Adhesion, Proliferation and Migration of NIH/3T3 Cells on Modified Polyaniline Surfaces
Abstract
:1. Introduction
2. Results and Discussion
2.1. Surface Energy
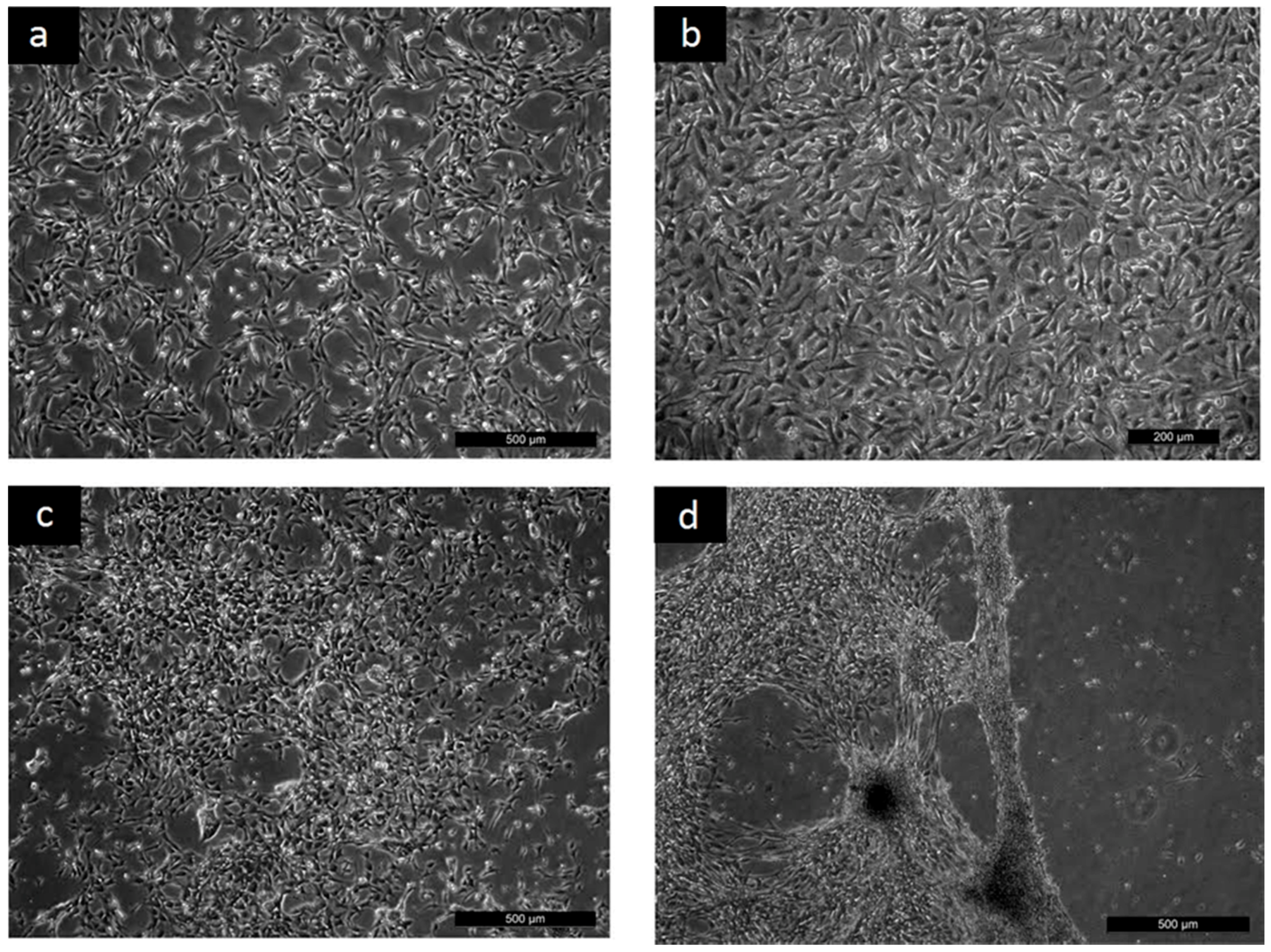
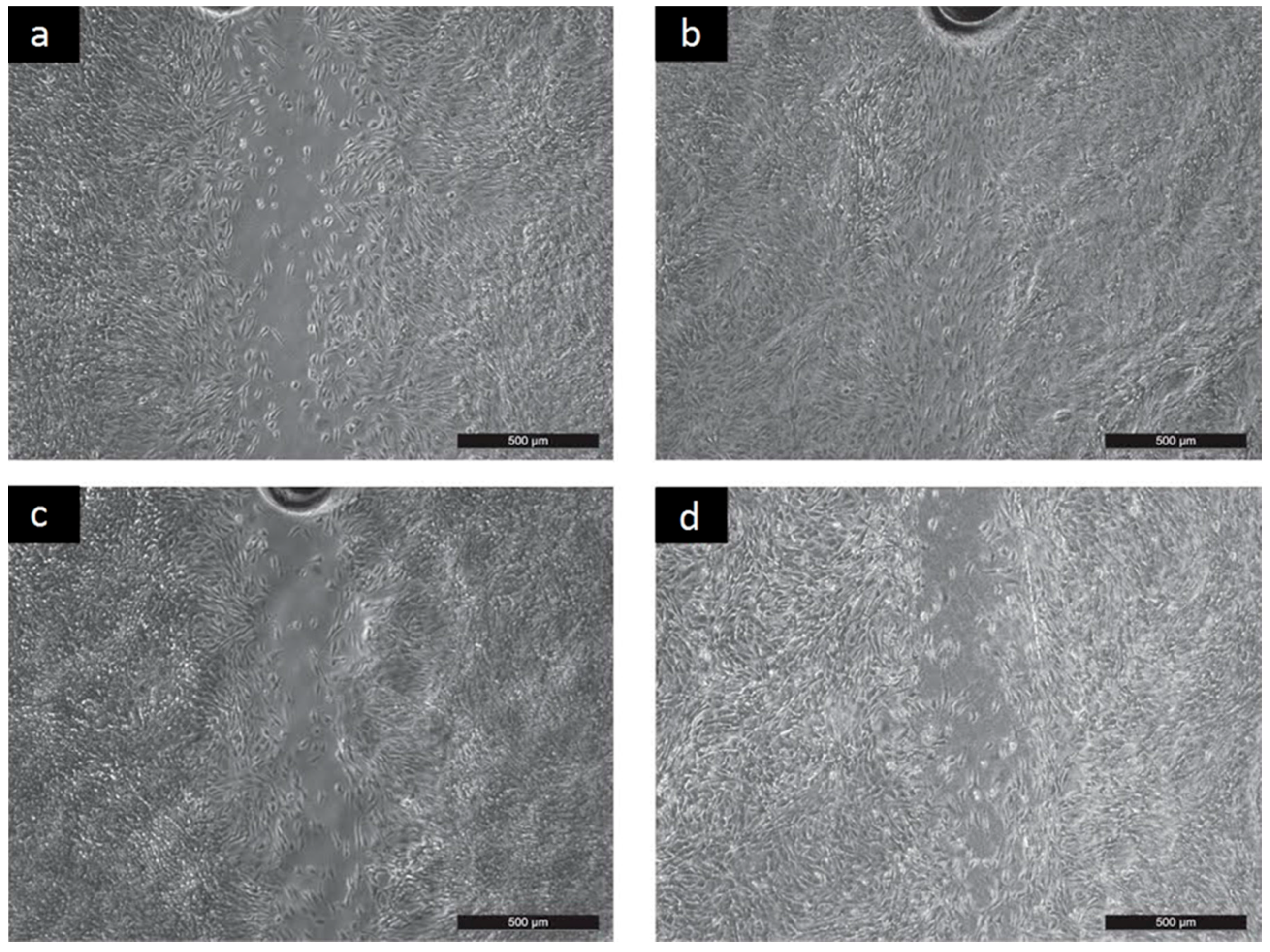
2.2. Cyto-Compatibility
3. Materials and Methods
3.1. Preparation of Polyaniline Films
3.2. Surface Energy
3.3. Cyto-Compatibility
- To reveal the ability of cells to adhere to the surfaces, the cells were seeded on reference culture dishes (TPP, Trasadingen, Switzerland) and the studied polymer films at a concentration of 1 × 107 cells·mL−1. After one hour, the cells were gently rinsed and micrographs were taken.
- Cell proliferation and morphology were evaluated on cells that had been seeded at an initial concentration of 1 × 105 cells·mL−1 and cultivated.
- Cell migration was determined by the scratch assay according to Liang et al. [22] with modification. The scratch assay was created in a confluent cell monolayer. After 48 h had passed, micrographs were captured with an Olympus inverted fluorescent microscope (Olympus, IX51, Tokyo, Japan) equipped with a digital color camera (Leica DFC480, Wetzlar, Germany).
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cate, A.T.; Gaspar, C.H.; Virtanen, H.L.K.; Stevens, R.S.A.; Koldeweij, R.B.J.; Olkkonen, J.T.; Rentrop, C.H.A.; Smolander, M.H. Printed electronic switch on flexible substrates using printed microcapsules. J. Mater. Sci. 2014, 49, 5831–5837. [Google Scholar] [CrossRef]
- Zhybak, M.; Beni, V.; Vagin, M.Y.; Dempsey, E.; Turner, A.P.F.; Korpan, Y. Creatinine and urea biosensors based on a novel ammonium ion-selective copper-polyaniline nano-composite. Biosens. Bioelectron. 2016, 77, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Qazi, T.H.; Rai, R.; Boccaccini, A.R. Tissue engineering of electrically responsive tissues using polyaniline based polymers: A review. Biomaterials 2014, 35, 9068–9086. [Google Scholar] [CrossRef] [PubMed]
- Ricotti, L.; Menciassi, A. Bio-hybrid muscle cell-based actuators. Biomed. Microdevices 2012, 14, 987–998. [Google Scholar] [CrossRef] [PubMed]
- Juarez-Hernandez, L.J.; Comella, N.; Pasquardini, L.; Battistoni, S.; Vidalino, L.; Vanzetti, L.; Caponi, S.; Dalla Serra, M.; Iannotta, S.; Pederzolli, C.; et al. Bio-hybrid interfaces to study neuromorphic functionalities: New multidisciplinary evidences of cell viability on poly(anyline) (PANI), a semiconductor polymer with memristive properties. Biophys. Chem. 2016, 208, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Bidez, P.R.; Li, S.X.; MacDiarmid, A.G.; Venancio, E.C.; Wei, Y.; Lelkes, P.I. Polyaniline, an electroactive polymer, supports adhesion and proliferation of cardiac myoblasts. J. Biomater. Sci. Polym. Ed. 2006, 17, 199–212. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Ji, L.; Li, D.; Wang, J. Characterization of nanostructure and cell compatibility of polyaniline films with different dopant acids. J. Phys. Chem. B 2008, 112, 2671–2677. [Google Scholar] [CrossRef] [PubMed]
- Bober, P.; Humpolicek, P.; Pachernik, J.; Stejskal, J.; Lindfors, T. Conducting polyaniline based cell culture substrate for embryonic stem cells and embryoid bodies. RSC Adv. 2015, 5, 50328–50335. [Google Scholar] [CrossRef]
- Prabhakar, P.K.; Raj, S.; Anuradha, P.R.; Sawant, S.N.; Doble, M. Biocompatibility studies on polyaniline and polyaniline-silver nanoparticle coated polyurethane composite. Colloids Surf. B Biointerfaces 2011, 86, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Chen, J.; Yang, J.; Xue, Q.; Miele, P. Fabrication of free-standing, electrochemically active, and biocompatible graphene oxide-polyaniline and graphene-polyaniline hybrid papers. ACS Appl. Mater. Interfaces 2010, 2, 2521–2529. [Google Scholar] [CrossRef] [PubMed]
- Humpolicek, P.; Kucekova, Z.; Kasparkova, V.; Pelkova, J.; Modic, M.; Junkar, I.; Trchova, M.; Bober, P.; Stejskal, J.; Lehocky, M. Blood coagulation and platelet adhesion on polyaniline films. Colloids Surf. B Biointerfaces 2015, 133, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Sivaraman, K.M.; Oezkale, B.; Ergeneman, O.; Luhmann, T.; Fortunato, G.; Zeeshan, M.A.; Nelson, B.J.; Pane, S. Redox cycling for passive modification of polypyrrole surface properties: Effects on cell adhesion and proliferation. Adv. Healthc. Mater. 2013, 2, 591–598. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Qi, H.; Wang, S.; Feng, L.; Ji, Y.; Tao, L.; Li, S.; Wei, Y. Cellular responses of aniline oligomers: A preliminary study. Toxicol. Res. 2012, 1, 201–205. [Google Scholar] [CrossRef]
- Humpolicek, P.; Radaszkiewicz, K.A.; Kasparkova, V.; Stejskal, J.; Trchova, M.; Kucekova, Z.; Vicarova, H.; Pachernik, J.; Lehocky, M.; Minarik, A. Stem cell differentiation on conducting polyaniline. RSC Adv. 2015, 5, 68796–68805. [Google Scholar] [CrossRef]
- Kasparkova, V.; Humpolicek, P.; Stejskal, J.; Kopecka, J.; Kucekova, Z.; Moucka, R. Conductivity, impurity profile, and cytotoxicity of solvent-extracted polyaniline. Polym. Adv. Technol. 2016, 27, 156–161. [Google Scholar] [CrossRef]
- Kucekova, Z.; Humpolicek, P.; Kasparkova, V.; Perecko, T.; Lehocky, M.; Hauerlandova, I.; Saha, P.; Stejskal, J. Colloidal polyaniline dispersions: Antibacterial activity, cytotoxicity and neutrophil oxidative burst. Colloids Surf. B: Biointerfaces 2014, 116, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Stejskal, J.; Hajna, M.; Kasparkova, V.; Humpolicek, P.; Zhigunov, A.; Trchova, M. Purification of a conducting polymer, polyaniline, for biomedical applications. Synth. Met. 2014, 195, 286–293. [Google Scholar] [CrossRef]
- Stejskal, J.; Sapurina, I. Polyaniline: Thin films and colloidal dispersions—(IUPAC Technical Report). Pure Appl. Chem. 2005, 77, 815–826. [Google Scholar] [CrossRef]
- Bayer, C.L.; Trenchard, I.J.; Peppas, N.A. Analyzing polyaniline-poly (2-acrylamido-2-methylpropane sulfonic acid) biocompatibility with 3T3 fibroblasts. J. Biomater. Sci. Polym. Ed. 2010, 21, 623–634. [Google Scholar] [CrossRef] [PubMed]
- Stejskal, J.; Sapurina, I.; Prokes, J.; Zemek, J. In-situ polymerized polyaniline films. Synth. Met. 1999, 105, 195–202. [Google Scholar] [CrossRef]
- Yoo, J.E.; Cross, J.L.; Bucholz, T.L.; Lee, K.S.; Espe, M.P.; Loo, Y. Improving the electrical conductivity of polymer acid-doped polyaniline by controlling the template molecular weight. J. Mater. Chem. 2007, 17, 1268–1275. [Google Scholar] [CrossRef]
- Liang, C.; Park, A.Y.; Guan, J. In vitro scratch assay: A convenient and inexpensive method for analysis of cell migration in vitro. Nat. Protoc. 2007, 2, 329–333. [Google Scholar] [CrossRef] [PubMed]

| Sample | Surface Energy Components (mN·m−1) | |||
|---|---|---|---|---|
| γtot | γLW | γAB | γdif | |
| PANI-S | 52.54 * | 46.05 * | 6.49 * | 3.33 |
| PANI-B | 50.88 * | 46.54 * | 4.35 * | 1.67 |
| PANI-SULF | 52.13 | 44.97 | 7.17 | 2.92 |
| PANI-PT | 51.89 | 47.39 | 4.50 | 2.68 |
| PANI-PAMPSA-1:1 | 41.85 | 40.98 | 0.87 | 7.36 |
| PANI-PAMPSA-2:1 | 56.35 | 43.91 | 12.45 | 7.14 |
| Cells | 49.21 | 23.21 | 26.00 | - |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rejmontová, P.; Capáková, Z.; Mikušová, N.; Maráková, N.; Kašpárková, V.; Lehocký, M.; Humpolíček, P. Adhesion, Proliferation and Migration of NIH/3T3 Cells on Modified Polyaniline Surfaces. Int. J. Mol. Sci. 2016, 17, 1439. https://doi.org/10.3390/ijms17091439
Rejmontová P, Capáková Z, Mikušová N, Maráková N, Kašpárková V, Lehocký M, Humpolíček P. Adhesion, Proliferation and Migration of NIH/3T3 Cells on Modified Polyaniline Surfaces. International Journal of Molecular Sciences. 2016; 17(9):1439. https://doi.org/10.3390/ijms17091439
Chicago/Turabian StyleRejmontová, Petra, Zdenka Capáková, Nikola Mikušová, Nela Maráková, Věra Kašpárková, Marián Lehocký, and Petr Humpolíček. 2016. "Adhesion, Proliferation and Migration of NIH/3T3 Cells on Modified Polyaniline Surfaces" International Journal of Molecular Sciences 17, no. 9: 1439. https://doi.org/10.3390/ijms17091439









