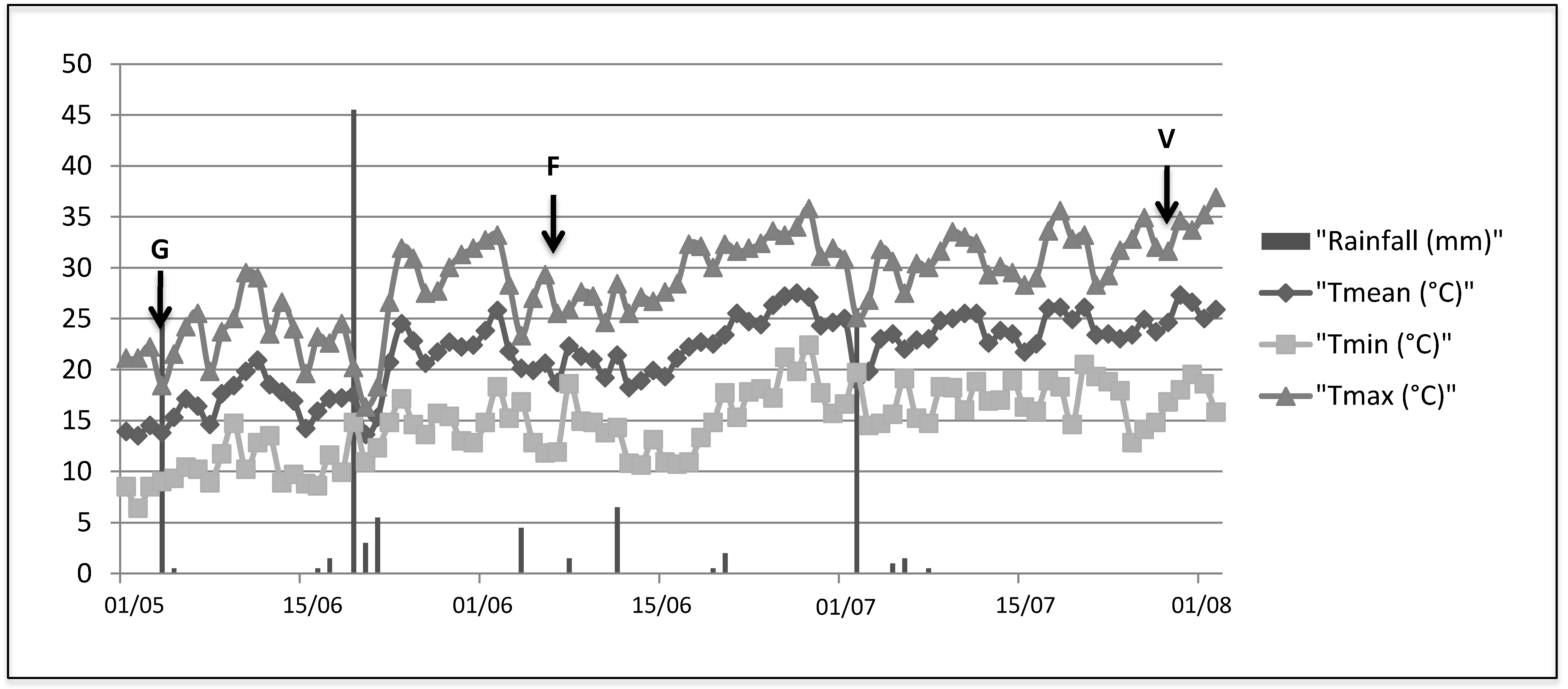
Flowering as the Most Highly Sensitive Period of Grapevine (Vitis vinifera L. cv Mourvèdre) to the Botryosphaeria Dieback Agents Neofusicoccum parvum and Diplodia seriata Infection
Abstract
:1. Introduction
2. Results and Discussion
2.1. Pathogenicity Tests
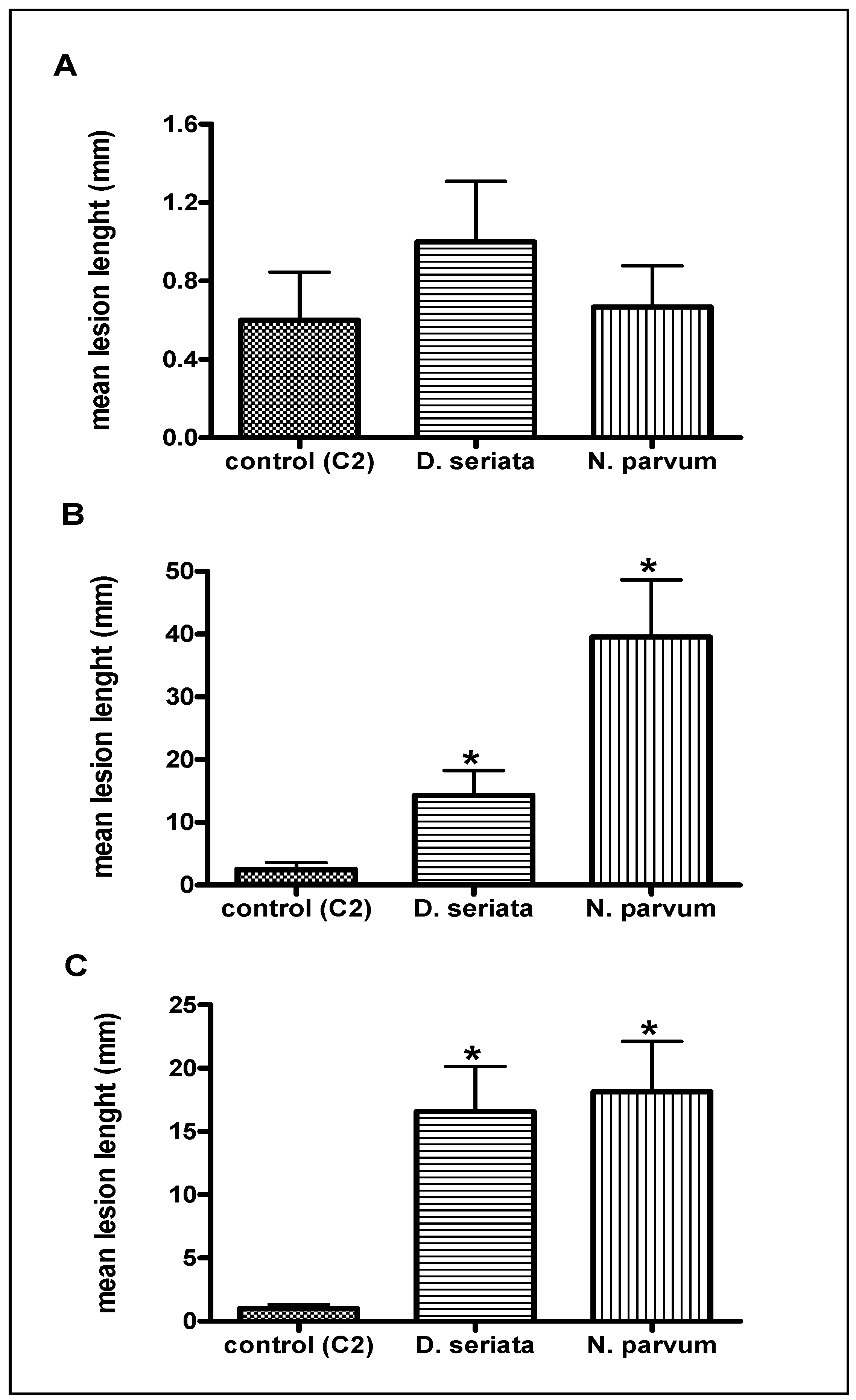
2.2. Two-Dimensional Gel Electrophoresis (2D) Analysis
2.2.1. Identification of Differentially Expressed Protein Spots by nanoLC-MS/MS Analysis

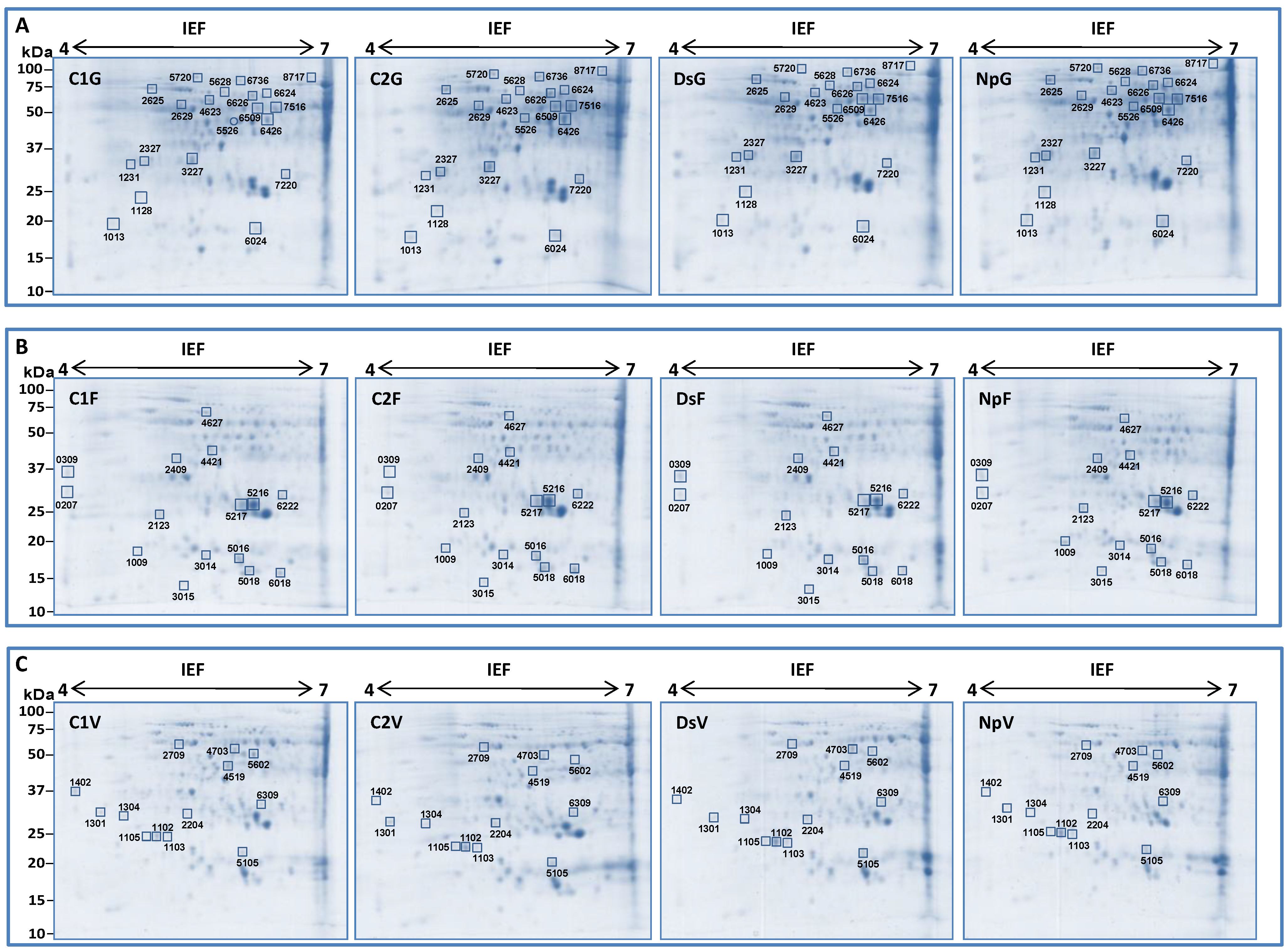
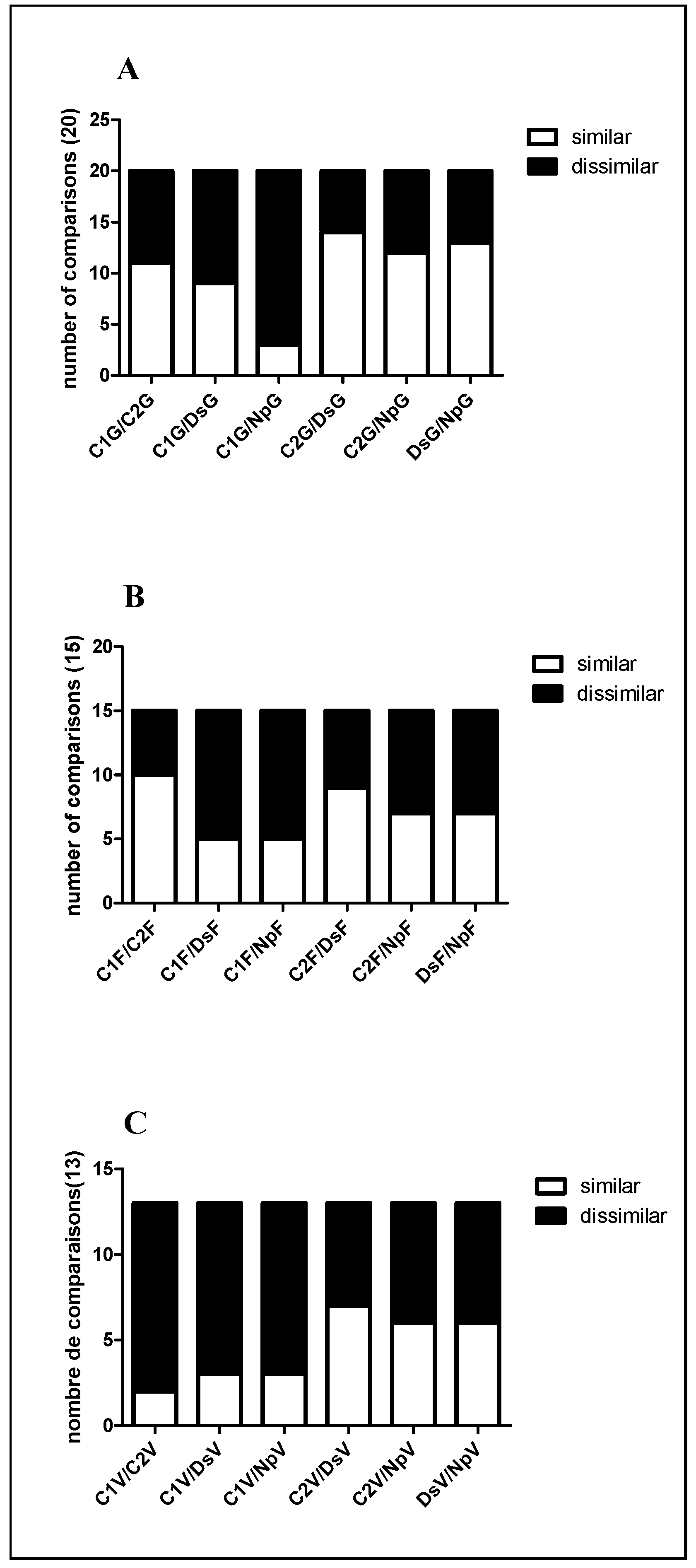
2.2.2. Differentially Expressed Proteins among the Treatments
G Stage (Separated Clusters)
Flowering
Veraison
| Spot a | Ratio to C1G b | Matched Protein c | Accession Number d | Cov. % e | MW f | Functional Category | ||
|---|---|---|---|---|---|---|---|---|
| C2G | DsG | NpG | ||||||
| 5526 | * | * | * | S-adenosylmethionine synthase 2 | gi|223635284 | 40 | 24.70 | Defense and cell rescue |
| 5526 | * | * | * | eukaryotic initiation factor 4A-2 (Vitis vinifera) | gi|225442221 | 46 | 53.30 | Protein synthesis |
| 5526 | * | * | * | protein disulfide isomerase-like 2-3 (Vitis vinifera) | gi|225447176 | 46 | 32.80 | Protein processing |
| 5526 | * | * | * | peroxidase 12-like (Vitis vinifera) | gi|359493149 | 39 | 33.10 | Defense and cell rescue |
| 1013 | 9.0 | 4.6 | 9.5 | actin-depolymerizing factor 2-like isoform 1 (Vitis vinifera) | gi|225435040 | 16 | 13.70 | Cytoskeleton |
| 1128 | 3.0 | 7.6 | 8.3 | 2-Cys peroxiredoxin (Vitis vinifera) | gi|147789752 | 30 | 52.70 | Defense and cell rescue |
| 1128 | 3.0 | 7.6 | 8.3 | oxygen-evolving enhancer protein 1, chloroplastic (Vitis vinifera) | gi|147791852 | 33 | 20.50 | Photosynthesis |
| 1231 | 4.1 | 12.3 | 4.5 | 60S acidic ribosomal protein P0 (Vitis vinifera) | gi|147843260 | 34 | 12.50 | Protein synthesis |
| 1231 | 4.1 | 12.3 | 4.5 | elongation factor 1-beta 1 (Vitis vinifera) | gi|29608391 | 27 | 38.30 | Protein synthesis |
| 1231 | 4.1 | 12.3 | 4.5 | 14-3-3 protein (Vitis vinifera) | gi|359492889 | 29 | 66.20 | Signal transduction |
| 6624 | 2.4 | 2.6 | 4.5 | phosphoglucomutase, cytoplasmic (Vitis vinifera) | gi|225424316 | 63 | 26.60 | Glycolysis/Gluconeogenesis |
| 6624 | 2.4 | 2.6 | 4.5 | succinate dehydrogenase (ubiquinone) flavoprotein subunit 1, mitochondrial isoform 1 (Vitis vinifera) | gi|225430776 | 73 | 38.80 | Citrate cycle |
| 6624 | 2.4 | 2.6 | 4.5 | 2,3-bisphosphoglycerate-independent phosphoglycerate mutase isoform 1 (Vitis vinifera) | gi|225439064 | 61 | 24.50 | Glycolysis/Gluconeogenesis |
| 6736 | 3.2 | 3.3 | 3.4 | eukaryotic initiation factor 4A-2 (Vitis vinifera) | gi|225442221 | 46 | 22.80 | Protein synthesis |
| 6736 | 3.2 | 3.3 | 3.4 | phospholipase D alpha 1 (Vitis vinifera) | gi|225442981 | 92 | 41.90 | Glycan metabolism |
| 6736 | 3.2 | 3.3 | 3.4 | ATP-dependent Clp protease ATP-binding subunit clpA homolog CD4A, chloroplastic-like (Vitis vinifera) | gi|225456471 | 102 | 46.50 | Cell growth and death |
| 8717 | 3.0 | 2.0 | 2.8 | oxygen-evolving enhancer protein 1, chloroplastic (Vitis vinifera) | gi|147791852 | 33 | 36.60 | Photosynthesis |
| 8717 | 3.0 | 2.0 | 2.8 | elongation factor 2-like isoform 1 (Vitis vinifera) | gi|225462164 | 93 | 44.70 | Protein synthesis |
| 6509 | 1.1 | 2.1 | 2.5 | S-adenosylmethionine synthase 2 | gi|223635284 | 43 | 44.00 | Defense and cell rescue |
| 6509 | 1.1 | 2.1 | 2.5 | UDP-glucose 6-dehydrogenase-like isoform 1 (Vitis vinifera) | gi|225423507 | 52 | 23.70 | Pentose phosphate cycle |
| 6509 | 1.1 | 2.1 | 2.5 | adenosylhomocysteinase isoform 1 (Vitis vinifera) | gi|225433506 | 53 | 30.50 | Amino acid metabolism |
| 6509 | 1.1 | 2.1 | 2.5 | enolase 1 (Vitis vinifera) | gi|225441000 | 47 | 56.80 | Glycolysis/Gluconeogenesis |
| 6509 | 1.1 | 2.1 | 2.5 | glutamate decarboxylase-like (Vitis vinifera) | gi|225462892 | 57 | 25.30 | Amino acid metabolism |
| 6024 | 1.3 | 3.0 | 3.5 | pathogenesis-related protein 10 (Vitis hybrid cultivar) | gi|163914213 | 17 | 49.40 | Defense and cell rescue |
| 6024 | 1.3 | 3.0 | 3.5 | universal stress protein A-like protein isoform 1 (Vitis vinifera) | gi|225431940 | 18 | 57.60 | Defense and cell rescue |
| 6024 | 1.3 | 3.0 | 3.5 | eukaryotic translation initiation factor 5A (Vitis vinifera) | gi|225468027 | 17 | 28.10 | Protein synthesis |
| 7516 | 1.2 | 2.0 | 2.4 | Ribulose bisphosphate carboxylase large chain (Vitis vinifera) | gi|134034997 | 6 | 40.00 | photosynthesis |
| 7516 | 1.2 | 2.0 | 2.4 | catalase (Vitis vinifera) | gi|19070130 | 56 | 12.20 | Glyoxylate and dicarboxylate metabolism |
| 7516 | 1.2 | 2.0 | 2.4 | ATPase subunit 1 (Vitis vinifera) | gi|224365668 | 55 | 19.30 | Metabolism and energy |
| 7516 | 1.2 | 2.0 | 2.4 | UDP-glucose 6-dehydrogenase-like isoform 1 (Vitis vinifera) | gi|225423507 | 52 | 24.20 | Pentose phosphate cycle |
| 7516 | 1.2 | 2.0 | 2.4 | adenosylhomocysteinase isoform 1 (Vitis vinifera) | gi|225433506 | 53 | 38.10 | Amino acid metabolism |
| 7516 | 1.2 | 2.0 | 2.4 | enolase 1 (Vitis vinifera) | gi|225441000 | 47 | 40.50 | Glycolysis/Gluconeogenesis |
| 5720 | 2.6 | 1.7 | 2.1 | phospholipase D alpha 1 (Vitis vinifera) | gi|225442981 | 92 | 25.50 | Glycan metabolism |
| 5720 | 2.6 | 1.7 | 2.1 | aminopeptidase N-like (Vitis vinifera) | gi|359474189 | 101 | 50.60 | Amino acid metabolism |
| 3227 | 2.0 | 0.7 | 0.8 | oxygen-evolving enhancer protein 1, chloroplastic (Vitis vinifera) | gi|147791852 | 33 | 57.70 | Photosynthesis |
| 3227 | 2.0 | 0.7 | 0.8 | isoflavone reductase homolog P3 (Vitis vinifera) | gi|225458243 | 33 | 34.40 | Secondary metabolism |
| 3227 | 2.0 | 0.7 | 0.8 | putative fructokinase-5-like (Vitis vinifera) | gi|225459906 | 34 | 23.20 | Carbohydrate metabolism |
| 2327 | 1.3 | 3.4 | 1.6 | ran-binding protein 1 homolog c (Vitis vinifera) | gi|225439378 | 24 | 37.60 | Defense and cell rescue |
| 2327 | 1.3 | 3.4 | 1.6 | 14-3-3 protein 7 (Vitis vinifera) | gi|225459292 | 28 | 66.30 | Signal transduction |
| 2327 | 1.3 | 3.4 | 1.6 | elongation factor 1-beta 1 (Vitis vinifera) | gi|296083911 | 27 | 55.30 | photosynthesis |
| 2327 | 1.3 | 3.4 | 1.6 | aspartic proteinase isoform 2 (Vitis vinifera) | gi|302144105 | 46 | 23.50 | Amino acid metabolism |
| 7220 | 1.1 | 1.6 | 9.4 | triosephosphate isomerase, cytosolic (Vitis vinifera) | gi|147784332 | 27 | 19.30 | Glycolysis/Gluconeogenesis |
| 7220 | 1.1 | 1.6 | 9.4 | l-ascorbate peroxidase 2, cytosolic (Vitis vinifera) | gi|225435177 | 27 | 64.80 | Carbohydrate metabolism |
| 7220 | 1.1 | 1.6 | 9.4 | 3-oxoacyl-(acyl-carrier-protein) reductase 1, chloroplastic (Vitis vinifera) | gi|225456248 | 34 | 24.80 | Fatty acid metabolism |
| 6426 | 1.3 | 1.3 | 2.4 | eukaryotic initiation factor 4A-3-like (Vitis vinifera) | gi|147785805 | 44 | 42.20 | Protein synthesis |
| 6426 | 1.3 | 1.3 | 2.4 | GDP-mannose 3,5-epimerase 1 isoform 1 (Vitis vinifera) | gi|147794688 | 42 | 38.30 | Carbohydrate metabolism |
| 6426 | 1.3 | 1.3 | 2.4 | I-adenosylmethionine synthase 2 | gi|223635284 | 43 | 83.00 | Defense and cell rescue |
| 6426 | 1.3 | 1.3 | 2.4 | alcohol dehydrogenase 1 (Vitis vinifera) | gi|225431505 | 41 | 18.40 | Glycolysis/Gluconeogenesis |
| 6426 | 1.3 | 1.3 | 2.4 | isocitrate dehydrogenase (NADP) (Vitis vinifera) | gi|225466253 | 42 | 50.00 | Citrate cycle |
| 5628 | 1.0 | 1.1 | 2.3 | heat shock 70 kDa protein, mitochondrial-like (Vitis vinifera) | gi|225429228 | 72 | 41.70 | Protein destination |
| 5628 | 1.0 | 1.1 | 2.3 | 2,3-bisphosphoglycerate-independent phosphoglycerate mutase isoform 1 (Vitis vinifera) | gi|225439064 | 61 | 22.20 | Glycolysis/Gluconeogenesis |
| 5628 | 1.0 | 1.1 | 2.3 | isoflavone reductase homolog P3 (Vitis vinifera) | gi|225458243 | 33 | 14.00 | Secondary metabolism |
| 2625 | 1.3 | 1.5 | 2.1 | luminal-binding protein 5-like (Vitis vinifera) | gi|359490716 | 73 | 42.40 | |
| 2625 | 1.3 | 1.5 | 2.1 | heat shock cognate 70 kDa protein isoform 2 (Vitis vinifera) | gi|359486799 | 75 | 38.60 | Protein destination |
| 2625 | 1.3 | 1.5 | 2.1 | stromal 70 kDa heat shock-related protein, chloroplastic-like (Vitis vinifera) | gi|225456004 | 75 | 53.50 | Protein destination |
| 2629 | 1.2 | 0.9 | 2.0 | protein disulfide-isomerase (Vitis vinifera) | gi|225459587 | 55 | 51.50 | Protein processing |
| 2629 | 1.2 | 0.9 | 2.0 | ruBisCO large subunit-binding protein subunit alpha, chloroplastic-like (Vitis vinifera) | gi|359479362 | 61 | 67.60 | photosynthesis |
| 6626 | 1.1 | 1.3 | 2.0 | succinate dehydrogenase (ubiquinone) flavoprotein subunit 1, mitochondrial isoform 1 (Vitis vinifera) | gi|225430776 | 73 | 12.10 | Citrate cycle |
| 6626 | 1.1 | 1.3 | 2.0 | 2,3-bisphosphoglycerate-independent phosphoglycerate mutase isoform 1 (Vitis vinifera) | gi|225439064 | 61 | 64.90 | Glycolysis/Gluconeogenesis |
| 6626 | 1.1 | 1.3 | 2.0 | beta-xylosidase/alpha-l-arabinofuranosidase 2-like (Vitis vinifera) | gi|297745522 | 80 | 16.60 | Carbohydrate metabolism |
| 4623 | 0.7 | 0.6 | 2.0 | d-3-phosphoglycerate dehydrogenase, chloroplastic-like (Vitis vinifera) | gi|225428898 | 62 | 10.60 | Glycolysis/Gluconeogenesis |
| 4623 | 0.7 | 0.6 | 2.0 | chaperonin CPN60-2, mitochondrial isoform 1 (Vitis vinifera) | gi|225433375 | 61 | 68.70 | Protein destination |
| 4623 | 0.7 | 0.6 | 2.0 | stromal 70 kDa heat shock-related protein, chloroplastic-like (Vitis vinifera) | gi|225456004 | 75 | 20.70 | Protein destination |
| Spot a | Ratio to C1F b | Matched Protein c | Accession Number d | Cov. % e | MW f | Functional Category | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C2F | DsF | NpF | |||||||||||||||
| 3015 | 4.56 | 4.69 | 2.95 | thioredoxin H-type isoform 1 (Vitis vinifera) | gi|225458147 | 62 | 12.80 | Protein folding | |||||||||
| 3014 | 2.80 | 26.5 | 1.78 | 18.2 kDa class I heat shock protein isoform 1 (Vitis vinifera) | gi|225449302 | 46 | 17.02 | Protein destination | |||||||||
| 2123 | 0.83 | 3.52 | 1.25 | 23.6 kDa heat shock protein, mitochondrial isoform 1(Vitis vinifera) | gi|225466111 | 42 | 23.74 | Protein destination | |||||||||
| 1009 | 0.58 | 0.96 | 3.47 | 18.2 kDa class I heat shock protein isoform 1 (Vitis vinifera) | gi|225449302 | 13 | 17.02 | Protein destination | |||||||||
| 6222 | 1.04 | 0.61 | 3.05 | l-ascorbate peroxidase 2, cytosolic (Vitis vinifera) | gi|225435177 | 65 | 27.56 | Carbohydrate metabolism | |||||||||
| 6222 | 1.04 | 0.61 | 3.05 | triosephosphate isomerase, cytosolic (Vitis vinifera) | gi|225449541 | 53 | 21.13 | Glycolysis/Gluconeogenesis | |||||||||
| 2409 | 0.70 | 1.37 | 2.10 | l-galactose dehydrogenase (Vitis vinifera) | gi|146432259 | 47 | 34.64 | Carbohydrate metabolism | |||||||||
| 2409 | 0.70 | 1.37 | 2.10 | pyruvate dehydrogenase E1 component subunit beta, mitochondrial-like isoform 1 (Vitis vinifera) | gi|225425166 | 42 | 39.49 | Glycolysis/Gluconeogenesis | |||||||||
| 2409 | 0.70 | 1.37 | 2.10 | PREDICTED: fructokinase-2 (Vitis vinifera) | gi|225433918 | 53 | 35.20 | Carbohydrate metabolism | |||||||||
| 2409 | 0.70 | 1.37 | 2.10 | isoflavone reductase homolog P3 (Vitis vinifera) | gi|225458243 | 36 | 33.81 | Secondary metabolism | |||||||||
| 5016 | 1.08 | 3.31 | 0.46 | 18.2 kDa class I heat shock protein (Vitis vinifera) | gi|225449250 | 49 | 18.15 | Protein destination | |||||||||
| 5016 | 1.08 | 3.31 | 0.46 | pathogenesis-related protein 10 (Vitis hybrid cultivar) | gi|163914213 | 14 | 17.11 | Defense and cell rescue | |||||||||
| 4627 | 0.43 | 1.08 | 1.38 | MLP-like protein 34 (Vitis vinifera) | gi|225424277 | 47 | 17.08 | Defense and cell rescue | |||||||||
| 4627 | 0.43 | 1.08 | 1.38 | chaperonin CPN60-2, mitochondrial isoform 1 (Vitis vinifera) | gi|225433375 | 45 | 61.37 | Protein destination | |||||||||
| 4627 | 0.43 | 1.08 | 1.38 | ruBisCO large subunit-binding protein subunit beta, chloroplastic (Vitis vinifera) | gi|225435794 | 42 | 64.61 | Photosynthesis | |||||||||
| 6018 | 1.90 | 0.24 | 1.12 | ubiquitin-conjugating enzyme E2 35 isoform 1 (Vitis vinifera) | gi|225461646 | 51 | 17.22 | Protein degradation | |||||||||
| 6018 | 1.90 | 0.24 | 1.12 | MLP-like protein 34 (Vitis vinifera) | gi|225424277 | 72 | 17.08 | Defense and cell rescue | |||||||||
| 6018 | 1.90 | 0.24 | 1.12 | chaperonin CPN60-2, mitochondrial isoform 1 (Vitis vinifera) | gi|225433375 | 27 | 61.37 | Protein destination | |||||||||
| 6018 | 1.90 | 0.24 | 1.12 | ubiquitin-conjugating enzyme E2 36 isoform 1 (Vitis vinifera) | gi|225446595 | 38 | 17.22 | Protein degradation | |||||||||
| 5018 | 1.65 | 0.17 | 0.65 | MLP-like protein 34 (Vitis vinifera) | gi|225424277 | 76 | 17.08 | Defense and cell rescue | |||||||||
| 5018 | 1.65 | 0.17 | 0.65 | ubiquitin-conjugating enzyme E2 36 isoform 1 (Vitis vinifera) | gi|225446595 | 38 | 17.22 | Protein degradation | |||||||||
| 5018 | 1.65 | 0.17 | 0.65 | glycine-rich RNA-binding protein GRP1A-like (Vitis vinifera) | gi|359475330 | 17 | 16.33 | Cell growth and death | |||||||||
| 5018 | 1.65 | 0.17 | 0.65 | superoxide dismutase (Cu-Zn) isoform 2 (Vitis vinifera) | gi|225451120 | 29 | 15.28 | Defense and cell rescue | |||||||||
| 5216 | 0.73 | 0.50 | 0.42 | putative transcription factor (Vitis vinifera) | gi|14582465 | 31 | 16.70 | Protein synthesis | |||||||||
| 5216 | 0.73 | 0.50 | 0.42 | ferritin-3, chloroplastic (Vitis vinifera) | gi|147784301 | 18 | 25.37 | Metabolism of cofactors and vitamins | |||||||||
| 5216 | 0.73 | 0.50 | 0.42 | triosephosphate isomerase, chloroplastic-like isoform 1 (Vitis vinifera) | gi|225427917 | 59 | 34.67 | Glycolysis/Gluconeogenesis | |||||||||
| 4421 | 0.52 | 0.47 | 0.33 | glutelin type-A 1 (Vitis vinifera) | gi|225435090 | 44 | 38.33 | - | |||||||||
| 4421 | 0.52 | 0.47 | 0.33 | glutamine synthetase nodule isozyme isoform 1 (Vitis vinifera) | gi|225451235 | 19 | 34.37 | Nitrogen metabolism | |||||||||
| 5217 | 0.61 | 0.32 | 0.19 | stem-specific protein TSJT1 (Vitis vinifera) | gi|225432548 | 46 | 25.25 | Defense and cell rescue | |||||||||
| 0207 | 0.20 | 0.10 | 0.24 | putative ripening-related protein (Vitis vinifera) | gi|7406667 | 23 | 15.39 | - | |||||||||
| 0309 | 0.35 | 0.24 | 0.16 | uncharacterized protein LOC100232885 (Vitis vinifera) | gi|225447003 | 78 | 18.41 | - | |||||||||
| Spot a | Ratio to C1V b | Matched Protein c | Accession Number d | Cov. % e | MW f | Functional Category | ||
|---|---|---|---|---|---|---|---|---|
| C2V | DsV | NpV | ||||||
| 1102 | 5.50 | 4.35 | 3.26 | small heat shock protein, chloroplastic (Vitis vinifera) | gi|225455238 | 55 | 25.03 | Protein destination |
| 1103 | 12.93 | 8.10 | 2.86 | small heat shock protein, chloroplastic (Vitis vinifera) | gi|225455238 | 48 | 25.03 | Protein destination |
| 1105 | 33.75 | 5.75 | 18.75 | small heat shock protein, chloroplastic (Vitis vinifera) | gi|225455238 | 54 | 25.03 | Protein destination |
| 5105 | 1.98 | 3.02 | 2.16 | S-adenosylmethionine synthase 5 | gi|223635289 | 10 | 42.79 | Defense and cell rescue |
| 5105 | 1.98 | 3.02 | 2.16 | 22.0 kDa heat shock protein (Vitis vinifera) | gi|225459900 | 46 | 21.12 | Protein destination |
| 6309 | 1.95 | 2.16 | 2.40 | l-ascorbate peroxidase 2, cytosolic (Vitis vinifera) | gi|225435177 | 61 | 27.56 | Other carboyhydrate metabolism |
| 2204 | 4.13 | 2.20 | 1.58 | uncharacterized protein LOC100254632 (Vitis vinifera) | gi|225441008 | 34 | 16.79 | |
| 2204 | 4.13 | 2.20 | 1.58 | glutathione S-transferase F9 (Vitis vinifera) | gi|225446791 | 29 | 24.91 | Defense and celle rescue |
| 2204 | 4.13 | 2.20 | 1.58 | uridylate kinase isoform 1 (Vitis vinifera) | gi|225454048 | 20 | 23.53 | Nucleotide metabolism |
| 4703 | 1.31 | 2.91 | 0.90 | peroxidase 12-like (Vitis vinifera) | gi|359493149 | 64 | 39.18 | Defense and cell rescue |
| 1301 | 2.15 | 1.29 | 0.29 | putative transcription factor (Vitis vinifera) | gi|14582465 | 14 | 16.70 | Protein synthesis |
| 1301 | 2.15 | 1.29 | 0.29 | nascent polypeptide-associated complex subunit alpha-like (Vitis vinifera) | gi|225470846 | 42 | 22.03 | Protein synthesis |
| 2709 | 0.00 | 1.34 | 0.20 | tubulin alpha-2 chain (Vitis vinifera) | gi|225458970 | 57 | 49.59 | Cytoskleton |
| 4519 | 0.20 | 0.70 | 0.50 | naringenin,2-oxoglutarate 3-dioxygenase (Vitis vinifera) | gi|225431140 | 65 | 40.81 | Secondary metabolism |
| 4519 | 0.20 | 0.70 | 0.50 | caffeic acid 3- O-methyltransferase 1-like isoform 1 (Vitis vinifera) | gi|359490763 | 53 | 39.50 | Secondary metabolism |
| 1304 | 0.44 | 0.07 | 0.48 | proteasome subunit alpha type-5 isoform 1 (Vitis vinifera) | gi|225441985 | 43 | 25.98 | Protein degradation |
| 1402 | 0.12 | 0.07 | 0.27 | uncharacterized protein LOC100232885 (Vitis vinifera) | gi|225447003 | 83 | 18.41 | |
| 5602 | 0.03 | 0.24 | 0.16 | S-adenosylmethionine synthase 5 | gi|223635289 | 75 | 42.79 | Defense and celle rescue |
3. Experimental Section
3.1. Plant Material, Fungal Strains and Pathogenicity Tests
| Condition | Phenological Stage | ||
|---|---|---|---|
| G stage | Flowering | Veraison | |
| Control 1 | C1G | C1F | C1V |
| Control 2 | C2G | C2F | C2V |
| D.seriata strain Bo98-1 | DsG | DsF | DsV |
| N.parvum strain Np SV | NpG | NpF | NpV |
3.2. Two-Dimensional Gel Electrophoresis (2D) Analysis
3.2.1. Protein Extraction
3.2.2. Image Analysis
3.2.3. Protein Identification by Mass Spectrometry
3.2.4. Functional Classification of Identified Proteins
4. Conclusions
Supplementary Files
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Crous, P.W.; Slippers, B.; Wingfield, M.J.; Rheeder, J.; Marasas, W.F.O.; Philips, A.J.L.; Alves, A.; Burgess, T.; Barber, P.; Groenewald, J.Z. Phylogenetic lineages in the Botryosphaeriaceae. Stud. Mycol. 2006, 55, 235–253. [Google Scholar]
- Slippers, B.; Wingfield, M.J. Botryosphaeriaceae as endophytes and latent pathogens of woody plants: Diversity, ecology and impact. Fungal Biol. Rev. 2007, 21, 90–106. [Google Scholar]
- Phillips, A.J.L.; Alves, A.; Abdollahzadeh, J.; Slippers, B.; Wingfield, M.; Groenewald, J.Z.; Crous, P.W. The Botryosphaeriaceae: Genera and species known from culture. Stud. Mycol. 2013, 76, 51–167. [Google Scholar]
- Úrbez-Torres, J.R.; Leavitt, G.M.; Voegel, T.M.; Gubler, W.D. Identification and distribution of Botryosphaeria spp. associated with grapevine cankers in California. Plant Dis. 2006, 90, 1490–1503. [Google Scholar]
- Pitt, W.M.; Huang, R.; Steel, C.C.; Savocchia, S. Identification, distribution and current taxonomy of Botryosphaeriaceae species associated with grapevine decline in New South Wales and South Australia. Aust. J. Grape Wine Res. 2010, 16, 258–271. [Google Scholar]
- Úrbez-Torres, J.R. The status of Botryosphaeriaceae species infecting grapevines. Phytopathol. Mediterr. 2011, 50, S5–S45. [Google Scholar]
- Pitt, W.M.; Huang, R.; Steel, C.C.; Savocchia, S. Pathogenicity and epidemiology of Botryosphaerriaceae species isolated from grapevines in Australia. Aust. Plant Pathol. 2013, 42, 573–582. [Google Scholar]
- Rolshausen, E.; Akgül, D.S.; Perez, R.; Eskalen, A.; Gispert, C. First report of wood canker caused by Neoscytalidium dimidiatum on grapevine in California. Plant Dis. 2013, 97, 1511–1511. [Google Scholar]
- Yan, J.-Y.; Xie, Y.; Zhang, W.; Wang, Y.; Liu, J.-K.; Hyde, K.D.; Seem, R.C.; Zhang, G.-Z.; Wang, Z.-Y.; Yao, S.-W.; et al. Species of Botryosphaeriaceae involved in grapevine dieback in China. Fungal Divers. 2013, 61, 221–236. [Google Scholar]
- Larignon, P.; Fulchic, R.; Cere, L.; Dubos, B. Observation on black dead arm in French vineyards. Phytopathol. Mediterr. 2001, 40, S336–S342. [Google Scholar]
- Bertsch, C.; Ramirez-Suero, M.; Magnin-Robert, M.; Larignon, P.; Chong, J.; Abou-Mansour, E.; Spagnolo, A.; Clément, C.; Fontaine, F. Grapevine trunk diseases: Complex and still poorly understood. Plant Pathol. 2013, 62, 243–265. [Google Scholar]
- Úrbez-Torres, J.R.; Leavitt, G.M.; Guerrero, J.C.; Guevara, J.; Gubler, W.D. Identification and pathogenicity of Lasiodiplodia theobromae and Diplodia seriata, the causal agents of Bot canker disease of grapevines in Mexico. Plant Dis. 2008, 92, 519–529. [Google Scholar]
- Mohammadi, H.; Gramaje, D.; Banishashemi, Z.; Armengol, J. Characterization of Diplodia seriata and Neofusicoccum parvum associated with grapevine decline in Iran. J. Agric. Sci. Technol. 2013, 15, 603–616. [Google Scholar]
- Epstein, L.K.; Sukhwinder, K.; Van der Gheynst, J.S. Botryosphaeria-related dieback and control investigated in noncoastal California grapevines. Calif. Agric. 2008, 62, 161–166. [Google Scholar]
- Larignon, P.; Coarer, M.; Larbre, C.; Girardon, K.; Vigues, V.; Yobregat, O. Identification sur le matériel végétal des sources d’inoculum des champignons associés aux maladies du bois. Phytoma 2009, 622–623, 46–48. [Google Scholar]
- van Niekerk, J.M.; Calitz, F.J.; Halleen, F.; Fourie, P.H. Temporal spore dispersal patterns of grapevine trunk pathogens in South Africa. Eur. J. Plant Pathol. 2010, 127, 375–390. [Google Scholar]
- Urbez-Torres, J.R.; Bruez, E.; Hutado, J.; Gubler, W.W. Effect of temperature on conidial germination of Botryosphaeriaceae species infecting grapevines. Plant Dis. 2010, 94, 1476–1484. [Google Scholar]
- Larignon, P.; Dubos, B. Le Black Dead Arm. Maladie nouvelle à ne pas confondre avec l’esca. Phytoma 2001, 538, 26–29. [Google Scholar]
- Kuntzmann, P.; Villaume, S.; Larignon, P.; Bertsch, C. Esca, BDA and Eutypiosis: Foliar symptoms, trunk lesions and fungi observed in diseased vinestocks in two vineyards in Alsace. Vitis 2010, 49, 71–76. [Google Scholar]
- Úrbez-Torres, J.R.; Adams, P.; Kamas, J.; Gubler, W.D. Identification, incidence, and pathogenicity of fungal species associated with grapevine dieback in Texas. Am. J. Enol. Vitic. 2009, 60, 497–507. [Google Scholar]
- Larignon, P. Studies on the role of pruning wounds in infection by Phaeocremonium aleophilum and Diplodia seriata in France. In Proceedings of the 8th International Workshop on Grapevine Trunk Diseases, Valencia, Spain, 18–21 June 2012.
- Serra, S.; Mannoni, M.A.; Ligios, V. Studies on the susceptibility of pruning wounds to infection by fungi involved in grapevine wood diseases in Italy. Phytopathol. Mediterr. 2008, 47, 234–246. [Google Scholar]
- Berger, S.; Sinha, A.K.; Roitsch, T. Plant physiology meets phytopathology: Plant primary metabolism and plant-pathogen interactions. J. Exp. Bot. 2007, 58, 4019–4026. [Google Scholar]
- Lebon, G.; Wojnarowiez, G.; Holzapfel, B.; Fontaine, F.; Vaillant-Gaveau, N.; Clément, C. Sugars and flowering in the grapevine (Vitis vinifera L.). J. Exp. Bot. 2008, 59, 2565–2578. [Google Scholar]
- Petit, A.N.; Baillieul, F.; Vaillant-Gaveau, N.; Jacquens, L.; Conreux, A.; Jeandet, P.; Clément, C.; Fontaine, F. Low responsiveness of grapevine flowers and berries at fruit set to UV-C irradiation. J. Exp. Bot. 2009, 60, 1155–1162. [Google Scholar]
- Keller, M.; Viret, O.; Cole, F.M. Botrytis cinerea infection in grape flowers: Defense reaction, latency, and disease expression. Phytopathology 2003, 93, 316–322. [Google Scholar]
- Lebon, G.; Duchêne, E.; Brun, O.; Magné, C.; Clément, C. Flower abscission and inflorescence carbohydrates in sensitive and non-sensitive cultivars of grapevine. Sex. Plant Reprod. 2004, 17, 71–79. [Google Scholar]
- Zappata, C.; Deléens, E.; Chaillou, S.; Magné, C. Partitioning and mobilization of starch and N reserves in grapevine (Vitis vinifera L.). J. Plant Physiol. 2004, 161, 1031–1040. [Google Scholar]
- Riou, C.; Freyssinet, G.; Fevre, M. Production of cell wall-degrading enzymes by the phytopathogenic fungus Sclerotinia sclerotiorum. Appl. Environ. Microbiol. 1991, 57, 1478–1484. [Google Scholar]
- Fasoli, M.; dal Santo, S.; Zenoni, S.; Tornielli, G.B.; Farina, L.; Zamboni, A.; Porceddu, A.; Venturini, L.; Bicego, M.; Murino, V.; et al. The grapevine expression atlas reveals a deep transcriptome shift driving the entire plant into a maturation program. Plant Cell 2012, 24, 3489–3505. [Google Scholar]
- Spagnolo, A.; Magnin-Robert, M.; Alayi, T.D.; Cilindre, C.; Schaeffer-Reiss, C.; van Dorsselaer, A.; Clément, C.; Larignon, P.; Suero-Ramirez, M.; Chong, J.; et al. Differential responses of three grapevine cultivars to Botryosphaeria dieback. Phytopathology 2014. [Google Scholar] [CrossRef]
- Bolton, M.D. Primary metabolism and plant defense-fuel for the fire. Mol. Plant Microbe Interact. 2009, 22, 487–497. [Google Scholar]
- Rojas, C.M.; Senthil-Kumar, M.; Vered, T.; Mysore, K.S. Regulation of primary plant metabolism during plant-pathogen interactions and its contribution to plant defense. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef]
- Forde, B.B.; Lea, P.J. Glutamate in plants: Metabolism, regulation, and signalling. J. Exp. Bot. 2007, 58, 2339–2358. [Google Scholar]
- Spagnolo, A.; Magnin-Robert, M.; Alayi, T.D.; Cilindre, C.; Mercier, L.; Schaeffer-Reiss, C.; van Dorsselaer, A.; Clément, C.; Fontaine, F. Physiological changes in green stems of Vitis vinifera L. cv. Chardonnay in response to esca proper and apoplexy revealed by proteomic and transcriptomic analyses. J. Proteome Res. 2012, 11, 461–475. [Google Scholar]
- Magnin-Robert, M.; Spagnolo, A.; Alayi, T.D.; Cilindre, C.; Mercier, L.; Schaeffer-Reiss, C.; van Dorsselaer, A.; Clément, C.; Fontaine, F. Proteomic insights into changes in wood of Vitis vinifera L. in response to esca proper and apoplexy. Phytopathol. Mediterr. 2014, 53, 173–192. [Google Scholar]
- Roje, S. S-Adenosyl-l-methionine: Beyond the universal methyl group donor. Phytochemistry 2006, 67, 1686–1698. [Google Scholar]
- Tsunezuka, H.; Fujiwara, M.; Kawasaki, T.; Shimamoto, K. Proteome analysis of programmed cell death and defense signaling using the rice lesion mimic mutant cdr2. Mol. Plant Microbe Interact. 2005, 18, 52–59. [Google Scholar]
- Figueiredo, A.; Fortes, A.M.; Ferreira, S.; Sebastiana, M.; Choi, Y.H.; Sousa, L.; Acioli-Santos, B.; Pessoa, F.; Verpoorte, R.; Pais, M.S. Transcriptional and metabolic profiling of grape (Vitis vinifera L.) leaves unravel possible innate resistance against pathogenic fungi. J. Exp. Bot. 2008, 59, 3371–3381. [Google Scholar]
- Camps, C.; Kappel, C.; Lecomte, P.; Leon, C.; Gomes, E.; Coutos-Thevenot, P.; Delrot, S. A transcriptomic study of grapevine (Vitis vinifera cv. Cabernet-Sauvignon) interaction with the vascular ascomycete fungus Eutypa lata. J. Exp. Bot. 2010, 61, 1719–1737. [Google Scholar]
- Margaria, P.; Abba, S.; Palmano, S. Novel aspects of grapevine response to phytoplasma infection investigated by a proteomic and phospho-proteomic approach with data integration into functional networks. BMC Genomics 2013, 14, 38–52. [Google Scholar]
- Robertson, D.; McCormick, B.A.; Bolwell, G.P. Cell wall polysaccharide biosynthesis and related metabolism in elicitor-stressed cells of French bean (Phaseolus vulgaris L.). Biochem. J. 1995, 30, 745–750. [Google Scholar]
- Tenhaken, R.; Thulke, O. Cloning of an enzyme that synthesizes a key nucleotide-sugar precursor of hemicellulose biosynthesis from soybean: UDP-glucose dehydrogenase. Plant Physiol. 1996, 112, 1127–1134. [Google Scholar]
- Reiter, W.D.; Vauzin, G.F. Molecular genetics of nucleotide sugar interconversion pathways in plants. Plant Mol. Biol. 2001, 47, 95–113. [Google Scholar]
- Chávez Montes, R.A.; Ranocha, P.; Martinez, Y.; Minic, Z.; Jouanin, L.; Marquis, M.; Saulnier, L.; Fulton, L.M.; Cobbett, C.S.; Bitton, F.; et al. Cell wall modifications in Arabidopsis plants with altered α-l-arabinofuranosidase activity. Plant Physiol. 2008, 147, 63–77. [Google Scholar]
- Gilbert, L.; Alhagdow, M.; Nunes-Nesi, A.; Quemener, B.; Guillon, F.; Bouchet, B.; Faurobert, M.; Gouble, B.; Page, D.; Garcia, V.; et al. GDP-d-mannose 3,5-epimerase (GME) plays a key role at the intersection of ascorbate and non-cellulosic cell-wall biosynthesis in tomato. Plant J. 2009, 60, 499–508. [Google Scholar]
- Valtaud, C.; Foyer, C.H.; Fleurat-Lessard, P.; Bourbouloux, A. Systemic effects on leaf glutathione metabolism and defence protein expression caused by esca infection in grapevines. Funct. Plant Biol. 2009, 36, 260–279. [Google Scholar]
- Letousey, P.; Baillieul, F.; Perrot, G.; Rabenoelina, F.; Boulay, M.; Vaillant-Gaveau, N.; Clément, C.; Fontaine, F. Early events prior to visual symptoms in the apoplectic form of grapevine esca disease. Phytopathology 2010, 100, 424–431. [Google Scholar]
- Magnin-Robert, M.; Letousey, P.; Spagnolo, A.; Rabenoelina, F.; Jacquens, L.; Mercier, L.; Clément, C.; Fontaine, F. Leaf strip of esca induces alteration of photosynthesis and defence reactions in presymptomatic leaves. Funct. Plant Biol. 2011, 38, 856–866. [Google Scholar]
- Mittler, R. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci. 2002, 7, 405–410. [Google Scholar]
- Lima, M.R.M.; Ferreres, F.; Dias, A.C.P. Response of Vitis vinifera cell cultures to Phaeomoniella chlamydospora: Changes in phenolic production, oxidative state and expression of defence-related genes. Eur. J. Plant Pathol. 2012, 132, 133–146. [Google Scholar]
- Sauter, M.; Rzewuski, G.; Marwedel, T.; Lorbiecke, R. The novel ethylene-regulated gene OsUsp1 from rice encodes a member of a plant protein family related to prokaryotic universal stress proteins. J. Exp. Bot. 2002, 53, 2523–2331. [Google Scholar]
- Mahomed, W.; van den Berg, N. EST sequencing and gene expression profiling of defence-related genes from infected with Phytophthora cinnamomi. BMC Plant Biol. 2011, 11, 167–180. [Google Scholar]
- Roberts, M.R.; Salinas, J.; Collinge, D.B. 14–3-3 proteins and the response to abiotic and biotic stress. Plant Mol. Biol. 2002, 50, 1031–1039. [Google Scholar]
- Wang, W.; Vinocur, B.; Shoseyov, O.; Altman, A. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci. 2004, 9, 244–252. [Google Scholar]
- Duan, Y.H.; Guo, J.; Ding, K.; Wang, S.-J.; Zhang, H.; Dai, X.-W.; Chen, Y.-Y.; Govers, F.; Huang, L.-L.; Kang, Z.-S. Characterization of a wheat HSP70 gene and its expression in response to stripe rust infection and abiotic stresses. Mol. Biol. Rep. 2011, 38, 310–307. [Google Scholar]
- Musetti, R.; di Toppi, L.S.; Ermacora, P.; Favali, M.A. Recovery in apple trees infected with the apple proliferation phytoplasma: An ultrastructural and biochemical study. Phytopathology 2004, 94, 203–208. [Google Scholar]
- Grenville-Briggs, L.J.; van West, P. Biotrophic stages of oomycete–plant interactions. Adv. Appl. Microbiol. 2005, 57, 217–243. [Google Scholar]
- Larignon, P.; Dubos, B. Fungi associated with esca disease in grapevine. Eur. J. Plant Pathol. 1997, 103, 147–157. [Google Scholar]
- Geer, L.Y.; Markey, S.P.; Kowalak, J.A.; Wagner, L.; Xu, M.; Maynard, D.M.; Yang, X.; Shi, W.; Bryant, S.H. Open mass spectrometry search algorithm. J. Proteome Res. 2004, 3, 958–964. [Google Scholar]
- Elias, J.E.; Gygi, S.P. Target-decoy search strategy for increased confidence in large-scale protein identifications by mass spectrometry. Nat. Methods 2007, 4, 207–214. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Spagnolo, A.; Larignon, P.; Magnin-Robert, M.; Hovasse, A.; Cilindre, C.; Van Dorsselaer, A.; Clément, C.; Schaeffer-Reiss, C.; Fontaine, F. Flowering as the Most Highly Sensitive Period of Grapevine (Vitis vinifera L. cv Mourvèdre) to the Botryosphaeria Dieback Agents Neofusicoccum parvum and Diplodia seriata Infection. Int. J. Mol. Sci. 2014, 15, 9644-9669. https://doi.org/10.3390/ijms15069644
Spagnolo A, Larignon P, Magnin-Robert M, Hovasse A, Cilindre C, Van Dorsselaer A, Clément C, Schaeffer-Reiss C, Fontaine F. Flowering as the Most Highly Sensitive Period of Grapevine (Vitis vinifera L. cv Mourvèdre) to the Botryosphaeria Dieback Agents Neofusicoccum parvum and Diplodia seriata Infection. International Journal of Molecular Sciences. 2014; 15(6):9644-9669. https://doi.org/10.3390/ijms15069644
Chicago/Turabian StyleSpagnolo, Alessandro, Philippe Larignon, Maryline Magnin-Robert, Agnès Hovasse, Clara Cilindre, Alain Van Dorsselaer, Christophe Clément, Christine Schaeffer-Reiss, and Florence Fontaine. 2014. "Flowering as the Most Highly Sensitive Period of Grapevine (Vitis vinifera L. cv Mourvèdre) to the Botryosphaeria Dieback Agents Neofusicoccum parvum and Diplodia seriata Infection" International Journal of Molecular Sciences 15, no. 6: 9644-9669. https://doi.org/10.3390/ijms15069644