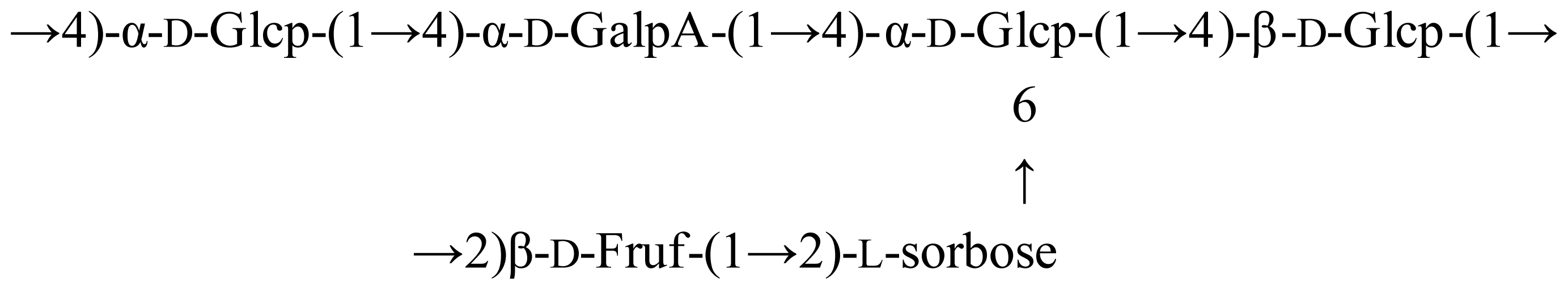1. Introduction
Dimocarpus longan pulp (longan) is a commercially available fruit widely distributed in southern China. Longan pulp has long been used in China to promote health and blood metabolism, soothe nerves, prevent amnesia, relieve insomnia and extend longevity [
1,
2]. Several recent studies have revealed that the alcohol extracts of longan pulp reduce serum prolactin levels in female rats [
3]. The water extracts of longan pulp show a measurable effect against the JTC26 cervical cancer cell line, as confirmed by Cai
et al. [
4]. In addition, longan pulp polysaccharides have demonstrated immunomodulatory activity, as reported by Chen
et al. [
5].
Polysaccharides, which are widely distributed in fruit, animals, plants and fungi, have drawn increasing attention from researchers and consumers, due to their obvious antitumor, antioxidant [
6], anti-HIV/AIDS and immunostimulatory activities [
7,
8], as well as the relatively low toxicity [
9] of the polysaccharides. Therefore, the discovery and evaluation of polysaccharides with antitumor and immunostimulatory properties has become an important focus of research in chemistry and biology [
10].
However, information regarding the polysaccharides from longan pulp and the in vitro immunomodulatory and antitumor properties of longan polysaccharides (LPs) is limited. Therefore, as reported in this paper, a purified fraction, referred to as LP1, was obtained from crude polysaccharide extract from longan pulp via diethylaminoethyl (DEAE)-cellulose anion-exchange and Sephacryl S-300 HR gel chromatography. The chemical structure of the polysaccharide and its antitumor activity in vitro and immunomodulatory activity in vivo were investigated.
3. Experimental Section
3.1. Materials
The human ovarian cancer cell lines, SKOV3 and HO8910, were supplied by the cell bank of the Chinese Academy of Sciences (Shanghai, China). SPF BALB/c male mice were provided by the experimental animal center of Guangxi Medical University (Nanning, China). PBS buffer was obtained from Wuhan Boster Bio-Engineering Co., Ltd. (Wuhan, China). Sodium chloride for injection was obtained from Kunming Yusi Pharmaceutical Co., Ltd. (Kunming, China). RPMI-1640 with an improved nutrient solution was purchased from Thermo Fisher Scientific (Beijing) Inc. (Beijing, China). Sword bean protein and lipopolysaccharide were purchased from Beijing Bo Ao Tuoda Technology Co., Ltd. (Beijing, China). 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and 0.25% trypsin (containing EDTA) were purchased from Sigma Chemicals Co., Perth, ND, USA. All other chemicals and solvents were analytical grade and used without further purification.
3.2. Isolation and Purification of Polysaccharides
First, the dried longan pulp (2.0 kg) was extracted with distilled water at 90 °C for 4 h (three times) and then filtered with cotton gauze. The extracted solutions were combined and concentrated by rotary evaporation under reduced pressure below 65 °C and subjected to the Sevag method [
49] to remove free proteins. The crude polysaccharide thus obtained was labeled LP. Then, the LP extract (2.00 g) was further purified by dissolution in distilled water and application to a column (Φ2.5 cm × 80 cm) of DEAE-cellulose. After the sample was loaded, the column was successively washed with distilled water, 0.125 M sodium chloride and 0.30 M sodium hydroxide aqueous solution at an elution rate of 0.4 mL/min. An improved phenol-sulfuric acid detector was used online, and the eluate was collected automatically [
50]. A total of four fractions of polysaccharide were obtained and labeled F-1, F-2, F-3 and F-4. Third, the largest NaCl-eluted fraction, F-1, was collected, dialyzed for 2 days, concentrated and lyophilized. The F-1 fraction was further purified by dissolution in distilled water using ultrasound and subjected to Sephacryl S-300 HR gel filtration chromatography (Φ2.0 cm × 50 cm) (Shanghai Stars Biological Technology Co., Ltd., Shanghai, China) after filtration with a 0.45-μm microporous membrane filter. The loading sample was washed with distilled water at an elution rate of 0.75 mL/min and monitored with a phenol-sulfuric acid detector. The purified fraction containing carbohydrate was collected, concentrated, precipitated with alcohol and dried under vacuum to yield a sample of purified polysaccharide, which was labeled LP1.
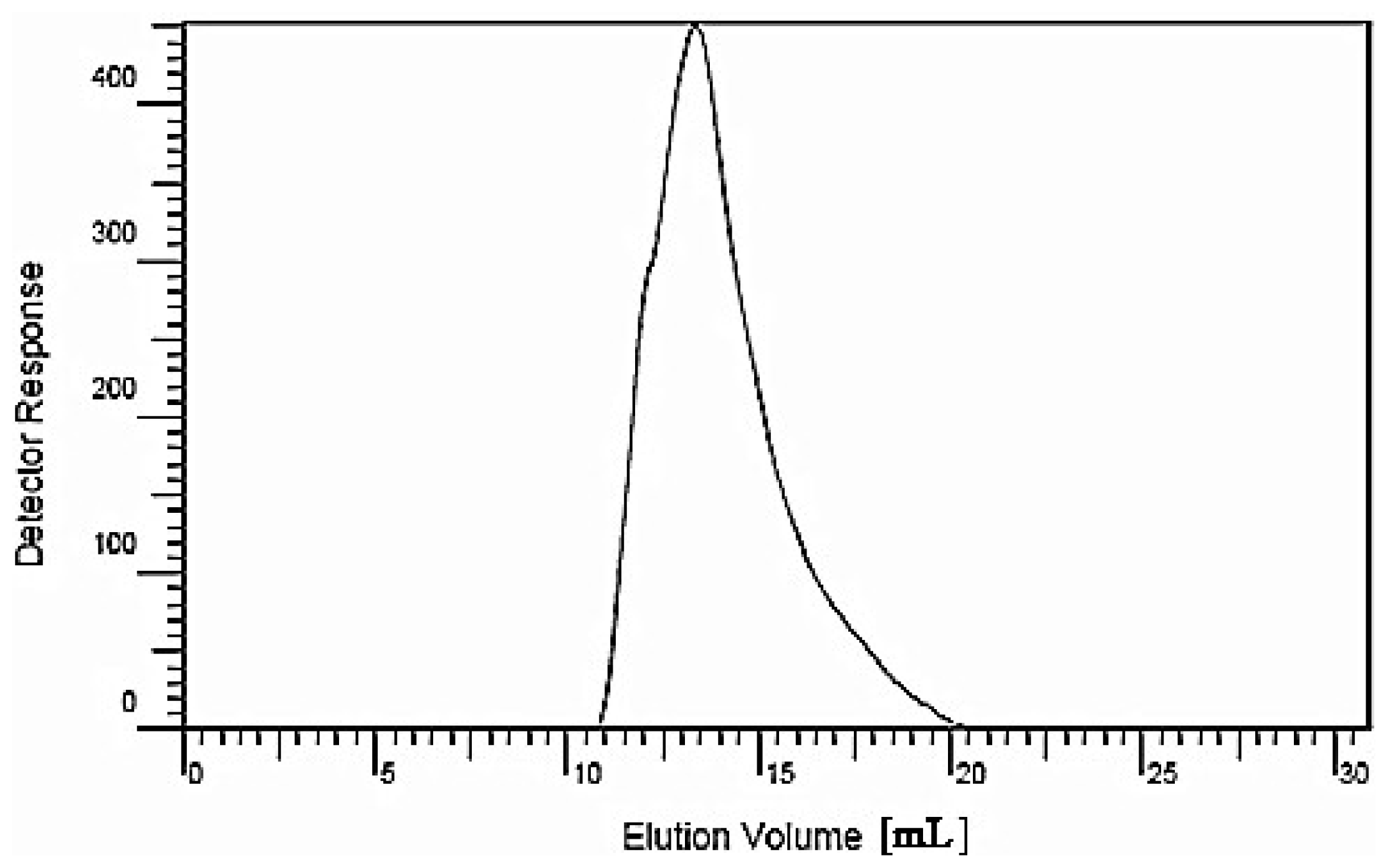
3.3. HPGPC to Determine the Molecular Weight of the Polysaccharide
The preparation of standard solutions and sample solutions was performed as follows: dextran 40, a series of standard glucans (molecular weights 5900, 11,800, 22,800, 47,300, 112,000, 212,000 and 404,000) and dried longan polysaccharide LP1 were accurately weighed; ultrapure water was then added, and the samples were dissolved with the aid of ultrasonication over 15 min. The final concentration of the standard solutions was 10 mg/mL, and the solutions were filtered with a 0.45-μm microporous filter.
The chromatographic conditions were as follows: a chromatographic column (TSK-4000PW, Φ7.5 mm × 300 mm, Tosoh Bioscience Shanghai Co., Ltd., Shanghai, China), a gel column (TSK-3000SW, Φ7.5 mm × 300 mm, Tosoh Bioscience Shanghai Co., Ltd.) and a guard column (TSK-Guard SW, Φ7.5 mm × 75 mm, Tosoh Bioscience Shanghai Co., Ltd.) were employed; the two gel columns and the guard column were used in series. The chromatographic column temperature was 23 °C with a flow velocity of 1 mL/min; detection was performed using a refractive index detector (Agilent G1362A, Agilent Technologies (China) Co., Ltd., Beijing, China). A volume of 20 μL each of the standard sample and LP1 were injected separately. The mobile phase was ultrapure water. The evolution volume (Ve) was measured, and through data analysis, (Agilent GPC software, Beijing, China), the standard curve was drawn with the Ve as the abscissa and the logarithm of the Mr as the vertical axis.
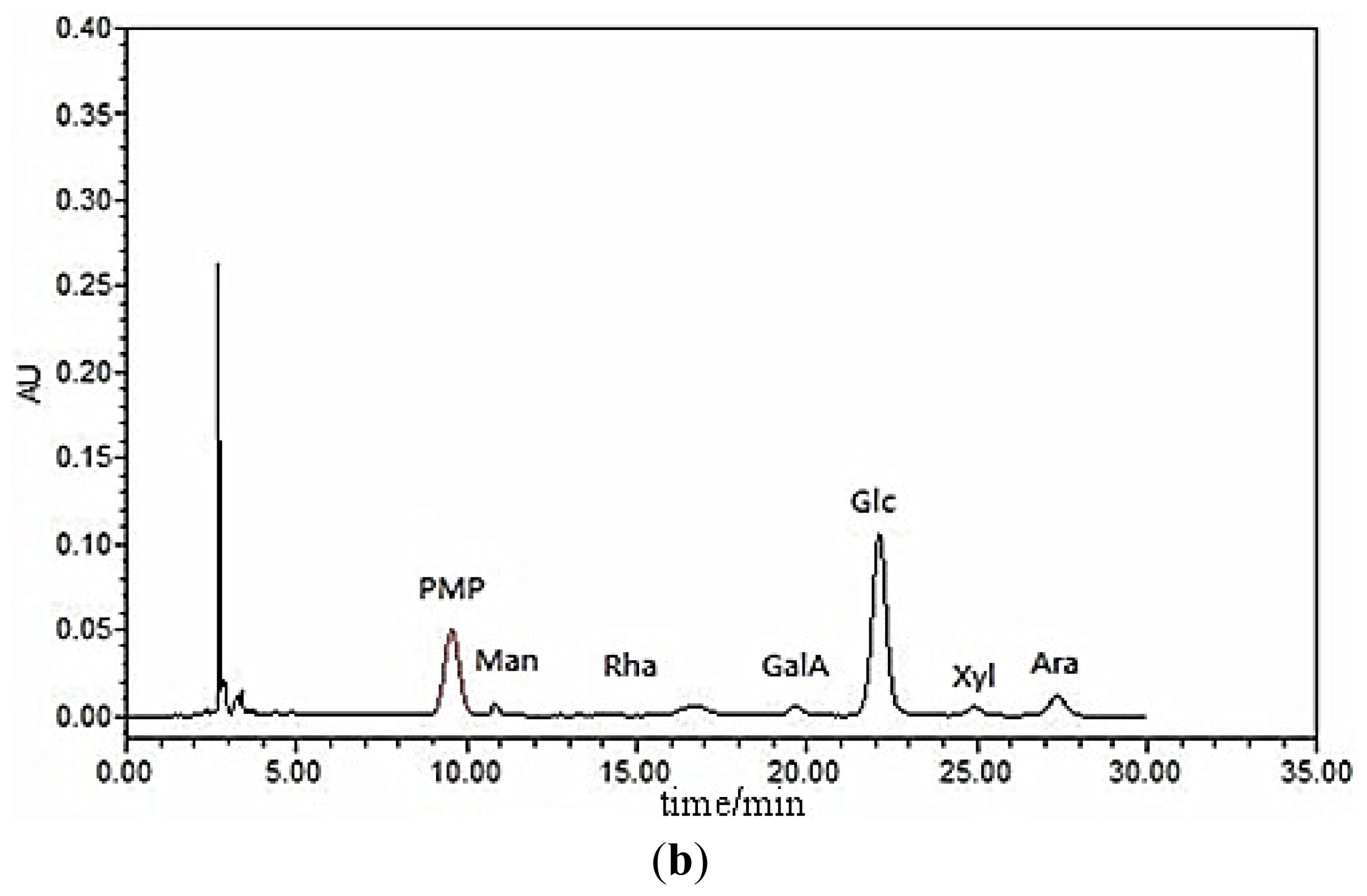
3.4. Monosaccharide Composition
The LP1 polysaccharide sample (250.0 mg) was hydrolyzed with 5 mL of 1.0 mol/L TFA at 102 °C for 6 h in a sealed ampule, cooled to room temperature, transferred to a 10-mL volumetric flask and then neutralized with a 3-mol/L NaOH solution until the pH reached 7.0. The sample was then diluted with distilled water, mixed thoroughly and then centrifuged for 10 min at 4000 rpm. The supernatant was collected, precisely measured and mixed with seven different monosaccharide solutions (mannose, rhamnose, galacturonic acid, glucose, galactose, xylose and arabinose); each solution contained 0.4 mL at 0.2 mg/mL in two test tubes. Next, 0.4 mL of a 0.5 mol/L 1-phenyl-3-methyl-5-pyrazolone (PMP) solution in methanol and 0.4 mL of a 0.4 mol/L NaOH solution were sequentially added. The solutions were mixed, heated in a 70 °C water bath for 100 min and then cooled to room temperature. This procedure was followed by neutralization with 0.5 mL of a 0.3-mol/L HCl solution, the addition of distilled water to a volume of 2.0 mL and extraction with 5.0 mL chloroform. The upper aqueous phase was then filtered through a 0.45-μm microporous membrane.
Chromatography was performed with an Agilent TC-C18 column (Φ4.6 mm × 250 mm, 5 μm) (Agilent Technologies (China) Co., Ltd.) under the following conditions: column temperature: 25 °C; UV detection: 250 nm, with a PD-10-a VP Plus detector (Agilent Technologies (China) Co., Ltd.); sample quantity: 10 μL; mobile phase: 80 mL of 0.05 mol/L ammonium acetate solution and 20 mL of acetonitrile; flow rate: 1.0 mL/min; and elution time: 35 min.
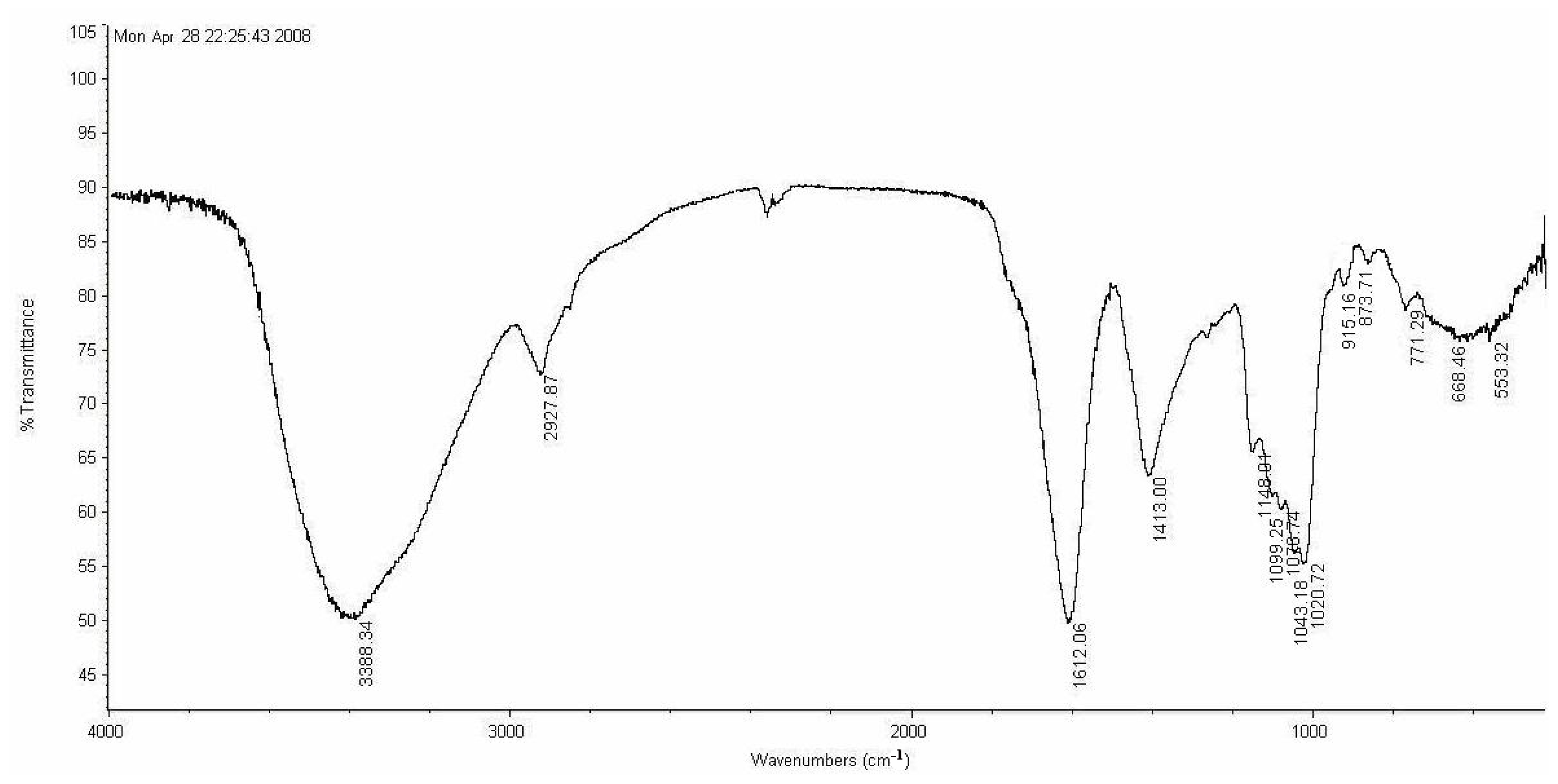
3.5. Characterization
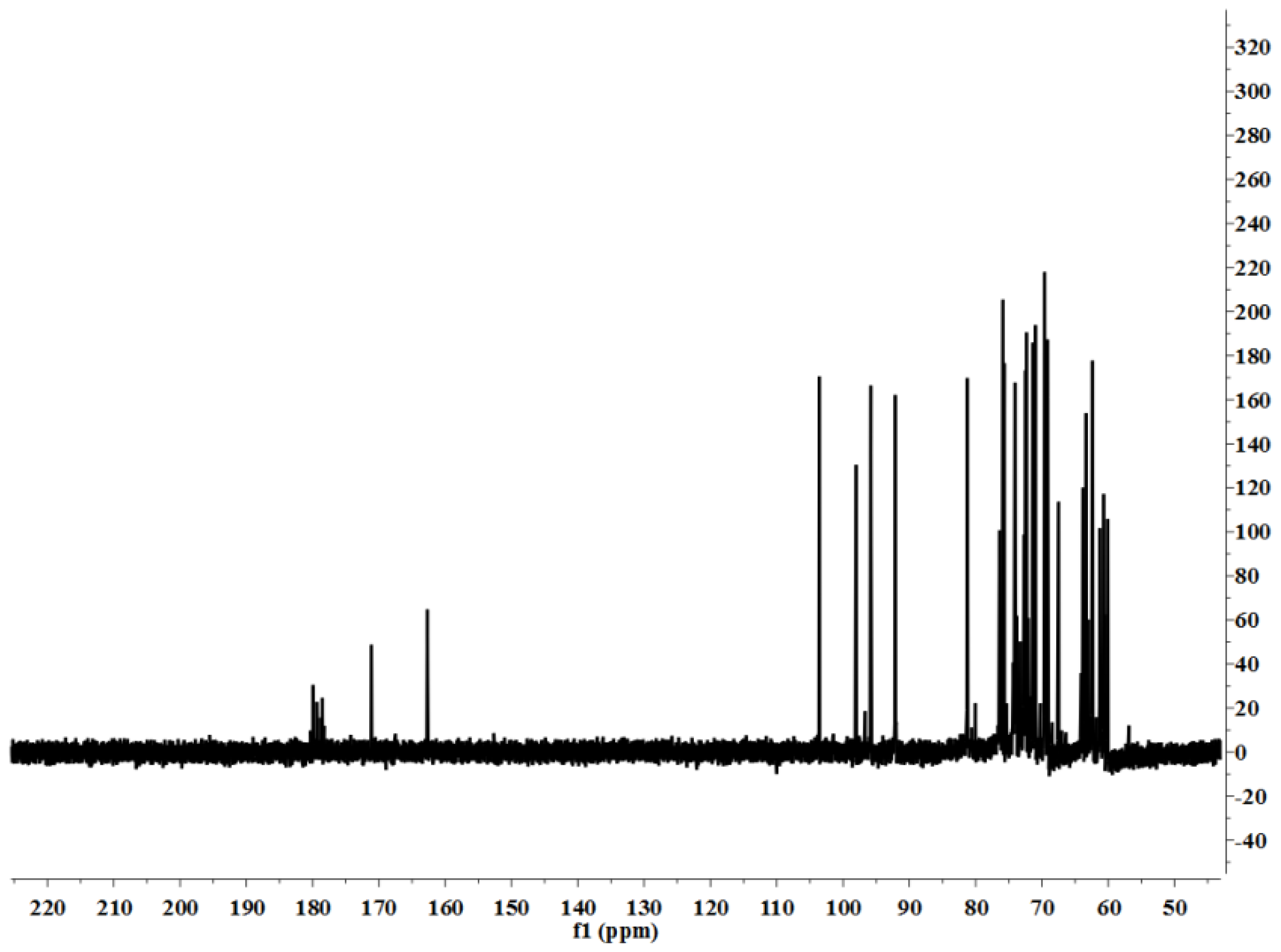
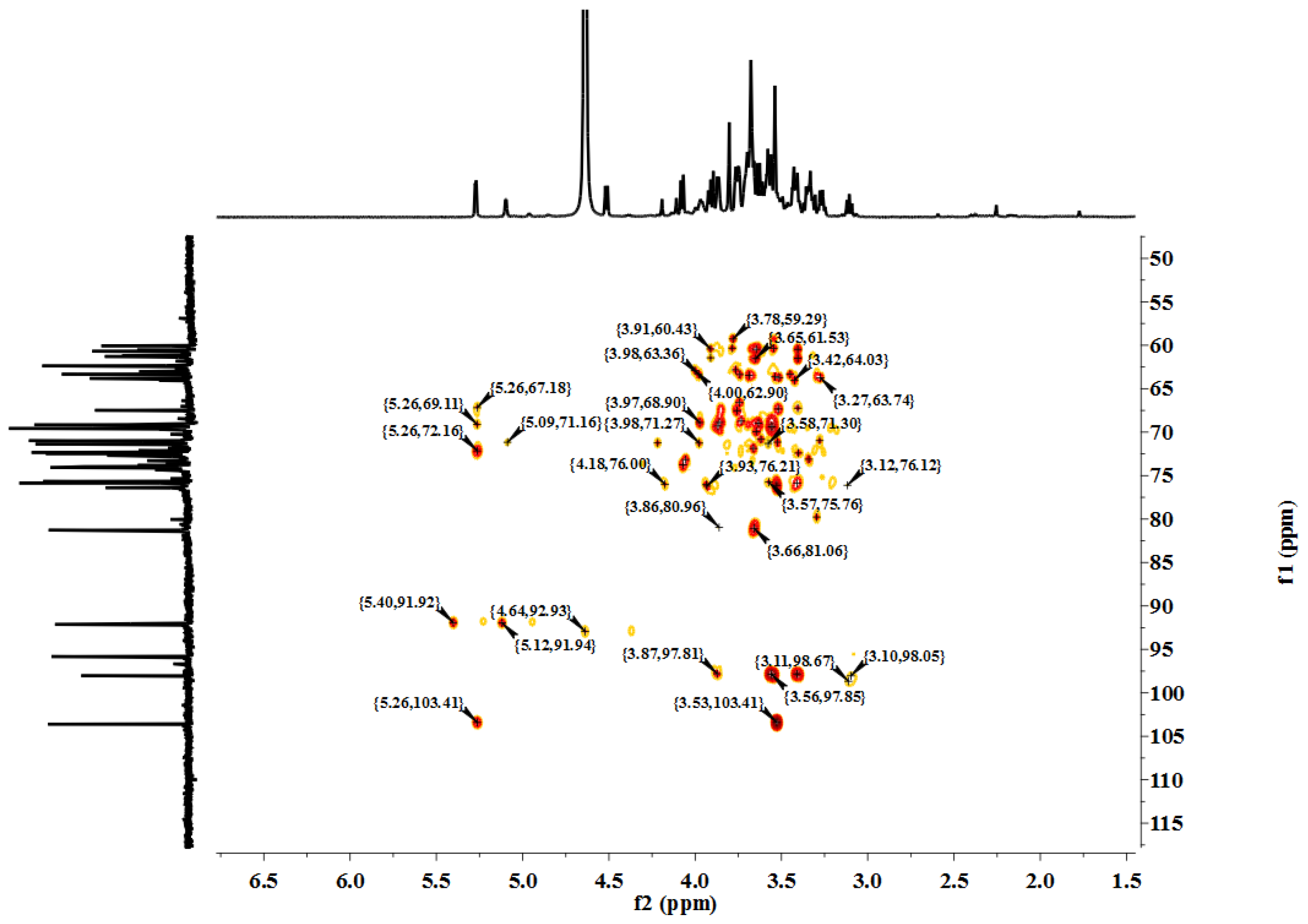
LP1 was placed in a 5-mm NMR tube and dissolved in D
2O (0.5 mL). The NMR spectra were recorded with a Bruker AVIII 600 NMR spectrometer (Bruker (Beijing) Scientific Technology Co., Ltd., Beijing, China) at room temperature. All chemical shifts are reported in parts per million relative to the internal standard TSP-d4. Infrared spectroscopy of the samples was performed with a Nicolet 170SX FT-IR spectrometer from 400–4000 cm
−1 with a DGTS detector and DMNIC 3.2 software (Thermo Fisher Scientific (Beijing) Inc., Beijing, China). Gas chromatography (GC) of the alditol acetate derivatives of the saccharides, as reported in the literature [
51], was carried out on an HP 6890 instrument (Hewlett Packard, Palo Alto, CA, USA) with a DB-225 column (Φ0.25 mm × 15 m) (Agilent Technologies (China) Co., Ltd.) from 180 to 220 °C at 4 °C/min.
3.6. Biological Assays
3.6.1. Anti-Tumor Activity in Vitro
3.6.1.1. Cell Lines and Culture
Human gastric carcinoma SGC-7901 cells were maintained in RPMI-1640 medium supplemented with 10% FBS, penicillin (100 U/mL) and streptomycin (100 mg/L) at 37 °C in a humidified atmosphere with 5% CO2. The culture was passaged every 2 or 3 days. The final cell concentration was 3 × 107 cells/mL.
3.6.1.2. Assay of the Inhibition of Tumor Cell Proliferation in Vitro
The inhibitory effects of LP1 on the proliferation of SKOV3 and HO8910 cells (3 × 10
7 cells/mL) were determined
in vitro with the colorimetric MTT assay [
52]. The doses of LP1 applied to the SKOV3 tumor cells were 5, 10, 20 and 40 mg/L. The doses of LP1 applied to the HO8910 tumor cells were 40, 80, 160 and 320 mg/L. The concentration of 5-fluorouracil used for the positive control was 5 mg/L. The absorbance was measured at a wavelength of 570 nm with a 96-well microplate ELISA reader (Bio-Tek Instruments, Inc., Winooski, VT, USA). All experiments were performed in triplicate. The inhibition percentage of tumor cell proliferation was calculated according to the formula below:
where OD and OD0 are the absorbances of the treated cells and the untreated cells, respectively.
3.6.1.3. Effect of LP1 on the Proliferation of Macrophages in Vitro
Isolation of peritoneal macrophages. Macrophages were prepared from BALB/c mice, as described previously [
53]. Briefly, peritoneal macrophages were harvested from 2 to 3 BALB/c mice that had been intraperitoneally injected with 3 mL of thioglycollate for three days before a sterile peritoneal lavage with 10 mL of Hank’s balanced salt solution. After centrifugation at 1000 rpm/min for 5 min, the cell pellets were suspended in RPMI-1640 supplemented with 10% FBS, penicillin (100 IU/mL) and streptomycin (100 μg/mL), seeded in a 96-well plate at a cell density of 2 × 10
6 cells/mL and allowed to adhere for 3 h at 37 °C in a 5% CO
2 humidified incubator. Non-adherent cells were removed by thorough washing, leaving monolayers of adherent cells consisting of ~99% macrophages. The viability of the adherent cells was assessed by the trypan blue exclusion test, and the proportion of macrophages was determined via cell morphology using a microscope (>95%).
Phagocytic assay with the neutral red method. [
54] The phagocytic ability of the macrophages was measured via neutral red uptake. The cells were cultured with LP1 (0, 25, 50 and 100 μg/mL) or LPS (10 μg/mL) at 37 °C and 5% CO
2 for 48 h. the culture media were removed, and 100 μL/well of 0.075% neutral red were added; the cells were then incubated in 96-well plates for 4 h. The supernatant was discarded, and the cells were washed twice with PBS to remove the neutral red that was not phagocytized by the macrophages. Cell lysis buffer (ethanol and 0.01% acetic acid at a ratio of 1:1, 100 μL/well) was added to lyse the cells. After the cells were incubated at room temperature overnight, the optical density at 540 nm was measured with a microplate reader.
Nitric oxide assay. Nitrite accumulation in the medium was used as an indicator of NO production as previously described [
55], and colorimetric assays [
56] with the Griess reagent were used to detect NO levels (Nanjing SunShine Biotechnology Co., Ltd., Nanjing, China). Adherent macrophages were cultured with various concentrations of LP1 (0, 25, 50 and 100 μg/mL) or LPS (10 μg/mL) at 37 °C for 48 h. At the end of the culture period, 100 μL of the isolated supernatant was allowed to react with the Griess reagent (1% sulfanilamide, 0.1% naphthylethylenediamine dihydrochloride and 2.5% H
3PO
4) at room temperature for 10 min. Nitrite production was determined by comparing the absorbance at 550 nm with a standard curve generated using NaNO
2.
Cytokine assays. [
57] Adherent macrophages were cultured with various concentrations of LP1 (0, 25, 50 and 100 μg/mL) or LPS (10 μg/mL) at 37 °C for 24 h. At the end of the culture period, the supernatants were collected and the level of the cytokines, TNF-α, IL-6 and IL-1β, were determined with ELISA kits (Guangxi Nanning Chujie Biological Technology Co., Ltd., Nanning, China), according to the manufacturer’s instructions.
3.6.2. Animal Immune Experiments
3.6.2.1. Chemicals and Animals
Kunming mice (SPF), weighing 20 ± 2 g and 6–8 weeks old, were provided by the Center of Animal Experimentation of Guangxi Medical University (Nanning, China) (experimental animal use license: SYKG Guangxi 2003-0005; laboratory animal production license: SCXKG Guangxi 2003-2003). CY for injection (No. 04120705) was purchased from Shanxi PuDe Pharmaceutical Co., Ltd. (Shanxi, China). The mouse IFN-γ ELISA kit (No. 978941214) and mouse IL-2 ELISA kit (No. 1208911214) were provided by Wuhan Boster Bio-Engineering Co. Ltd. (Wuhan, China). Neutral red (No. E895), concanavalin A (No. c-2010) and LPS (No. L2880), as a B-cell mitogen, were purchased from Beijing Bo Ao Tuoda Technology Co., Ltd. (Beijing, China). MTT (No. 20110613) and 0.25% trypsin (with EDTA) were purchased from Solarbio (Beijing, China).
3.6.2.2. Cyclophosphamide-Induced Immunosuppression Mouse Model
After one day of adaptability feeding, the mice were randomly allocated to a blank group, a cyclophosphamide (CY) inhibition group and an LP1 group, according to weight. Ten mice were in each experimental group (half male and half female), including 3 groups with different doses of LP1: CY + LP1 (I) (320 mg/kg), CY + LP1 (II) (160 mg/kg) and CY + LP1 (III) (80 mg/kg). The mice in the blank control group were given an intraperitoneal injection of saline. The mice in the inhibition group and the experimental group were given an intraperitoneal injection of cyclophosphamide once every three days at a dose of 20 mg/kg and a capacity of 10 mL/kg. Administration was intragastric. The blank control group was given the same amount of physiological saline, with a capacity of 10 mL/kg, once per day over a period of 21 days [
58,
59].
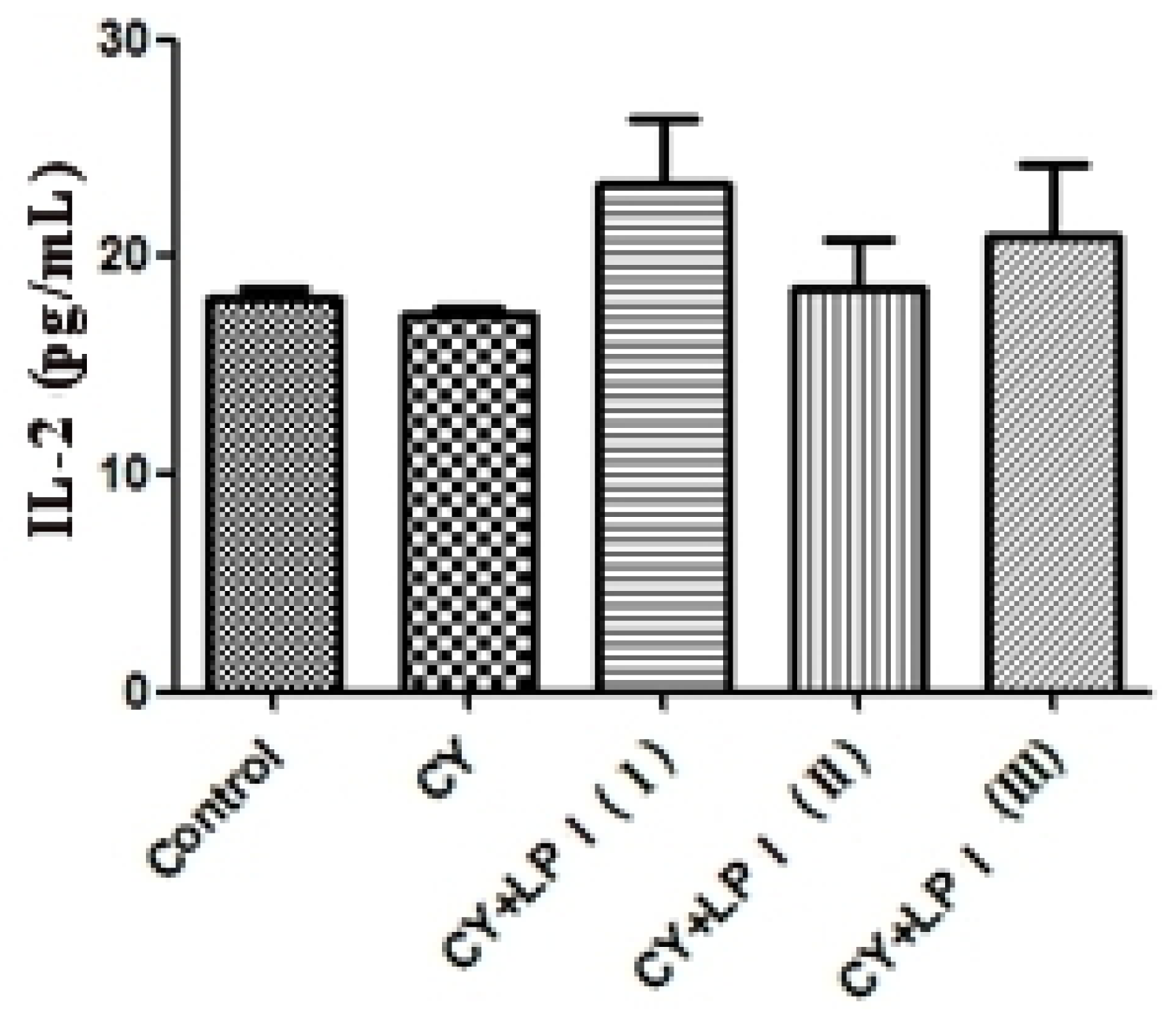
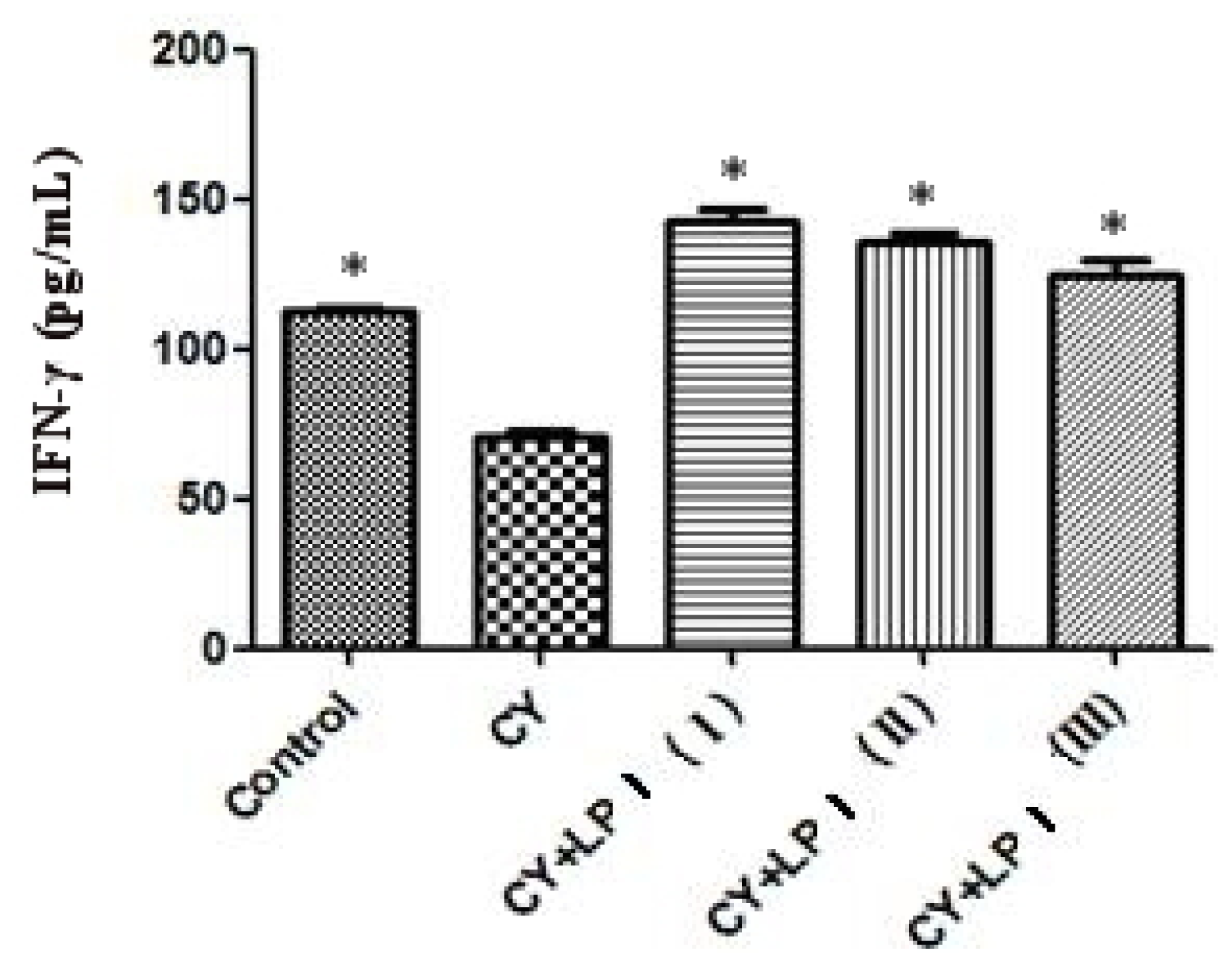
3.6.2.3. Measurement of IL-2 and IFN-γ
Blood was taken from the eyeballs of the mice and put into a 2 mL EP tube. It was then centrifuged for 10 min at 3500 rpm/min at 4 °C. The upper, clarified serum was drawn and the amount of IL-2 and IFN-γ in the serum was measured via ELISA [
60].
3.6.2.4. Macrophage Phagocytosis Assay
Macrophages were prepared from BALB/c mice as described previously [
61]. After removing the eyeballs and soaking the mice with 75% ethanol for 2 min, 5 mL sterile PBS solution was injected into the celiac region of the mice, followed by kneading for 2 min. The peritoneal fluid was then removed with a syringe (while avoiding the abdominal viscera, which could result in bleeding), and the abdominal fluid was centrifuged at 2000 rpm/min for 15 min. The centrifugal supernatant was discarded, and PRMI-1640 with 10% fetal bovine serum (FBS) was then added to the cell pellet. The cells were dispersed by gentle trituration. The cells were seeded at a density of 1 × 10
6 cells/mL in a 96-well plate, with each well containing 100 μL of suspension, and were incubated at 37 °C in a humidified 5% CO
2 atmosphere for 4 h. All cell culture solutions were then discarded, and the non-adherent cells were removed by washing. To each well, 100 μL 0.1% neutral red dye was then added (0.2500 g neutral red in 250 mL 0.9% saline), and the plates were incubated at 37 °C in a 5% CO
2 incubator for another 30 min. Excess neutral red solution was discarded. The cells in each well were washed with PBS solution three times, and 200 μL of a cell lysis solution was added (acetic acid:anhydrous ethanol, 1:1). The plate was gently oscillated and then placed in a 4 °C environment overnight. The optical density at 570 nm was measured for each well with an enzyme marking instrument.
3.6.2.5. Analysis of B- and T-Cell Proliferation
The spleen was weighed, crushed into 1-mm
3 pieces and digested at 37 °C with 0.25% pancreatic enzymes. PRMI-1640 containing 10% calf serum was then added at twice the volume, followed by gentle tapping to mix and render the pancreatic enzymes inactive. This action was performed to reduce the influence of the pancreatic enzymes on the living cells. The material was then passed through a 200-mesh cellular sieve, filtered and centrifuged for 8 min at 1000 rpm/min. The supernatant was discarded, leaving the pellet, which was composed of spleen cells. PRMI-1640 culture medium was then added to prepare a single spleen cell suspension. One hundred microliters of the spleen cell suspension were placed into each well of a 96-well plate at a concentration of 2 × 10
6 cells/mL, and then, 100 μL LPS (30 μg/mL) was added to each well as the B-lymphocyte transformation irritant. This was repeated for each sample in three wells. The plates were incubated at 37 °C in a 5% CO
2 for 66 h; 20 μL of MTT was then added to each well followed by incubation at 37 °C for another 4 h. The supernatants were discarded; 150 μL of dimethyl sulfoxide (DMSO) was then added, and the plates were oscillated. The
A490 was recorded as an index of stimulating the proliferation of the B-cells in an enzyme marking instrument. The value for the experiments that did not involve stimulation with LPS was set at 100%, and the values for the other experiments were converted into a percentage expression. A statistical analysis was performed. For monitoring the proliferation of T-cells, the procedure was the same as that described for B-lymphocyte cell proliferation above, except that concanavalin A (ConA) replaced LPS for the stimulation [
62].
3.7. Statistics
All the experiments were performed at least in duplicate, and the analyses of all the samples were run in triplicate and averaged. The statistical analysis utilized SPSS 13.0 software (SPSS Inc., Chicago, IL, USA) for processing. The results are presented as the means of three determinations, χ̄ ± SD. The results were analyzed using a one-way analysis of variance (ANOVA) for the mean differences among the samples. A p-value <0.05 was considered to be statistically significant.
4. Conclusions
The water-soluble polysaccharide LP1 was successfully isolated from the Dimocarpus longan pulp, purified and partially characterized. LP1 contained α- and β-d-glucans. The apparent mean Mr of the sample was 1.1 × 105 Da. The sample exhibited a significant inhibition of the growth of SKOV3 and HO8910 cells, with the inhibition percentage of HO8910 cells exceeding 50%. LP1 also stimulated spleen lymphocyte proliferation and macrophage function in a dose-dependent manner. The experimental results suggest that LP1 has significant potential as a safe and effective reagent to prevent the development of tumor cells by killing the tumor cells directly and, more importantly, by improving immunocompetence, as observed in the in vitro and murine experiments, respectively. Further investigation of LP1 is important and will be reported elsewhere, as no medicine currently exists that can effectively treat cancer without side effects.