Intracrine Endorphinergic Systems in Modulation of Myocardial Differentiation
Abstract
:1. Introduction
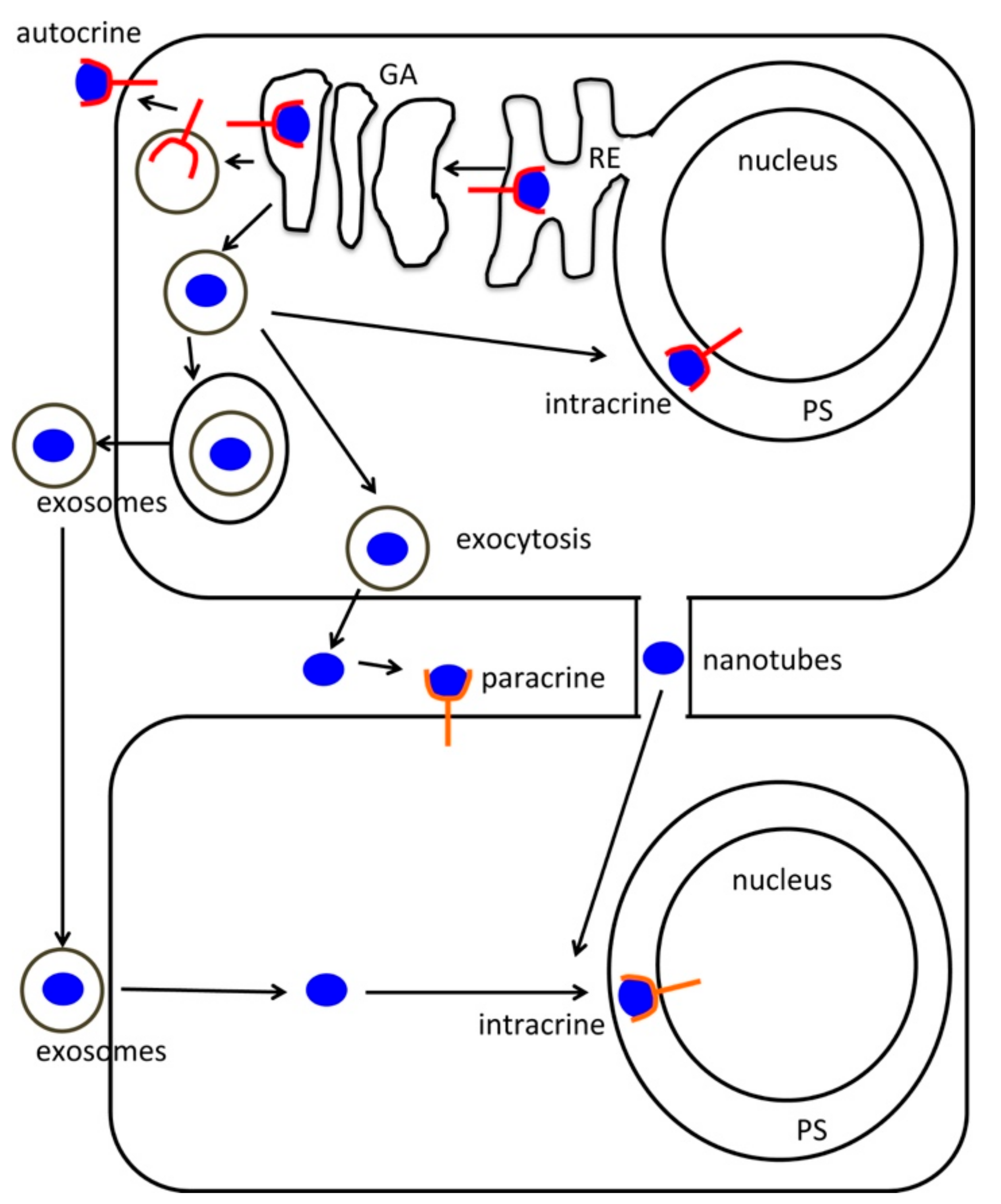
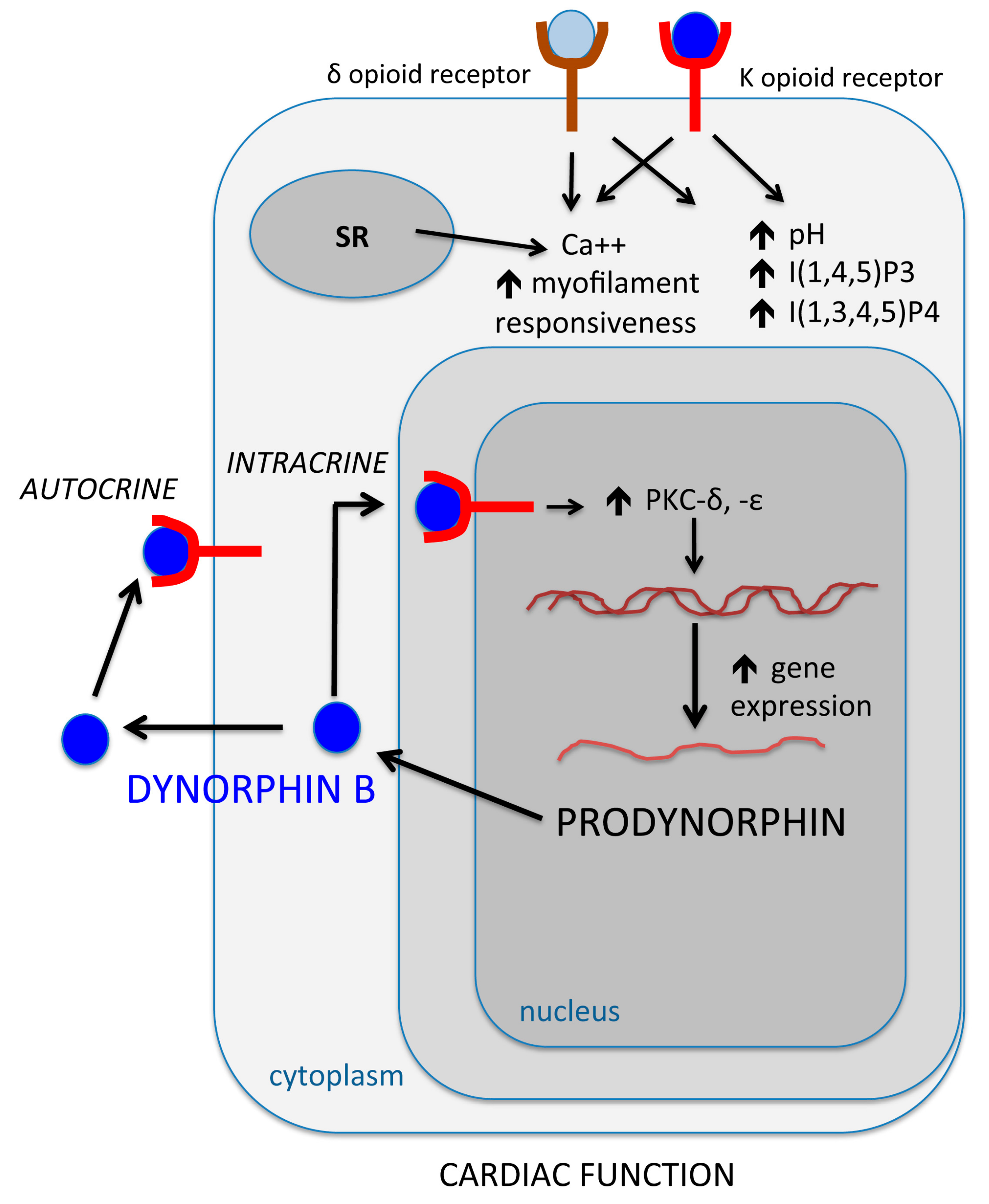
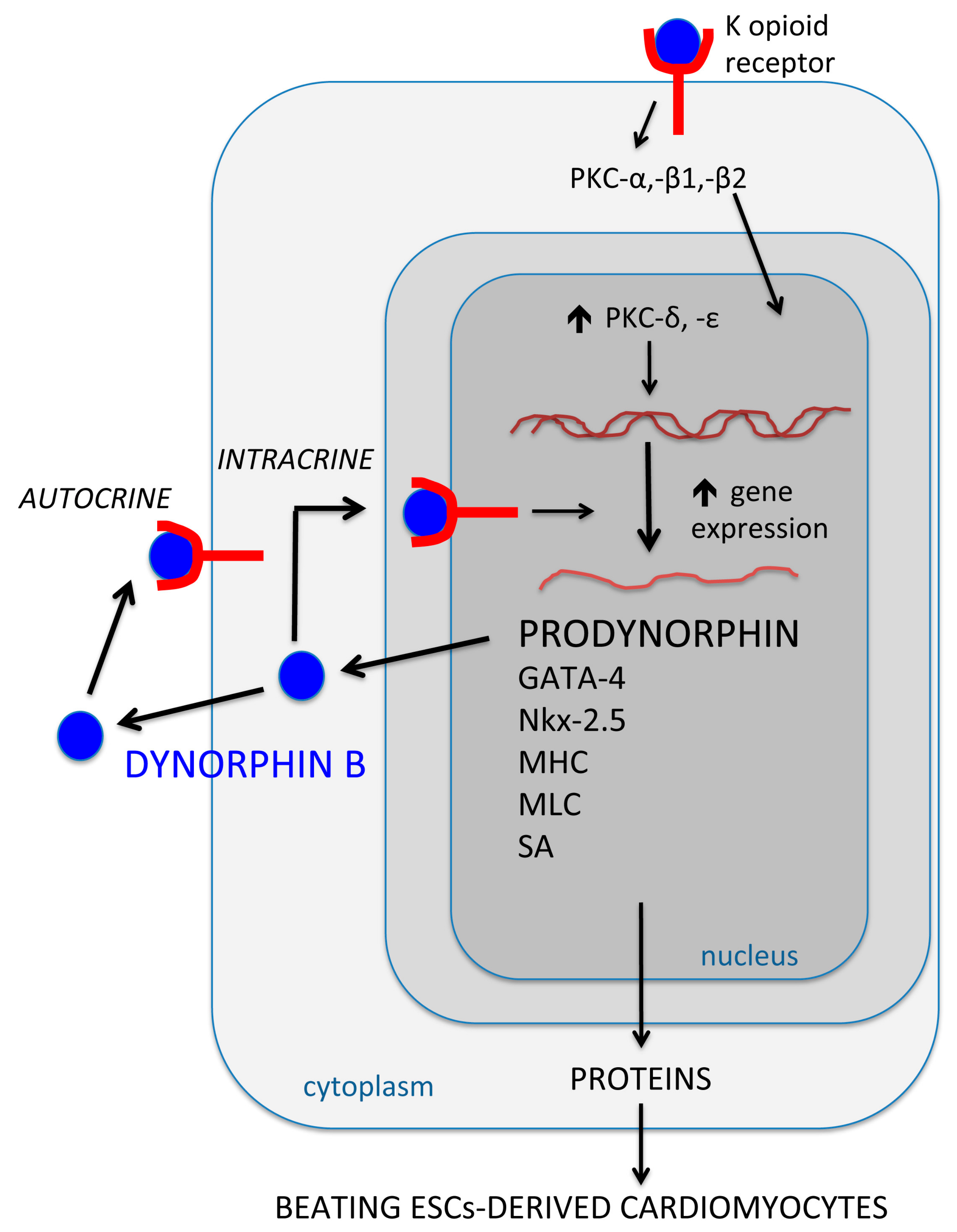
2. Opioid Peptides and the Intracrine Regulation of Cellular Dynamics: A Pattern Highly Oriented Towards Stem Cell Cardiogenesis
3. Hyaluronan Mixed Esters of Butyric and Retinoic Acids (HBR): A Synthetic Intracrine Promoting Prodynorphin Gene and Dynorphin B Expression, Stem Cell Cardiogenesis and Cardiac Repair
4. The Use of Electromagnetic Energy to Afford Efficient Increase in Prodynorphin Gene and Dynorphin B Expression. Implications in Myocardial Cell Growth and Stem Cell Cardiogenesis
5. Conclusions and Future Perspective
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACE | Angiotensin Converting Enzyme |
| AD | Adipose tissue |
| CPCs | Cardiac progenitor cells |
| DMSO | Dimethyl sulfoxide |
| ELF-MF | Extremely-low frequency magnetic fields |
| ESCs | Embryonic stem cells |
| FGF1 | Fibroblast growth factor 1 |
| FM | Fetal membranes |
| HBR | Hyaluronan mixed esters of butyric and retinoic acids |
| HEL | Human erythroleukemia cells |
| HGF | Hepatocyte growth factor |
| hMSCs | Human mesenchymal stem cells |
| HSCs | Hematopoietic stem cells |
| Ins(1,3,4,5)P4 | Inositol 1,3,4,5-tetrakisphosphate |
| Ins(1,4,5)P3 | Inositol 1,4,5-trisphosphate |
| KCl | Potassium chloride |
| KDR | Kinase insert domain receptor |
| MF | Magnetic fields |
| MHC | Myosin heavy chain |
| MKs | megakaryocytes |
| MLC | Myosin light chain |
| PKC | Protein kinase C |
| PMA | Phorbol 12-myristate 13-acetate |
| REAC | Radioelectric asymmetric conveyer |
| SA | Sarcomeric actinin |
| VEGF | Vascular endothelial growth factor |
References
- Re, R.; Parab, M. Effect of angiotensin II on RNA synthesis by isolated nuclei. Life Sci. 1984, 34, 647–651. [Google Scholar] [CrossRef]
- Re, R.N.; Vizard, D.L.; Brown, J.; Bryan, S.E. Angiotensin II receptors in chromatin fragments generated by micrococcal nuclease. Biochem. Biophys. Res. Commun. 1984, 119, 220–227. [Google Scholar] [CrossRef]
- Re, R.N. The nature of intracrine peptide hormone action. Hypertension 1999, 34, 534–538. [Google Scholar] [CrossRef] [PubMed]
- Re, R.N. On the biological actions of intracellular angiotensin. Hypertension 2000, 35, 1189–1190. [Google Scholar] [CrossRef] [PubMed]
- Re, R.N. The intracrine hypothesis and intracellular peptide hormone action. Bioessays 2003, 25, 401–409. [Google Scholar] [CrossRef]
- Re, R.N.; Cook, J.L. Senescence, apoptosis, and stem cell biology: The rationale for an expanded view of intracrine action. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H893–H901. [Google Scholar] [CrossRef]
- Gerber, H.P.; Malik, A.K.; Solar, G.P.; Sherman, D.; Liang, X.H.; Meng, G.; Hong, K.; Marsters, J.C.; Ferrara, N. VEGF regulates haematopoietic stem cell survival by an internal autocrine loop mechanism. Nature 2002, 417, 954–958. [Google Scholar] [CrossRef]
- Murray, L.M.A.; Krasnodembskaya, A.D. Concise Review: Intercellular Communication Via Organelle Transfer in the Biology and Therapeutic Applications of Stem Cells. Stem Cells 2019, 37, 14–25. [Google Scholar] [CrossRef]
- Zhang, J.; Whitehead, J.; Liu, Y.; Yang, Q.; Leach, J.K.; Liu, G.Y. Direct Observation of Tunneling Nanotubes within Human Mesenchymal Stem Cell Spheroids. J. Phys. Chem. B 2018, 122, 9920–9926. [Google Scholar] [CrossRef]
- Sartori-Rupp, A.; Cordero Cervantes, D.; Pepe, A.; Gousset, K.; Delage, E.; Corroyer-Dulmont, S.; Schmitt, C.; Krijnse-Locker, J.; Zurzolo, C. Correlative cryo-electron microscopy reveals the structure of TNTs in neuronal cells. Nat. Commun. 2019, 10, 342. [Google Scholar] [CrossRef]
- Da Silva Novaes, A.; Ribeiro, R.; Pereira, L.G.; Borges, F.T.; Boim, M.A. Intracrine action of angiotensin II in mesangial cells: Subcellular distribution of angiotensin II receptor subtypes AT1 and AT2. Mol. Cell. Biochem. 2018, 448, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Gerber, H.P.; Ferrara, N. The role of VEGF in normal and neoplastic hematopoiesis. J. Mol. Med. (Berl.) 2003, 81, 20–31. [Google Scholar] [CrossRef] [PubMed]
- Kirito, K.; Fox, N.; Komatsu, N.; Kaushansky, K. Thrombopoietin enhances expression of vascular endothelial growth factor (VEGF) in primitive hematopoietic cells through induction of HIF-1alpha. Blood 2005, 105, 4258–4263. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.C.; Dias, S. Internal and external autocrine VEGF/KDR loops regulate survival of subsets of acute leukemia through distinct signaling pathways. Blood 2004, 103, 3883–3889. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.; Chen, T.T.; Barber, C.L.; Jordan, M.C.; Murdock, J.; Desai, S.; Ferrara, N.; Nagy, A.; Roos, K.P.; Iruela-Arispe, M.L. Autocrine VEGF signaling is required for vascular homeostasis. Cell 2007, 130, 691–703. [Google Scholar] [CrossRef]
- Zhang, Z.; Neiva, K.G.; Lingen, M.W.; Ellis, L.M.; Nör, J.E. VEGF-dependent tumor angiogenesis requires inverse and reciprocal regulation of VEGFR1 and VEGFR2. Cell Death Differ. 2010, 17, 499–512. [Google Scholar] [CrossRef]
- Bhattacharya, R.; Ye, X.C.; Wang, R.; Ling, X.; McManus, M.; Fan, F.; Boulbes, D.; Ellis, L.M. Intracrine VEGF Signaling Mediates the Activity of Prosurvival Pathways in Human Colorectal Cancer Cells. Cancer Res. 2016, 76, 3014–3024. [Google Scholar] [CrossRef] [Green Version]
- Josefsson, E.C.; James, C.; Henley, K.J.; Debrincat, M.A.; Rogers, K.L.; Dowling, M.R.; White, M.J.; Kruse, E.A.; Lane, R.M.; Ellis, S.; et al. Megakaryocytes possess a functional intrinsic apoptosis pathway that must be restrained to survive and produce platelets. J. Exp. Med. 2011, 208, 2017–2031. [Google Scholar] [CrossRef]
- Debrincat, M.A.; Josefsson, E.C.; James, C.; Henley, K.J.; Ellis, S.; Lebois, M.; Betterman, K.L.; Lane, R.M.; Rogers, K.L.; White, M.J.; et al. Mcl-1 and Bcl-x(L) coordinately regulate megakaryocyte survival. Blood 2012, 119, 5850–5858. [Google Scholar] [CrossRef]
- Kodama, T.; Hikita, H.; Kawaguchi, T.; Shigekawa, M.; Shimizu, S.; Hayashi, Y.; Li, W.; Miyagi, T.; Hosui, A.; Tatsumi, T.; et al. Mcl-1 and Bcl-xL regulate Bak/Bax-dependent apoptosis of the megakaryocytic lineage at multistages. Cell Death Differ. 2012, 19, 1856–1869. [Google Scholar] [CrossRef] [Green Version]
- Sarvothaman, S.; Undi, R.B.; Pasupuleti, S.R.; Gutti, U.; Gutti, R.K. Apoptosis: Role in myeloid cell development. Blood Res. 2015, 50, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Delmas, E.; Jah, N.; Pirou, C.; Bouleau, S.; Le Floch, N.; Vayssière, J.L.; Mignotte, B.; Renaud, F. FGF1 C-terminal domain and phosphorylation regulate intracrine FGF1 signaling for its neurotrophic and anti-apoptotic activities. Cell Death Dis. 2016, 7, e2079. [Google Scholar] [CrossRef] [PubMed]
- Tadevosyan, A.; Létourneau, M.; Folch, B.; Doucet, N.; Villeneuve, L.R.; Mamarbachi, A.M.; Pétrin, D.; Hébert, T.E.; Fournier, A.; Chatenet, D.; et al. Photoreleasable ligands to study intracrine angiotensin II signalling. J. Physiol. 2015, 593, 521–539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Merlen, C.; Villeneuve, L.R.; Allen, B.G. Using caged ligands to study intracrine endothelin signaling in intact cardiac myocytes. Methods Mol. Biol. 2015, 1234, 31–41. [Google Scholar] [CrossRef]
- Reyes, S.; Varagic, J.; Ahmad, S.; VonCannon, J.; Kon, N.D.; Wang, H.; Groban, L.; Cheng, C.P.; Dell’Italia, L.J.; Ferrario, C.M. Novel Cardiac Intracrine Mechanisms Based on Ang-(1-12)/Chymase Axis Require a Revision of Therapeutic Approaches in Human Heart Disease. Curr. Hypertens. Rep. 2017, 19, 16. [Google Scholar] [CrossRef]
- De Mello, W.C. Exchange of chemical signals between cardiac cells. Fundamental role on cell communication and metabolic cooperation. Exp. Cell Res. 2016, 346, 130–136. [Google Scholar] [CrossRef]
- Gaddis, R.; Dixon, W.R. Modulation of peripheral adrenergic neurotransmission by methionine-enkephalin. J. Pharmacol. Exp. Ther. 1982, 221, 282–288. [Google Scholar]
- Hughes, J.; Kosterlitz, H.W.; Leslie, F.M. Effect of morphine on adrenergic transmission in the mouse vas deferens. Assessment of agonist and antagonist potencies of narcotic analgesics. Br. J. Pharmacol. 1975, 53, 371–381. [Google Scholar] [CrossRef]
- Illes, P.; Ramme, D.; Starke, K. Inhibition of neuroeffector transmission in the rabbit mesenteric artery by [Met5] enkephalin. Eur. J. Pharmacol. 1985, 107, 397–398. [Google Scholar] [CrossRef]
- Wong-Dusting, H.K.; Rand, M.J. Effect of [D-Ala2, Met5] enkephalinamide and [D-Ala2, D-Leu5] enkephalin on cholinergic and noradrenergic neurotransmission in isolated atria. Eur. J. Pharmacol. 1985, 111, 65–72. [Google Scholar] [CrossRef]
- Konishi, S.; Tsunoo, A.; Otsuka, M. Enkephalin as a transmitter for presynaptic inhibition in sympathetic ganglia. Nature 1981, 294, 80–82. [Google Scholar] [CrossRef] [PubMed]
- Bixby, J.L.; Spitzer, N.C. Enkephalin reduces quantal content at the frog neuromuscular junction. Nature 1983, 301, 431–432. [Google Scholar] [CrossRef] [PubMed]
- Cherubini, E.; North, R.A. Mu and kappa opioids inhibit transmitter release by different mechanisms. Proc. Natl. Acad. Sci. USA 1985, 82, 1860–1863. [Google Scholar] [CrossRef] [PubMed]
- Wong-Dusting, H.; Rand, M.J. Effects of the opioid peptides [Met5] enkephalin-Arg6-Phe7 and [Met5] enkephalin-Arg6-Gly7-Leu8 on cholinergic neurotransmission in the rabbit isolated atria. Clin. Exp. Pharmacol. Physiol. 1987, 14, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Yang, H.Y.; Costa, E. Distribution of met5-enkephalin-Arg6-Phe7 (MEAP) in various tissues of rats and guinea pigs. Life Sci. 1982, 31, 2303–2306. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.E.; Hermann, K.; Dietz, R.; Gaida, W.; Ganten, D.; Kraft, K.; Unger, T. Evidence for the presence of enkephalins in the heart. Life Sci. 1983, 32, 399–406. [Google Scholar] [CrossRef]
- Weihe, E.; McKnight, A.T.; Corbett, A.D.; Hartschuh, W.; Reinecke, M.; Kosterlitz, H.W. Characterization of opioid peptides in guinea-pig heart and skin. Life Sci. 1983, 33 (Suppl. 1), 711–714. [Google Scholar] [CrossRef]
- Xiang, J.Z.; Archelos, J.; Lang, R.E. Enkephalins in the heart. Clin. Exp. Hypertens. A 1984, 6, 1883–1888. [Google Scholar]
- Weihe, E.; McKnight, A.T.; Corbett, A.D.; Kosterlitz, H.W. Proenkephalin- and prodynorphin- derived opioid peptides in guinea-pig heart. Neuropeptides 1985, 5, 453–456. [Google Scholar] [CrossRef]
- Ventura, C.; Bastagli, L.; Bernardi, P.; Caldarera, C.M.; Guarnieri, C. Opioid receptors in rat cardiac sarcolemma: Effect of phenylephrine and isoproterenol. Biochim. Biophys. Acta 1989, 987, 69–74. [Google Scholar] [CrossRef]
- Ventura, C.; Guarnieri, C.; Stefanelli, C.; Cirielli, C.; Lakatta, E.G.; Capogrossi, M.C. Comparison between alpha-adrenergic- and K-opioidergic-mediated inositol (1,4,5) P3/inositol (1,3,4,5) P4 formation in adult cultured rat ventricular cardiomyocytes. Biochem. Biophys. Res. Commun. 1991, 179, 972–978. [Google Scholar] [CrossRef]
- Ventura, C.; Capogrossi, M.C.; Spurgeon, H.A.; Lakatta, E.G. Kappa-opioid peptide receptor stimulation increases cytosolic pH and myofilament responsiveness to Ca2+ in cardiac myocytes. Am. J. Physiol. 1991, 261, H1671–H1674. [Google Scholar] [CrossRef] [PubMed]
- Ventura, C.; Spurgeon, H.; Lakatta, E.G.; Guarnieri, C.; Capogrossi, M.C. Kappa and delta opioid receptor stimulation affects cardiac myocyte function and Ca2+ release from an intracellular pool in myocytes and neurons. Circ. Res. 1992, 70, 66–81. [Google Scholar] [CrossRef] [PubMed]
- Clô, C.; Muscari, C.; Tantini, B.; Pignatti, C.; Bernardi, P.; Ventura, C. Reduced mechanical activity of perfused rat heart following morphine or enkephalin peptides administration. Life Sci. 1985, 37, 1327–1333. [Google Scholar] [CrossRef]
- Ventura, C.; Muscari, C.; Spampinato, S.; Bernardi, P.; Caldarera, C.M. Effects of naloxone on the mechanical activity of isolated rat hearts perfused with morphine or opioid peptides. Peptides 1987, 8, 695–699. [Google Scholar] [CrossRef]
- Caffrey, J.L.; Gaugl, J.F.; Jones, C.E. Local endogenous opiate activity in dog myocardium: Receptor blockade with naloxone. Am. J. Physiol. 1985, 248 Pt 2, H382–H388. [Google Scholar] [CrossRef]
- Laurent, S.; Marsh, J.D.; Smith, T.W. Enkephalins have a direct positive inotropic effect on cultured cardiac myocytes. Proc. Natl. Acad. Sci. USA 1985, 82, 5930–5934. [Google Scholar] [CrossRef]
- Laurent, S.; Marsh, J.D.; Smith, T.W. Enkephalins increase cyclic adenosine monophosphate content, calcium uptake, and contractile state in cultured chick embryo heart cells. J. Clin. Investig. 1986, 77, 1436–1440. [Google Scholar] [CrossRef]
- Howells, R.D.; Kilpatrick, D.L.; Bailey, L.C.; Noe, M.; Udenfriend, S. Proenkephalin mRNA in rat heart. Proc. Natl. Acad. Sci. USA 1986, 83, 1960–1963. [Google Scholar] [CrossRef]
- Springhorn, J.P.; Claycomb, W.C. Preproenkephalin mRNA expression in developing rat heart and in cultured ventricular cardiac muscle cells. Biochem. J. 1989, 258, 73–78. [Google Scholar] [CrossRef] [Green Version]
- Spampinato, S.; Canossa, M.; Ventura, C.; Bachetti, T.; Venturini, R.; Bastagli, L.; Bernardi, P.; Ferri, S. Heterogeneity of immunoreactive dynorphin B-like material in human, rat, rabbit and guinea-pig heart. Life Sci. 1991, 48, 551–559. [Google Scholar] [CrossRef]
- Ventura, C.; Guarnieri, C.; Vaona, I.; Campana, G.; Pintus, G.; Spampinato, S. Dynorphin gene expression and release in the myocardial cell. J. Biol. Chem. 1994, 269, 5384–5386. [Google Scholar] [PubMed]
- Ventura, C.; Pintus, G.; Vaona, I.; Bennardini, F.; Pinna, G.; Tadolini, B. Phorbol ester regulation of opioid peptide gene expression in myocardial cells. Role of nuclear protein kinase. J. Biol. Chem. 1995, 270, 30115–30120. [Google Scholar] [CrossRef] [PubMed]
- Ventura, C.; Maioli, M.; Pintus, G.; Posadino, A.M.; Tadolini, B. Nuclear opioid receptors activate opioid peptide gene transcription in isolated myocardial nuclei. J. Biol. Chem. 1998, 273, 13383–13386. [Google Scholar] [CrossRef] [PubMed]
- Ventura, C.; Maioli, M. Opioid peptide gene expression primes cardiogenesis in embryonal pluripotent stem cells. Circ. Res. 2000, 87, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Ventura, C.; Pintus, G.; Fiori, M.G.; Bennardini, F.; Pinna, G.; Gaspa, L. Opioid peptide gene expression in the primary hereditary cardiomyopathy of the Syrian hamster. (1) Regulation of prodynorphin gene expression by nuclear protein kinase C. J. Biol. Chem. 1997, 272, 6685–6692. [Google Scholar] [CrossRef] [PubMed]
- Ventura, C.; Pintus, G.; Tadolini, B. Opioid peptide gene expression in the primary hereditary cardiomyopathy of the Syrian hamster. (2) Role of intracellular calcium loading. J. Biol. Chem. 1997, 272, 6693–6698. [Google Scholar] [CrossRef]
- Ventura, C.; Pintus, G. Opioid peptide gene expression in the primary hereditary cardiomyopathy of the Syrian hamster. (3) Autocrine stimulation of prodynorphin gene expression by dynorphin B. J. Biol. Chem. 1997, 272, 6699–6705. [Google Scholar] [CrossRef]
- Maioli, M.; Asara, Y.; Pintus, A.; Ninniri, S.; Bettuzzi, S.; Scaltriti, M.; Galimi, F.; Ventura, C. Creating prodynorphin-expressing stem cells alerted for a high-throughput of cardiogenic commitment. Regen. Med. 2007, 2, 193–202. [Google Scholar] [CrossRef]
- Ventura, C.; Zinellu, E.; Maninchedda, E.; Fadda, M.; Maioli, M. Protein kinase C signaling transduces endorphin-primed cardiogenesis in GTR1 embryonic stem cells. Circ. Res. 2003, 92, 617–622. [Google Scholar] [CrossRef]
- Ventura, C.; Zinellu, E.; Maninchedda, E.; Maioli, M. Dynorphin B is an agonist of nuclear opioid receptors coupling nuclear protein kinase C activation to the transcription of cardiogenic genes in GTR1 embryonic stem cells. Circ. Res. 2003, 92, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Feridooni, T.; Pasumarthi, K.B.S. Fractionation of embryonic cardiac progenitor cells and evaluation of their differentiation potential. Differentiation 2019, 105, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, N.; Diehl, N.; Yaswen, L.; Brennan, M.B.; Hochgeschwender, U. Generation of dynorphin knockout mice. Brain Res. Mol. Brain Res. 2001, 86, 70–75. [Google Scholar] [CrossRef]
- Baldock, P.A.; Driessler, F.; Lin, S.; Wong, I.P.; Shi, Y.; Yulyaningsih, E.; Castillo, L.; Janmaat, S.; Enriquez, R.F.; Zengin, A.; et al. The endogenous opioid dynorphin is required for normal bone homeostasis in mice. Neuropeptides 2012, 46, 383–394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, L.; Zhang, S.; Shkhyan, R.; Lee, S.; Gullo, F.; Eliasberg, C.D.; Petrigliano, F.A.; Ba, K.; Wang, J.; Lin, Y.; et al. Kappa opioid receptor signaling protects cartilage tissue against posttraumatic degeneration. JCI Insight 2017, 2, e88553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamamizu, K.; Furuta, S.; Katayama, S.; Narita, M.; Kuzumaki, N.; Imai, S.; Nagase, H.; Suzuki, T.; Narita, M.; Yamashita, J.K. The κ opioid system regulates endothelial cell differentiation and pathfinding in vascular development. Blood 2011, 118, 775–785. [Google Scholar] [CrossRef]
- Nguyen, A.D.; Slack, K.; Schwarzer, C.; Lee, N.J.; Boey, D.; Macia, L.; Yulyaningsih, E.; Enriquez, R.F.; Zhang, L.; Lin, S.; et al. Double deletion of orexigenic neuropeptide Y and dynorphin results in paradoxical obesity in mice. Neuropeptides 2014, 48, 143–151. [Google Scholar] [CrossRef] [Green Version]
- Tsai, N.P.; Tsui, Y.C.; Pintar, J.E.; Loh, H.H.; Wei, L.N. Kappa opioid receptor contributes to EGF-stimulated neurite extension in development. Proc. Natl. Acad. Sci. USA 2010, 107, 3216–3221. [Google Scholar] [CrossRef] [Green Version]
- McLaughlin, P.J. Exposure to the opioid antagonist naltrexone throughout gestation alters postnatal heart development. Biol. Neonate 2002, 82, 207–216. [Google Scholar] [CrossRef]
- Mel’nikova, N.P.; Timoshin, S.S.; Tsygankov, V.I.; Murzina, N.B.; Obukhova, G.G.; Bespalov, Z.D. Dynorphin administered to newborn rats modulates morphogenesis of the heart. Bull. Exp. Biol. Med. 2000, 129, 527–529. [Google Scholar] [CrossRef]
- Ventura, C.; Maioli, M.; Asara, Y.; Santoni, D.; Scarlata, I.; Cantoni, S.; Perbellini, A. Butyric and retinoic mixed ester of hyaluronan. A novel differentiating glycoconjugate affording a high throughput of cardiogenesis in embryonic stem cells. J. Biol. Chem. 2004, 279, 23574–23579. [Google Scholar] [CrossRef]
- Wheatley, S.C.; Isacke, C.M.; Crossley, P.H. Restricted expression of the hyaluronan receptor, CD44, during postimplantation mouse embryogenesis suggests key roles in tissue formation and patterning. Development 1993, 119, 295–306. [Google Scholar] [PubMed]
- Camenisch, T.D.; Spicer, A.P.; Brehm-Gibson, T.; Biesterfeldt, J.; Augustine, M.L.; Calabro, A., Jr.; Kubalak, S.; Klewer, S.E.; McDonald, J.A. Disruption of hyaluronan synthase-2 abrogates normal cardiac morphogenesis and hyaluronan-mediated transformation of epithelium to mesenchyme. J. Clin. Investig. 2000, 106, 349–360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Takahashi, Y.; Li, L.; Kamiryo, M.; Asteriou, T.; Moustakas, A.; Yamashita, H.; Heldin, P. Hyaluronan fragments induce endothelial cell differentiation in a CD44- and CXCL1/GRO1-dependent manner. J. Biol. Chem. 2005, 280, 24195–24204. [Google Scholar] [CrossRef] [PubMed]
- Savani, R.C.; Cao, G.; Pooler, P.M.; Zaman, A.; Zhou, Z.; DeLisser, H.M. Differential involvement of the hyaluronan (HA) receptors CD44 and receptor for HA-mediated motility in endothelial cell function and angiogenesis. J. Biol. Chem. 2001, 276, 36770–36778. [Google Scholar] [CrossRef] [PubMed]
- Tammi, R.; Rilla, K.; Pienimaki, J.P.; MacCallum, D.K.; Hogg, M.; Luukkonen, M.; Hascall, V.C.; Tammi, M. Hyaluronan enters keratinocytes by a novel endocytic route for catabolism. J. Biol. Chem. 2001, 276, 35111–35122. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, M.; Meenakshi, J.; Goswami, S.K.; Datta, K. Hyaluronan binding protein 1 (HABP1)/C1QBP/p32 is an endogenous substrate for MAP kinase and is translocated to the nucleus upon mitogenic stimulation. Biochem. Biophys. Res. Commun. 2002, 291, 829–837. [Google Scholar] [CrossRef]
- Grammatikakis, N.; Grammatikakis, A.; Yoneda, M.; Yu, Q.; Banerjee, S.D.; Toole, B.P. A novel glycosaminoglycan-binding protein is the vertebrate homologue of the cell cycle control protein, Cdc37. J. Biol. Chem. 1995, 270, 16198–16205. [Google Scholar] [CrossRef]
- Wolffe, A.P.; Pruss, D. Targeting chromatin disruption: Transcription regulators that acetylate histones. Cell 1996, 84, 817–819. [Google Scholar] [CrossRef]
- Illi, B.; Scopece, A.; Nanni, S.; Farsetti, A.; Morgante, L.; Biglioli, P.; Capogrossi, M.C.; Gaetano, C. Epigenetic histone modification and cardiovascular lineage programming in mouse embryonic stem cells exposed to laminar shear stress. Circ. Res. 2005, 96, 501–508. [Google Scholar] [CrossRef]
- Kastner, P.; Grondona, J.M.; Mark, M.; Gansmuller, A.; LeMeur, M.; Decimo, D.; Vonesch, J.L.; Dollé, P.; Chambon, P. Genetic analysis of RXR alpha developmental function: Convergence of RXR and RAR signaling pathways in heart and eye morphogenesis. Cell 1994, 78, 987–1003. [Google Scholar] [CrossRef]
- Sucov, H.M.; Dyson, E.; Gumeringer, C.L.; Price, J.; Chien, K.R.; Evans, R.M. RXR alpha mutant mice establish a genetic basis for vitamin A signaling in heart morphogenesis. Genes Dev. 1994, 8, 1007–1018. [Google Scholar] [CrossRef] [PubMed]
- Wobus, A.M.; Kaomei, G.; Shan, J.; Wellner, M.C.; Rohwedel, J.; Ji, G.; Fleischmann, B.; Katus, H.A.; Hescheler, J.; Franz, W.M. Retinoic acid accelerates embryonic stem cell-derived cardiac differentiation and enhances development of ventricular cardiomyocytes. J. Mol. Cell. Cardiol. 1997, 29, 1525–1539. [Google Scholar] [CrossRef] [PubMed]
- Lai, L.; Bohnsack, B.L.; Niederreither, K.; Hirschi, K.K. Retinoic acid regulates endothelial cell proliferation during vasculogenesis. Development 2003, 130, 6465–6474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ventura, C.; Cantoni, S.; Bianchi, F.; Lionetti, V.; Cavallini, C.; Scarlata, I.; Foroni, L.; Maioli, M.; Bonsi, L.; Alviano, F.; et al. Hyaluronan mixed esters of butyric and retinoic acid drive cardiac and endothelial fate in term placenta human mesenchymal stem cells and enhance cardiac repair in infarcted rat hearts. J. Biol. Chem. 2007, 282, 14243–14252. [Google Scholar] [CrossRef]
- Ventura, C. Multi-source hMSCs, including those that had been isolated from the bone marrow, the dental pulp and term placenta, HBR lack any effect on the expression of neurogenin 1, coding for a neurogenic transcription factor; Department of Experimental, Diagnostic and Specialty Medicine (DIMES), University of Bologna: Bologna, Italy, 2007. [Google Scholar]
- Simioniuc, A.; Campan, M.; Lionetti, V.; Marinelli, M.; Aquaro, G.D.; Cavallini, C.; Valente, S.; Di Silvestre, D.; Cantoni, S.; Bernini, F.; et al. Placental stem cells pre-treated with a hyaluronan mixed ester of butyric and retinoic acid to cure infarcted pig hearts: A multimodal study. Cardiovasc. Res. 2011, 90, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Lionetti, V.; Cantoni, S.; Cavallini, C.; Bianchi, F.; Valente, S.; Frascari, I.; Olivi, E.; Aquaro, G.D.; Bonavita, F.; Scarlata, I.; et al. Hyaluronan mixed esters of butyric and retinoic acid affording myocardial survival and repair without stem cell transplantation. J. Biol. Chem. 2010, 285, 9949–9961. [Google Scholar] [CrossRef]
- Maioli, M.; Santaniello, S.; Montella, A.; Bandiera, P.; Cantoni, S.; Cavallini, C.; Bianchi, F.; Lionetti, V.; Rizzolio, F.; Marchesi, I.; et al. Hyaluronan esters drive Smad gene expression and signaling enhancing cardiogenesis in mouse embryonic and human mesenchymal stem cells. PLoS ONE 2010, 5, e15151. [Google Scholar] [CrossRef]
- Maioli, M.; Contini, G.; Santaniello, S.; Bandiera, P.; Pigliaru, G.; Sanna, R.; Rinaldi, S.; Delitala, A.P.; Montella, A.; Bagella, L.; et al. Amniotic fluid stem cells morph into a cardiovascular lineage: Analysis of a chemically induced cardiac and vascular commitment. Drug Des. Dev. Ther. 2013, 7, 1063–1073. [Google Scholar] [CrossRef]
- Cadossi, R.; Bersani, F.; Cossarizza, A.; Zucchini, P.; Emilia, G.; Torelli, G.; Franceschi, C. Lymphocytes and low-frequency electromagnetic fields. FASEB J. 1992, 6, 2667–2674. [Google Scholar]
- Cossarizza, A.; Monti, D.; Bersani, F.; Cantini, M.; Cadossi, R.; Sacchi, A.; Franceschi, C. Extremely low frequency pulsed electromagnetic fields increase cell proliferation in lymphocytes from young and aged subjects. Biochem. Biophys. Res. Commun. 1989, 160, 692–698. [Google Scholar] [CrossRef] [PubMed]
- Cossarizza, A.; Monti, D.; Bersani, F.; Paganelli, R.; Montagnani, G.; Cadossi, R.; Cantini, M.; Franceschi, C. Extremely low frequency pulsed electromagnetic fields increase interleukin-2 (IL-2) utilization and IL-2 receptor expression in mitogen-stimulated human lymphocytes from old subjects. FEBS Lett. 1989, 248, 141–144. [Google Scholar] [CrossRef] [Green Version]
- Thomas, A.W.; Kavaliers, M.; Prato, F.S.; Ossenkopp, K.P. Pulsed magnetic field induced “analgesia” in the land snail, Cepaea nemoralis, and the effects of mu, delta, and kappa opioid receptor agonists/antagonists. Peptides 1997, 18, 703–709. [Google Scholar] [CrossRef] [PubMed]
- Vorobyov, V.V.; Sosunov, E.A.; Kukushkin, N.I.; Lednev, V.V. Weak combined magnetic field affects basic and morphine-induced rat’s EEG. Brain Res. 1998, 781, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Rojavin, M.A.; Ziskin, M.C. Electromagnetic millimeter waves increase the duration of anaesthesia caused by ketamine and chloral hydrate in mice. Int. J. Radiat. Biol. 1997, 72, 475–480. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.; Carino, M.A.; Horita, A.; Guy, A.W. Effects of a 60 Hz magnetic field on central cholinergic systems of the rat. Bioelectromagnetics 1993, 14, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Lai, H.; Carino, M.A.; Horita, A.; Guy, A.W. Opioid receptor subtypes that mediate a microwave-induced decrease in central cholinergic activity in the rat. Bioelectromagnetics 1992, 13, 237–246. [Google Scholar] [CrossRef]
- Sandyk, R.; Iacono, R.P. Naltrexone attenuates the antiparkinsonian effects of picoTesla range magnetic fields. Int. J. Neurosci. 1994, 78, 111–122. [Google Scholar] [CrossRef]
- Ventura, C.; Maioli, M.; Pintus, G.; Gottardi, G.; Bersani, F. Elf-pulsed magnetic fields modulate opioid peptide gene expression in myocardial cells. Cardiovasc. Res. 2000, 45, 1054–1064. [Google Scholar] [CrossRef]
- Ventura, C.; Maioli, M.; Asara, Y.; Santoni, D.; Mesirca, P.; Remondini, D.; Bersani, F. Turning on stem cell cardiogenesis with extremely low frequency magnetic fields. FASEB J. 2005, 19, 155–157. [Google Scholar] [CrossRef]
- Maioli, M.; Rinaldi, S.; Santaniello, S.; Castagna, A.; Pigliaru, G.; Gualini, S.; Fontani, V.; Ventura, C. Radiofrequency energy loop primes cardiac, neuronal, and skeletal muscle differentiation in mouse embryonic stem cells: A new tool for improving tissue regeneration. Cell Transplant. 2012, 21, 1225–1233. [Google Scholar] [CrossRef] [PubMed]
- Maioli, M.; Rinaldi, S.; Migheli, R.; Pigliaru, G.; Rocchitta, G.; Santaniello, S.; Basoli, V.; Castagna, A.; Fontani, V.; Ventura, C.; et al. Neurological morphofunctional differentiation induced by REAC technology in PC12. A neuro protective model for Parkinson’s disease. Sci. Rep. 2015, 15, 10439. [Google Scholar] [CrossRef] [PubMed]
- Collodel, G.; Fioravanti, A.; Pascarelli, N.A.; Lamboglia, A.; Fontani, V.; Maioli, M.; Santaniello, S.; Pigliaru, G.; Castagna, A.; Moretti, E.; et al. Effects of regenerative radioelectric asymmetric conveyer treatment on human normal and osteoarthritic chondrocytes exposed to il-1β. A biochemical and morphological study. Clin. Interv. Aging 2013, 8, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Maioli, M.; Rinaldi, S.; Santaniello, S.; Castagna, A.; Pigliaru, G.; Delitala, A.; Bianchi, F.; Tremolada, C.; Fontani, V.; Ventura, C. Radioelectric asymmetric conveyed fields and human adipose-derived stem cells obtained with a nonenzymatic method and device: A novel approach to multipotency. Cell Transplant. 2014, 23, 1489–1500. [Google Scholar] [CrossRef] [PubMed]
- Maioli, M.; Rinaldi, S.; Santaniello, S.; Castagna, A.; Pigliaru, G.; Gualini, S.; Cavallini, C.; Fontani, V.; Ventura, C. Radio electric conveyed fields directly reprogram human dermal skin fibroblasts toward cardiac, neuronal, and skeletal muscle-like lineages. Cell Transplant. 2013, 22, 1227–1235. [Google Scholar] [CrossRef]
- Rinaldi, S.; Maioli, M.; Santaniello, S.; Castagna, A.; Pigliaru, G.; Gualini, S.; Margotti, M.L.; Carta, A.; Fontani, V.; Ventura, C. Regenerative treatment using a radioelectric asymmetric conveyor as a novel tool in antiaging medicine: An in vitro beta-galactosidase study. Clin. Interv. Aging 2012, 7, 191–194. [Google Scholar] [CrossRef]
- Rinaldi, S.; Maioli, M.; Pigliaru, G.; Castagna, A.; Santaniello, S.; Basoli, V.; Fontani, V.; Ventura, C. Stem cell senescence. Effects of REAC technology on telomerase-independent and telomerase-dependent pathways. Sci. Rep. 2014, 4, 6373. [Google Scholar] [CrossRef] [Green Version]
- Maioli, M.; Rinaldi, S.; Santaniello, S.; Castagna, A.; Pigliaru, G.; Delitala, A.; Lotti Margotti, M.; Bagella, L.; Fontani, V.; Ventura, C. Anti-senescence efficacy of radio-electric asymmetric conveyer technology. Age (Dordr) 2014, 36, 9–20. [Google Scholar] [CrossRef]
- Maioli, M.; Rinaldi, S.; Pigliaru, G.; Santaniello, S.; Basoli, V.; Castagna, A.; Fontani, V.; Ventura, C. REAC technology and hyaluron synthase 2, an interesting network to slow down stem cell senescence. Sci. Rep. 2016, 6, 28682. [Google Scholar] [CrossRef] [Green Version]
| Stimulus | Species | Cell Type | Biological Effects | Ref. |
|---|---|---|---|---|
| KCl1 60 mM (for 4 or 24 h of treatment) | Rat | Ventricular Cardiomyocytes | Increased expression of the prodynorphin gene Increase of dynorphin B At the intracellular level and in the culture medium | [52] |
| PMA2 100 nM (from 1 to 24 h of treatment) | Rat | Cardiomyocytes and nuclei isolated from cardiomyocytes | Activation of the nuclear protein kinases C-δ and -ε Subsequent increased expression of the prodynorphin gene Increase of dynorphin B At the intracellular level and in the culture medium | [53] |
| DMSO3 1% (for 2 or 4 days of treatment) | Mouse | P19 ESCs4 | Increased expression of the prodynorphin gene Following an increase of GATA-4 and Nkx-2.5 expression Increase of dynorphin B At the intracellular level and in the culture medium Counteracted by opioid receptor antagonism or targeted inhibition of prodynorphin gene expression | [55] |
| Stimulus | Species | Cell Type | Biological Effects | Ref. |
|---|---|---|---|---|
| HBR1 0.75 mg/mL for five days of treatment | Mouse | ESCs | Overexpression of the prodynorphin gene Increase in GATA-4 and Nkx-2.5 transcription Increase of dynorphin B at the intracellular level and in the culture medium Increase of the number of spontaneously beating myocardial cells Expression of α-MHC and MLC-2V in spontaneously beating cardiomyocytes Absence of effect on skeletal myogenic or neurogenic markers | [71] |
| HBR 1.5 mg/mL for seven days of treatment (fourteen days for analyses of cardiac markers) | Human | MSCs2 isolated from different sources, including the bone marrow, dental pulp, and FM6 | Improvement of VEGF3, KDR4, and HGF5 gene expression Increase of VEGF and HGF secretion Increase in GATA-4 and Nkx-2.5 transcription Expression of cardiac markers Absence of effect on skeletal myogenic or neurogenic markers | [85] |
| Transplantation of HBR-preconditioned FMhMSCs (1.5 mg/mL for 14 days) in animals affected by acute myocardial infarction | Rat and Pig | Cardiomyocytes | Enhancement of myocardial vascularization and contractility Reduction of the infarct scar size | [85,87] |
| HBR injection (0.2 mg/100 g of rat weight) in heart affected by acute myocardial infarction | Rat | Cardiomyocytes | Presence of cardiovascular repair, reducing the infarct size and affording a full recovery of myocardial performance Increase of capillary density, and decrease of the number of apoptotic cardiomyocytes Increase of histone H4 acetylation at the level of myocardial nuclei | [88] |
| HBR 2 mg/mL (from 1 to 10 days of treatment) | Human | MSCs | Enhancement of Smad1, 3, and 4 gene and protein expression Downregulation of Smad7 Increase of transcription rates of Smad4 in nuclei isolated from HBR treated cells correlated to Nkx-2.5 expression | [89,90] |
| Stimulus | Species | Cell Type | Biological Effects | Ref. |
|---|---|---|---|---|
| ELF-MF1 of 50 Hz, 0.8 mTRSM (for 4 h of treatment) | rat | Nuclei isolated from ventricular cardiac myocytes or intact cells | Increase in prodynorphin gene transcription Increase in intracellular levels of dynorphin B Enhancement in dynorphin B secretion in the culture medium | [100] |
| ELF-MF of 50 Hz, 0.8 mTRSM(for three and ten days of treatment) | mouse | ESCs | Induction of GATA-4 and Nkx-2.5 gene expression Increase in the number of spontaneously beating ESC-derived cardiomyocytes Increase in the transcription rate of the prodynorphin gene Increase in intracellular levels of dynorphin B Enhancement in dynorphin B secretion in the culture medium | [101] |
| Radiofrequency of 2.4 GHz emitted by REAC2 (for treatment details see Ref.) | mouse and human | ESCs and AD-MSCs | Modulation in the transcription of stemness genes Increase in the yield of cells differentiating into cardiac, neural, skeletal and myogenic lineages Association of cardiac differentiation with GATA-4, Nkx-2.5 and prodynorphin gene transcription | [102,103,104,105] |
| Radiofrequency of 2.4 GHz emitted by REAC (from 2 to 72 h of treatment and 72 h of treatment + four or seven days of recovery) | human | Dermal skin fibroblasts | Early transcriptional increase of Oct4, Sox2, c-Myc, Nanog, and Klf4, stemness genes followed by their downregulation Increase in the yield of cells differentiating into cardiac, neural and myogenic lineages Association of cardiac differentiation with: Expression of Mef2c, Tbx5, GATA-4 and Nkx-2.5 genes; expression of MHC and SA proteins; increase in the expression of prodynorphin gene | [106] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canaider, S.; Facchin, F.; Tassinari, R.; Cavallini, C.; Olivi, E.; Taglioli, V.; Zannini, C.; Bianconi, E.; Maioli, M.; Ventura, C. Intracrine Endorphinergic Systems in Modulation of Myocardial Differentiation. Int. J. Mol. Sci. 2019, 20, 5175. https://doi.org/10.3390/ijms20205175
Canaider S, Facchin F, Tassinari R, Cavallini C, Olivi E, Taglioli V, Zannini C, Bianconi E, Maioli M, Ventura C. Intracrine Endorphinergic Systems in Modulation of Myocardial Differentiation. International Journal of Molecular Sciences. 2019; 20(20):5175. https://doi.org/10.3390/ijms20205175
Chicago/Turabian StyleCanaider, Silvia, Federica Facchin, Riccardo Tassinari, Claudia Cavallini, Elena Olivi, Valentina Taglioli, Chiara Zannini, Eva Bianconi, Margherita Maioli, and Carlo Ventura. 2019. "Intracrine Endorphinergic Systems in Modulation of Myocardial Differentiation" International Journal of Molecular Sciences 20, no. 20: 5175. https://doi.org/10.3390/ijms20205175







