Glycosphingolipid–Protein Interaction in Signal Transduction
Abstract
:1. Introduction
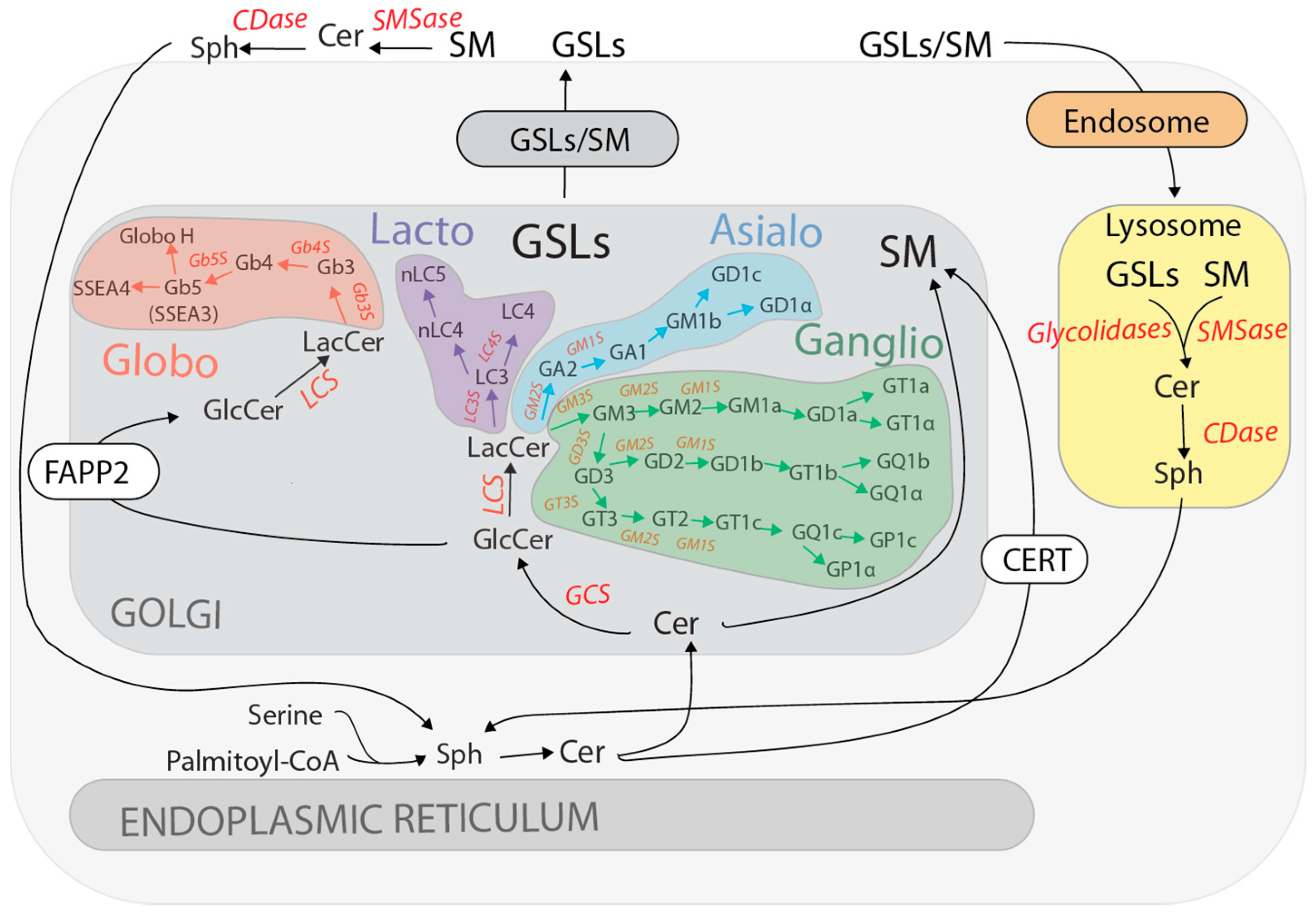
2. GSL Synthesis and Turnover
3. GSL-Dependent Regulation of Signal Transduction
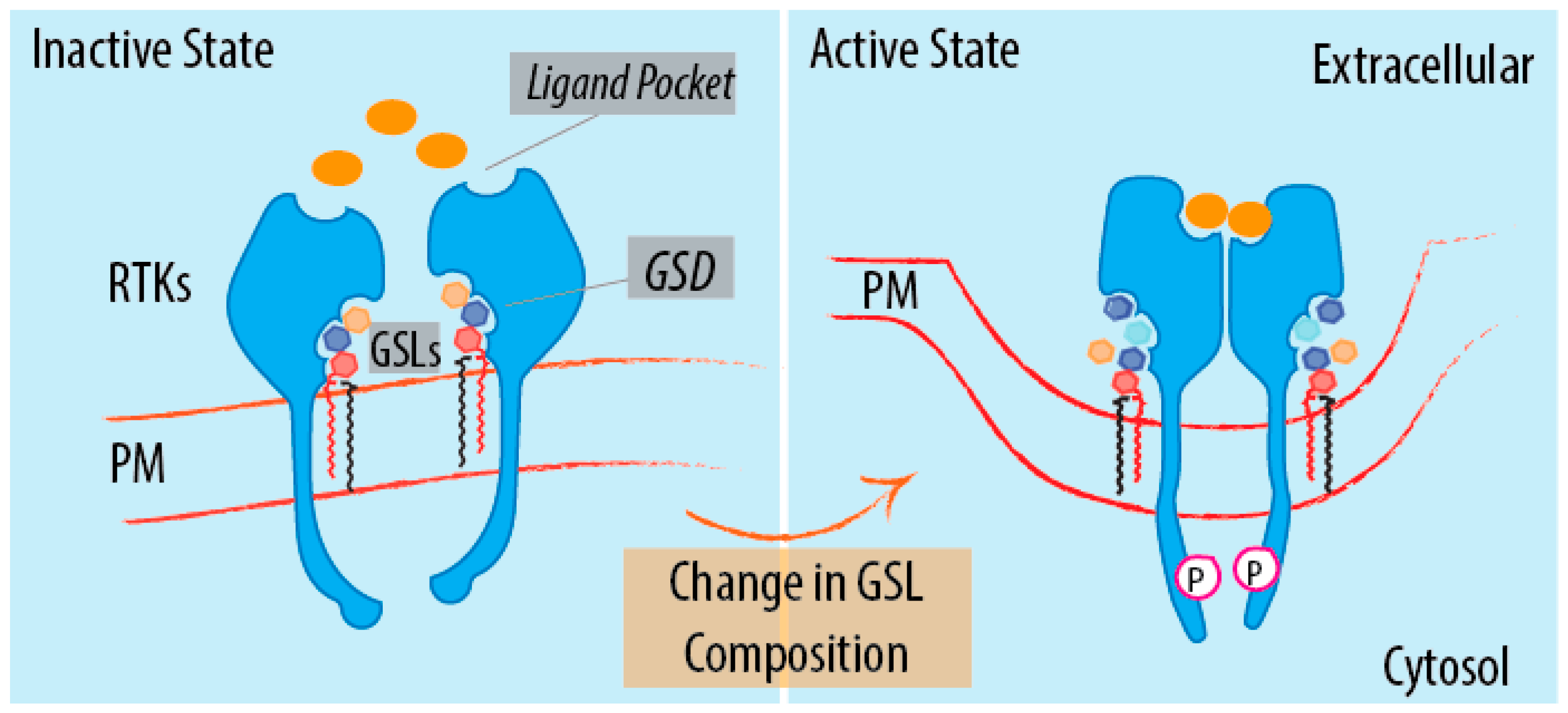
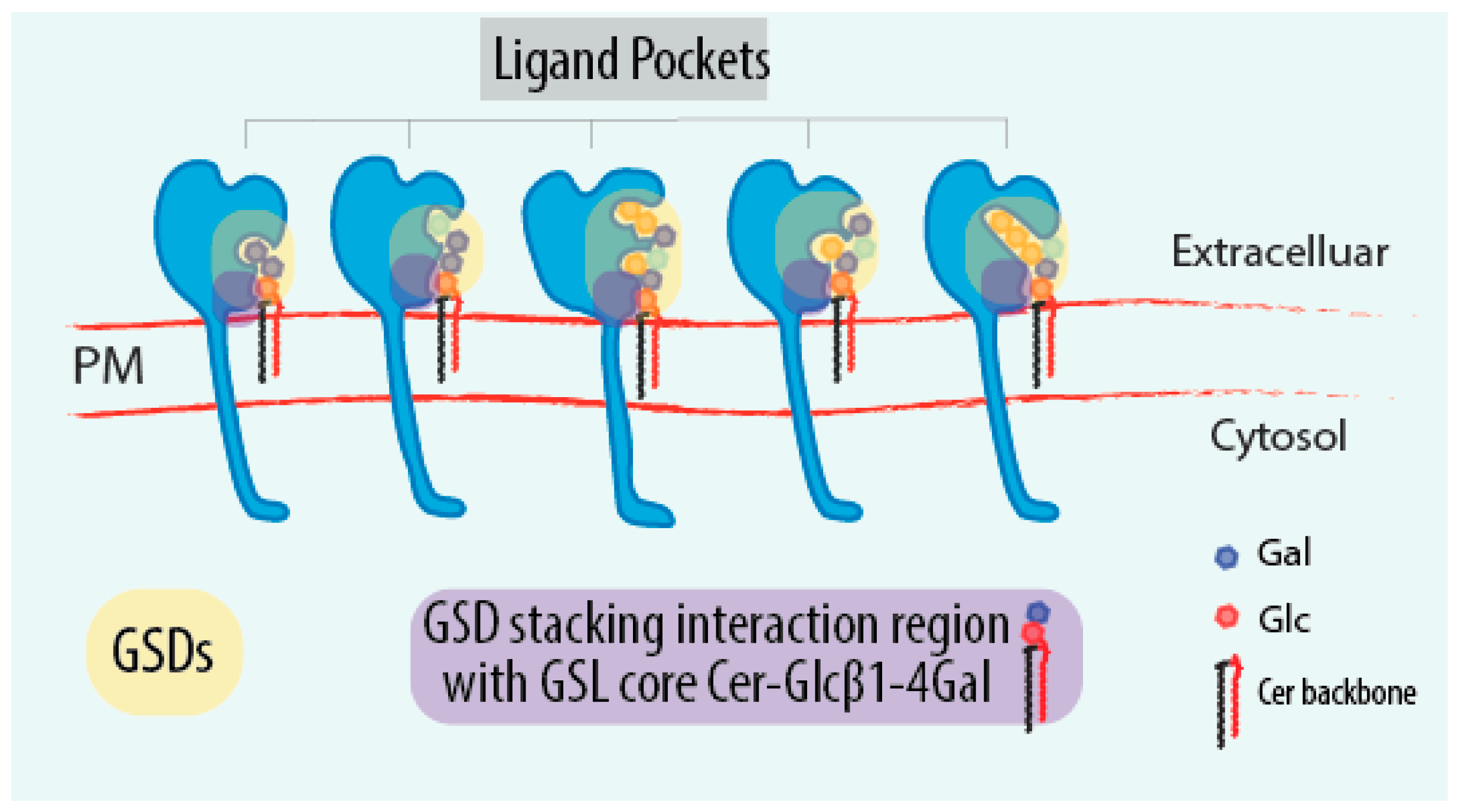
4. GSL-Sensing Domains (GSDs) as Sensors of GSLs
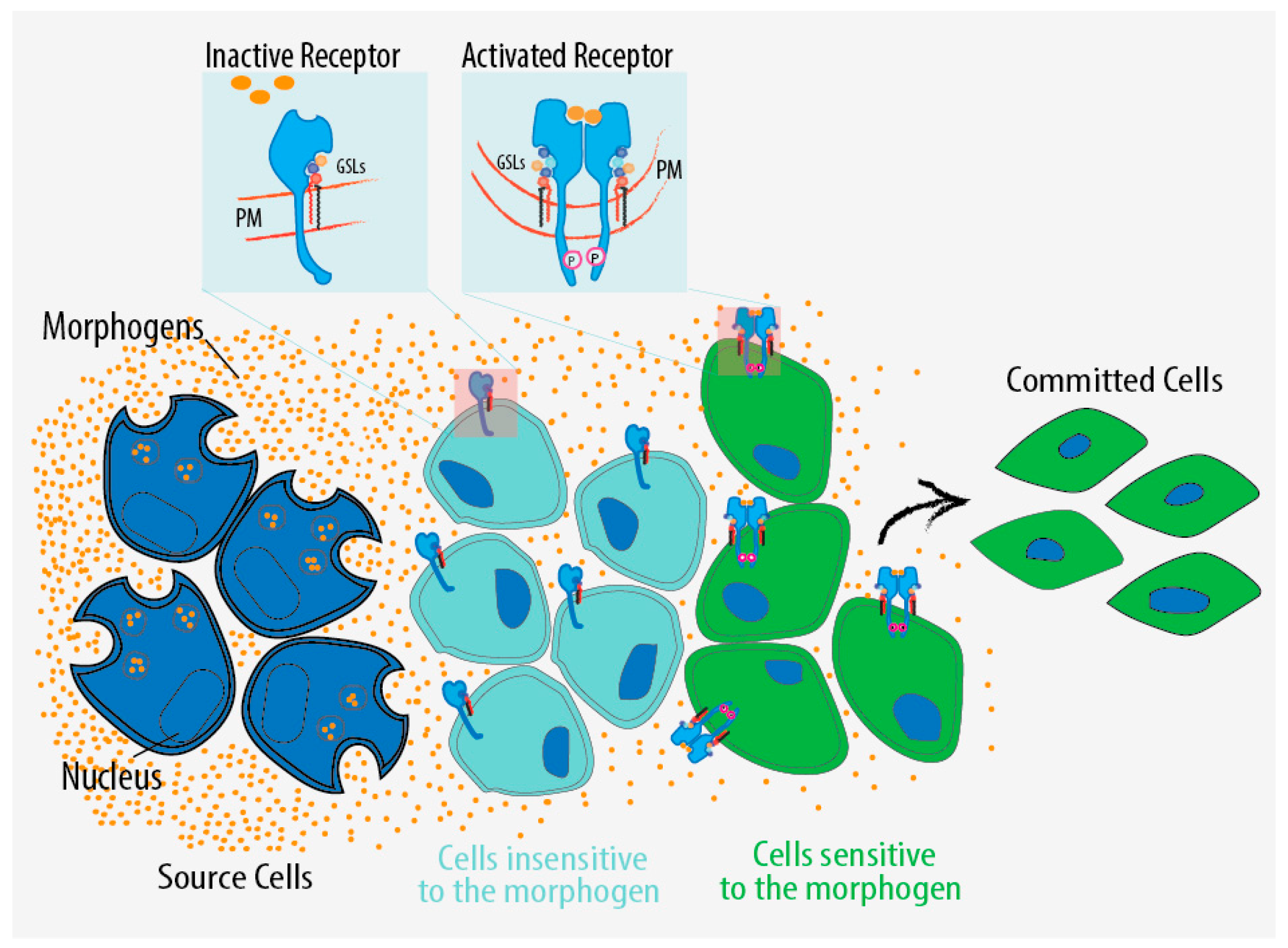
5. GSL Regulation in Development
6. Open Questions
6.1. What Are the Molecular Rules Driving GSL Sensing?
6.2. Which Are the Targets of GSL-Dependent Regulation?
6.3. What Is the Role of the Hydrophobic GSL Portion in the Regulation of Signal Transduction?
6.4. How Is GSL Metabolism Regulated?
6.5. Which Is the Role of the GSL-Dependent Regulation in Development?
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Merrill, A.H., Jr. Sphingolipid and glycosphingolipid metabolic pathways in the era of sphingolipidomics. Chem. Rev. 2011, 111, 6387–6422. [Google Scholar] [CrossRef] [PubMed]
- Coskun, U.; Grzybek, M.; Drechsel, D.; Simons, K. Regulation of human EGF receptor by lipids. Proc. Natl. Acad. Sci. USA 2011, 108, 9044–9048. [Google Scholar] [CrossRef] [PubMed]
- Vyas, K.A.; Patel, H.V.; Vyas, A.A.; Schnaar, R.L. Segregation of gangliosides GM1 and GD3 on cell membranes, isolated membrane rafts, and defined supported lipid monolayers. Biol. Chem. 2001, 382, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Kiyokawa, E.; Baba, T.; Otsuka, N.; Makino, A.; Ohno, S.; Kobayashi, T. Spatial and functional heterogeneity of sphingolipid-rich membrane domains. J. Biol. Chem. 2005, 280, 24072–24084. [Google Scholar] [CrossRef] [PubMed]
- Lindquist, S.; Karitkina, D.; Langnaese, K.; Posevitz-Fejfar, A.; Schraven, B.; Xavier, R.; Seed, B.; Lindquist, J.A. Phosphoprotein associated with glycosphingolipid-enriched microdomains differentially modulates SRC kinase activity in brain maturation. PLoS ONE 2011, 6, e23978. [Google Scholar] [CrossRef] [PubMed]
- Ideo, H.; Seko, A.; Ishizuka, I.; Yamashita, K. The N-terminal carbohydrate recognition domain of galectin-8 recognizes specific glycosphingolipids with high affinity. Glycobiology 2003, 13, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Sprong, H.; Degroote, S.; Nilsson, T.; Kawakita, M.; Ishida, N.; van der Sluijs, P.; van Meer, G. Association of the Golgi UDP-galactose transporter with UDP-galactose:ceramide galactosyltransferase allows UDP-galactose import in the endoplasmic reticulum. Mol. Biol. Cell 2003, 14, 3482–3493. [Google Scholar] [CrossRef] [PubMed]
- Schulze, H.; Sandhoff, K. Lysosomal lipid storage diseases. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef] [PubMed]
- Hamel, S.; Fantini, J.; Schweisguth, F. Notch ligand activity is modulated by glycosphingolipid membrane composition in Drosophila melanogaster. J. Cell Biol. 2010, 188, 581–594. [Google Scholar] [CrossRef] [PubMed]
- Minoguchi, K.; Swaim, W.D.; Berenstein, E.H.; Siraganian, R.P. Src family tyrosine kinase p53/56lyn, a serine kinase and Fc epsilon RI associate with α-galactosyl derivatives of ganglioside GD1b in rat basophilic leukemia RBL-2H3 cells. J. Biol. Chem. 1994, 269, 5249–5254. [Google Scholar] [PubMed]
- Kasahara, K.; Watanabe, Y.; Yamamoto, T.; Sanai, Y. Association of Src family tyrosine kinase Lyn with ganglioside GD3 in rat brain: Possible regulation of Lyn by glycosphingolipid in caveolae-like domains. J. Biol. Chem. 1997, 272, 29947–29953. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, S.; Handa, K.; Hakomori, S. A close association of GM3 with c-Src and Rho in GM3-enriched microdomains at the B16 melanoma cell surface membrane: A preliminary note. Biochem. Biophys. Res. Commun. 1997, 236, 218–222. [Google Scholar] [CrossRef] [PubMed]
- Iwabuchi, K.; Yamamura, S.; Prinetti, A.; Handa, K.; Hakomori, S. GM3-enriched microdomain involved in cell adhesion and signal transduction through carbohydrate-carbohydrate interaction in mouse melanoma B16 cells. J. Biol. Chem. 1998, 273, 9130–9138. [Google Scholar] [CrossRef] [PubMed]
- Holleran, W.M.; Takagi, Y.; Menon, G.K.; Legler, G.; Feingold, K.R.; Elias, P.M. Processing of epidermal glucosylceramides is required for optimal mammalian cutaneous permeability barrier function. J. Clin. Investig. 1993, 91, 1656–1664. [Google Scholar] [CrossRef] [PubMed]
- Holleran, W.M.; Ginns, E.I.; Menon, G.K.; Grundmann, J.U.; Fartasch, M.; McKinney, C.E.; Elias, P.M.; Sidransky, E. Consequences of β-glucocerebrosidase deficiency in epidermis. Ultrastructure and permeability barrier alterations in Gaucher disease. J. Clin. Investig. 1994, 93, 1756–1764. [Google Scholar] [CrossRef] [PubMed]
- Jennemann, R.; Sandhoff, R.; Langbein, L.; Kaden, S.; Rothermel, U.; Gallala, H.; Sandhoff, K.; Wiegandt, H.; Gröne, H.J. Integrity and barrier function of the epidermis critically depend on glucosylceramide synthesis. J. Biol. Chem. 2007, 282, 3083–3094. [Google Scholar] [CrossRef] [PubMed]
- Lingwood, C.A. Glycosphingolipid functions. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef]
- Hakomori, S.I. Structure and function of glycosphingolipids and sphingolipids: Recollections and future trends. Biochim. Biophys. Acta 2008, 1780, 325–346. [Google Scholar] [CrossRef] [PubMed]
- Mirkin, B.L.; Clark, S.H.; Zhang, C. Inhibition of human neuroblastoma cell proliferation and EGF receptor phosphorylation by gangliosides GM1, GM3, GD1A and GT1B. Cell Prolif. 2002, 35, 105–115. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Kwak, C.Y.; Shayman, J.A.; Kim, J.H. Globoside promotes activation of ERK by interaction with the epidermal growth factor receptor. Biochim. Biophys. Acta 2012, 1820, 1141–1148. [Google Scholar] [CrossRef] [PubMed]
- Guan, F.; Handa, K.; Hakomori, S.I. Regulation of epidermal growth factor receptor through interaction of ganglioside GM3 with GlcNAc of N-linked glycan of the receptor: Demonstration in ldlD cells. Neurochem. Res. 2011, 36, 1645–1653. [Google Scholar] [CrossRef] [PubMed]
- Bremer, E.G.; Hakomori, S. GM3 ganglioside induces hamster fibroblast growth inhibition in chemically-defined medium: Ganglioside may regulate growth factor receptor function. Biochem. Biophys. Res. Commun. 1982, 106, 711–718. [Google Scholar] [CrossRef]
- Bremer, E.G.; Hakomori, S.; Bowen-Pope, D.F.; Raines, E.; Ross, R. Ganglioside-mediated modulation of cell growth, growth factor binding, and receptor phosphorylation. J. Biol. Chem. 1984, 259, 6818–6825. [Google Scholar] [PubMed]
- Mutoh, T.; Tokuda, A.; Miyadai, T.; Hamaguchi, M.; Fujiki, N. Ganglioside GM1 binds to the Trk protein and regulates receptor function. Proc. Natl. Acad. Sci. USA 1995, 92, 5087–5091. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M.; Hidari, K.I.; Suzuki, T.; Miyamoto, D.; Suzuki, Y. Engagement of endogenous ganglioside GM1a induces tyrosine phosphorylation involved in neuron-like differentiation of PC12 cells. Glycobiology 2001, 11, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Kabayama, K.; Sato, T.; Saito, K.; Loberto, N.; Prinetti, A.; Sonnino, S.; Kinjo, M.; Igarashi, Y.; Inokuchi, J. Dissociation of the insulin receptor and caveolin-1 complex by ganglioside GM3 in the state of insulin resistance. Proc. Natl. Acad. Sci. USA 2007, 104, 13678–13683. [Google Scholar] [CrossRef] [PubMed]
- Tagami, S.; Inokuchi Ji, J.; Kabayama, K.; Yoshimura, H.; Kitamura, F.; Uemura, S.; Ogawa, C.; Ishii, A.; Saito, M.; Ohtsuka, Y.; et al. Ganglioside GM3 participates in the pathological conditions of insulin resistance. J. Biol. Chem. 2002, 277, 3085–3092. [Google Scholar] [CrossRef] [PubMed]
- Kondo, Y.; Ikeda, K.; Tokuda, N.; Nishitani, C.; Ohto, U.; Akashi-Takamura, S.; Ito, Y.; Uchikawa, M.; Kuroki, Y.; Taguchi, R.; et al. TLR4-MD-2 complex is negatively regulated by an endogenous ligand, globotetraosylceramide. Proc. Natl. Acad. Sci. USA 2013, 110, 4714–4719. [Google Scholar] [CrossRef] [PubMed]
- Chakrabandhu, K.; Huault, S.; Garmy, N.; Fantini, J.; Stebe, E.; Mailfert, S.; Marguet, D.; Hueber, A.O. The extracellular glycosphingolipid-binding motif of Fas defines its internalization route, mode and outcome of signals upon activation by ligand. Cell Death Differ. 2008, 15, 1824–1837. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.S.; Son, K.Y.; Koo, K.M.; Kim, J.Y.; Alfajaro, M.M.; Park, J.G.; Hosmillo, M.; Soliman, M.; Baek, Y.B.; Cho, E.H.; et al. Porcine sapelovirus uses α2,3-linked sialic acid on GD1a ganglioside as a receptor. J. Virol. 2016, 90, 4067–4077. [Google Scholar] [CrossRef] [PubMed]
- Rydell, G.E.; Dahlin, A.B.; Hook, F.; Larson, G. QCM-D studies of human norovirus VLPs binding to glycosphingolipids in supported lipid bilayers reveal strain-specific characteristics. Glycobiology 2009, 19, 1176–1184. [Google Scholar] [CrossRef] [PubMed]
- Le Pendu, J.; Ruvoen-Clouet, N.; Kindberg, E.; Svensson, L. Mendelian resistance to human norovirus infections. Semin. Immunol. 2006, 18, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Rydell, G.E.; Svensson, L.; Larson, G.; Johannes, L.; Romer, W. Human GII.4 norovirus VLP induces membrane invaginations on giant unilamellar vesicles containing secretor gene dependent α1,2-fucosylated glycosphingolipids. Biochim. Biophys. Acta 2013, 1828, 1840–1845. [Google Scholar] [CrossRef] [PubMed]
- Taube, S.; Perry, J.W.; Yetming, K.; Patel, S.P.; Auble, H.; Shu, L.; Nawar, H.F.; Lee, C.H.; Connell, T.D.; Shayman, J.A.; et al. Ganglioside-linked terminal sialic acid moieties on murine macrophages function as attachment receptors for murine noroviruses. J. Virol. 2009, 83, 4092–4101. [Google Scholar] [CrossRef] [PubMed]
- Taube, S.; Perry, J.W.; McGreevy, E.; Yetming, K.; Perkins, C.; Henderson, K.; Wobus, C.E. Murine noroviruses bind glycolipid and glycoprotein attachment receptors in a strain-dependent manner. J. Virol. 2012, 86, 5584–5593. [Google Scholar] [CrossRef] [PubMed]
- Zakhour, M.; Ruvoen-Clouet, N.; Charpilienne, A.; Langpap, B.; Poncet, D.; Peters, T.; Bovin, N.; Le Pendu, J. The αGal epitope of the histo-blood group antigen family is a ligand for bovine norovirus Newbury2 expected to prevent cross-species transmission. PLoS Pathog. 2009, 5, e1000504. [Google Scholar] [CrossRef] [PubMed]
- Ruvoen-Clouet, N.; Ganiere, J.P.; Andre-Fontaine, G.; Blanchard, D.; Le Pendu, J. Binding of rabbit hemorrhagic disease virus to antigens of the ABH histo-blood group family. J. Virol. 2000, 74, 11950–11954. [Google Scholar] [CrossRef] [PubMed]
- Nilsson, E.C.; Storm, R.J.; Bauer, J.; Johansson, S.M.; Lookene, A.; Angstrom, J.; Hedenstrom, M.; Eriksson, T.L.; Frangsmyr, L.; Rinaldi, S.; et al. The GD1a glycan is a cellular receptor for adenoviruses causing epidemic keratoconjunctivitis. Nat. Med. 2011, 17, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Stencel-Baerenwald, J.; Reiss, K.; Blaum, B.S.; Colvin, D.; Li, X.N.; Abel, T.; Boyd, K.; Stehle, T.; Dermody, T.S. Glycan engagement dictates hydrocephalus induction by serotype 1 reovirus. mBio 2015, 6, e02356. [Google Scholar] [CrossRef] [PubMed]
- Rolsma, M.D.; Gelberg, H.B.; Kuhlenschmidt, M.S. Assay for evaluation of rotavirus-cell interactions: Identification of an enterocyte ganglioside fraction that mediates group A porcine rotavirus recognition. J. Virol. 1994, 68, 258–268. [Google Scholar] [PubMed]
- Rolsma, M.D.; Kuhlenschmidt, T.B.; Gelberg, H.B.; Kuhlenschmidt, M.S. Structure and function of a ganglioside receptor for porcine rotavirus. J. Virol. 1998, 72, 9079–9091. [Google Scholar] [PubMed]
- Haselhorst, T.; Fleming, F.E.; Dyason, J.C.; Hartnell, R.D.; Yu, X.; Holloway, G.; Santegoets, K.; Kiefel, M.J.; Blanchard, H.; Coulson, B.S.; et al. Sialic acid dependence in rotavirus host cell invasion. Nat. Chem. Biol. 2009, 5, 91–93. [Google Scholar] [CrossRef] [PubMed]
- Martinez, M.A.; Lopez, S.; Arias, C.F.; Isa, P. Gangliosides have a functional role during rotavirus cell entry. J. Virol. 2013, 87, 1115–1122. [Google Scholar] [CrossRef] [PubMed]
- Delorme, C.; Brussow, H.; Sidoti, J.; Roche, N.; Karlsson, K.A.; Neeser, J.R.; Teneberg, S. Glycosphingolipid binding specificities of rotavirus: Identification of a sialic acid-binding epitope. J. Virol. 2001, 75, 2276–2287. [Google Scholar] [CrossRef] [PubMed]
- Dormitzer, P.R.; Sun, Z.Y.; Blixt, O.; Paulson, J.C.; Wagner, G.; Harrison, S.C. Specificity and affinity of sialic acid binding by the rhesus rotavirus VP8* core. J. Virol. 2002, 76, 10512–10517. [Google Scholar] [CrossRef] [PubMed]
- Fukudome, K.; Yoshie, O.; Konno, T. Comparison of human, simian, and bovine rotaviruses for requirement of sialic acid in hemagglutination and cell adsorption. Virology 1989, 172, 196–205. [Google Scholar] [CrossRef]
- Guo, C.T.; Nakagomi, O.; Mochizuki, M.; Ishida, H.; Kiso, M.; Ohta, Y.; Suzuki, T.; Miyamoto, D.; Hidari, K.I.; Suzuki, Y. Ganglioside GMia on the cell surface is involved in the infection by human rotavirus KUN and MO strains. J. Biochem. 1999, 126, 683–688. [Google Scholar] [CrossRef] [PubMed]
- Fleming, F.E.; Bohm, R.; Dang, V.T.; Holloway, G.; Haselhorst, T.; Madge, P.D.; Deveryshetty, J.; Yu, X.; Blanchard, H.; von Itzstein, M.; et al. Relative roles of GM1 ganglioside, N-acylneuraminic acids, and α2β1 integrin in mediating rotavirus infection. J. Virol. 2014, 88, 4558–4571. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Coulson, B.S.; Fleming, F.E.; Dyason, J.C.; von Itzstein, M.; Blanchard, H. Novel structural insights into rotavirus recognition of ganglioside glycan receptors. J. Mol. Biol. 2011, 413, 929–939. [Google Scholar] [CrossRef] [PubMed]
- Stroh, L.J.; Gee, G.V.; Blaum, B.S.; Dugan, A.S.; Feltkamp, M.C.; Atwood, W.J.; Stehle, T. Trichodysplasia spinulosa-associated polyomavirus uses a displaced binding site on VP1 to engage sialylated glycolipids. PLoS Pathog. 2015, 11, e1005112. [Google Scholar] [CrossRef] [PubMed]
- Tsai, B.; Gilbert, J.M.; Stehle, T.; Lencer, W.; Benjamin, T.L.; Rapoport, T.A. Gangliosides are receptors for murine polyoma virus and SV40. EMBO J. 2003, 22, 4346–4355. [Google Scholar] [CrossRef] [PubMed]
- You, J.; O’Hara, S.D.; Velupillai, P.; Castle, S.; Levery, S.; Garcea, R.L.; Benjamin, T. Ganglioside and non-ganglioside mediated host responses to the mouse polyomavirus. PLoS Pathog. 2015, 11, e1005175. [Google Scholar] [CrossRef] [PubMed]
- Ewers, H.; Romer, W.; Smith, A.E.; Bacia, K.; Dmitrieff, S.; Chai, W.; Mancini, R.; Kartenbeck, J.; Chambon, V.; Berland, L.; et al. GM1 structure determines SV40-induced membrane invagination and infection. Nat. Cell Biol. 2010, 12, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Neu, U.; Woellner, K.; Gauglitz, G.; Stehle, T. Structural basis of GM1 ganglioside recognition by simian virus 40. Proc. Natl. Acad. Sci. USA 2008, 105, 5219–5224. [Google Scholar] [CrossRef] [PubMed]
- Low, J.A.; Magnuson, B.; Tsai, B.; Imperiale, M.J. Identification of gangliosides GD1b and GT1b as receptors for BK virus. J. Virol. 2006, 80, 1361–1366. [Google Scholar] [CrossRef] [PubMed]
- Komagome, R.; Sawa, H.; Suzuki, T.; Suzuki, Y.; Tanaka, S.; Atwood, W.J.; Nagashima, K. Oligosaccharides as receptors for JC virus. J. Virol. 2002, 76, 12992–13000. [Google Scholar] [CrossRef] [PubMed]
- Erickson, K.D.; Garcea, R.L.; Tsai, B. Ganglioside GT1b is a putative host cell receptor for the Merkel cell polyomavirus. J. Virol. 2009, 83, 10275–10279. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.E.; Anderson, S.M.; Young, N.S. Erythrocyte P antigen: Cellular receptor for B19 parvovirus. Science 1993, 262, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Nasir, W.; Nilsson, J.; Olofsson, S.; Bally, M.; Rydell, G.E. Parvovirus B19 VLP recognizes globoside in supported lipid bilayers. Virology 2014, 456–457, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.E.; Liu, Z.; Gallinella, G.; Wong, S.; Mills, I.P.; O’Sullivan, M.G. Simian parvovirus infection: A potential zoonosis. J. Infect. Dis. 2004, 190, 1900–1907. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Chiorini, J.A. Gangliosides are essential for bovine adeno-associated virus entry. J. Virol. 2006, 80, 5516–5522. [Google Scholar] [CrossRef] [PubMed]
- Hammache, D.; Pieroni, G.; Yahi, N.; Delezay, O.; Koch, N.; Lafont, H.; Tamalet, C.; Fantini, J. Specific interaction of HIV-1 and HIV-2 surface envelope glycoproteins with monolayers of galactosylceramide and ganglioside GM3. J. Biol. Chem. 1998, 273, 7967–7971. [Google Scholar] [CrossRef] [PubMed]
- Hammache, D.; Yahi, N.; Pieroni, G.; Ariasi, F.; Tamalet, C.; Fantini, J. Sequential interaction of CD4 and HIV-1 gp120 with a reconstituted membrane patch of ganglioside GM3: Implications for the role of glycolipids as potential HIV-1 fusion cofactors. Biochem. Biophys. Res. Commun. 1998, 246, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Hammache, D.; Yahi, N.; Maresca, M.; Pieroni, G.; Fantini, J. Human erythrocyte glycosphingolipids as alternative cofactors for human immunodeficiency virus type 1 (HIV-1) entry: Evidence for CD4-induced interactions between HIV-1 gp120 and reconstituted membrane microdomains of glycosphingolipids (Gb3 and GM3). J. Virol. 1999, 73, 5244–5248. [Google Scholar] [PubMed]
- Hug, P.; Lin, H.M.; Korte, T.; Xiao, X.; Dimitrov, D.S.; Wang, J.M.; Puri, A.; Blumenthal, R. Glycosphingolipids promote entry of a broad range of human immunodeficiency virus type 1 isolates into cell lines expressing CD4, CXCR4, and/or CCR5. J. Virol. 2000, 74, 6377–6385. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.; Spitalnik, S.L.; Gonzalez-Scarano, F.; Silberberg, D.H. Galactosyl ceramide or a derivative is an essential component of the neural receptor for human immunodeficiency virus type 1 envelope glycoprotein gp120. Proc. Natl. Acad. Sci. USA 1991, 88, 7131–7134. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.; Mettus, R.V.; Reddy, E.P.; Ugen, K.E.; Srikanthan, V.; Williams, W.V.; Weiner, D.B. The galactosyl ceramide/sulfatide receptor binding region of HIV-1 gp120 maps to amino acids 206–275. AIDS Res. Hum. Retrovir. 1993, 9, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Rawat, S.S.; Johnson, B.T.; Puri, A. Sphingolipids: Modulators of HIV-1 infection and pathogenesis. Biosci. Rep. 2005, 25, 329–343. [Google Scholar] [CrossRef] [PubMed]
- Cook, D.G.; Fantini, J.; Spitalnik, S.L.; Gonzalez-Scarano, F. Binding of human immunodeficiency virus type I (HIV-1) gp120 to galactosylceramide (GalCer): Relationship to the V3 loop. Virology 1994, 201, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Wang, J.; Sun, T.; Bian, G.; Pan, W.; Feng, T.; Wang, P.; Li, Y.; Dai, J. Glycosphingolipid GM3 is Indispensable for Dengue Virus Genome Replication. Int. J. Biol. Sci. 2016, 12, 872–883. [Google Scholar] [CrossRef] [PubMed]
- Hidari, K.I.; Abe, T.; Suzuki, T. Carbohydrate-related inhibitors of dengue virus entry. Viruses 2013, 5, 605–618. [Google Scholar] [CrossRef] [PubMed]
- Wichit, S.; Jittmittraphap, A.; Hidari, K.I.; Thaisomboonsuk, B.; Petmitr, S.; Ubol, S.; Aoki, C.; Itonori, S.; Morita, K.; Suzuki, T.; et al. Dengue virus type 2 recognizes the carbohydrate moiety of neutral glycosphingolipids in mammalian and mosquito cells. Microbiol. Immunol. 2011, 55, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Meisen, I.; Dzudzek, T.; Ehrhardt, C.; Ludwig, S.; Mormann, M.; Rosenbruck, R.; Lumen, R.; Kniep, B.; Karch, H.; Muthing, J. The human H3N2 influenza viruses A/Victoria/3/75 and A/Hiroshima/52/2005 preferentially bind to α2–3-sialylated monosialogangliosides with fucosylated poly-N-acetyllactosaminyl chains. Glycobiology 2012, 22, 1055–1076. [Google Scholar] [CrossRef] [PubMed]
- Perino, J.; Crouzier, D.; Spehner, D.; Debouzy, J.C.; Garin, D.; Crance, J.M.; Favier, A.L. Lung surfactant DPPG phospholipid inhibits vaccinia virus infection. Antivir. Res. 2011, 89, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Perino, J.; Foo, C.H.; Spehner, D.; Cohen, G.H.; Eisenberg, R.J.; Crance, J.M.; Favier, A.L. Role of sulfatide in vaccinia virus infection. Biol. Cell 2011, 103, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, L.; Villar, E.; Munoz-Barroso, I. Gangliosides and N-glycoproteins function as Newcastle disease virus receptors. Int. J. Biochem. Cell Biol. 2004, 36, 2344–2356. [Google Scholar] [CrossRef] [PubMed]
- Epand, R.M.; Nir, S.; Parolin, M.; Flanagan, T.D. The role of the ganglioside GD1a as a receptor for Sendai virus. Biochemistry 1995, 34, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Portner, A.; Scroggs, R.A.; Uchikawa, M.; Koyama, N.; Matsuo, K.; Suzuki, Y.; Takimoto, T. Receptor specificities of human respiroviruses. J. Virol. 2001, 75, 4604–4613. [Google Scholar] [CrossRef] [PubMed]
- Jobling, M.G.; Holmes, R.K. Mutational analysis of ganglioside GM1-binding ability, pentamer formation, and epitopes of cholera toxin B (CTB) subunits and CTB/heat-labile enterotoxin B subunit chimeras. Infect. Immun. 2002, 70, 1260–1271. [Google Scholar] [CrossRef] [PubMed]
- Heyningen, S.V. Cholera toxin: Interaction of subunits with ganglioside GM1. Science 1974, 183, 656–657. [Google Scholar] [CrossRef]
- Lencer, W.I.; Saslowsky, D. Raft trafficking of AB5 subunit bacterial toxins. Biochim. Biophys. Acta 2005, 1746, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Jacewicz, M.; Clausen, H.; Nudelman, E.; Donohue-Rolfe, A.; Keusch, G.T. Pathogenesis of shigella diarrhea. XI. Isolation of a shigella toxin-binding glycolipid from rabbit jejunum and HeLa cells and its identification as globotriaosylceramide. J. Exp. Med. 1986, 163, 1391–1404. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, A.A.; Brown, J.E.; Stromberg, N.; Westling-Ryd, M.; Schultz, J.E.; Karlsson, K.A. Identification of the carbohydrate receptor for Shiga toxin produced by Shigella dysenteriae type 1. J. Biol. Chem. 1987, 262, 1779–1785. [Google Scholar] [PubMed]
- Keusch, G.T.; Jacewicz, M.; Acheson, D.W.; Donohue-Rolfe, A.; Kane, A.V.; McCluer, R.H. Globotriaosylceramide, Gb3, is an alternative functional receptor for Shiga-like toxin 2e. Infect. Immun. 1995, 63, 1138–1141. [Google Scholar] [PubMed]
- Okuda, T.; Tokuda, N.; Numata, S.; Ito, M.; Ohta, M.; Kawamura, K.; Wiels, J.; Urano, T.; Tajima, O.; Furukawa, K.; et al. Targeted disruption of Gb3/CD77 synthase gene resulted in the complete deletion of globo-series glycosphingolipids and loss of sensitivity to verotoxins. J. Biol. Chem. 2006, 281, 10230–10235. [Google Scholar] [CrossRef] [PubMed]
- Brunger, A.T.; Rummel, A. Receptor and substrate interactions of clostridial neurotoxins. Toxicon 2009, 54, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, K.; Kohda, T.; Shibata, Y.; Tsukamoto, K.; Arimitsu, H.; Hayashi, M.; Mukamoto, M.; Sasakawa, N.; Kozaki, S. Unique biological activity of botulinum D/C mosaic neurotoxin in murine species. Infect. Immun. 2012, 80, 2886–2893. [Google Scholar] [CrossRef] [PubMed]
- Fukuta, S.; Magnani, J.L.; Twiddy, E.M.; Holmes, R.K.; Ginsburg, V. Comparison of the carbohydrate-binding specificities of cholera toxin and Escherichia coli heat-labile enterotoxins LTh-I, LT-IIa, and LT-IIb. Infect. Immun. 1988, 56, 1748–1753. [Google Scholar] [PubMed]
- Nakayama, H.; Ogawa, H.; Takamori, K.; Iwabuchi, K. GSL-enriched membrane microdomains in innate immune responses. Arch. Immunol. Ther. Exp. 2013, 61, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Duan, J.; Zhang, J.; Zhao, Y.; Yang, F.; Zhang, X. Ganglioside GM2 modulates the erythrocyte Ca2+-ATPase through its binding to the calmodulin-binding domain and its ‘receptor’. Arch. Biochem. Biophys. 2006, 454, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Boscher, C.; Zheng, Y.Z.; Lakshminarayan, R.; Johannes, L.; Dennis, J.W.; Foster, L.J.; Nabi, I.R. Galectin-3 protein regulates mobility of N-cadherin and GM1 ganglioside at cell-cell junctions of mammary carcinoma cells. J. Biol. Chem. 2012, 287, 32940–32952. [Google Scholar] [CrossRef] [PubMed]
- Handa, K.; Hakomori, S.I. Carbohydrate to carbohydrate interaction in development process and cancer progression. Glycoconj. J. 2012, 29, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Heuss, S.F.; Tarantino, N.; Fantini, J.; Ndiaye-Lobry, D.; Moretti, J.; Israel, A.; Logeat, F. A glycosphingolipid binding domain controls trafficking and activity of the mammalian notch ligand delta-like 1. PLoS ONE 2013, 8, e74392. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Paila, Y.D.; Chattopadhyay, A. Role of glycosphingolipids in the function of human serotonin(1)A receptors. J. Neurochem. 2012, 123, 716–724. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Wada, R.; Sasaki, T.; Deng, C.; Bierfreund, U.; Sandhoff, K.; Proia, R.L. A vital role for glycosphingolipid synthesis during development and differentiation. Proc. Natl. Acad. Sci. USA 1999, 96, 9142–9147. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Wu, Y.P.; Sandhoff, R.; Werth, N.; Mizukami, H.; Ellis, J.M.; Dupree, J.L.; Geyer, R.; Sandhoff, K.; Proia, R.L. Interruption of ganglioside synthesis produces central nervous system degeneration and altered axon-glial interactions. Proc. Natl. Acad. Sci. USA 2005, 102, 2725–2730. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, T.; Tanaka, M.; Yokoyama, M.; Sato, T.; Shinkai, T.; Furukawa, K. Early lethality of β-1,4-galactosyltransferase V-mutant mice by growth retardation. Biochem. Biophys. Res. Commun. 2009, 379, 456–459. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Hashiramoto, A.; Haluzik, M.; Mizukami, H.; Beck, S.; Norton, A.; Kono, M.; Tsuji, S.; Daniotti, J.L.; Werth, N.; et al. Enhanced insulin sensitivity in mice lacking ganglioside GM3. Proc. Natl. Acad. Sci. USA 2003, 100, 3445–3449. [Google Scholar] [CrossRef] [PubMed]
- Takamiya, K.; Yamamoto, A.; Furukawa, K.; Yamashiro, S.; Shin, M.; Okada, M.; Fukumoto, S.; Haraguchi, M.; Takeda, N.; Fujimura, K.; et al. Mice with disrupted GM2/GD2 synthase gene lack complex gangliosides but exhibit only subtle defects in their nervous system. Proc. Natl. Acad. Sci. USA 1996, 93, 10662–10667. [Google Scholar] [CrossRef] [PubMed]
- Sheikh, K.A.; Sun, J.; Liu, Y.; Kawai, H.; Crawford, T.O.; Proia, R.L.; Griffin, J.W.; Schnaar, R.L. Mice lacking complex gangliosides develop Wallerian degeneration and myelination defects. Proc. Natl. Acad. Sci. USA 1999, 96, 7532–7537. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Lu, Z.H.; Kulkarni, N.; Amin, R.; Ledeen, R.W. Mice lacking major brain gangliosides develop parkinsonism. Neurochem. Res. 2011, 36, 1706–1714. [Google Scholar] [CrossRef] [PubMed]
- Linn, S.C.; Kim, H.S.; Keane, E.M.; Andras, L.M.; Wang, E.; Merrill, A.H., Jr. Regulation of de novo sphingolipid biosynthesis and the toxic consequences of its disruption. Biochem. Soc. Trans. 2001, 29, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Mullen, T.D.; Hannun, Y.A.; Obeid, L.M. Ceramide synthases at the centre of sphingolipid metabolism and biology. Biochem. J. 2012, 441, 789–802. [Google Scholar] [CrossRef] [PubMed]
- Holthuis, J.C.; Pomorski, T.; Raggers, R.J.; Sprong, H.; van Meer, G. The organizing potential of sphingolipids in intracellular membrane transport. Physiol. Rev. 2001, 81, 1689–1723. [Google Scholar] [PubMed]
- Hanada, K.; Kumagai, K.; Yasuda, S.; Miura, Y.; Kawano, M.; Fukasawa, M.; Nishijima, M. Molecular machinery for non-vesicular trafficking of ceramide. Nature 2003, 426, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Funakoshi, T.; Yasuda, S.; Fukasawa, M.; Nishijima, M.; Hanada, K. Reconstitution of ATP- and cytosol-dependent transport of de novo synthesized ceramide to the site of sphingomyelin synthesis in semi-intact cells. J. Biol. Chem. 2000, 275, 29938–29945. [Google Scholar] [CrossRef] [PubMed]
- Maccioni, H.J.; Quiroga, R.; Ferrari, M.L. Cellular and molecular biology of glycosphingolipid glycosylation. J. Neurochem. 2011, 117, 589–602. [Google Scholar] [CrossRef] [PubMed]
- Buton, X.; Herve, P.; Kubelt, J.; Tannert, A.; Burger, K.N.; Fellmann, P.; Muller, P.; Herrmann, A.; Seigneuret, M.; Devaux, P.F. Transbilayer movement of monohexosylsphingolipids in endoplasmic reticulum and Golgi membranes. Biochemistry 2002, 41, 13106–13115. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, G.; Polishchuk, E.; di Tullio, G.; Santoro, M.; fi Campli, A.; Godi, A.; West, G.; Bielawski, J.; Chuang, C.C.; van der Spoel, A.C.; et al. Glycosphingolipid synthesis requires FAPP2 transfer of glucosylceramide. Nature 2007, 449, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Halter, D.; Neumann, S.; van Dijk, S.M.; Wolthoorn, J.; de Maziere, A.M.; Vieira, O.V.; Mattjus, P.; Klumperman, J.; van Meer, G.; Sprong, H. Pre- and post-Golgi translocation of glucosylceramide in glycosphingolipid synthesis. J. Cell Biol. 2007, 179, 101–115. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, G.; Uemura, T.; Chuang, C.C.; Polishchuk, E.; Santoro, M.; Ohvo-Rekila, H.; Sato, T.; di Tullio, G.; Varriale, A.; D’Auria, S.; et al. Vesicular and non-vesicular transport feed distinct glycosylation pathways in the Golgi. Nature 2013, 501, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, T.; Sato, T.; Natsuka, S.; Kobayashi, Y.; Zhou, D.; Shinkai, T.; Hayakawa, S.; Furukawa, K. Involvement of murine β-1,4-galactosyltransferase V in lactosylceramide biosynthesis. Glycoconj. J. 2010, 27, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Nomura, T.; Takizawa, M.; Aoki, J.; Arai, H.; Inoue, K.; Wakisaka, E.; Yoshizuka, N.; Imokawa, G.; Dohmae, N.; Takio, K.; et al. Purification, cDNA cloning, and expression of UDP-Gal: Glucosylceramide β-1,4-galactosyltransferase from rat brain. J. Biol. Chem. 1998, 273, 13570–13577. [Google Scholar] [CrossRef] [PubMed]
- Ishii, A.; Ohta, M.; Watanabe, Y.; Matsuda, K.; Ishiyama, K.; Sakoe, K.; Nakamura, M.; Inokuchi, J.; Sanai, Y.; Saito, M. Expression cloning and functional characterization of human cDNA for ganglioside GM3 synthase. J. Biol. Chem. 1998, 273, 31652–31655. [Google Scholar] [CrossRef] [PubMed]
- Kojima, Y.; Fukumoto, S.; Furukawa, K.; Okajima, T.; Wiels, J.; Yokoyama, K.; Suzuki, Y.; Urano, T.; Ohta, M.; Furukawa, K. Molecular cloning of globotriaosylceramide/CD77 synthase, a glycosyltransferase that initiates the synthesis of globo series glycosphingolipids. J. Biol. Chem. 2000, 275, 15152–15156. [Google Scholar] [CrossRef] [PubMed]
- Biellmann, F.; Hulsmeier, A.J.; Zhou, D.; Cinelli, P.; Hennet, T. The Lc3-synthase gene B3gnt5 is essential to pre-implantation development of the murine embryo. BMC Dev. Biol. 2008, 8. [Google Scholar] [CrossRef] [PubMed]
- Nagata, Y.; Yamashiro, S.; Yodoi, J.; Lloyd, K.O.; Shiku, H.; Furukawa, K. Expression cloning of β 1,4 N-acetylgalactosaminyltransferase cDNAs that determine the expression of GM2 and GD2 gangliosides. J. Biol. Chem. 1992, 267, 12082–12089. [Google Scholar] [PubMed]
- Hidari, J.K.; Ichikawa, S.; Furukawa, K.; Yamasaki, M.; Hirabayashi, Y. β 1–4N-acetylgalactosaminyltransferase can synthesize both asialoglycosphingolipid GM2 and glycosphingolipid GM2 in vitro and in vivo: Isolation and characterization of a β 1–4N-acetylgalactosaminyltransferase cDNA clone from rat ascites hepatoma cell line AH7974F. Biochem. J. 1994, 303, 957–965. [Google Scholar] [PubMed]
- Maccioni, H.J.; Quiroga, R.; Spessott, W. Organization of the synthesis of glycolipid oligosaccharides in the Golgi complex. FEBS Lett. 2011, 585, 1691–1698. [Google Scholar] [CrossRef] [PubMed]
- Uliana, A.S.; Crespo, P.M.; Martina, J.A.; Daniotti, J.L.; Maccioni, H.J. Modulation of GalT1 and SialT1 sub-Golgi localization by SialT2 expression reveals an organellar level of glycolipid synthesis control. J. Biol. Chem. 2006, 281, 32852–32860. [Google Scholar] [CrossRef] [PubMed]
- Aureli, M.; Loberto, N.; Chigorno, V.; Prinetti, A.; Sonnino, S. Remodeling of sphingolipids by plasma membrane associated enzymes. Neurochem. Res. 2011, 36, 1636–1644. [Google Scholar] [CrossRef] [PubMed]
- Kitatani, K.; Idkowiak-Baldys, J.; Hannun, Y.A. The sphingolipid salvage pathway in ceramide metabolism and signaling. Cell Signal. 2008, 20, 1010–1018. [Google Scholar] [CrossRef] [PubMed]
- Ginzburg, L.; Kacher, Y.; Futerman, A.H. The pathogenesis of glycosphingolipid storage disorders. Semin. Cell Dev. Biol. 2004, 15, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Brunngraber, E.G.; Berra, B.; Zambotti, V. Altered levels of tissue glycoproteins, gangliosides, glycosaminoglycans and lipids in Niemann–Pick’s disease. Clin. Chim. Acta 1973, 48, 173–181. [Google Scholar] [CrossRef]
- Fahy, E.; Subramaniam, S.; Murphy, R.C.; Nishijima, M.; Raetz, C.R.; Shimizu, T.; Spener, F.; van Meer, G.; Wakelam, M.J.; Dennis, E.A. Update of the LIPID MAPS comprehensive classification system for lipids. J. Lipid Res. 2009, 50, S9–S14. [Google Scholar] [CrossRef] [PubMed]
- Bremer, E.G.; Schlessinger, J.; Hakomori, S. Ganglioside-mediated modulation of cell growth. Specific effects of GM3 on tyrosine phosphorylation of the epidermal growth factor receptor. J. Biol. Chem. 1986, 261, 2434–2440. [Google Scholar] [PubMed]
- Hanai, N.; Nores, G.A.; MacLeod, C.; Torres-Mendez, C.R.; Hakomori, S. Ganglioside-mediated modulation of cell growth: Specific effects of GM3 and lyso-GM3 in tyrosine phosphorylation of the epidermal growth factor receptor. J. Biol. Chem. 1988, 263, 10915–10921. [Google Scholar] [PubMed]
- Kawashima, N.; Yoon, S.J.; Itoh, K.; Nakayama, K. Tyrosine kinase activity of epidermal growth factor receptor is regulated by GM3 binding through carbohydrate to carbohydrate interactions. J. Biol. Chem. 2009, 284, 6147–6155. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, R.; Ladisch, S. Exogenous ganglioside GD1a enhances epidermal growth factor receptor binding and dimerization. J. Biol. Chem. 2004, 279, 36481–36489. [Google Scholar] [CrossRef] [PubMed]
- Meuillet, E.; Cremel, G.; Dreyfus, H.; Hicks, D. Differential modulation of basic fibroblast and epidermal growth factor receptor activation by ganglioside GM3 in cultured retinal Muller glia. Glia 1996, 17, 206–216. [Google Scholar] [CrossRef]
- Toledo, M.S.; Suzuki, E.; Handa, K.; Hakomori, S. Cell growth regulation through GM3-enriched microdomain (glycosynapse) in human lung embryonal fibroblast WI38 and its oncogenic transformant VA13. J. Biol. Chem. 2004, 279, 34655–34664. [Google Scholar] [CrossRef] [PubMed]
- Toledo, M.S.; Suzuki, E.; Handa, K.; Hakomori, S. Effect of ganglioside and tetraspanins in microdomains on interaction of integrins with fibroblast growth factor receptor. J. Biol. Chem. 2005, 280, 16227–16234. [Google Scholar] [CrossRef] [PubMed]
- Rabin, S.J.; Mocchetti, I. GM1 ganglioside activates the high-affinity nerve growth factor receptor trkA. J. Neurochem. 1995, 65, 347–354. [Google Scholar] [CrossRef] [PubMed]
- Duchemin, A.M.; Ren, Q.; Mo, L.; Neff, N.H.; Hadjiconstantinou, M. GM1 ganglioside induces phosphorylation and activation of Trk and Erk in brain. J. Neurochem. 2002, 81, 696–707. [Google Scholar] [CrossRef] [PubMed]
- Cazet, A.; Lefebvre, J.; Adriaenssens, E.; Julien, S.; Bobowski, M.; Grigoriadis, A.; Tutt, A.; Tulasne, D.; le Bourhis, X.; Delannoy, P. GD3 synthase expression enhances proliferation and tumor growth of MDA-MB-231 breast cancer cells through c-Met activation. Mol. Cancer Res. 2010, 8, 1526–1535. [Google Scholar] [CrossRef] [PubMed]
- Cazet, A.; Groux-Degroote, S.; Teylaert, B.; Kwon, K.M.; Lehoux, S.; Slomianny, C.; Kim, C.H.; le Bourhis, X.; Delannoy, P. GD3 synthase overexpression enhances proliferation and migration of MDA-MB-231 breast cancer cells. Biol. Chem. 2009, 390, 601–609. [Google Scholar] [CrossRef] [PubMed]
- Cazet, A.; Bobowski, M.; Rombouts, Y.; Lefebvre, J.; Steenackers, A.; Popa, I.; Guerardel, Y.; le Bourhis, X.; Tulasne, D.; Delannoy, P. The ganglioside GD2 induces the constitutive activation of c-Met in MDA-MB-231 breast cancer cells expressing the GD3 synthase. Glycobiology 2012, 22, 806–816. [Google Scholar] [CrossRef] [PubMed]
- Yates, A.J.; Saqr, H.E.; van Brocklyn, J. Ganglioside modulation of the PDGF receptor. A model for ganglioside functions. J. Neurooncol. 1995, 24, 65–73. [Google Scholar] [CrossRef] [PubMed]
- Hynds, D.L.; Summers, M.; van Brocklyn, J.; O’Dorisio, M.S.; Yates, A.J. Gangliosides inhibit platelet-derived growth factor-stimulated growth, receptor phosphorylation, and dimerization in neuroblastoma SH-SY5Y cells. J. Neurochem. 1995, 65, 2251–2258. [Google Scholar] [CrossRef] [PubMed]
- Lang, Z.; Guerrera, M.; Li, R.; Ladisch, S. Ganglioside GD1a enhances VEGF-induced endothelial cell proliferation and migration. Biochem. Biophys. Res. Commun. 2001, 282, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Chung, T.W.; Kim, S.J.; Choi, H.J.; Kim, K.J.; Kim, M.J.; Kim, S.H.; Lee, H.J.; Ko, J.H.; Lee, Y.C.; Suzuki, A.; et al. Ganglioside GM3 inhibits VEGF/VEGFR-2-mediated angiogenesis: Direct interaction of GM3 with VEGFR-2. Glycobiology 2009, 19, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Kabayama, K.; Sato, T.; Kitamura, F.; Uemura, S.; Kang, B.W.; Igarashi, Y.; Inokuchi, J. TNFα-induced insulin resistance in adipocytes as a membrane microdomain disorder: Involvement of ganglioside GM3. Glycobiology 2005, 15, 21–29. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, A.; Hata, K.; Suzuki, S.; Sawada, M.; Wada, T.; Yamaguchi, K.; Obinata, M.; Tateno, H.; Suzuki, H.; Miyagi, T. Overexpression of plasma membrane-associated sialidase attenuates insulin signaling in transgenic mice. J. Biol. Chem. 2003, 278, 27896–27902. [Google Scholar] [CrossRef] [PubMed]
- Kaucic, K.; Liu, Y.; Ladisch, S. Modulation of growth factor signaling by gangliosides: Positive or negative? Methods Enzymol. 2006, 417, 168–185. [Google Scholar] [PubMed]
- Farooqui, T.; Franklin, T.; Pearl, D.K.; Yates, A.J. Ganglioside GM1 enhances induction by nerve growth factor of a putative dimer of TrkA. J. Neurochem. 1997, 68, 2348–2355. [Google Scholar] [CrossRef] [PubMed]
- Mutoh, T.; Hamano, T.; Tokuda, A.; Kuriyama, M. Unglycosylated Trk protein does not co-localize nor associate with ganglioside GM1 in stable clone of PC12 cells overexpressing Trk (PCtrk cells). Glycoconj. J. 2000, 17, 233–237. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, N.; Iwabuchi, K.; Kurihara, H.; Ishii, K.; Kobayashi, T.; Sasaki, T.; Hattori, N.; Mizuno, Y.; Hozumi, K.; Yamada, Y.; et al. Binding of laminin-1 to monosialoganglioside GM1 in lipid rafts is crucial for neurite outgrowth. J. Cell Sci. 2009, 122, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Maglione, V.; Marchi, P.; Di Pardo, A.; Lingrell, S.; Horkey, M.; Tidmarsh, E.; Sipione, S. Impaired ganglioside metabolism in Huntington’s disease and neuroprotective role of GM1. J. Neurosci. 2010, 30, 4072–4080. [Google Scholar] [CrossRef] [PubMed]
- Di Pardo, A.; Maglione, V.; Alpaugh, M.; Horkey, M.; Atwal, R.S.; Sassone, J.; Ciammola, A.; Steffan, J.S.; Fouad, K.; Truant, R.; et al. Ganglioside GM1 induces phosphorylation of mutant huntingtin and restores normal motor behavior in Huntington disease mice. Proc. Natl. Acad. Sci. USA 2012, 109, 3528–3533. [Google Scholar] [CrossRef] [PubMed]
- Nojiri, H.; Stroud, M.; Hakomori, S. A specific type of ganglioside as a modulator of insulin-dependent cell growth and insulin receptor tyrosine kinase activity: Possible association of ganglioside-induced inhibition of insulin receptor function and monocytic differentiation induction in HL-60 cells. J. Biol. Chem. 1991, 266, 4531–4537. [Google Scholar] [PubMed]
- Sato, T.; Nihei, Y.; Nagafuku, M.; Tagami, S.; Chin, R.; Kawamura, M.; Miyazaki, S.; Suzuki, M.; Sugahara, S.; Takahashi, Y.; et al. Circulating levels of ganglioside GM3 in metabolic syndrome: A pilot study. Obes. Res. Clin. Pract. 2008, 2. [Google Scholar] [CrossRef] [PubMed]
- Veillon, L.; Go, S.; Matsuyama, W.; Suzuki, A.; Nagasaki, M.; Yatomi, Y.; Inokuchi, J. Identification of ganglioside GM3 molecular species in human serum associated with risk factors of metabolic syndrome. PLoS ONE 2015, 10, e0129645. [Google Scholar] [CrossRef] [PubMed]
- Merrill, A.H., Jr.; Stokes, T.H.; Momin, A.; Park, H.; Portz, B.J.; Kelly, S.; Wang, E.; Sullards, M.C.; Wang, M.D. Sphingolipidomics: A valuable tool for understanding the roles of sphingolipids in biology and disease. J. Lipid Res. 2009, 50, S97–S102. [Google Scholar] [CrossRef] [PubMed]
- Hannun, Y.A.; Obeid, L.M. Many ceramides. J. Biol. Chem. 2011, 286, 27855–27862. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Hakomori, S.; Handa, K.; Iwabuchi, K.; Yamamura, S.; Prinetti, A. New insights in glycosphingolipid function: “glycosignaling domain”, a cell surface assembly of glycosphingolipids with signal transducer molecules,involved in cell adhesion coupled with signaling. Glycobiology 1998, 8, xi–xix. [Google Scholar] [CrossRef] [PubMed]
- Regina Todeschini, A.; Hakomori, S.I. Functional role of glycosphingolipids and gangliosides in control of cell adhesion, motility, and growth, through glycosynaptic microdomains. Biochim. Biophys. Acta 2008, 1780, 421–433. [Google Scholar] [CrossRef] [PubMed]
- Mahfoud, R.; Garmy, N.; Maresca, M.; Yahi, N.; Puigserver, A.; Fantini, J. Identification of a common sphingolipid-binding domain in Alzheimer, prion, and HIV-1 proteins. J. Biol. Chem. 2002, 277, 11292–11296. [Google Scholar] [CrossRef] [PubMed]
- Lingwood, C.A.; Branch, D.R. The role of glycosphingolipids in HIV/AIDS. Discov. Med. 2011, 11, 303–313. [Google Scholar] [PubMed]
- Nehete, P.N.; Vela, E.M.; Hossain, M.M.; Sarkar, A.K.; Yahi, N.; Fantini, J.; Sastry, K.J. A post-CD4-binding step involving interaction of the V3 region of viral gp120 with host cell surface glycosphingolipids is common to entry and infection by diverse HIV-1 strains. Antivir. Res. 2002, 56, 233–251. [Google Scholar] [CrossRef]
- Delezay, O.; Hammache, D.; Fantini, J.; Yahi, N. SPC3, a V3 loop-derived synthetic peptide inhibitor of HIV-1 infection, binds to cell surface glycosphingolipids. Biochemistry 1996, 35, 15663–15671. [Google Scholar] [CrossRef] [PubMed]
- Fantini, J.; Yahi, N. Molecular basis for the glycosphingolipid-binding specificity of α-synuclein: Key role of tyrosine 39 in membrane insertion. J. Mol. Biol. 2011, 408, 654–669. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, A.; Paila, Y.D.; Shrivastava, S.; Tiwari, S.; Singh, P.; Fantini, J. Sphingolipid-binding domain in the serotonin1A receptor. Adv. Exp. Med. Biol. 2012, 749, 279–293. [Google Scholar] [PubMed]
- Maresca, M.; Derghal, A.; Caravagna, C.; Dudin, S.; Fantini, J. Controlled aggregation of adenine by sugars: Physicochemical studies, molecular modelling simulations of sugar-aromatic CH-pi stacking interactions, and biological significance. Phys. Chem. Chem. Phys. 2008, 10, 2792–2800. [Google Scholar] [CrossRef] [PubMed]
- Soltyk, A.M.; MacKenzie, C.R.; Wolski, V.M.; Hirama, T.; Kitov, P.I.; Bundle, D.R.; Brunton, J.L. A mutational analysis of the globotriaosylceramide-binding sites of verotoxin VT1. J. Biol. Chem. 2002, 277, 5351–5359. [Google Scholar] [CrossRef] [PubMed]
- Ling, H.; Boodhoo, A.; Hazes, B.; Cummings, M.D.; Armstrong, G.D.; Brunton, J.L.; Read, R.J. Structure of the shiga-like toxin I B-pentamer complexed with an analogue of its receptor Gb3. Biochemistry 1998, 37, 1777–1788. [Google Scholar] [CrossRef] [PubMed]
- Maloney, M.D.; Lingwood, C.A. CD19 has a potential CD77 (globotriaosyl ceramide)-binding site with sequence similarity to verotoxin B-subunits: Implications of molecular mimicry for B cell adhesion and enterohemorrhagic Escherichia coli pathogenesis. J. Exp. Med. 1994, 180, 191–201. [Google Scholar] [CrossRef] [PubMed]
- D’Angelo, G.; Capasso, S.; Sticco, L.; Russo, D. Glycosphingolipids: Synthesis and functions. FEBS J. 2013, 280, 6338–6353. [Google Scholar] [CrossRef] [PubMed]
- Ichikawa, S.; Nakajo, N.; Sakiyama, H.; Hirabayashi, Y. A mouse B16 melanoma mutant deficient in glycolipids. Proc. Natl. Acad. Sci. USA 1994, 91, 2703–2707. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.J.; Kuo, H.H.; Lin, C.H.; Chen, Y.Y.; Yang, B.C.; Cheng, Y.Y.; Yu, A.L.; Khoo, K.H.; Yu, J. Switching of the core structures of glycosphingolipids from globo- and lacto- to ganglio-series upon human embryonic stem cell differentiation. Proc. Natl. Acad. Sci. USA 2010, 107, 22564–22569. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Haynes, C.A.; Nairn, A.V.; Kulik, M.; Dalton, S.; Moremen, K.; Merrill, A.H., Jr. Transcript profiling and lipidomic analysis of ceramide subspecies in mouse embryonic stem cells and embryoid bodies. J. Lipid Res. 2010, 51, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.J.; Yang, B.C.; Chen, J.M.; Lin, Y.H.; Huang, C.L.; Cheng, Y.Y.; Hsu, C.Y.; Khoo, K.H.; Shen, C.N.; Yu, J. Changes in glycosphingolipid composition during differentiation of human embryonic stem cells to ectodermal or endodermal lineages. Stem Cells 2011, 29, 1995–2004. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.K.; Tsai, Y.T.; Ariga, T. Functional roles of gangliosides in neurodevelopment: An overview of recent advances. Neurochem. Res. 2012, 37, 1230–1244. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, Y.; Yanagisawa, M.; Yagi, H.; Nakatani, Y.; Yu, R.K. Involvement of β1-integrin up-regulation in basic fibroblast growth factor- and epidermal growth factor-induced proliferation of mouse neuroepithelial cells. J. Biol. Chem. 2010, 285, 18443–18451. [Google Scholar] [CrossRef] [PubMed]
- Tsai, Y.T.; Yu, R.K. Epigenetic activation of mouse ganglioside synthase genes: Implications for neurogenesis. J. Neurochem. 2014, 128, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Gurdon, J.B.; Bourillot, P.Y. Morphogen gradient interpretation. Nature 2001, 413, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Wegner, M.S.; Schiffmann, S.; Parnham, M.J.; Geisslinger, G.; Grosch, S. The enigma of ceramide synthase regulation in mammalian cells. Prog. Lipid Res. 2016, 63, 93–119. [Google Scholar] [CrossRef] [PubMed]
- Borbat, P.P.; Costa-Filho, A.J.; Earle, K.A.; Moscicki, J.K.; Freed, J.H. Electron spin resonance in studies of membranes and proteins. Science 2001, 291, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Marsh, D.; Horvath, L.I. Structure, dynamics and composition of the lipid-protein interface: Perspectives from spin-labelling. Biochim. Biophys. Acta 1998, 1376, 267–296. [Google Scholar] [CrossRef]
- Ionova, I.V.; Livshits, V.A.; Marsh, D. Phase diagram of ternary cholesterol/palmitoylsphingomyelin/palmitoyloleoyl-phosphatidylcholine mixtures: Spin-label EPR study of lipid-raft formation. Biophys. J. 2012, 102, 1856–1865. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Lindahl, P.A. Effect of sodium sulfide on Ni-containing carbon monoxide dehydrogenases. J. Am. Chem. Soc. 2004, 126, 9094–9100. [Google Scholar] [CrossRef] [PubMed]
- Esmann, M.; Marsh, D.; Schwarzmann, G.; Sandhoff, K. Ganglioside-protein interactions: Spin-label electron spin resonance studies with (Na+, K+)-ATPase membranes. Biochemistry 1988, 27, 2398–2403. [Google Scholar]
- Haberkant, P.; Schmitt, O.; Contreras, F.X.; Thiele, C.; Hanada, K.; Sprong, H.; Reinhard, C.; Wieland, F.T.; Brugger, B. Protein-sphingolipid interactions within cellular membranes. J. Lipid Res. 2008, 49, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Haberkant, P.; Stein, F.; Hoglinger, D.; Gerl, M.J.; Brugger, B.; van Veldhoven, P.P.; Krijgsveld, J.; Gavin, A.C.; Schultz, C. Bifunctional sphingosine for cell-based analysis of protein-sphingolipid interactions. ACS Chem. Biol. 2016, 11, 222–230. [Google Scholar] [CrossRef] [PubMed]
| Virus | Glycosphingolipid Receptors | References |
| Picornaviridae | ||
| Porcine sapelovirus (PSV) | GD1a | [30] |
| Caliciviridae | ||
| Human Norovirus (HuNoV) | Type 1, 2, 3 HBGA | [31,32] |
| Human Norovirus (HuNoV): GII.4 strain | H, B, and A type 1 Lewis b | [33] |
| Murine Norovirus (MNV): MNV-1 and CR3 strains | GD1a; GT1b | [34,35] |
| Bovine Norovirus (BoNoV) | HBGA | [36] |
| Rabbit Hemorrhagic Disease Virus (RHDV) | A and H Type 2 HBGA | [37] |
| Adenoviridae | ||
| Adenovirus type 37 (Ad37) | GD1a | [38] |
| Reoviridae | ||
| Reovirus serotype 1 (T1) | GM2 | [39] |
| Porcine Rotavirus: OSU strain | GM3 | [40,41] |
| Porcine Rotavirus: CRW-8 strain | GD1a | [42] |
| Porcine Rotavirus: TFR-41 strain | Unknown ganglioside | [43] |
| Simian Rotavirus: SA11 strain | NeuGcGM3, IV3NeuAcLc4, GM2, GD1a | [44] |
| Simian Rhesus Rotavirus: RRV strain | Unknown ganglioside | [43,45] |
| Bovine Rotavirus: NCVD strain | NeuGcGM3, IV3NeuAcLc4, GM2, GD1a | [44,46] |
| Bovine Rotavirus: UK strain | NeuGcGM3, GM1, GD1a, GM2, IV3NeuAcLc4 | [43,46] |
| Human Rotavirus: KU, MO, DS-1 and Wa strains | GM3, GM1 | [42,43,47,48,49] |
| Polyomaviridae | ||
| Trichodysplasia spinulosa-associated Polyomavirus (TSPyV) | GM1 | [50] |
| Murine Polyomavirus (MPyV) | GD1a, GT1b | [51,52] |
| Simian Virus 40 (SV40) | GM1 | [44,53,54] |
| BK Virus (BKV) | GD1b, GT1b | [55] |
| JC Virus (JCV) | GT1b | [56] |
| Merkel Cell Polyomavirus (MCPyV) | GT1b | [57] |
| Parvoviridae | ||
| Human Parvovirus B19 | Gb4, SSEA-3, SSEA-4, nLc4 | [58,59] |
| Simian Parvovirus | Gb4; Forssmann antigen | [60] |
| Bovine Adeno-associated Virus (BAAV) | Unknown ganglioside | [61] |
| Retroviridae | ||
| Human Immunodeficiency Virus (HIV) | Gb3, GM3, GalCer, GD3, SM4 sulfatide | [62,63,64,65,66,67,68,69] |
| Flaviviridae | ||
| Dengue virus (DENV) type 2 | GM3, nLc4 | [70,71,72] |
| Orthomyxoviridae | ||
| Influenza A virus, subtype H3N2: A/Victoria/3/75 strain | Ganglioside with Neu5Acα2-3Galβ1-4 (Fucα1-3) GlcNAc epitope; nLc8, nLc10 and nLc12 | [73] |
| Influenza A virus, subtype H3N2: A/Hiroshima/52/2005 strain | ||
| Poxviridae | ||
| Vaccinia virus (VACV): Western-Reserve strain | SM4 sulfatide | [74,75] |
| Paramyxoviridae | ||
| ParamyxoVirus 1 (Newcastle Disease) | GM3, GM2, GM1, GD1a | [76] |
| Sendai virus (SV) (murine parainfluenza virus type 1) | GD1a, GQ1b, IV3NeuAcLc4, nLC4 | [77,78] |
| Human parainfluenza virus types 1 (Hpiv-1) | IV3NeuAcLc4, Nlc4 | [78] |
| Human parainfluenza virus types 3 (hPIV-3) | ||
| Bacterial Toxin | Glycosphingolipid Receptors | References |
| Cholera toxin Vibrio cholera | GM1 | [79,80] |
| Heat labile toxin 1 Escherichia coli | GM1 | [81] |
| Shiga Toxin Shighella dysenteriae | Gb3 | [82,83] |
| Shiga-like toxins (SLT1 and SLT 2) Escherichia coli (Verotoxins) | Gb4 | [84,85] |
| Tetanus neurotoxin (TeNT) Clostridium tetani | GT1b, GD1b | [86] |
| Botulinium toxin BoNT Clostridium botulinum | GT1b, GD1a | [87] |
| Heat labile toxin IIB Escherichia coli | GD1a | [88] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Russo, D.; Parashuraman, S.; D’Angelo, G. Glycosphingolipid–Protein Interaction in Signal Transduction. Int. J. Mol. Sci. 2016, 17, 1732. https://doi.org/10.3390/ijms17101732
Russo D, Parashuraman S, D’Angelo G. Glycosphingolipid–Protein Interaction in Signal Transduction. International Journal of Molecular Sciences. 2016; 17(10):1732. https://doi.org/10.3390/ijms17101732
Chicago/Turabian StyleRusso, Domenico, Seetharaman Parashuraman, and Giovanni D’Angelo. 2016. "Glycosphingolipid–Protein Interaction in Signal Transduction" International Journal of Molecular Sciences 17, no. 10: 1732. https://doi.org/10.3390/ijms17101732
APA StyleRusso, D., Parashuraman, S., & D’Angelo, G. (2016). Glycosphingolipid–Protein Interaction in Signal Transduction. International Journal of Molecular Sciences, 17(10), 1732. https://doi.org/10.3390/ijms17101732





