Environmental Conditions Influence Induction of Key ABC-Transporter Genes Affecting Glyphosate Resistance Mechanism in Conyza canadensis
Abstract
:1. Introduction
2. Results
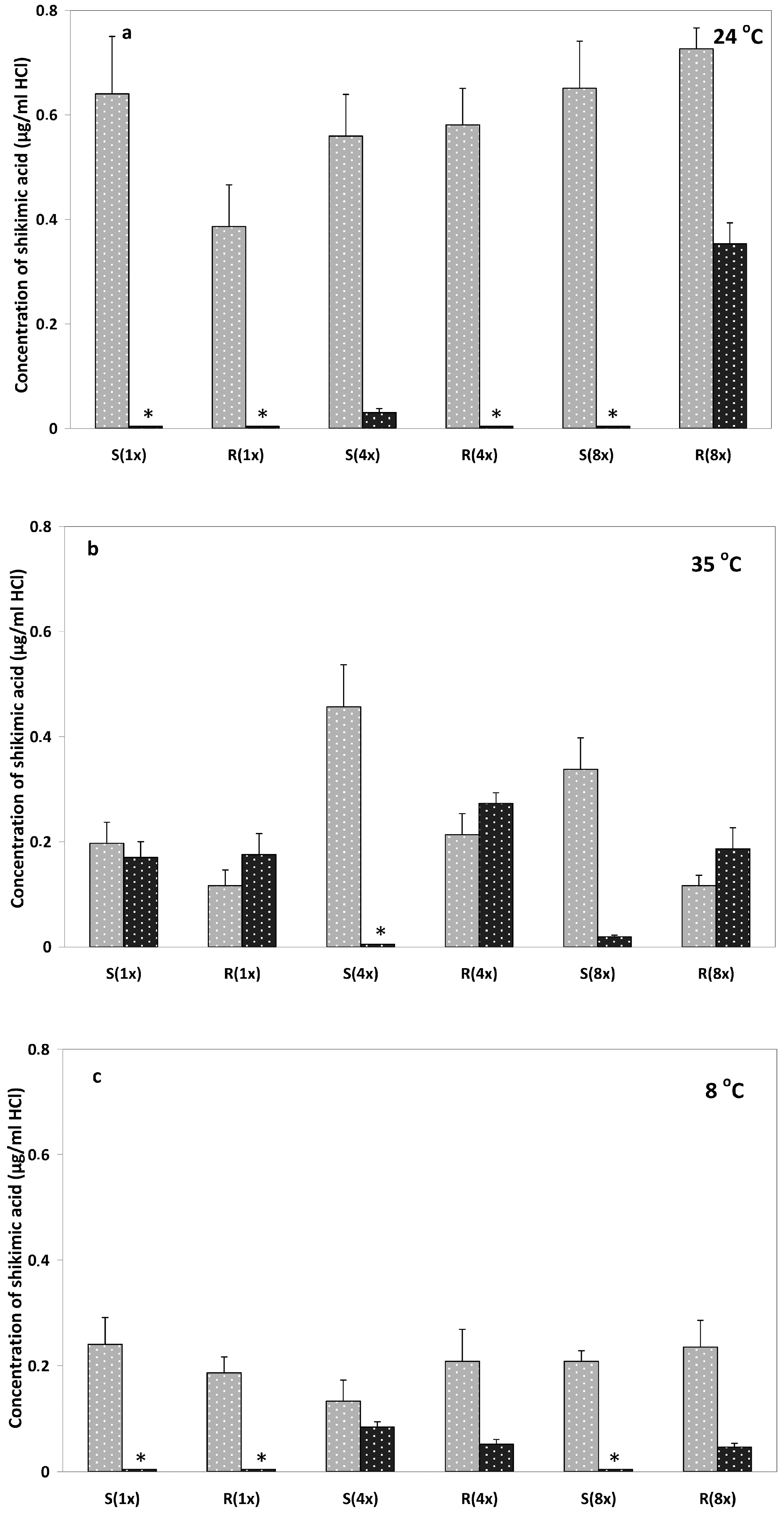
2.1. Shikimate Measurements
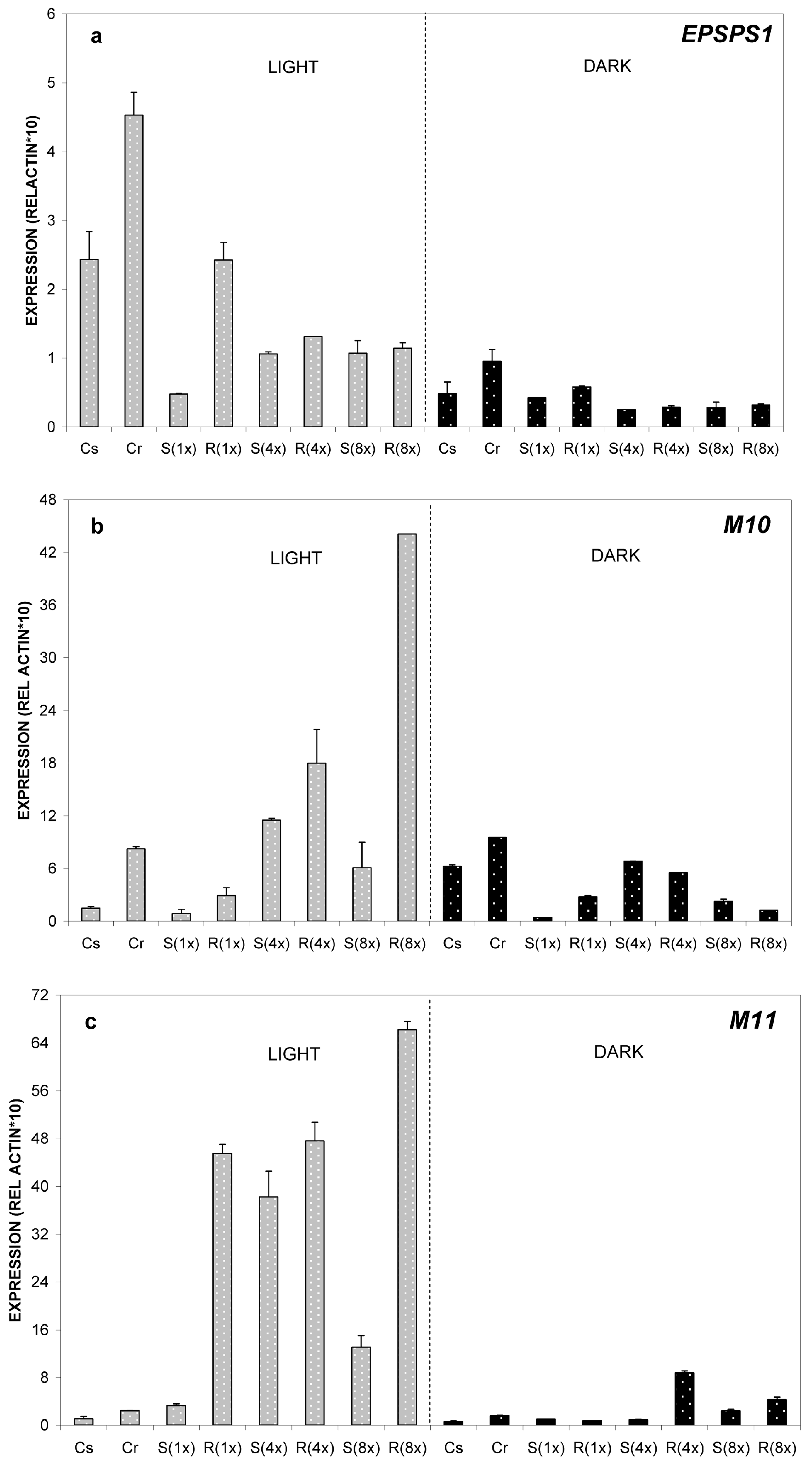
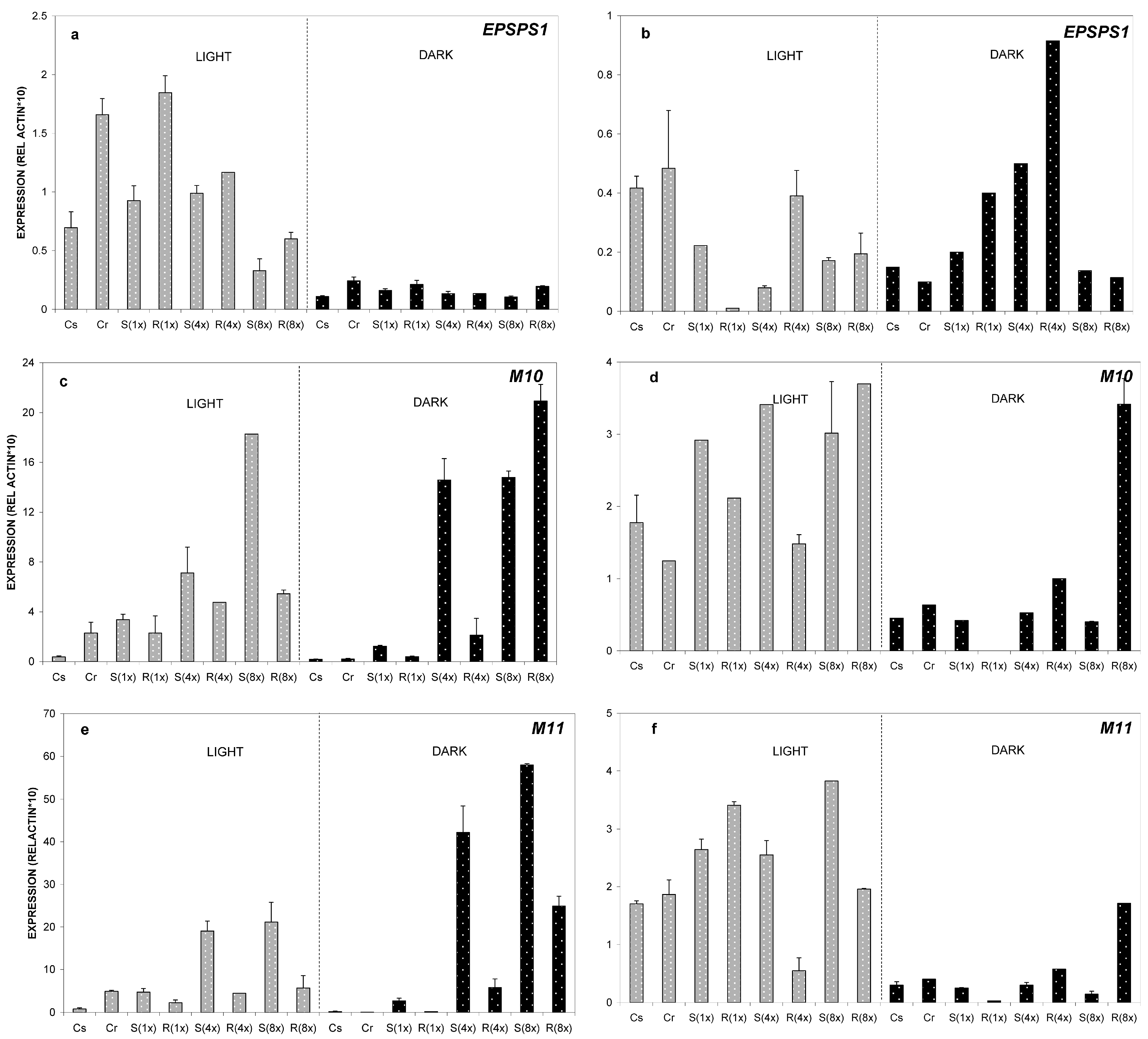
2.2. Expression Analysis of ABC-Transporter-Like Genes and EPSPS Gene
2.2.1. Relative Expression of M10, M11 and EPSPS Genes at 24 °C
2.2.2. Relative Expression of M10, M11 and EPSPS Genes at 35 °C
2.2.3. Relative Expression of M10, M11 and EPSPS Genes at 8 °C
3. Discussion
4. Experimental Section
4.1. Plant Material
4.2. Herbicide and Temperature Treatments
4.3. Shikimate Measurement
4.4. RNA Isolation, cDNA Synthesis and qRT-PCRexperiments
4.5. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Amrhein, N.; Deus, P.; Gehrke, P.; Steinrucken, H. The site of inhibition of the shikimate pathway by glyphosate. II. Interference of glyphosate with chorismate formation in vivo and in vitro. Plant Physiol. 1980, 66, 830–834. [Google Scholar] [CrossRef] [PubMed]
- Bromilow, R.H.; Chamberlain, K. The herbicide glyphosate and related molecules: Physicochemical and structural factors determining their mobility in phloem. Pest Manag. Sci. 2000, 56, 368–373. [Google Scholar] [CrossRef]
- Duke, S.O.; Powles, S.B. Glyphosate: A once-in-a-century herbicide. Pest Manag. Sci. 2008, 64, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Heap, I. Global perspective of herbicide-resistant weeds. Pest Manag. Sci. 2014, 70, 1306–1315. [Google Scholar] [CrossRef] [PubMed]
- Service, R.F. A growing threat down on the farm. Science 2007, 316, 1114–1117. [Google Scholar] [CrossRef] [PubMed]
- Hanson, B.D.; Shrestha, A.; Shaner, D.L. Distribution of glyphosate-resistant horseweed (Conyza canadensis) and relationship to cropping systems in the central valley of California. Weed Sci. 2009, 57, 48–53. [Google Scholar] [CrossRef]
- VanGessel, M.J. Glyphosate-resistant horseweed from Delaware. Weed Sci. 2001, 49, 703–705. [Google Scholar] [CrossRef]
- Chachalis, D.; Travlos, I.S. Glyphosate resistant weeds in Southern Europe: Current status, control strategies and future challenges. In Handbook of Herbicides: Biological Activity, Classification, and Health and Environmental Implications; Kobayashi, D., Watanabe, E., Eds.; Nova publishers: New York, NY, USA, 2014; pp. 175–191. [Google Scholar]
- Travlos, I.S.; Chachalis, D. Glyphosate-resistant hairy fleabane (Conyza bonariensis) reported in Greece. Weed Technol. 2010, 24, 569–573. [Google Scholar] [CrossRef]
- Travlos, I.S.; Chachalis, D. Assessment of glyphosate-resistant horseweed (Conyza canadensis L. Cronq.) and fleabane (Conyza albida Willd. ex Spreng) populations from perennial crops in Greece. Int. J. Plant Prod. 2013, 4, 665–676. [Google Scholar]
- Sammons, R.D.; Gaines, T.A. Glyphosate resistance: State of knowledge. Pest Manag. Sci. 2014, 70, 1367–1377. [Google Scholar] [CrossRef] [PubMed]
- Baerson, S.R.; Rodriguez, D.J.; Tran, M.; Feng, Y.; Biest, N.A.; Dill, G.M. Glyphosate-resistant goosegrass. Identification of a mutation in the target enzyme 5-enolpyruvylshikimate-3-phosphate synthase. Plant Physiol. 2002, 129, 1265–1275. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.H.; Wickneswari, R.; Salmijah, S.; Teng, Y.T.; Ismail, B.S. Gene polymorphisms in glyphosate-resistant and -susceptible biotypes of Eleusine indica from Malaysia. Weed Res. 2003, 43, 108–115. [Google Scholar] [CrossRef]
- Gaines, T.A.; Zhang, W.; Wang, D.; Bukun, B.; Chisholm, S.T.; Shaner, D.L.; Nissen, S.J.; Patzoldt, W.L.; Tranel, P.J.; Culpeper, A.S.; et al. Gene amplification confers glyphosate resistance in Amaranthus palmeri. Proc. Natl. Acad. Sci. USA 2010, 107, 1029–1034. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.C.C.; Tran, M.; Chiu, T.; Sammons, R.D.; Heck, G.R.; CaJacob, C.A. Investigations into glyphosate-resistant horseweed (Conyza canadensis): Retention, uptake, translocation, and metabolism. Weed Sci. 2004, 52, 498–505. [Google Scholar] [CrossRef]
- Vila-Aiub, M.M.; Balbi, M.C.; Distefano, A.J.; Fernandez, L.; Hopp, E.; Yu, Q.; Powles, S. Glyphosate resistance in perennial Sorghum halepense (Johnsongrass), endowed by reduced glyphosate translocation and leaf uptake. Pest Manag. Sci. 2012, 68, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; d'Avignon, D.A.; Ackerman, J.J.H.; Sammons, R.D. Rapid vacuolar sequestration: The horseweed glyphosate resistance mechanism. Pest Manag. Sci. 2010, 66, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Torralva, F.; Rojano-Delgado, A.M.; Luque de Castro, M.D.; Mülleder, N.; de Prado, R. Two non-target mechanisms are involved in glyphosate-resistant horseweed (Conyza canadensis L. Cronq.) biotypes. J. Plant Physiol. 2012, 169, 1673–1679. [Google Scholar] [CrossRef] [PubMed]
- Brabham, C.B.; Gerber, C.K.; Johnson, W.G. Fate of glyphosate-resistant giant ragweed (Ambrosia trifida) in the presence and absence of glyphosate. Weed Sci. 2011, 59, 506–511. [Google Scholar] [CrossRef]
- Tani, E.; Chachalis, D.; Travlos, I.S. A glyphosate resistance mechanism in Conyza canadensis involves synchronization of EPSPS and ABC-transporter genes. Plant Mol. Biol. Rep. 2015, 33. [Google Scholar] [CrossRef]
- Franz, J.E.; Mao, M.K.; Sikorski, J.A. Glyphosate: A Unique Global Herbicide; American Chemical Society: Washington, DC, USA, 1997; p. 653. [Google Scholar]
- Coupland, D. Influence of light, temperature and humidity on the translocation and activity of glyphosate in Elymus repens (= Agropyron repens). Weed Res. 1983, 23, 347–355. [Google Scholar] [CrossRef]
- Masiunas, J.B.; Weller, S.C. Glyphosate activity in potato (Solanum tuberosum) under different temperature regimes and light levels. Weed Sci. 1988, 36, 137–140. [Google Scholar]
- Ge, X.; d'Avignon, D.A.; Ackerman, J.J.; Duncan, B.; Spaur, M.B.; Sammons, R.D. Glyphosate-resistant horseweed made sensitive to glyphosate: low-temperature suppression of glyphosate vacuolar sequestration revealed by P-31 NMR. Pest Manag. Sci. 2011, 67, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- Yu, Q.; Huang, S.; Powles, S. Direct measurement of paraquat in leaf protoplasts indicates vacuolar paraquat sequestration as a resistance mechanism in Lolium rigidum. Pestic. Biochem. Physiol. 2010, 98, 104–109. [Google Scholar] [CrossRef]
- Sharkhuu, A.; Narasimhan, M.L.; Merzaban, J.S.; Bressan, R.A.; Weller, S.; Gehring, C. A red and far-red light receptor mutation confers resistance to the herbicide glyphosate. Plant J. 2014, 78, 916–926. [Google Scholar] [CrossRef] [PubMed]
- Chachalis, D.; Reddy, K.N. Factors affecting sprouting and glyphosate translocation in rootstocks of redvine (Brunnichia ovata) and trumpetcreeper (Campsis radicans). Weed Technol. 2005, 19, 141–147. [Google Scholar] [CrossRef]
- Hollander, H.; Amrhein, N. The site of the inhibition of the shikimate pathway by glyphosate. I. Inhibition by glyphosate of phenylpropanoid synthesis in buckwheat (Fagopyrum esculentum Moench). Plant Physiol. 1980, 66, 823–829. [Google Scholar] [PubMed]
- Nandula, V.K.; Reddy, K.N.; Rimando, A.M.; Duke, S.O.; Poston, D.H. Glyphosate-resistant and -susceptible soybean (Glycine max) and canola (Brassica napus) dose response and metabolism relationships with glyphosate. J. Agric. Food Chem. 2007, 55, 3540–3545. [Google Scholar] [CrossRef] [PubMed]
- Nol, N.; Tsikou, D.; Eid, M.; Livieratos, I.C.; Giannopolitis, C.N. Shikimate leaf disc assay for early detection of glyphosate resistance in Conyza canadensis and relative transcript levels of EPSPS and ABC transporter genes. Weed Res. 2012, 52, 233–241. [Google Scholar] [CrossRef]
- Dinelli, G.; Marotti, I.; Bonetti, A.; Catizone, P.; Urbano, J.M.; Barnes, J. Physiological and molecular bases of glyphosate resistance in Conyza bonariensis biotypes from Spain. Weed Res. 2008, 48, 257–265. [Google Scholar] [CrossRef]
- Dinelli, G.; Marotti, I.; Bonetti, A.; Minelli, M.; Catizone, P.; Barnes, J. Physiological and molecular insight on the mechanisms of resistance to glyphosate in Conyza canadensis (L.) Cronq.biotypes. Pestic. Biochem. Physiol. 2006, 86, 30–41. [Google Scholar] [CrossRef]
- Shaner, D.L.; Nadler-Hassar, T.; Henry, W.B.; Koger, C.H. A rapid in vivo shikimate accumulation assay with excised leaf discs. Weed Sci. 2005, 53, 769–774. [Google Scholar] [CrossRef]
- Peng, Y.; Abercrombie, L.L.; Yuan, J.S.; Riggins, C.W.; Sammons, R.D.; Tranel, P.J.; Stewart, C.N., Jr. Characterization of the horseweed (Conyza canadensis) transcriptome using GS-FLX 454 pyrosequencing and its application for expression analysis of candidate non-target herbicide resistance genes. Pest Manag. Sci. 2010, 66, 1053–1062. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tani, E.; Chachalis, D.; Travlos, I.S.; Bilalis, D. Environmental Conditions Influence Induction of Key ABC-Transporter Genes Affecting Glyphosate Resistance Mechanism in Conyza canadensis. Int. J. Mol. Sci. 2016, 17, 342. https://doi.org/10.3390/ijms17040342
Tani E, Chachalis D, Travlos IS, Bilalis D. Environmental Conditions Influence Induction of Key ABC-Transporter Genes Affecting Glyphosate Resistance Mechanism in Conyza canadensis. International Journal of Molecular Sciences. 2016; 17(4):342. https://doi.org/10.3390/ijms17040342
Chicago/Turabian StyleTani, Eleni, Demosthenis Chachalis, Ilias S. Travlos, and Dimitrios Bilalis. 2016. "Environmental Conditions Influence Induction of Key ABC-Transporter Genes Affecting Glyphosate Resistance Mechanism in Conyza canadensis" International Journal of Molecular Sciences 17, no. 4: 342. https://doi.org/10.3390/ijms17040342







