Poly(lactide-co-trimethylene carbonate) and Polylactide/Polytrimethylene Carbonate Blown Films
Abstract
:1. Introduction
2. Results and Discussion
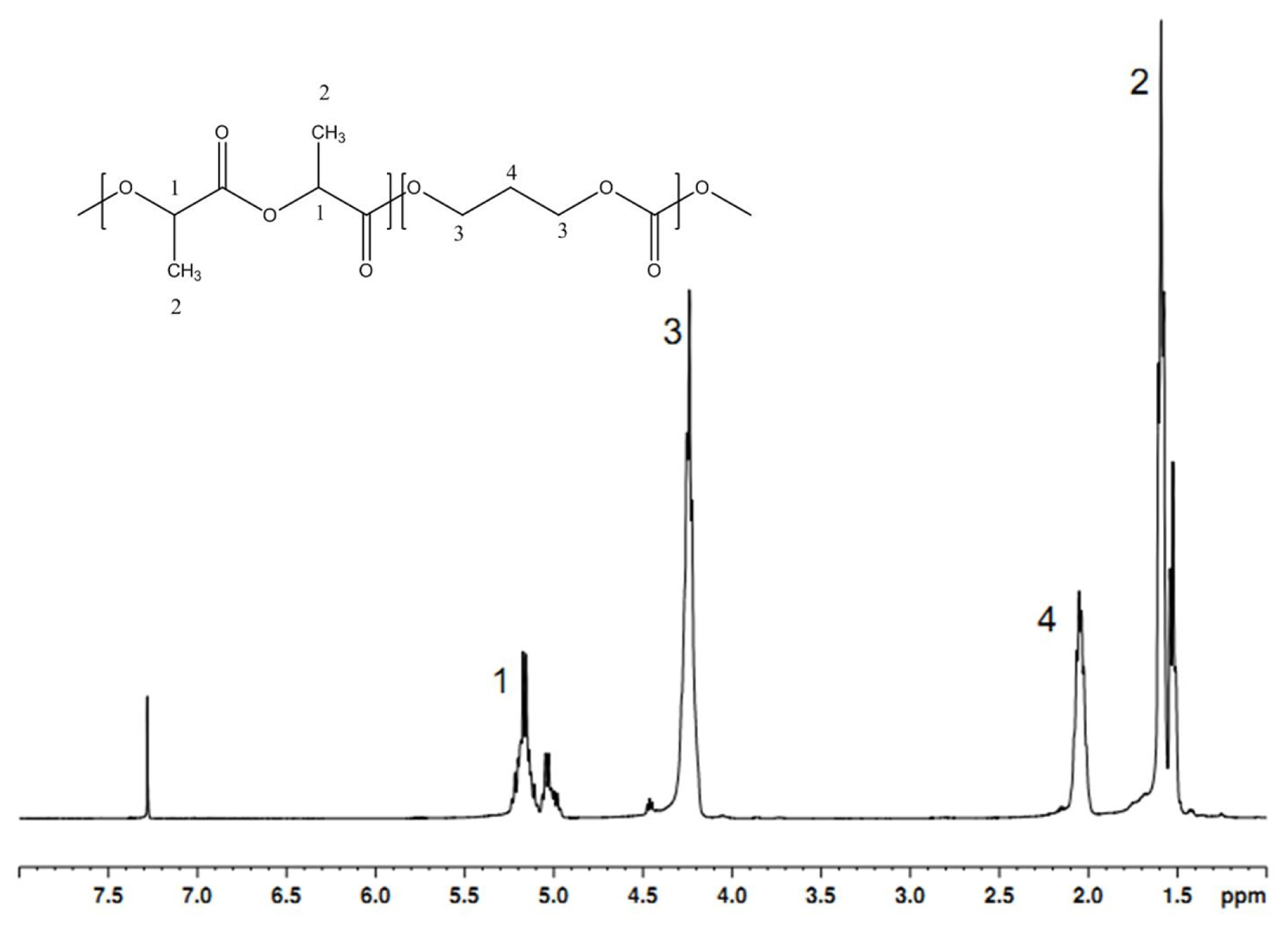
2.1. The Characteristics of the PTMC and P(LA-TMC) Polymers
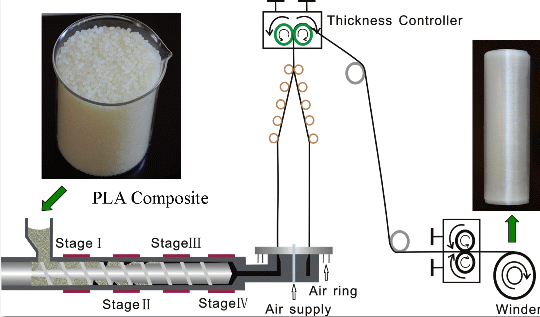
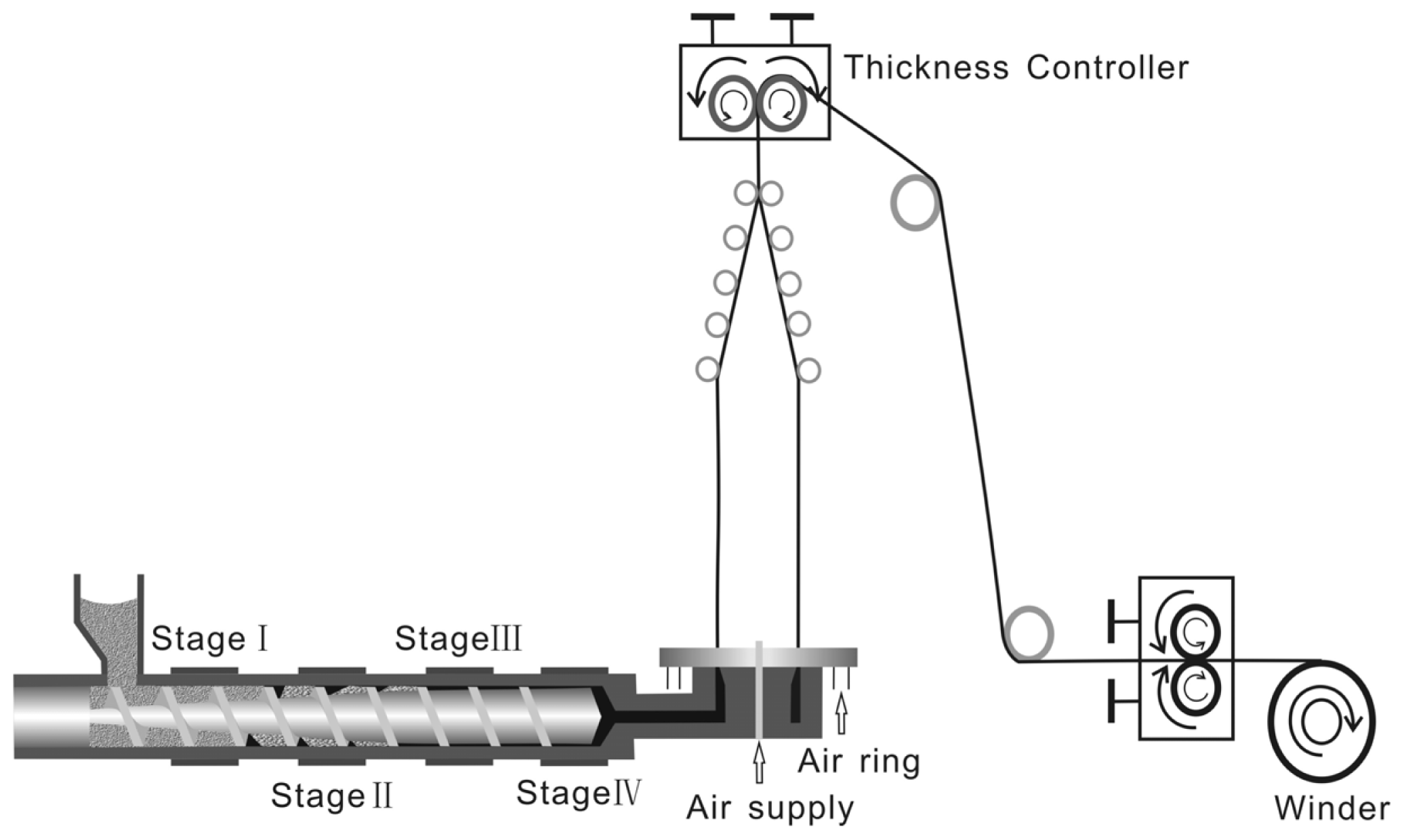
2.2. The Processing Parameters of Film Blowing
2.3. FTIR Measurements
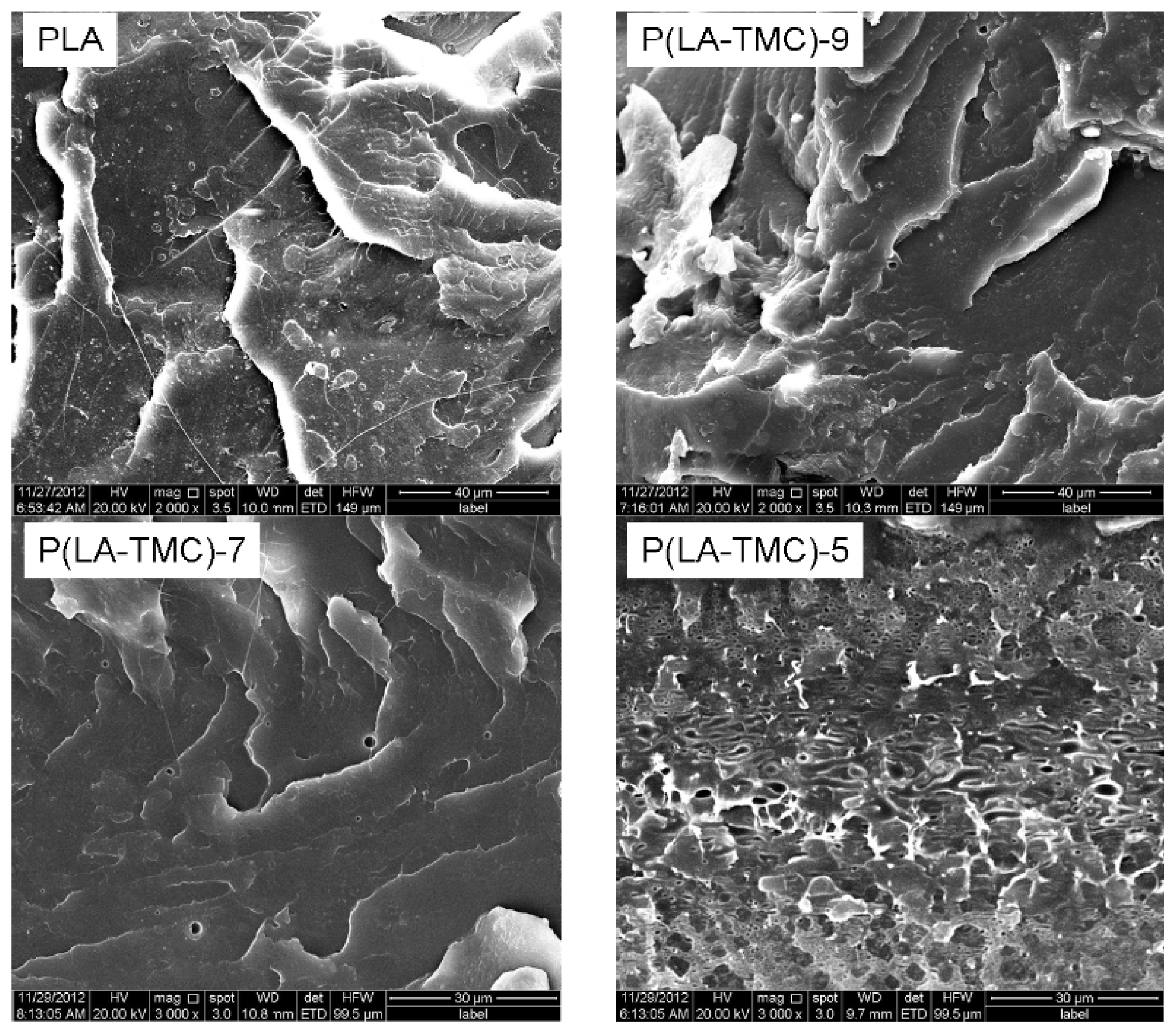
2.4. SEM Measurements
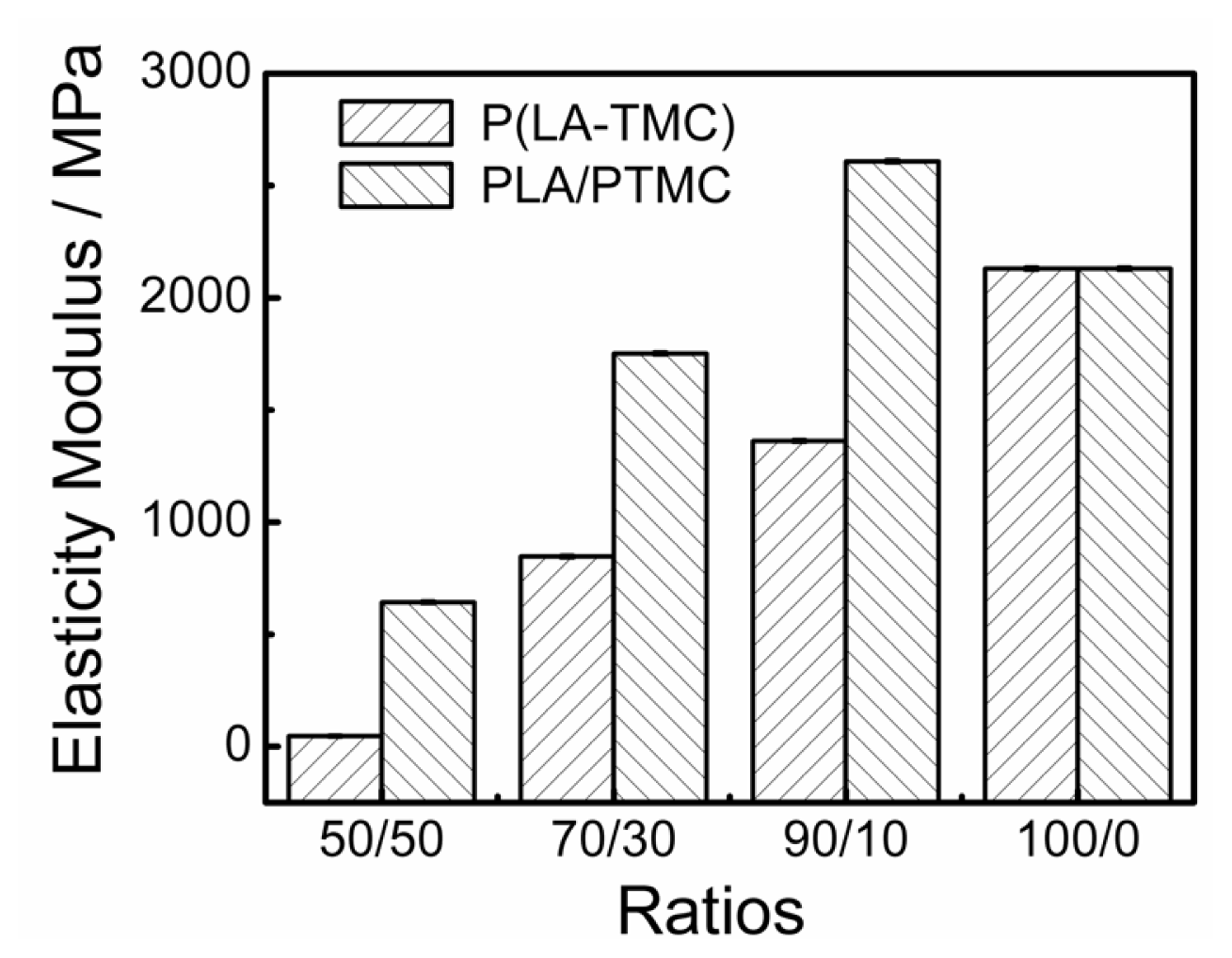
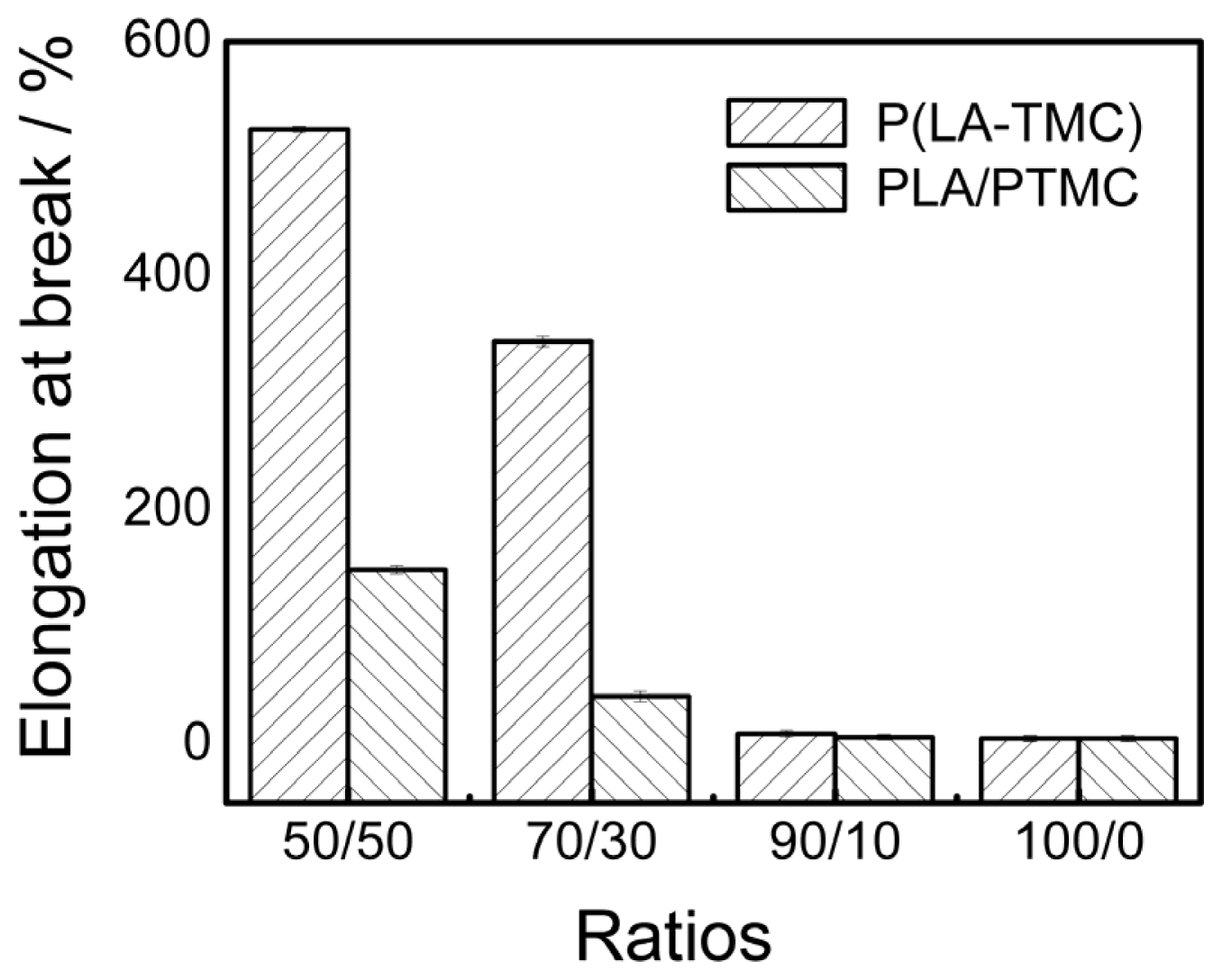
2.5. Mechanical Properties Analysis
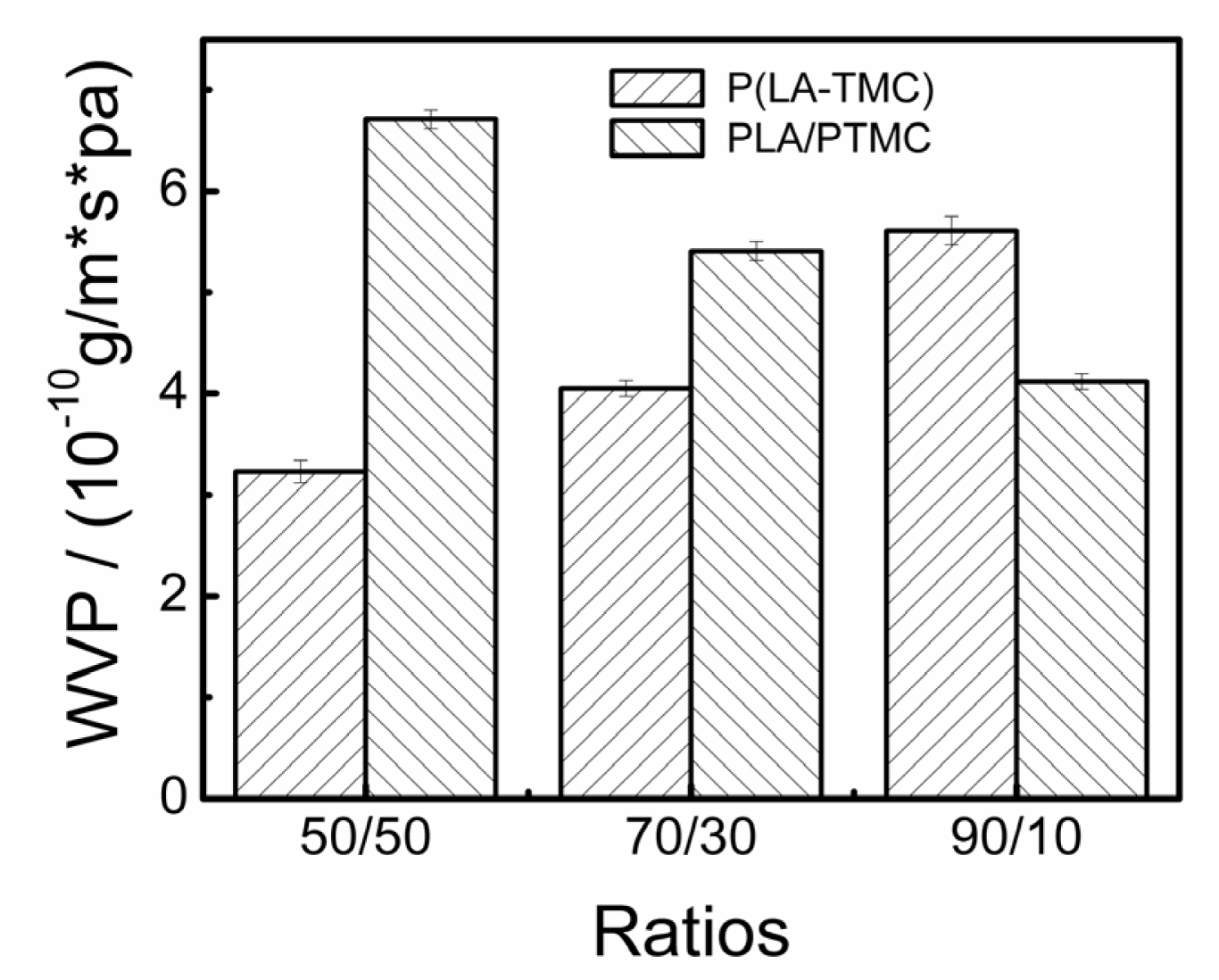
2.6. WVP Measurement
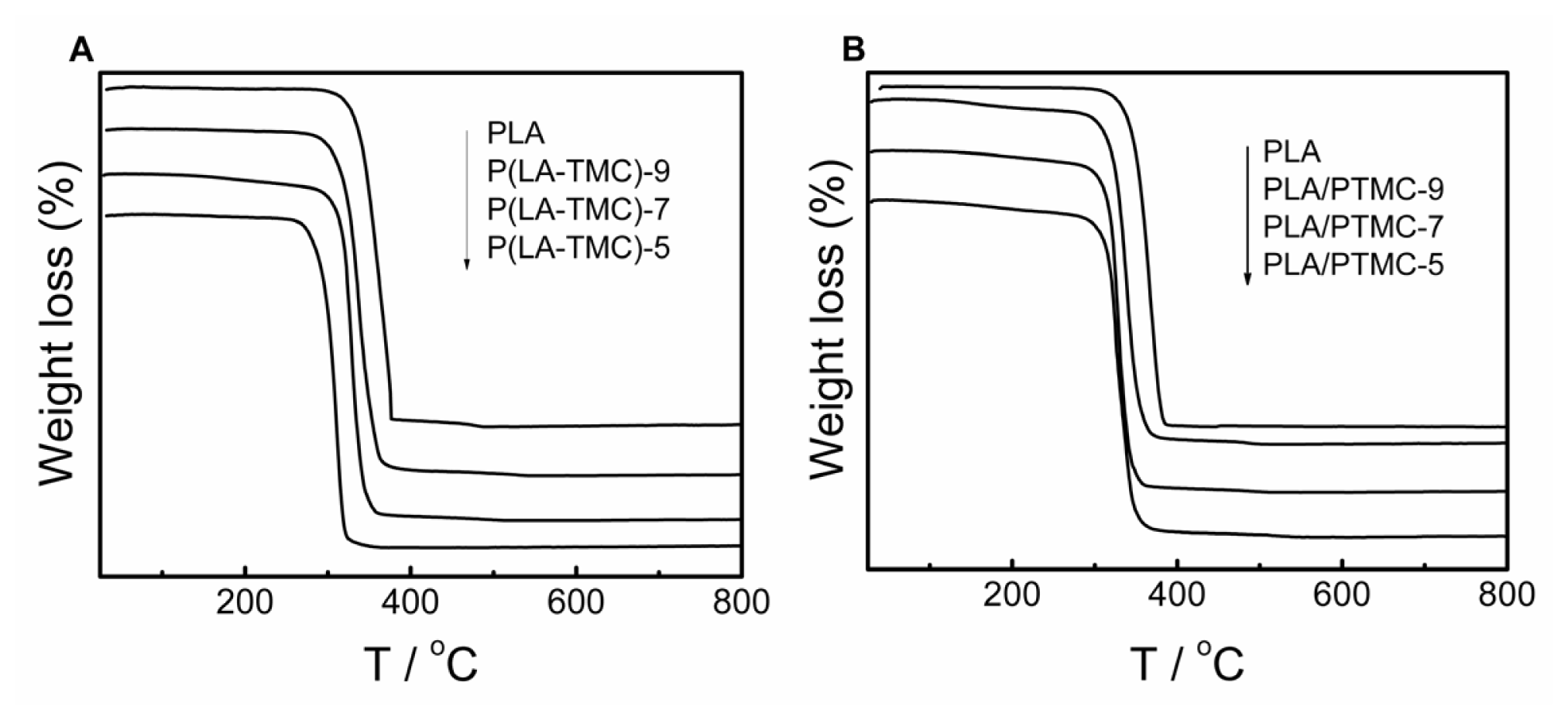
2.7. TG Measurements
2.8. Effects of P(LA-TMC) Film and PLA/PTMC Film on the Shelf Life of Apples
3. Experimental Section
3.1. Materials
3.2. Polymerization of Poly(lactide-co-trimethylene carbonate)
3.3. Preparation of Blown Films
3.4. 1H-NMR Measurement
3.5. GPC Measurements
3.6. FTIR Spectra Measurements
3.7. SEM Measurements
3.8. Mechanical Properties
3.9. Water Vapor Permeability (WVP) Measurement
3.10. TG Measurements
3.11. Effects of P(LA-TMC) Film and PLA/PTMC Film on the Shelf Life of Apples
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Sirisinha, K.; Somboom, W. Melt characteristics, mechanical, and thermal properties of blown film from modified blends of poly(butylenes adipate-co-terephthalate) and poly(lactide). J. Appl. Polym. Sci 2012, 124, 4986–4992. [Google Scholar]
- Siracusa, V.; Rocculi, P.; Romani, S.; Rosa, M.D. Biodegradable polymers for food packaging: A review. Trends Food Sci. Technol 2008, 19, 634–643. [Google Scholar]
- Odent, J.; Leclère, P.; Raquez, J.M.; Dubois, P. Toughening of polylactide by tailoring phase-morphology with P[CL-co-LA] random copolyesters as biodegradable impact modifiers. Eur. Polym. J 2013, 49, 914–922. [Google Scholar]
- Terao, K.; Miyake, J.; Watanabe, J.; Ikeda, Y. Regulation of protein loading on poly(trimethylene carbonate), poly(l-lactic acid), and their copolymer: Effect of surface enrichment by polymer crystallinity. Mater. Sci. Eng. C 2012, 32, 988–993. [Google Scholar]
- Zhang, C.M.; Wang, W.W.; Huang, Y.; Pan, Y.H.; Jiang, L.; Dan, Y.; Luo, Y.Y.; Peng, Z. Thermal, mechanical and rheological properties of polylactide toughened by expoxidized natural rubber. Mater. Des 2013, 45, 198–205. [Google Scholar]
- Patel, R.; Ruehle, D.A.; Dorgan, J.R.; Halley, P.; Martin, D. Biorenewable blends of polyamide-11 and polylactide. Polym. Eng. Sci 2013. [Google Scholar] [CrossRef]
- Park, K.; Uk Ha, J.; Xanthos, M. Ionic liquids as plasticizers/lubricants for polylactic acid. Polym. Eng. Sci 2010, 50, 1105–1110. [Google Scholar]
- Wang, X.M.; Zhuang, Y.G.; Dong, L.S. Study of biodegradable polylactide/poly(butylene carbonate) blend. J. Appl. Polym. Sci 2013, 127, 471–477. [Google Scholar]
- Miguel, A.; Timoneda, M.; Engel, E.; Petra, H.; Oscar, C.; Josep, P.; Soledad, A.; Perales, C. The effect of the composition of PLA films and lactate release on glial and neuronal maturation and the maintenance of the neuronal progenitor niche. Biomaterials 2013, 34, 2221–2233. [Google Scholar]
- Bajpai, P.K.; Singh, I.; Madaan, J. Tribological behavior of natural fiber reinforced PLA composites. Wear 2013, 297, 829–840. [Google Scholar]
- Tyson, T.; Finne, A.; Albertsson, C. Degradable porous scaffolds from various l-Lactide and trimethylene carbonate copolymers obtained by a simple and effective method. Biomacromolecules 2009, 10, 149–154. [Google Scholar]
- Song, Y.; Kamphuis, M.M.J.; Zhang, Z.; Sterk, L.M.T.; Vermes, I.; Poot, A.A.; Feijena, J.; Grijpma, D.W. Flexible and elastic porous poly(trimethylene carbonate) structures for use in vascular tissue engineering. Acta Biomater 2010, 6, 1269–1277. [Google Scholar]
- Ajiro, H.; Takahashi, Y.; Akashi, M.; Fujiwara, T. Polylactide block copolymers using trimethylene carbonate with methoxyethoxy side groups for dual modification of hydrophilicity and biodegradability. Macromol. Biosci 2012, 12, 1315–1320. [Google Scholar]
- Sharifi, S.; Blanquer, S.B.G.; van Kooten, T.G.; Grijpma, D.W. Biodegradable nanocomposite hydrogel structures with enhanced mechanical properties prepared by photo-crosslinking solutions of poly(trimethylene carbonate)-poly(ethylene glycol)-poly(trimethylene carbonate) macromonomers and nanoclay particles. Acta Biomater 2012, 8, 4233–4243. [Google Scholar]
- Zhang, H.H.; Sun, B.W.; Chen, Y.Q.; Wang, J.L. Synthesis of Y-shaped poly(N,N-dimethylamino-2-ethylmethacrylate) and poly(trimethylene carbonate) from a new heterofunctional initiator. Polym. Eng. Sci 2011, 51, 776–784. [Google Scholar]
- Dargaville, B.L.; Vaquette, C.; Rasoul, F.; Cooper-White, J.J.; Campbell, J.H.; Whittaker, A.K. Electrospinning and crosslinking of low-molecular-weight poly(trimethylene carbonate-co-(l)-lactide) as an elastomeric scaffold for vascular engineering. Acta Biomater 2013, 9, 6885–6897. [Google Scholar]
- Van Leeuwen, A.C.; Bos, R.R.M.; Grijpma, D.W. Composite materials based on poly(trimethylene carbonate) and β-tricalcium phosphate for orbital floor and wall reconstruction. J. Biomed. Mater. Res. B 2012, 100B, 1610–1620. [Google Scholar]
- Yang, L.Q.; He, B.; Meng, S.; Zhang, J.Z.; Li, M.; Guo, J.; Guan, Y.M.; Li, J.X.; Gu, Z.W. Biodegradable cross-linked poly(trimethylene carbonate) networks for implant applications: Synthesis and properties. Polymer 2013, 54, 2668–2675. [Google Scholar]
- Dargaville, B.L.; Vaquette, C.; Peng, H.; Rasoul, F.; Chau, Y.Q.; Cooper-White, J.J.; Campbell, J.H.; Whittaker, A.K. Cross-linked poly(trimethylene carbonate-co-l-lactide) as a biodegradable, elastomeric scaffold for vascular engineering applications. Biomacromolecules 2011, 12, 3856–3869. [Google Scholar]
- Tong, R.; Cheng, J.J. Drug-initiated, controlled ring-opening polymerization for the synthesis of polymer—Drug conjugates. Macromolecules 2012, 45, 2225–2232. [Google Scholar]
- Adamus, A.; Wach, R.A.; Olejnik, A.K.; Dzierzawska, J.; Rosia, J.M. Degradation of nerve guidance channels based on a poly(l-lactic acid) poly(trimethylene carbonate) biomaterial. Polym. Degrad. Stabil 2012, 97, 532–540. [Google Scholar]
- Auras, R.; Harte, B.; Selke, S. An overview of polylactides as packaging materials. Macromol. Biosci 2004, 4, 835–864. [Google Scholar]
- Siracusa, V.; Blanco, I.; Romani, S.; Tylewicz, U.; Rocculi, P.; Rosa, M.D. Poly(lactic acid)-modified films for food packaging application: Physical, mechanical, and barrier behavior. J. Appl. Polym. Sci 2012, 125, 390–401. [Google Scholar]
- Bourtoom, T.; Chinnan, M.S. Preparation and properties of rice starch-chitosan blend biodegradable film. LWT-Food Sci. Technol 2008, 41, 1633–1641. [Google Scholar]
- Van Puijenbroek, A.; Josephus, M. Biodegradable Laminate and Packaging Foil Comprising such a Laminate WO1999043497A1, 2 September 1999.
- Zenkiewicz, M.; Richert, J.; Rozanski, A. Effect of blow molding ratio on barrier properties of polylactide nanocomposite films. Polym. Test 2010, 29, 251–257. [Google Scholar]
- Márquez, Y.; Franco, L.; Puiggalí, J. Thermal degradation studies of poly(trimethylene carbonate) blends with either polylactide or polycaprolactone. Thermochim. Acta 2012, 550, 65–75. [Google Scholar]
- He, L.R.; Mu, C.D.; Shi, J.B.; Zhang, Q.; Shi, B.; Lin, W. Modification of collagen with a natural cross-linker, procyanidin. Int. J. Biol. Macromol 2011, 48, 354–359. [Google Scholar]


| Polymer | Mn [kDa] | Mw [kDa] | PDI |
|---|---|---|---|
| PTMC | 6.7 | 10.8 | 1.62 |
| P(LA-TMC)-5 | 51.6 | 83.4 | 1.62 |
| P(LA-TMC)-7 | 58.6 | 94.7 | 1.59 |
| P(LA-TMC)-9 | 91 | 143 | 1.57 |
| Stage I (°C) | Stage II (°C) | Stage III (°C) | Stage IV (°C) | Screw speed (pr/min) | Macroscopic morphology of film |
|---|---|---|---|---|---|
| 100 | 140 | 140 | 130 | 40 | Film with white points |
| 110 | 150 | 150 | 137 | 40 | Smooth film |
| 120 | 160 | 160 | 140 | 40 | Unformed film |
| 110 | 150 | 150 | 137 | 20 | Unformed film |
| 110 | 150 | 150 | 137 | 60 | Porous film |
| StageI (°C) | Stage II (°C) | Stage III (°C) | Stage IV (°C) | Screw speed (pr/min) | Macroscopic morphology of film |
|---|---|---|---|---|---|
| 160 | 175 | 175 | 165 | 40 | Film with white points |
| 170 | 185 | 185 | 175 | 40 | Smooth film |
| 180 | 195 | 195 | 185 | 40 | Unformed film |
| 170 | 185 | 185 | 175 | 20 | Unformed film |
| 170 | 185 | 185 | 175 | 60 | Porous film |
| Polymer | Initial decomposition temperature/°C | Decomposition temperature/°C |
|---|---|---|
| PLA | 315 | 364 |
| P(LA-TMC)-9 | 275 | 340 |
| P(LA-TMC)-7 | 265 | 328 |
| P(LA-TMC)-5 | 259 | 310 |
| PLA/PTMC-9 | 284 | 339 |
| PLA/PTMC-7 | 283 | 329 |
| PLA/PTMC-5 | 276 | 325 |
| Film | P(LA-TMC)-7 | PLA/PTMC-7 | LDPE | Control | |
|---|---|---|---|---|---|
| Weight loss (%) | 0 | 0 | 0 | 0 | 0 |
| 7 | 1.9 ± 0.4 a | 1.8 ± 0.2 a | 2.0 ± 0.1 a | 5.6 ± 0.4 b | |
| 14 | 3.0 ± 0.2 a | 3.2 ± 0.2 a | 2.9 ± 0.0 a | 9.2 ± 0.6 b | |
| 21 | 3.9 ± 0.1 a | 3.6 ± 0.1 a | 3.6 ± 0.3 a | 14.3 ± 0.5 b | |
| 28 | 7.4 ± 0.3 a | 7.2 ± 0.6 a | 6.7 ± 0.5 a | 20.8 ± 1.3 b | |
| Firmness (N) | 0 | 22.6 ± 0.2 a | 22.6 ± 0.2 a | 22.6 ± 0.2 a | 22.6 ± 0.2 a |
| 7 | 21.3 ± 0.1 b | 21.9 ± 0.7 b | 21.8 ± 1.1 b | 19.2 ± 0.3 a | |
| 14 | 19.4 ± 0.4 b | 19.2 ± 0.2 b | 19.8 ± 0.9 b | 17.3 ± 0.5 a | |
| 21 | 18.5 ± 0.8 b | 18.0 ± 0.8 b | 18.7 ± 0.7 b | 10.5 ± 0.6 a | |
| 28 | 16.5 ± 1.0 a | 16.1 ± 0.4 a | 15.9 ± 0.1 a | -- | |
| Acidity | 0 | 1.24 ± 0.03 a | 1.24 ± 0.03 a | 1.24 ± 0.03 a | 1.24 ± 0.03 a |
| 7 | 1.21 ± 0.11 b | 1.20 ± 0.09 b | 1.21 ± 0.02 b | 1.16 ± 0.05 a | |
| 14 | 1.15 ± 0.07 b | 1.14 ± 0.07 b | 1.14 ± 0.12 b | 1.07 ± 0.13 a | |
| 21 | 1.12 ± 0.08 b | 1.11 ± 0.15 b | 1.12 ± 0.06 b | 1.05 ± 0.09 a | |
| 28 | 1.10 ± 0.06 a | 1.09 ± 0.07 a | 1.09 ± 0.07 a | -- | |
| Sensory evaluation | 0 | 9.8 ± 0.2 a | 9.8 ± 0.2 a | 9.8 ± 0.2 a | 9.8 ± 0.2 a |
| 7 | 8.9 ± 0.4 b | 8.8 ± 0.8 b | 9.0 ± 0.6 b | 7.5 ± 0.2 a | |
| 14 | 8.1 ± 0.6 b | 8.1 ± 0.2 b | 7.9 ± 0.7 b | 6.1 ± 0.8 a | |
| 21 | 6.5 ± 0.7 b | 6.8 ± 0.4 b | 7.0 ± 0.3 b | 3.3 ± 0.1 a | |
| 28 | 5.8 ± 0.1 a | 5.9 ± 0.5 a | 6.2 ± 0.6 a | -- |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, H.; Chang, J.; Qin, Y.; Wu, Y.; Yuan, M.; Zhang, Y. Poly(lactide-co-trimethylene carbonate) and Polylactide/Polytrimethylene Carbonate Blown Films. Int. J. Mol. Sci. 2014, 15, 2608-2621. https://doi.org/10.3390/ijms15022608
Li H, Chang J, Qin Y, Wu Y, Yuan M, Zhang Y. Poly(lactide-co-trimethylene carbonate) and Polylactide/Polytrimethylene Carbonate Blown Films. International Journal of Molecular Sciences. 2014; 15(2):2608-2621. https://doi.org/10.3390/ijms15022608
Chicago/Turabian StyleLi, Hongli, Jiangping Chang, Yuyue Qin, Yan Wu, Minglong Yuan, and Yingjie Zhang. 2014. "Poly(lactide-co-trimethylene carbonate) and Polylactide/Polytrimethylene Carbonate Blown Films" International Journal of Molecular Sciences 15, no. 2: 2608-2621. https://doi.org/10.3390/ijms15022608