Stimuli in the Mesolimbic Dopamine System. Neuron 2019, 101, 133–151. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Wang, J.; Yang, Y.-F.; Shen, Y.; Qu, W.-M.; Huang, Z.-L. Dorsal Striatum Dopamine Levels Fluctuate Across the Sleep-Wake Cycle and Respond to Salient Stimuli in Mice. Front Neurosci. 2019, 13, 242. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Tian, M.; He, F.; Li, J.; Xie, H.; Liu, W.; Zhang, Y.; Zhang, R.; Yi, M.; Che, F.; et al. Mutations in ASH1L confer susceptibility to Tourette syndrome. Mol. Psychiatry 2020, 25, 476–490. [Google Scholar] [CrossRef] [PubMed]
- Robinson, J.E.; Coughlin, G.M.; Hori, A.M.; Cho, J.R.; Mackey, E.D.; Turan, Z.; Patriarchi, T.; Tian, L.; Gradinaru, V. Optical dopamine monitoring with dLight1 reveals mesolimbic phenotypes in a mouse model of neurofibromatosis type 1. eLife 2019, 8, e48983. [Google Scholar] [CrossRef]
- Graves, S.M.; Xie, Z.; Stout, K.A.; Zampese, E.; Burbulla, L.F.; Shih, J.C.; Kondapalli, J.; Patriarchi, T.; Tian, L.; Brichta, L.; et al. Dopamine metabolism by a monoamine oxidase mitochondrial shuttle activates the electron transport chain. Nat. Neurosci. 2020, 23, 15–20. [Google Scholar] [CrossRef]
- Ziegler, N.; Bätz, J.; Zabel, U.; Lohse, M.J.; Hoffmann, C. FRET-based sensors for the human M1-, M3-, and M5-acetylcholine receptors. Bioorganic Med. Chem. 2011, 19, 1048–1054. [Google Scholar] [CrossRef]
- Ravotto, L.; Duffet, L.; Zhou, X.; Weber, B.; Patriarchi, T. A Bright and Colorful Future for G-Protein Coupled Receptor Sensors. Front. Cell. Neurosci. 2020, 14, 67. [Google Scholar] [CrossRef]
- Andreoni, A.; Davis, C.M.O.; Tian, L. Measuring brain chemistry using genetically encoded fluorescent sensors. Curr. Opin. Biomed. Eng. 2019, 12, 59–67. [Google Scholar] [CrossRef]
- Marvin, J.S.; Scholl, B.; Wilson, D.E.; Podgorski, K.; Kazemipour, A.; Müller, J.A.; Schoch, S.; Quiroz, F.J.U.; Rebola, N.; Bao, H.; et al. Stability, affinity, and chromatic variants of the glutamate sensor iGluSnFR. Nat. Methods 2018, 15, 936–939. [Google Scholar] [CrossRef]
- Marvin, J.S.; Shimoda, Y.; Magloire, V.; Leite, M.; Kawashima, T.; Jensen, T.P.; Kolb, I.; Knott, E.L.; Novak, O.; Podgorski, K.; et al. A genetically encoded fluorescent sensor for in vivo imaging of GABA. Nat. Methods 2019, 16, 763–770. [Google Scholar] [CrossRef]
- Borden, P.M.; Zhang, P.; Shivange, A.V.; Marvin, J.S.; Cichon, J.; Dan, C.; Podgorski, K.; Figueiredo, A.; Novak, O.; Tanimoto, M.; et al. A fast genetically encoded fluorescent sensor for faithful in vivo acetylcholine detection in mice, fish, worms and flies. bioRxiv 2020. [Google Scholar] [CrossRef]
- Mizuno, G.O.; Unger, E.K.; Tian, L. Real time monitoring of neuromodulators in behaving animals using genetically encoded indicators. In Compendium of In Vivo Monitoring in Real-Time Molecular Neuroscience; World Scientific: Singapore, 2019; pp. 1–18. ISBN 9789811206221. [Google Scholar]
- Harada, K.; Ito, M.; Wang, X.; Tanaka, M.; Wongso, D.; Konno, A.; Hirai, H.; Hirase, H.; Tsuboi, T.; Kitaguchi, T. Red fluorescent protein-based cAMP indicator applicable to optogenetics and in vivo imaging. Sci. Rep. 2017, 7, 7351. [Google Scholar] [CrossRef] [PubMed]
- Kroeze, W.K.; Sassano, M.F.; Huang, X.-P.; Lansu, K.; McCorvy, J.D.; Giguère, P.M.; Sciaky, N.; Roth, B.L. PRESTO-Tango as an open-source resource for interrogation of the druggable human GPCRome. Nat. Struct. Mol. Biol. 2015, 22, 362–369. [Google Scholar] [CrossRef]
- Steinmetz, N.A.; Buetfering, C.; Lecoq, J.; Lee, C.R.; Peters, A.J.; Jacobs, E.A.K.; Coen, P.; Ollerenshaw, D.R.; Valley, M.T.; de Vries, S.E.J.; et al. Aberrant Cortical Activity in Multiple GCaMP6-Expressing Transgenic Mouse Lines. eNeuro 2017, 4. [Google Scholar] [CrossRef]
- Mosharov, E.V.; Borgkvist, A.; Sulzer, D. Presynaptic effects of L-DOPA and their possible role in dyskinesia. Mov. Disord. 2015, 30, 45–53. [Google Scholar] [CrossRef]
- Cragg, S.J.; Rice, M.E. DAncing past the DAT at a DA synapse. Trends Neurosci. 2004, 27, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Schultz, W. Multiple Dopamine Functions at Different Time Courses. Annu. Rev. Neurosci. 2007, 30, 259–288. [Google Scholar] [CrossRef]
- Björklund, A.; Hökfelt, T. Handbook of Chemical Neuroanatomy: Classical Transmitters in the CNS; Elsevier: Amsterdam, The Netherlands, 1984; Volume 2. [Google Scholar]
- Matsuda, W.; Furuta, T.; Nakamura, K.C.; Hioki, H.; Fujiyama, F.; Arai, R.; Kaneko, T. Single Nigrostriatal Dopaminergic Neurons Form Widely Spread and Highly Dense Axonal Arborizations in the Neostriatum. J. Neurosci. 2009, 29, 444–453. [Google Scholar] [CrossRef]
- Debeir, T.; Ginestet, L.; François, C.; Laurens, S.; Martel, J.-C.; Chopin, P.; Marien, M.; Colpaert, F.; Raisman-Vozari, R. Effect of intrastriatal 6-OHDA lesion on dopaminergic innervation of the rat cortex and globus pallidus. Exp. Neurol. 2005, 193, 444–454. [Google Scholar] [CrossRef]
- Moreno, M.; Decara, J.; Pavon, F.J.; Stouffer, D.G.; Edwards, S.; Serrano, A.; Suárez, J.; Parsons, L.H.; de Fonseca, F.R. Cannabinoid dependence induces sustained changes in GABA release in the globus pallidus without affecting dopamine release in the dorsal striatum: A dual microdialysis probe study. Addict. Biol. 2018, 23, 1251–1261. [Google Scholar] [CrossRef]
- Gomez-Sintes, R.; Bortolozzi, A.; Artigas, F.; Lucas, J.J. Reduced striatal dopamine DA D2 receptor function in dominant-negative GSK-3 transgenic mice. Eur. Neuropsychopharmacol. 2014, 24, 1524–1533. [Google Scholar] [CrossRef] [PubMed]
- Cederfjäll, E.; Sahin, G.; Kirik, D.; Björklund, T. Design of a Single AAV Vector for Coexpression of TH and GCH1 to Establish Continuous DOPA Synthesis in a Rat Model of Parkinson’s Disease. Mol. Ther. 2012, 20, 1315–1326. [Google Scholar] [CrossRef] [PubMed]
- Pazo, J.H.; Höcht, C.; Barceló, A.C.; Fillipini, B.; Lomastro, M.J. Effect of electrical and chemical stimulation of the subthalamic nucleus on the release of striatal dopamine. Synapse 2010, 64, 905–915. [Google Scholar] [CrossRef]
- Li, X.; Li, J.; Gardner, E.L.; Xi, Z.-X. Activation of mGluR7s inhibits cocaine-induced reinstatement of drug-seeking behavior by a nucleus accumbens glutamate-mGluR2/3 mechanism in rats. J. Neurochem. 2010, 114, 1368–1380. [Google Scholar] [CrossRef]
- Korkmaz, O.T.; Tunçel, N.; Tunçel, M.; Öncü, E.M.; Şahintürk, V.; Çelik, M. Vasoactive Intestinal Peptide (VIP) Treatment of Parkinsonian Rats Increases Thalamic Gamma-Aminobutyric Acid (GABA) Levels and Alters the Release of Nerve Growth Factor (NGF) by Mast Cells. J. Mol. Neurosci. 2010, 41, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Andersson, D.R.; Björnsson, E.; Bergquist, F.; Nissbrandt, H. Motor activity-induced dopamine release in the substantia nigra is regulated by muscarinic receptors. Exp. Neurol. 2010, 221, 251–259. [Google Scholar] [CrossRef]
- Andersson, D.R.; Nissbrandt, H.; Bergquist, F. Partial depletion of dopamine in substantia nigra impairs motor performance without altering striatal dopamine neurotransmission. Eur. J. Neurosci. 2006, 24, 617–624. [Google Scholar] [CrossRef]
- Yamamura, S.; Ohoyama, K.; Nagase, H.; Okada, M. Zonisamide enhances delta receptor-associated neurotransmitter release in striato-pallidal pathway. Neuropharmacology 2009, 57, 322–331. [Google Scholar] [CrossRef]
- Shou, M.; Ferrario, C.R.; Schultz, K.N.; Robinson, T.E.; Kennedy, R.T. Monitoring Dopamine in Vivo by Microdialysis Sampling and On-Line CE-Laser-Induced Fluorescence. Anal. Chem. 2006, 78, 6717–6725. [Google Scholar] [CrossRef]
- Schwarz, A.J.; Zocchi, A.; Reese, T.; Gozzi, A.; Garzotti, M.; Varnier, G.; Curcuruto, O.; Sartori, I.; Girlanda, E.; Biscaro, B.; et al. Concurrent pharmacological MRI and in situ microdialysis of cocaine reveal a complex relationship between the central hemodynamic response and local dopamine concentration. NeuroImage 2004, 23, 296–304. [Google Scholar] [CrossRef]
- Meissner, W.; Harnack, D.; Hoessle, N.; Bezard, E.; Winter, C.; Morgenstern, R.; Kupsch, A. High frequency stimulation of the entopeduncular nucleus has no effect on striatal dopaminergic transmission. Neurochem. Int. 2004, 44, 281–286. [Google Scholar] [CrossRef]
- Lee, C.R.; Abercrombie, E.D.; Tepper, J.M. Pallidal control of substantia nigra dopaminergic neuron firing pattern and its relation to extracellular neostriatal dopamine levels. Neuroscience 2004, 129, 481–489. [Google Scholar] [CrossRef] [PubMed]
- Heeringa, M.J.; Abercrombie, E.D. Biochemistry of Somatodendritic Dopamine Release in Substantia Nigra: An In Vivo Comparison with Striatal Dopamine Release. J. Neurochem. 1995, 65, 192–200. [Google Scholar] [CrossRef]
- Meissner, W.; Paul, G.; Reum, T.; Reese, R.; Sohr, R.; Morgenstern, R.; Kupsch, A. The influence of pallidal deep brain stimulation on striatal dopaminergic metabolism in the rat. Neurosci. Lett. 2000, 296, 149–152. [Google Scholar] [CrossRef]
- Grimm, J.W.; See, R.E. Chronic haloperidol-induced alterations in pallidal GABA and striatal D1-mediated dopamine turnover as measured by dual probe microdialysis in rats. Neuroscience 2000, 100, 507–514. [Google Scholar] [CrossRef]
- Balcioglu, A.; Wurtman, R.J. Sibutramine, a serotonin uptake inhibitor, increases dopamine concentrations in rat striatal and hypothalamic extracellular fluid. Neuropharmacology 2000, 39, 2352–2359. [Google Scholar] [CrossRef]
- Opacka-Juffry, J.; Ashworth, S.; Ahier, R.G.; Hume, S.P. Modulatory effects of L-DOPA on D2 dopamine receptors in rat striatum, measured using in vivo microdialysis and PET. J. Neural. Transm. 1998, 105, 349. [Google Scholar] [CrossRef] [PubMed]
- Semba, J.; Wakuta, M.S. Regional Differences in the Effects of Glutamate Uptake Inhibitor l-Trans-Pyrrolidine-2,4-Dicarboxylic Acid on Extracellular Amino Acids and Dopamine in Rat Brain: An In Vivo Microdialysis Study. Gen. Pharmacol. Vasc. Syst. 1998, 31, 399–404. [Google Scholar] [CrossRef]
- Grimm, J.W.; See, R.E. Unique activation of extracellular striato-pallidal neurotransmitters in rats following acute risperidone. Brain Res. 1998, 801, 182–189. [Google Scholar] [CrossRef]
- Ferraro, L.; O’Connor, W.T.; Antonelli, T.; Fuxe, K.; Tanganelli, S. Differential Effects of lntrastriatal Neurotensin (1–13) and Neurotensin (8–13) on Striatal Dopamine and Pallidal GABA Release. A Dual-probe Microdialysis Study in the Awake Rat. Eur. J. Neurosci. 1997, 9, 1838–1846. [Google Scholar] [CrossRef]
- Rimondini, R.; O’Connor, W.T.; Sillard, R.; Mutt, V.; Ungerstedt, U.; Fuxe, K. The secretory trypsin inhibitor like-peptide, PEC-60 increases dopamine D2 receptor agonist induced inhibition of GABA release in the dorsolateral neostriatum of the awake freely moving rat. An in vivo microdialysis study. Regul. Pept. 1996, 61, 111–117. [Google Scholar] [CrossRef]
- Rimondini, R.; O’Connor, W.T.; Ferré, S.; Sillard, R.; Agerberth, B.; Mutt, V.; Ungerstedt, U.; Fuxe, K. PEC-60 increases dopamine but not GABA release in the dorsolateral neostriatum of the halothane anaesthetized rat. An in vivo microdialysis study. Neurosci. Lett. 1994, 177, 53–57. [Google Scholar] [CrossRef]
- Ferré, S. Antagonistic interaction between adenosine A2A receptors and dopamine D2 receptors in the ventral striopallidal system. Implications for the treatment of schizophrenia. Neuroscience 1994, 63, 765–773. [Google Scholar] [CrossRef]
- Drew, K.L.; O’Connor, W.T.; Kehr, J.; Ungerstedt, U. Regional specific effects of clozapine and haloperidol on GABA and dopamine release in rat basal ganglia. Eur. J. Pharmacol. 1990, 187, 385–397. [Google Scholar] [CrossRef]
- Drew, K.L.; O’Connor, W.T.; Kehr, J.; Ungerstedt, U. Characterization of gamma-aminobutyric acid and dopamine overflow following acute implantation of a microdialysis probe. Life Sci. 1989, 45, 1307–1317. [Google Scholar] [CrossRef]
- Di Chiara, G.; Imperato, A. Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc. Natl. Acad. Sci. USA 1988, 85, 5274–5278. [Google Scholar] [CrossRef]
- Wydra, K.; Golembiowska, K.; Zaniewska, M.; Kamińska, K.; Ferraro, L.; Fuxe, K.; Filip, M. Accumbal and pallidal dopamine, glutamate and GABA overflow during cocaine self-administration and its extinction in rats. Addict. Biol. 2013, 18, 307–324. [Google Scholar] [CrossRef] [PubMed]
- Borgkvist, A.; Malmlöf, T.; Feltmann, K.; Lindskog, M.; Schilström, B. Dopamine in the hippocampus is cleared by the norepinephrine transporter. Int. J. Neuropsychopharmacol. 2012, 15, 531–540. [Google Scholar] [CrossRef]
- Sotomayor-Zárate, R.; Araya, K.A.; Pereira, P.; Blanco, E.; Quiroz, G.; Pozo, S.; Carreño, P.; Andrés, M.E.; Forray, M.I.; Gysling, K. Activation of GABA-B receptors induced by systemic amphetamine abolishes dopamine release in the rat lateral septum. J. Neurochem. 2010, 114, 1678–1686. [Google Scholar] [CrossRef]
- Smith, D.G.; Tzavara, E.T.; Shaw, J.; Luecke, S.; Wade, M.; Davis, R.; Salhoff, C.; Nomikos, G.G.; Gehlert, D.R. Mesolimbic Dopamine Super-Sensitivity in Melanin-Concentrating Hormone-1 Receptor-Deficient Mice. J. Neurosci. 2005, 25, 914–922. [Google Scholar] [CrossRef]
- Anagnostakis, Y.; Kastellakis, A.; Spyraki, C. Dizocilpine (MK-801) and tetrodotoxin influence accumbal dopamine release evoked by intrapallidal morphine. Eur. Neuropsychopharmacol. 1998, 8, 47–53. [Google Scholar] [CrossRef]
- Ferre, S.; Cortes, R.; Artigas, F. Dopaminergic regulation of the serotonergic raphe-striatal pathway: Microdialysis studies in freely moving rats. J. Neurosci. 1994, 14, 4839–4846. [Google Scholar] [CrossRef]
- Klitenick, M.A.; Kalivas, P.W. Behavioral and neurochemical studies of opioid effects in the pedunculopontine nucleus and mediodorsal thalamus. J Pharmacol. Exp. Ther. 1994, 269, 437–448. [Google Scholar] [PubMed]
- Parsons, L.H.; Justice, J.B. Serotonin and Dopamine Sensitization in the Nucleus Accumbens, Ventral Tegmental Area, and Dorsal Raphe Nucleus Following Repeated Cocaine Administration. J. Neurochem. 1993, 61, 1611–1619. [Google Scholar] [CrossRef]
- Hurd, Y.L.; McGrego, A.; Pontén, M. In vivo Amygdala Dopamine Levels Modulate Cocaine Self-administration Behaviour in the Rat: D1 Dopamine Receptor Involvement. Eur. J. Neurosci. 1997, 9, 2541–2548. [Google Scholar] [CrossRef]
- Hurd, Y.L.; Pontén, M. Cocaine self-administration behavior can be reduced or potentiated by the addition of specific dopamine concentrations in the nucleus accumbens and amygdala using in vivo microdialysis. Behav. Brain Res. 2000, 116, 177–186. [Google Scholar] [CrossRef]
- Reid, M.S.; Herrera-Marschitz, M.; Ungerstedt, U. Effects of Intranigral Substance P and Neurokinin A Injections on Extracellular Dopamine Levels Measured with Microdialysis in the Striatum and Frontoparietal Cortex of Rats. J. Neurochem. 1991, 57, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Marschitz, M.; Goiny, M.; Utsumi, H.; Ungerstedt, U. Mesencephalic dopamine innervation of the frontoparietal (sensorimotor) cortex of the rat: A microdialysis study. Neurosci. Lett. 1989, 97, 266–270. [Google Scholar] [CrossRef]
- Kretschmer, B.D.; Goiny, M.; Herrera-Marschitz, M. Effect of Intracerebral Administration of NMDA and AMPA on Dopamine and Glutamate Release in the Ventral Pallidum and on Motor Behavior. J. Neurochem. 2000, 74, 2049–2057. [Google Scholar] [CrossRef] [PubMed]
- Sizemore, G.M.; Co, C.; Smith, J.E. Ventral pallidal extracellular fluid levels of dopamine, serotonin, gamma amino butyric acid, and glutamate during cocaine self-administration in rats. Psychopharmacology 2000, 150, 391–398. [Google Scholar] [CrossRef]
- Fuchs, H.; Hauber, W. Reverse microdialysis of ionotropic glutamate receptor agonists in the rat globus pallidus increased extracellular dopamine. Neurosci. Lett. 2003, 343, 37–40. [Google Scholar] [CrossRef]
- Fuchs, H.; Hauber, W. Changes in extracellular dopamine in the rat globus pallidus induced by typical and atypical antipsychotic drugs. Neurochem. Int. 2004, 45, 1029–1038. [Google Scholar] [CrossRef]
- Fuchs, H.; Nagel, J.; Hauber, W. Effects of physiological and pharmacological stimuli on dopamine release in the rat globus pallidus. Neurochem. Int. 2005, 47, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Grasshoff, C.; Herrera-Marschitz, M.; Goiny, M.; Kretschmer, B.D. Modulation of ventral pallidal dopamine and glutamate release by the intravenous anesthetic propofol studied by in vivo microdialysis. Amino Acids 2005, 28, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Melendez, R.I.; Rodd, Z.A.; McBride, W.J.; Murphy, J.M. Dopamine receptor regulation of ethanol intake and extracellular dopamine levels in the ventral pallidum of alcohol preferring (P) rats. Drug Alcohol Depend. 2005, 77, 293–301. [Google Scholar] [CrossRef]
- Cauli, O.; Mlili, N.; Rodrigo, R.; Felipo, V. Hyperammonaemia alters the mechanisms by which metabotropic glutamate receptors in nucleus accumbens modulate motor function. J. Neurochem. 2007, 103, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Ding, Z.-M.; Oster, S.M.; Hall, S.R.; Engleman, E.A.; Hauser, S.R.; McBride, W.J.; Rodd, Z.A. The stimulating effects of ethanol on ventral tegmental area dopamine neurons projecting to the ventral pallidum and medial prefrontal cortex in female Wistar rats: Regional difference and involvement of serotonin-3 receptors. Psychopharmacology 2011, 216, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Orosco, M.; Rough, C.; Meile, M.J.; Nicolaidis, S. Spontaneous feeding-related monoamine changes in rostromedial hypothalamus of the obese Zucker rat: A microdialysis study. Physiol. Behav. 1995, 57, 1103–1106. [Google Scholar] [CrossRef]
- Timmerman, W.; Poelman, R.T.; Westerink, B.H.C.; Schuiling, G.A. Semicircadian Rhythm of Dopamine Release in the Mediobasal Hypothalamus in Awake Rats during Pseudopregnancy: Evidence that a Thyrotropin-Releasing Hormone Analogue Stimulates Dopamine Release and thereby Inhibits Prolactin Secretion. NEN 1995, 62, 434–443. [Google Scholar] [CrossRef]
- Fetissov, S.O.; Meguid, M.M.; Chen, C.; Miyata, G. Synchronized release of dopamine and serotonin in the medial and lateral hypothalamus of rats. Neuroscience 2000, 101, 657–663. [Google Scholar] [CrossRef]
- Flik, G.; Folgering, J.H.A.; Cremers, T.I.H.F.; Westerink, B.H.C.; Dremencov, E. Interaction Between Brain Histamine and Serotonin, Norepinephrine, and Dopamine Systems: In Vivo Microdialysis and Electrophysiology Study. J. Mol. Neurosci. 2015, 56, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Ortega, J.E.; Katner, J.; Davis, R.; Wade, M.; Nisenbaum, L.; Nomikos, G.G.; Svensson, K.A.; Perry, K.W. Modulation of neurotransmitter release in orexin/hypocretin-2 receptor knockout mice: A microdialysis study. J. Neurosci. Res. 2012, 90, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Nisijima, K.; Kuboshima, K.; Shioda, K.; Yoshino, T.; Iwamura, T.; Kato, S. Memantine attenuates 3,4-methylenedioxymethamphetamine-induced hyperthermia in rats. Neurosci. Lett. 2012, 531, 198–203. [Google Scholar] [CrossRef]
- Swanson, C.J.; Perry, K.W.; Koch-Krueger, S.; Katner, J.; Svensson, K.A.; Bymaster, F.P. Effect of the attention deficit/hyperactivity disorder drug atomoxetine on extracellular concentrations of norepinephrine and dopamine in several brain regions of the rat. Neuropharmacology 2006, 50, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Melis, M.R.; Succu, S.; Mascia, M.S.; Cortis, L.; Argiolas, A. Extra-cellular dopamine increases in the paraventricular nucleus of male rats during sexual activity. Eur. J. Neurosci. 2003, 17, 1266–1272. [Google Scholar] [CrossRef]
- Gören, M.Z.; Aker, R.; Yananlı, H.R.; Onat, F.Y. Extracellular Concentrations of Catecholamines and Amino Acids in the Dorsomedial Hypothalamus of Kindled Rats. Pharmacology 2003, 68, 190–197. [Google Scholar] [CrossRef]
- Obi, K.; Amano, I.; Takatsuru, Y. Role of dopamine on functional recovery in the contralateral hemisphere after focal stroke in the somatosensory cortex. Brain Res. 2018, 1678, 146–152. [Google Scholar] [CrossRef]
- Caruana, D.A.; Sorge, R.E.; Stewart, J.; Chapman, C.A. Dopamine Has Bidirectional Effects on Synaptic Responses to Cortical Inputs in Layer II of the Lateral Entorhinal Cortex. J. Neurophysiology 2006, 96, 3006–3015. [Google Scholar] [CrossRef]
- Pum, M.E.; Huston, J.P.; De Souza Silva, M.A.; Müller, C.P. Visual sensory-motor gating by serotonin activation in the medial prefrontal and occipital, but not in the rhinal, cortices in rats. Neuroscience 2008, 153, 361–372. [Google Scholar] [CrossRef]
- Devoto, P.; Flore, G.; Pani, L.; Gessa, G.L. Evidence for co-release of noradrenaline and dopamine from noradrenergic neurons in the cerebral cortex. Mol. Psychiatry 2001, 6, 657–664. [Google Scholar] [CrossRef]
- Müller, C.P.; Huston, J.P. Dopamine activity in the occipital and temporal cortices of rats: Dissociating effects of sensory but not pharmacological stimulation. Synapse 2007, 61, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Ash, E.S.; Heal, D.J.; Stanford, S.C. Contrasting changes in extracellular dopamine and glutamate along the rostrocaudal axis of the anterior cingulate cortex of the rat following an acute d-amphetamine or dopamine challenge. Neuropharmacology 2014, 87, 180–187. [Google Scholar] [CrossRef][Green Version]
- Mabrouk, O.S.; Semaan, D.Z.; Mikelman, S.; Gnegy, M.E.; Kennedy, R.T. Amphetamine stimulates movement through thalamocortical glutamate release. J. Neurochem. 2014, 128, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.-C.; Li, Z.; Dai, J.; Meltzer, H.Y.; Ichikawa, J. Clozapine increases both acetylcholine and dopamine release in rat ventral hippocampus: Role of 5-HT1A receptor agonism. Brain Res. 2004, 1023, 54–63. [Google Scholar] [CrossRef]
- Muraoka, H.; Oshibuchi, H.; Kawano, M.; Kawano, T.; Tsutsumi, T.; Yamada, M.; Ishigooka, J.; Nishimura, K.; Inada, K. Escitalopram attenuates fear stress-induced increase in amygdalar dopamine following methamphetamine-induced sensitisation: Implications of fine-tuning action of selective serotonin reuptake inhibitors on emotional processing. Eur. J. Pharmacol. 2018, 834, 1–9. [Google Scholar] [CrossRef]
- Kawano, T.; Oshibuchi, H.; Kawano, M.; Muraoka, H.; Tsutsumi, T.; Yamada, M.; Ishigooka, J.; Nishimura, K.; Inada, K. Diazepam suppresses the stress-induced dopaminergic release in the amygdala of methamphetamine-sensitized rat. Eur. J. Pharmacol. 2018, 833, 247–254. [Google Scholar] [CrossRef]
- Del Arco, A.; Ronzoni, G.; Mora, F. Hypofunction of prefrontal cortex NMDA receptors does not change stress-induced release of dopamine and noradrenaline in amygdala but disrupts aversive memory. Psychopharmacology 2015, 232, 2577–2586. [Google Scholar] [CrossRef] [PubMed]
- Izumo, N.; Ishibashi, Y.; Ohba, M.; Morikawa, T.; Manabe, T. Decreased voluntary activity and amygdala levels of serotonin and dopamine in ovariectomized rats. Behav. Brain Res. 2012, 227, 1–6. [Google Scholar] [CrossRef]
- Oshibuchi, H.; Inada, K.; Sugawara, H.; Ishigooka, J. Aripiprazole and haloperidol suppress excessive dopamine release in the amygdala in response to conditioned fear stress, but show contrasting effects on basal dopamine release in methamphetamine-sensitized rats. Eur. J. Pharmacol. 2009, 615, 83–90. [Google Scholar] [CrossRef][Green Version]
- Barr, G.A.; Moriceau, S.; Shionoya, K.; Muzny, K.; Gao, P.; Wang, S.; Sullivan, R.M. Transitions in infant learning are modulated by dopamine in the amygdala. Nat. Neurosci. 2009, 12, 1367–1369. [Google Scholar] [CrossRef]
- Tor-Agbidye, J.; Yamamoto, B.; Bowyer, J.F. Seizure Activity and Hyperthermia Potentiate the Increases in Dopamine and Serotonin Extracellular Levels in the Amygdala during Exposure to d-Amphetamine. Toxicol. Sci. 2001, 60, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Sotomayor-Zárate, R.; Renard, G.M.; Araya, K.A.; Carreño, P.; Fuentealba, J.A.; Andrés, M.E.; Gysling, K. Long-term loss of dopamine release mediated by CRF-1 receptors in the rat lateral septum after repeated cocaine administration. Behav. Brain Res. 2013, 250, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Chefer, V.I.; Denoroy, L.; Zapata, A.; Shippenberg, T.S. Mu opioid receptor modulation of somatodendritic dopamine overflow: GABAergic and glutamatergic mechanisms. Eur. J. Neurosci. 2009, 30, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Cobb, W.S.; Abercrombie, E.D. Relative involvement of globus pallidus and subthalamic nucleus in the regulation of somatodendritic dopamine release in substantia nigra is dopamine-dependent. Neuroscience 2003, 119, 777–786. [Google Scholar] [CrossRef]
- Yamamuro, Y.; Hori, K.; Tanaka, J.; Iwano, H.; Nomura, M. Septo-hippocampal cholinergic system under the discrimination learning task in the rat: A microdialysis study with the dual-probe approach. Brain Res. 1995, 684, 1–7. [Google Scholar] [CrossRef]
- Miyazaki, K.W.; Miyazaki, K.; Doya, K. Activation of the central serotonergic system in response to delayed but not omitted rewards. Eur. J. Neurosci. 2011, 33, 153–160. [Google Scholar] [CrossRef]
- Neugebauer, F.; Korz, V.; Frey, J.U. Modulation of extracellular monoamine transmitter concentrations in the hippocampus after weak and strong tetanization of the perforant path in freely moving rats. Brain Res. 2009, 1273, 29–38. [Google Scholar] [CrossRef]
- Smolders, I.; Clinckers, R.; Meurs, A.; De Bundel, D.; Portelli, J.; Ebinger, G.; Michotte, Y. Direct enhancement of hippocampal dopamine or serotonin levels as a pharmacodynamic measure of combined antidepressant–anticonvulsant action. Neuropharmacology 2008, 54, 1017–1028. [Google Scholar] [CrossRef]
- Venton, B.J.; Zhang, H.; Garris, P.A.; Phillips, P.E.M.; Sulzer, D.; Wightman, R.M. Real-time decoding of dopamine concentration changes in the caudate–putamen during tonic and phasic firing. J. Neurochem. 2003, 87, 1284–1295. [Google Scholar] [CrossRef]
- Paxinos, G.; Watson, C. The Rat Brain in Stereotaxic Coordinates: Hard Cover Edition, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Davies, M.I.; Cooper, J.D.; Desmond, S.S.; Lunte, C.E.; Lunte, S.M. Analytical considerations for microdialysis sampling. Adv. Drug Deliv. Rev. 2000, 45, 169–188. [Google Scholar] [CrossRef]
- Robinson, D.L.; Heien, M.L.A.V.; Wightman, R.M. Frequency of Dopamine Concentration Transients Increases in Dorsal and Ventral Striatum of Male Rats during Introduction of Conspecifics. J. Neurosci. 2002, 22, 10477–10486. [Google Scholar] [CrossRef] [PubMed]
- Hamid, A.A.; Pettibone, J.R.; Mabrouk, O.S.; Hetrick, V.L.; Schmidt, R.; Vander Weele, C.M.; Kennedy, R.T.; Aragona, B.J.; Berke, J.D. Mesolimbic dopamine signals the value of work. Nat. Neurosci. 2016, 19, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Roitman, M.F. Dopamine Operates as a Subsecond Modulator of Food Seeking. J. Neurosci. 2004, 24, 1265–1271. [Google Scholar] [CrossRef] [PubMed]
- Phillips, P.E.M.; Stuber, G.D.; Heien, M.L.A.V.; Wightman, R.M.; Carelli, R.M. Subsecond dopamine release promotes cocaine seeking. Nature 2003, 422, 614–618. [Google Scholar] [CrossRef] [PubMed]
- Howe, M.W.; Tierney, P.L.; Sandberg, S.G.; Phillips, P.E.M.; Graybiel, A.M. Prolonged dopamine signalling in striatum signals proximity and value of distant rewards. Nature 2013, 500, 575–579. [Google Scholar] [CrossRef] [PubMed]
- Wightman, R.M.; Heien, M.L.A.V.; Wassum, K.M.; Sombers, L.A.; Aragona, B.J.; Khan, A.S.; Ariansen, J.L.; Cheer, J.F.; Phillips, P.E.M.; Carelli, R.M. Dopamine release is heterogeneous within microenvironments of the rat nucleus accumbens. Eur. J. Neurosci. 2007, 26, 2046–2054. [Google Scholar] [CrossRef] [PubMed]
- Oleson, E.B.; Salek, J.; Bonin, K.D.; Jones, S.R.; Budygin, E.A. Real-time voltammetric detection of cocaine-induced dopamine changes in the striatum of freely moving mice. Neurosci. Lett. 2009, 467, 144–146. [Google Scholar] [CrossRef]
- Bass, C.E.; Grinevich, V.P.; Vance, Z.B.; Sullivan, R.P.; Bonin, K.D.; Budygin, E.A. Optogenetic control of striatal dopamine release in rats. J. Neurochem. 2010, 114, 1344–1352. [Google Scholar] [CrossRef] [PubMed]
- Vickrey, T.L.; Condron, B.; Venton, B.J. Rapid Detection of Endogenous Dopamine Changes in Drosophila melanogaster using Fast-Scan Cyclic Voltammetry. Anal. Chem. 2009, 81, 9306–9313. [Google Scholar] [CrossRef]
- Pisano, F.; Pisanello, M.; Lee, S.J.; Lee, J.; Maglie, E.; Balena, A.; Sileo, L.; Spagnolo, B.; Bianco, M.; Hyun, M.; et al. Depth-resolved fiber photometry with a single tapered optical fiber implant. Nat. Methods 2019, 16, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Kesner, A.J.; Shin, R.; Calva, C.B.; Don, R.F.; Junn, S.; Potter, C.T.; Ramsey, L.A.; Abou-Elnaga, A.F.; Cover, C.G.; Wang, D.V.; et al. Supramammillary neurons projecting to the septum regulate dopamine and motivation for environmental interaction. bioRxiv 2020. [Google Scholar] [CrossRef]
- Lefevre, E.M.; Pisansky, M.T.; Toddes, C.; Baruffaldi, F.; Pravetoni, M.; Tian, L.; Kono, T.J.Y.; Rothwell, P.E. Interruption of continuous opioid exposure exacerbates drug-evoked adaptations in the mesolimbic dopamine system. Neuropsychopharmacology 2020, 45, 1781–1792. [Google Scholar] [CrossRef] [PubMed]
- Kramer, P.F.; Twedell, E.L.; Shin, J.H.; Zhang, R.; Khaliq, Z.M. Axonal mechanisms mediating GABA-A receptor inhibition of striatal dopamine release. bioRxiv 2020. [Google Scholar] [CrossRef]
- Tang, W.; Kochubey, O.; Kintscher, M.; Schneggenburger, R. Dopamine in the basal amygdala signals salient somatosensory events during fear learning. bioRxiv 2019, 716589. [Google Scholar] [CrossRef]
- Eisenhofer, G.; Kopin, I.J.; Goldstein, D.S. Catecholamine Metabolism: A Contemporary View with Implications for Physiology and Medicine. Pharmacol. Rev. 2004, 56, 331–349. [Google Scholar] [CrossRef] [PubMed]
- Berridge, C.W.; Waterhouse, B.D. The locus coeruleus–noradrenergic system: Modulation of behavioral state and state-dependent cognitive processes. Brain Res. Revs. 2003, 42, 33–84. [Google Scholar] [CrossRef]
- Léna, I.; Parrot, S.; Deschaux, O.; Muffat-Joly, S.; Sauvinet, V.; Renaud, B.; Suaud-Chagny, M.-F.; Gottesmann, C. Variations in extracellular levels of dopamine, noradrenaline, glutamate, and aspartate across the sleep–wake cycle in the medial prefrontal cortex and nucleus accumbens of freely moving rats. J. Neurosci. Res. 2005, 81, 891–899. [Google Scholar] [CrossRef]
- Ihalainen, J.A.; Riekkinen Jr, P.; Feenstra, M.G.P. Comparison of dopamine and noradrenaline release in mouse prefrontal cortex, striatum and hippocampus using microdialysis. Neurosci. Lett. 1999, 277, 71–74. [Google Scholar] [CrossRef]
- Finlay, J.M.; Zigmond, M.J.; Abercrombie, E.D. Increased dopamine and norepinephrine release in medial prefrontal cortex induced by acute and chronic stress: Effects of diazepam. Neuroscience 1995, 64, 619–628. [Google Scholar] [CrossRef]
- Li, X.-M.; Perry, K.W.; Wong, D.T.; Bymaster, F.P. Olanzapine increases in vivo dopamine and norepinephrine release in rat prefrontal cortex, nucleus accumbens and striatum. Psychopharmacology 1998, 136, 153–161. [Google Scholar] [CrossRef]
- Sabatini, B.L. The impact of reporter kinetics on the interpretation of data gathered with fluorescent reporters. bioRxiv 2019, 834895. [Google Scholar] [CrossRef]
- Paoletti, P.; Ellis-Davies, G.C.R.; Mourot, A. Optical control of neuronal ion channels and receptors. Nat. Rev. Neurosci. 2019, 20, 514–532. [Google Scholar] [CrossRef] [PubMed]
- Horton, N.G.; Wang, K.; Kobat, D.; Clark, C.G.; Wise, F.W.; Schaffer, C.B.; Xu, C. In vivo three-photon microscopy of subcortical structures within an intact mouse brain. Nat. Photonics 2013, 7, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Gomez, J.L.; Bonaventura, J.; Lesniak, W.; Mathews, W.B.; Sysa-Shah, P.; Rodriguez, L.A.; Ellis, R.J.; Richie, C.T.; Harvey, B.K.; Dannals, R.F.; et al. Chemogenetics revealed: DREADD occupancy and activation via converted clozapine. Science 2017, 357, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Nagai, Y.; Miyakawa, N.; Takuwa, H.; Hori, Y.; Oyama, K.; Ji, B.; Takahashi, M.; Huang, X.-P.; Slocum, S.T.; DiBerto, J.F.; et al. Deschloroclozapine, a potent and selective chemogenetic actuator enables rapid neuronal and behavioral modulations in mice and monkeys. Nat. Neurosci. 2020, 1–11. [Google Scholar] [CrossRef]
- Frohlich, J.; Van Horn, J.D. Reviewing the ketamine model for schizophrenia. J. Psychopharmacol. 2014, 28, 287–302. [Google Scholar] [CrossRef]
- Martianova, E.; Aronson, S.; Proulx, C.D. Multi-Fiber Photometry to Record Neural Activity in Freely-Moving Animals. JoVE 2019, 60278. [Google Scholar] [CrossRef]
- Bruno, C.A.; O’Brien, C.; Bryant, S.; Mejaes, J.; Pizzano, C.; Estrin, D.J.; Barker, D.J. pMAT: An Open-Source, Modular Software Suite for the Analysis of Fiber Photometry Calcium Imaging. bioRxiv 2020. [Google Scholar] [CrossRef]






| Sensor | Molecular Scaffold | Affinity (Kd/EC50) | Dynamic Range (dFFmax) | Molecular Specificity vs. NE | On Kinetics: t1/2 Rise Time | Off Kinetics: t1/2 Decay Time | 1-Photon Exc/Emis | Source |
|---|---|---|---|---|---|---|---|---|
| dLight1.1 | DRD1 | 330 nM * | 230% * | 70-fold | 10 ms ** | 100 ms ** | 490/517 nm | [58] |
| dLight1.2 | DRD1 | 765 nM * | 340% * | ND | 9.5 ms ** | 90 ms ** | 490/517 nm | [58] |
| dLight1.3a | DRD1 | 2300 nM * | 660% * | ND | ND | ND | ND | [58] |
| dLight1.3b | DRD1 | 1600 nM * | 930% * | 270-fold | ND | ND | ND | [58,101] |
| dLight1.4 | DRD4 | 4 nM * | 170% * | ND | ND | ND | ND | [58] |
| dLight1.5 | DRD2 | 110 nM * | 180% * | ND | ND | ND | ND | [58] |
| RdLight1 (red) | DRD1 | 860 nM * | 250% * | 60-fold | 14 ms ** | 400 ms ** | 560/588 nm | [59] |
| YdLight1 (yellow) | DRD1 | 1630 nM * | 310% * | ND | ND | ND | 514/525 nm | [59] |
| GRAB-DA1m | DRD2 | 130 nM * | 90% * | 10-fold | 60 ms * | 710 ms * | 490/510 nm | [60] |
| GRAB-DA1h | DRD2 | 10 nM * | 90% * | 10-fold | 140 ms * | 2520 ms * | 490/510 nm | [60] |
| GRAB-DA2m | DRD2 | 90 nM * | 340% * | 22-fold | 40 ms * | 1300 ms * | ND | [61] † |
| GRAB-DA2h | DRD2 | 7 nM * | 280% * | 15-fold | 50 ms * | 7300 ms * | 500/520 nm | [61] † |
| GRAB-rDA1m (red-shifted) | DRD2 | 95 nM * | 150% * | 15-fold | 80 ms * | 770 ms * | 565/595 nm | [61] † |
| GRAB-rDA1h (red-shifted) | DRD2 | 4 nM * | 100% * | 10-fold | 60 ms * | 2150 ms * | 565/595 nm | [61] † |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Labouesse, M.A.; Cola, R.B.; Patriarchi, T. GPCR-Based Dopamine Sensors—A Detailed Guide to Inform Sensor Choice for In Vivo Imaging. Int. J. Mol. Sci. 2020, 21, 8048. https://doi.org/10.3390/ijms21218048
Labouesse MA, Cola RB, Patriarchi T. GPCR-Based Dopamine Sensors—A Detailed Guide to Inform Sensor Choice for In Vivo Imaging. International Journal of Molecular Sciences. 2020; 21(21):8048. https://doi.org/10.3390/ijms21218048
Chicago/Turabian StyleLabouesse, Marie A., Reto B. Cola, and Tommaso Patriarchi. 2020. "GPCR-Based Dopamine Sensors—A Detailed Guide to Inform Sensor Choice for In Vivo Imaging" International Journal of Molecular Sciences 21, no. 21: 8048. https://doi.org/10.3390/ijms21218048
APA StyleLabouesse, M. A., Cola, R. B., & Patriarchi, T. (2020). GPCR-Based Dopamine Sensors—A Detailed Guide to Inform Sensor Choice for In Vivo Imaging. International Journal of Molecular Sciences, 21(21), 8048. https://doi.org/10.3390/ijms21218048





