1. Introduction
Hepatocellular carcinoma (HCC), which usually develops in the cirrhotic liver, is one of the most common malignancies worldwide [
1,
2]. It is essential to be able to predict the patient’s prognosis in order to select the most adequate treatments for malignancies such as HCC. However, outcome prediction is extremely complicated for HCC because the liver functional reserve influences prognosis, sometimes to a greater extent than does the progression of HCC itself [
3]. Further, the estimation of tumor stage alone is insufficient for predicting the prognosis of HCC since this malignancy very frequently recurs after curative treatment [
4]. In order to predict the prognosis of HCC more precisely, several prognostic staging systems have been developed, such as the Barcelona Clinic Liver Cancer (BCLC) [
5], Cancer of the Liver Italian Program (CLIP) [
6], and Japan Integrated Staging (JIS) [
7] systems. These staging systems are suitable for predicting HCC prognosis because they incorporate both tumor stages and liver functional reserves. However, it is also vital to continually make efforts to identify additional useful and simple factors that predict the prognosis of HCC.
Skeletal muscle depletion, or sarcopenia, was initially defined as the loss of skeletal muscle mass that occurs with aging [
8]. Skeletal muscle depletion has been critically implicated in a variety of pathological conditions, such as malnutrition, advanced organ failure, and inflammatory disease [
9]. It has also been identified as a poor prognostic factor for various types of human malignancies [
10,
11,
12,
13]. Particularly, cases of liver cirrhosis and HCC are frequently complicated by sarcopenia, which is associated with a poor prognosis for these diseases [
14,
15,
16,
17]. In a study of 217 consecutive patients with all stages of HCC, we found a strong relationship between skeletal muscle depletion and poor prognosis [
16]. These previous findings, including our own data, suggest that skeletal muscle mass could be a useful and objective indicator for predicting the prognosis of patients with HCC.
Sorafenib (Nexavar, Bayer HealthCare Pharmaceuticals, Leverkusen, Germany) is the first orally active multi-kinase inhibitor that has been confirmed to be efficacious against advanced HCC [
18,
19]. Sorafenib has become the standard first-line treatment for patients with advanced HCC, but it sometimes causes severe side effects, such as hand-foot syndrome and liver dysfunction, which may limit the patient’s ability to continue full-dose treatment [
20,
21]. A recent clinical trial showed that sarcopenia predicts early dose-limiting toxicities in patients who have advanced HCC with Child-Pugh A liver cirrhosis [
20]. Therefore, we hypothesized that the poor prognosis of patients with sarcopenia and HCC might be caused by early dose reductions or the discontinuation of sorafenib due to its increased toxicity.
In the present study, we used computed tomography (CT) to measure the skeletal muscle cross-sectional areas of patients with HCC just before they began to receive treatment with sorafenib. Further, we designed a retrospective analysis to identify factors, including skeletal muscle mass, that could influence the overall survival of consecutive HCC patients treated with sorafenib. Finally, we have discussed the mechanisms by which skeletal muscle depletion affects mortality in HCC and the actions that should be taken to prevent sarcopenia in HCC patients treated with sorafenib
3. Discussion
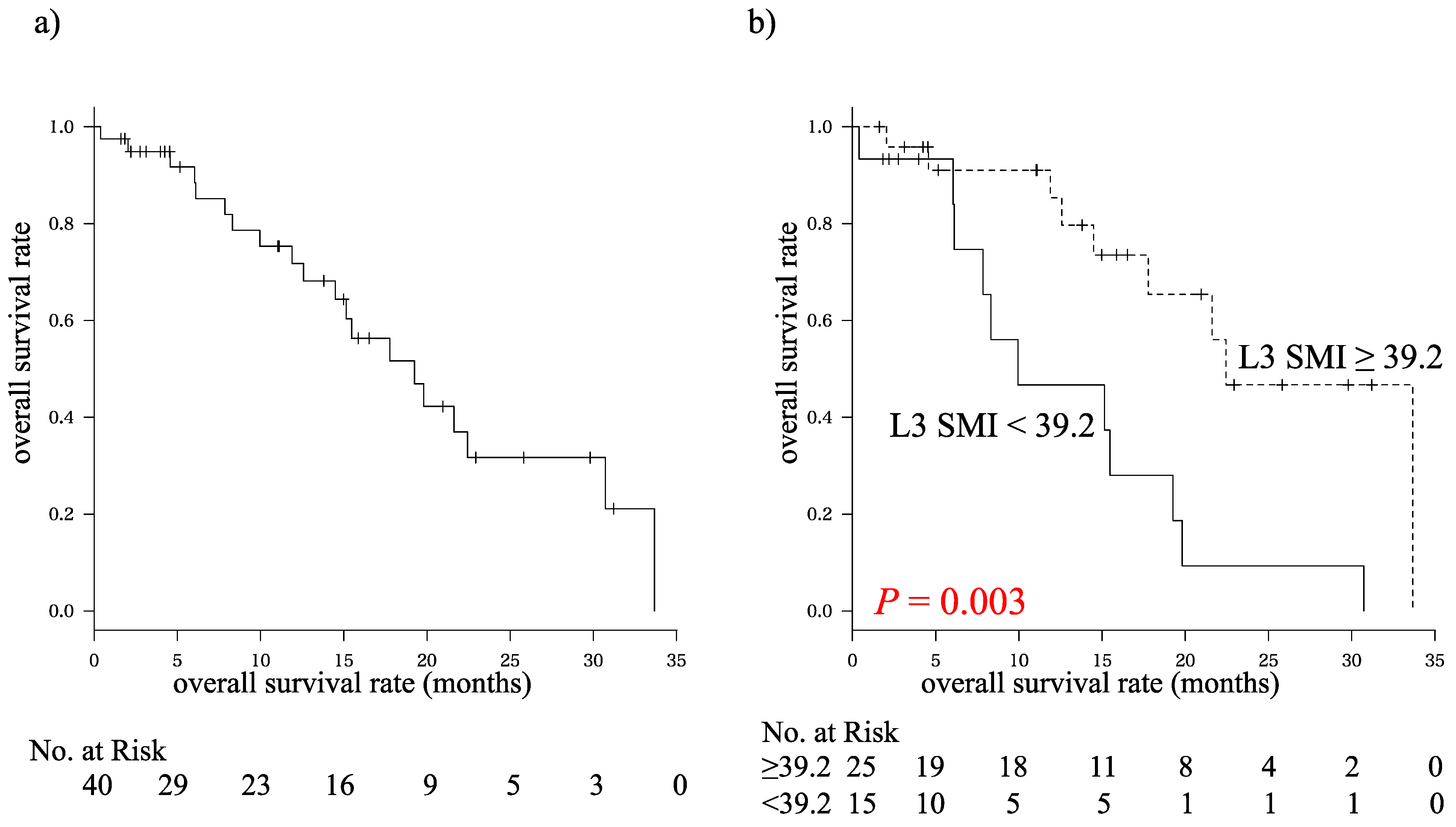
The results of the present study provide the first clear evidence that L3 SMI, an indicator of skeletal muscle mass or sarcopenia, is an independent prognostic factor for patients who have HCC and are being treated with sorafenib. These findings agree with previous studies that have shown significant relationships between skeletal muscle depletion and poor prognosis in patients with various clinical stages of HCC [
14,
15,
16,
17].
In the present study, we also obtained a regression equation that can be used to estimate L3 SMI from three explanatory factors: age, Child-Pugh score, and log (AFP). It has previously been reported that these factors may be associated with loss of skeletal muscle mass [
9,
22,
23]. This analysis indicates that L3 SMI decreases in correspondence with increasing age, worsening liver functional reserve, and elevated AFP levels in patients with HCC who receive sorafenib. The interaction effect between log (AFP) and Child-Pugh score was quite small and can be ignored because of the large differences between the coefficient of the interaction effect and the coefficients of the associated main effects. Indeed, the combined value of the log (AFP) term, the Child-Pugh score term, and the interaction term (1.6139 × (log (AFP)) × (Child-Pugh score) − 10.3441 × (Child-Pugh score) − 9.3922 × (log (AFP))) was far less than 0 in all patients (ranging from −66.46 to −52.82). The regression equation could present the key to explaining the association between skeletal muscle depletion and poor prognosis because liver functional reserve and AFP levels are both well-known prognostic factors for HCC [
3]. Moreover, L3 SMI was affected by ageing, which itself affects the prognosis of patients with most medical conditions, even though existing prognostic staging systems for HCC (such as BCLC, CLIP, and JIS) do not include an aging parameter [
5,
6,
7]. Thus, the evaluation of skeletal muscle mass using CT imaging could present a useful alternative method of predicting the prognosis of patients with HCC.
A recent clinical study reported that sarcopenia predicts early dose-limiting sorafenib toxicities, which might be involved in dose reductions or the discontinuation of this drug [
20]. Therefore, we initially speculated that the more favorable prognosis of HCC patients without skeletal muscle depletion was associated with the greater extent of continuous sorafenib treatment that they received. However, contrary to this expectation, the loss of L3 SMI did not affect dose reductions or the discontinuation of sorafenib in the present study. We consider this finding to be explained by the initial dose of sorafenib, which was comparatively small. Specifically, the dose was less than 400 mg per day in most cases (95%). These low doses might strengthen the patients’ adherence to sorafenib and, consequently, improve the prognosis of these patients in the present study. On the other hand, it should be noted that the initial dose was a risk factor for dose reduction. Further, we found that log (AFP) was a risk factor for the discontinuation of sorafenib. Therefore, it is necessary to be especially careful when facing cases of sorafenib-treated HCC that involve initial doses of 800 mg per day or high AFP levels. In such cases, adverse events that require dose reductions or discontinuation may occur with a high probability.
Importantly, the survival outcomes of the present study were quite favorable as compared with a previous phase III trial by Llovet
et al. In the present and previous studies, the median overall survival times were 19.5 and 10.7 months, respectively, and the survival rates at 1 year were 71.7% and 44.0%, respectively [
18]. The positive outcomes in our study may result from several factors. First, sorafenib was introduced at earlier tumor stage in some cases. Indeed, in three cases, it was used before obvious evidence of recurrent lesion had emerged, although pathological assessments of the resected specimen revealed either vascular invasion or focal positivity. Second, a combination treatment using sorafenib was administered in 29 cases (72.5%). It is also interesting that, among the patients who received combination treatment, six cases (20.6%) showed increased levels of skeletal muscle mass during this study, whereas only one case (9.1%) showed this phenomenon in the group of patients who were treated with sorafenib alone. Combined treatment with sorafenib and TACE has a strong scientific rationale because the ischemia that is induced by TACE results in local and systemic increases in vascular endothelial growth factor (VEGF), whereas sorafenib inhibits the activity of the VEGF receptor [
24]. Several meta-analyses have shown a positive outcome in HCC patients treated with TACE and sorafenib, as compared with HCC patients treated with TACE alone [
25,
26,
27]. Currently, several randomized, controlled phase III trials of the safety and efficacy of sorafenib-based combination treatment are ongoing. These basic and clinical studies may provide new evidence that combination treatment with sorafenib is a promising approach for improving the prognosis of patients with HCC. However, additional careful investigation is necessary to verify the safety and efficacy of this combination treatment.
As demonstrated in the present study, skeletal muscle depletion seems to be an inevitable consequence of aging. On the other hand, it is possible that the prevention of skeletal muscle depletion might be an effective strategy for improving the prognosis of HCC; this approach should be considered further. Two possible interventions could prevent sarcopenia: nutritional and exercise therapies. Both of these interventions have been shown to improve outcomes for cirrhotic patients [
28,
29]. In particular, oral supplementation with branched-chain amino acids (BCAA; leucine, isoleucine and valine) is an effective nutritional therapy that can ameliorate protein energy malnutrition and improve event-free survival in cirrhotic patients [
30,
31,
32]. BCAA supplementation is one of the most promising methods because it has been observed to improve the survival of sarcopenic patients with liver cirrhosis [
33]. In a study of patients with alcoholic cirrhosis, Tsien C
et al. demonstrated that impaired mTOR1 signaling and increased autophagy in skeletal muscle, which contribute progression of sarcopenia, are reversed by BCAA enriched with leucine [
34]. Furthermore, the combination of a low intensity exercise and leucine-rich essential amino acid protein administration is effective for enhancing not only muscle strength, but also muscle mass and walking speed in sarcopenic women [
35]. These reports suggest that BCAA supplementation, taking proper exercise, and the combination of these interventions might be effective for preventing sarcopenia. Future large-scale interventional studies should be conducted to determine whether nutritional and exercise therapies can prevent skeletal muscle depletion and possibly improve the prognosis of patients with HCC.
In summary, this study provides the first demonstration that L3 SMI, an indicator of skeletal muscle mass, is independently associated with poor prognosis among patients who have HCC and receive sorafenib treatment. In contrast, we found that L3 SMI was not a significant risk factor for dose reduction or the discontinuation of sorafenib. In addition, a multiple linear regression analysis suggested that L3 SMI was inversely associated with age, log (AFP), and Child-Pugh score, which indicated that these L3 SMI-correlated clinical features are also critical prognostic factors for patients with HCC. In conclusion, the evaluation of skeletal muscle mass using CT imaging could present a useful method of predicting the prognosis of patients with HCC, including those who are treated with sorafenib.