Adenosine Receptors: Expression, Function and Regulation
Abstract
:1. Introduction
2. Desensitization of Adenosine Receptors
2.1. A1AR
2.2. A2AAR
2.3. A2BAR
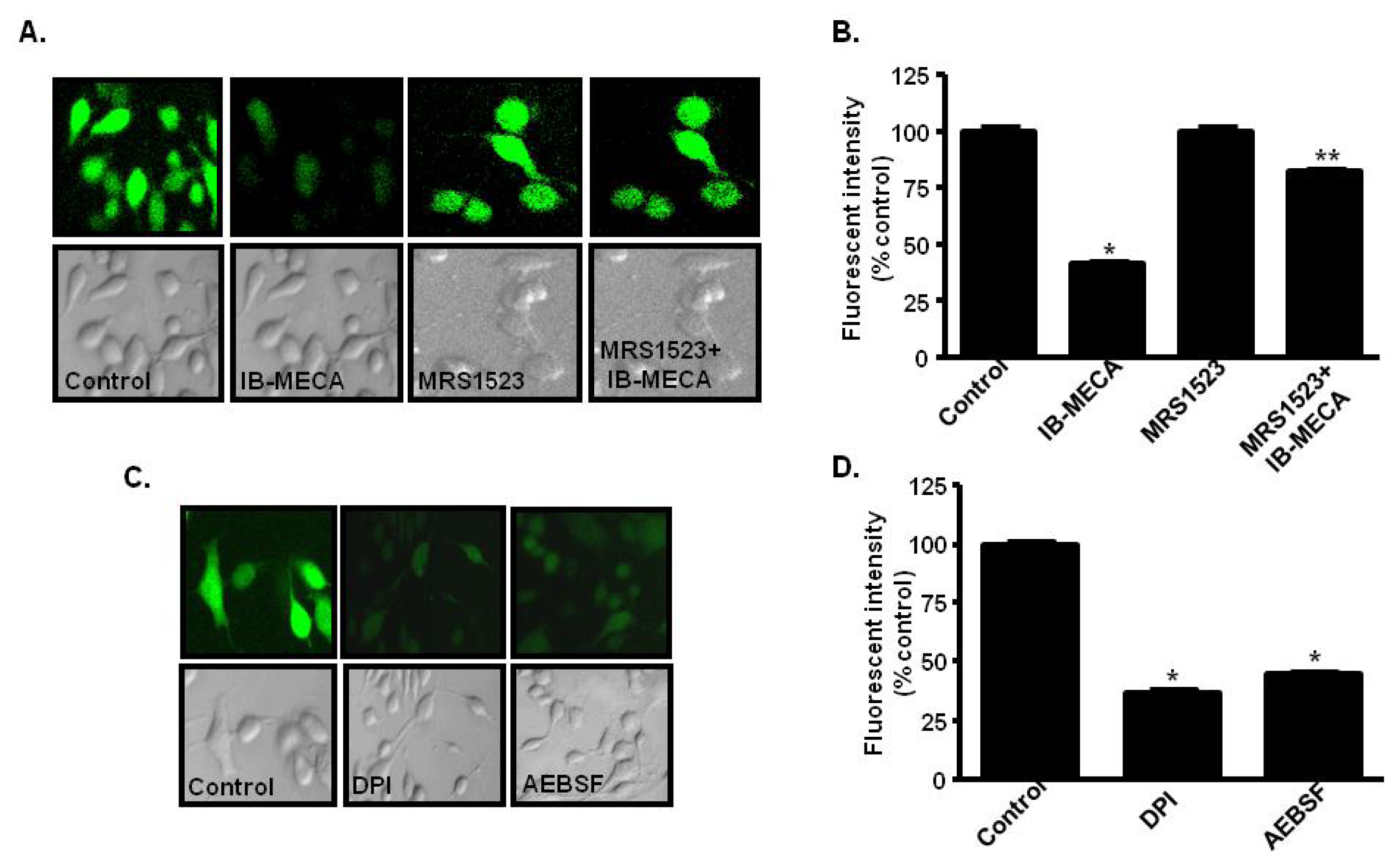
2.4. A3AR
3. NF-κB Regulation of A1AR and A2AAR Expression
4. Adenosine Receptor Oligomers
5. Adenosine Receptors in the Control of Sleep
6. Adenosine Receptor and Protection against Hearing Loss
7. Adenosine Receptors in Cancer
8. Conclusions
Acknowledgments
Conflicts of Interest
References
- Sebastiao, A.M.; Ribeiro, J.A. Fine-tuning neuromodulation by adenosine. Trends Pharmacol. Sci 2000, 21, 341–346. [Google Scholar]
- de Mendoncca, A.; Ribeiro, J.A. Adenosine and synaptic plasticity. Drug Dev. Res 2001, 52, 283–290. [Google Scholar]
- Cunha, R.A. Adenosine as a neuromodulator and as a homeostatic regulator in the nervous system: Different roles, different sources and different receptors. Neurochem. Int 2001, 38, 107–125. [Google Scholar]
- Ferreira, J.M.; Paes-de-Carvalho, R. Long-term activation of adenosine A(2a) receptors blocks glutamate excitotoxicity in cultures of avian retinal neurons. Brain Res 2001, 900, 169–176. [Google Scholar]
- Fredholm, B.B.; AP, I.J.; Jacobson, K.A.; Linden, J.; Muller, C.E. International Union of Basic and Clinical Pharmacology. LXXXI. Nomenclature and classification of adenosine receptors—An update. Pharmacol. Rev 2011, 63, 1–34. [Google Scholar]
- Li, J.; Fenton, R.A.; Wheeler, H.B.; Powell, C.C.; Peyton, B.D.; Cutler, B.S.; Dobson, J.G., Jr. Adenosine A2a receptors increase arterial endothelial cell nitric oxide. J. Surg. Res 1998, 80, 357–364. [Google Scholar]
- Hasko, G.; Pacher, P. A2A receptors in inflammation and injury: Lessons learned from transgenic animals. J. Leukoc. Biol 2008, 83, 447–455. [Google Scholar]
- Bouma, M.G.; Stad, R.K.; van den Wildenberg, F.A.; Buurman, W.A. Differential regulatory effects of adenosine on cytokine release by activated human monocytes. J. Immunol 1994, 153, 4159–4168. [Google Scholar]
- Van der Graaf, P.H.; Van Schaick, E.A.; Visser, S.A.; De Greef, H.J.; Ijzerman, A.P.; Danhof, M. Mechanism-based pharmacokinetic-pharmacodynamic modeling of antilipolytic effects of adenosine A(1) receptor agonists in rats: Prediction of tissue-dependent efficacy in vivo. J. Pharmacol. Exp. Ther. 1999, 290, 702–709. [Google Scholar]
- Latini, S.; Pedata, F. Adenosine in the central nervous system: Release mechanisms and extracellular concentrations. J. Neurochem 2001, 79, 463–484. [Google Scholar]
- Zimmermann, H. Extracellular metabolism of ATP and other nucleotides. Naunyn. Schmiedebergs Arch. Pharmacol 2000, 362, 299–309. [Google Scholar]
- Paes-De-Carvalho, R. Adenosine as a signaling molecule in the retina: Biochemical and developmental aspects. An. Acad. Bras. Cienc 2002, 74, 437–451. [Google Scholar]
- Jarvis, S.M.; Young, J.D. Nucleoside transport in rat erythrocytes: Two components with differences in sensitivity to inhibition by nitrobenzylthioinosine and p-chloromercuriphenyl sulfonate. J Membr. Biol 1986, 93, 1–10. [Google Scholar]
- Anderson, C.M.; Xiong, W.; Geiger, J.D.; Young, J.D.; Cass, C.E.; Baldwin, S.A.; Parkinson, F.E. Distribution of equilibrative, nitrobenzylthioinosine-sensitive nucleoside transporters (ENT1) in brain. J. Neurochem 1999, 73, 867–873. [Google Scholar]
- Ritzel, M.W.; Yao, S.Y.; Huang, M.Y.; Elliott, J.F.; Cass, C.E.; Young, J.D. Molecular cloning and functional expression of cDNAs encoding a human Na+-nucleoside cotransporter (hCNT1). Am. J. Physiol 1997, 272, C707–C714. [Google Scholar]
- Griffith, D.A.; Jarvis, S.M. Nucleoside and nucleobase transport systems of mammalian cells. Biochim. Biophys. Acta 1996, 1286, 153–181. [Google Scholar]
- Baldwin, J.M. Structure and function of receptors coupled to G proteins. Curr. Opin. Cell Biol 1994, 6, 180–190. [Google Scholar]
- Schoneberg, T.; Schulz, A.; Gudermann, T. The structural basis of G-protein-coupled receptor function and dysfunction in human diseases. Rev. Physiol. Biochem. Pharmacol 2002, 144, 143–227. [Google Scholar]
- Ramkumar, V.; Stiles, G.L.; Beaven, M.A.; Ali, H. The A3 adenosine receptor is the unique adenosine receptor which facilitates release of allergic mediators in mast cells. J. Biol. Chem 1993, 268, 16887–16890. [Google Scholar]
- Ali, H.; Muller, C.E.; Daly, J.W.; Beaven, M.A. Methylxanthines block antigen-induced responses in RBL-2H3 cells independently of adenosine receptors or cyclic AMP: Evidence for inhibition of antigen binding to IgE. J. Pharmacol. Exp. Ther 1991, 258, 954–962. [Google Scholar]
- Jacobson, K.A.; Gao, Z.G. Adenosine receptors as therapeutic targets. Nat. Rev. Drug Discov 2006, 5, 247–264. [Google Scholar]
- Dickenson, J.M.; Blank, J.L.; Hill, S.J. Human adenosine A1 receptor and P2Y2-purinoceptormediated activation of the mitogen-activated protein kinase cascade in transfected CHO cells. Br. J. Pharmacol 1998, 124, 1491–1499. [Google Scholar]
- Schulte, G.; Fredholm, B.B. Human adenosine A(1), A(2A), A(2B), and A(3) receptors expressed in Chinese hamster ovary cells all mediate the phosphorylation of extracellular-regulated kinase 1/2. Mol. Pharmacol 2000, 58, 477–482. [Google Scholar]
- Faure, M.; Voyno-Yasenetskaya, T.A.; Bourne, H.R. cAMP and beta gamma subunits of heterotrimeric G proteins stimulate the mitogen-activated protein kinase pathway in COS-7 cells. J. Biol. Chem 1994, 269, 7851–7854. [Google Scholar]
- Socodato, R.; Brito, R.; Calaza, K.C.; Paes-de-Carvalho, R. Developmental regulation of neuronal survival by adenosine in the in vitro and in vivo avian retina depends on a shift of signaling pathways leading to CREB phosphorylation or dephosphorylation. J. Neurochem 2011, 116, 227–239. [Google Scholar]
- Biber, K.; Klotz, K.N.; Berger, M.; Gebicke-Harter, P.J.; van Calker, D. Adenosine A1 receptor-mediated activation of phospholipase C in cultured astrocytes depends on the level of receptor expression. J. Neurosci 1997, 17, 4956–4964. [Google Scholar]
- Othman, T.; Yan, H.; Rivkees, S.A. Oligodendrocytes express functional A1 adenosine receptors that stimulate cellular migration. Glia 2003, 44, 166–172. [Google Scholar]
- Gebicke-Haerter, P.J.; Christoffel, F.; Timmer, J.; Northoff, H.; Berger, M.; Van Calker, D. Both adenosine A1- and A2-receptors are required to stimulate microglial proliferation. Neurochem. Int 1996, 29, 37–42. [Google Scholar]
- Li, X.X.; Nomura, T.; Aihara, H.; Nishizaki, T. Adenosine enhances glial glutamate efflux via A2a adenosine receptors. Life Sci 2001, 68, 1343–1350. [Google Scholar]
- Melani, A.; Cipriani, S.; Vannucchi, M.G.; Nosi, D.; Donati, C.; Bruni, P.; Giovannini, M.G.; Pedata, F. Selective adenosine A2a receptor antagonism reduces JNK activation in oligodendrocytes after cerebral ischaemia. Brain 2009, 132, 1480–1495. [Google Scholar]
- Hettinger, B.D.; Lee, A.; Linden, J.; Rosin, D.L. Ultrastructural localization of adenosine A2A receptors suggests multiple cellular sites for modulation of GABAergic neurons in rat striatum. J. Comp. Neurol 2001, 431, 331–346. [Google Scholar]
- Cunha, R.A.; Almeida, T.; Ribeiro, J.A. Modification by arachidonic acid of extracellular adenosine metabolism and neuromodulatory action in the rat hippocampus. J. Biol. Chem 2000, 275, 37572–37581. [Google Scholar]
- Lopes, L.V.; Cunha, R.A.; Kull, B.; Fredholm, B.B.; Ribeiro, J.A. Adenosine A(2A) receptor facilitation of hippocampal synaptic transmission is dependent on tonic A(1) receptor inhibition. Neuroscience 2002, 112, 319–329. [Google Scholar]
- Rebola, N.; Oliveira, C.R.; Cunha, R.A. Transducing system operated by adenosine A(2A) receptors to facilitate acetylcholine release in the rat hippocampus. Eur. J. Pharmacol 2002, 454, 31–38. [Google Scholar]
- Rebola, N.; Rodrigues, R.J.; Oliveira, C.R.; Cunha, R.A. Different roles of adenosine A1, A2A and A3 receptors in controlling kainate-induced toxicity in cortical cultured neurons. Neurochem. Int 2005, 47, 317–325. [Google Scholar]
- Allaman, I.; Lengacher, S.; Magistretti, P.J.; Pellerin, L. A2B receptor activation promotes glycogen synthesis in astrocytes through modulation of gene expression. Am. J. Physiol. Cell Physiol 2003, 284, C696–C704. [Google Scholar]
- Sebastiao, A.M.; Ribeiro, J.A. Adenosine A2 receptor-mediated excitatory actions on the nervous system. Prog. Neurobiol 1996, 48, 167–189. [Google Scholar]
- Brand, A.; Vissiennon, Z.; Eschke, D.; Nieber, K. Adenosine A(1) and A(3) receptors mediate inhibition of synaptic transmission in rat cortical neurons. Neuropharmacology 2001, 40, 85–95. [Google Scholar]
- Hammarberg, C.; Schulte, G.; Fredholm, B.B. Evidence for functional adenosine A3 receptors in microglia cells. J. Neurochem 2003, 86, 1051–1054. [Google Scholar]
- Wittendorp, M.C.; Boddeke, H.W.; Biber, K. Adenosine A3 receptor-induced CCL2 synthesis in cultured mouse astrocytes. Glia 2004, 46, 410–418. [Google Scholar]
- Krupnick, J.G.; Benovic, J.L. The role of receptor kinases and arrestins in G protein-coupled receptor regulation. Annu. Rev. Pharmacol. Toxicol 1998, 38, 289–319. [Google Scholar]
- Bunemann, M.; Hosey, M.M. G-protein coupled receptor kinases as modulators of G-protein signalling. J. Physiol 1999, 517, 5–23. [Google Scholar]
- Goodman, O.B., Jr.; Krupnick, J.G.; Santini, F.; Gurevich, V.V.; Penn, R.B.; Gagnon, A.W.; Keen, J.H.; Benovic, J.L. Beta-arrestin acts as a clathrin adaptor in endocytosis of the beta2-adrenergic receptor. Nature 1996, 383, 447–450. [Google Scholar]
- Tsao, P.; von Zastrow, M. Downregulation of G protein-coupled receptors. Curr. Opin. Neurobiol 2000, 10, 365–369. [Google Scholar]
- Klaasse, E.C.; Ijzerman, A.P.; de Grip, W.J.; Beukers, M.W. Internalization and desensitization of adenosine receptors. Purinergic Signal 2008, 4, 21–37. [Google Scholar]
- Parsons, W.J.; Stiles, G.L. Heterologous desensitization of the inhibitory A1 adenosine receptor-adenylate cyclase system in rat adipocytes. Regulation of both Ns and Ni. J. Biol. Chem 1987, 262, 841–847. [Google Scholar]
- Green, A. Adenosine receptor down-regulation and insulin resistance following prolonged incubation of adipocytes with an A1 adenosine receptor agonist. J. Biol. Chem 1987, 262, 15702–15707. [Google Scholar]
- Longabaugh, J.P.; Didsbury, J.; Spiegel, A.; Stiles, G.L. Modification of the rat adipocyte A1 adenosine receptor-adenylate cyclase system during chronic exposure to an A1 adenosine receptor agonist: Alterations in the quantity of GS alpha and Gi alpha are not associated with changes in their mRNAs. Mol. Pharmacol 1989, 36, 681–688. [Google Scholar]
- Ramkumar, V.; Olah, M.E.; Jacobson, K.A.; Stiles, G.L. Distinct pathways of desensitization of A1- and A2-adenosine receptors in DDT1 MF-2 cells. Mol. Pharmacol 1991, 40, 639–647. [Google Scholar]
- Nie, Z.; Mei, Y.; Ramkumar, V. Short term desensitization of the A1 adenosine receptors in DDT1MF-2 cells. Mol. Pharmacol 1997, 52, 456–464. [Google Scholar]
- Palmer, T.M.; Benovic, J.L.; Stiles, G.L. Molecular basis for subtype-specific desensitization of inhibitory adenosine receptors. Analysis of a chimeric A1–A3 adenosine receptor. J. Biol. Chem 1996, 271, 15272–15278. [Google Scholar]
- Saura, C.A.; Mallol, J.; Canela, E.I.; Lluis, C.; Franco, R. Adenosine deaminase and A1 adenosine receptors internalize together following agonist-induced receptor desensitization. J. Biol. Chem 1998, 273, 17610–17617. [Google Scholar]
- Hettinger-Smith, B.D.; Leid, M.; Murray, T.F. Chronic exposure to adenosine receptor agonists and antagonists reciprocally regulates the A1 adenosine receptor-adenylyl cyclase system in cerebellar granule cells. J. Neurochem 1996, 67, 1921–1930. [Google Scholar]
- Coelho, J.E.; Rebola, N.; Fragata, I.; Ribeiro, J.A.; de Mendonca, A.; Cunha, R.A. Hypoxia-induced desensitization and internalization of adenosine A1 receptors in the rat hippocampus. Neuroscience 2006, 138, 1195–1203. [Google Scholar]
- Jajoo, S.; Mukherjea, D.; Kumar, S.; Sheth, S.; Kaur, T.; Rybak, L.P.; Ramkumar, V. Role of beta-arrestin1/ERK MAP kinase pathway in regulating adenosine A1 receptor desensitization and recovery. Am. J. Physiol. Cell Physiol 2010, 298, C56–C65. [Google Scholar]
- Pereira, M.R.; Hang, V.R.; Vardiero, E.; de Mello, F.G.; Paes-de-Carvalho, R. Modulation of A1 adenosine receptor expression by cell aggregation and long-term activation of A2a receptors in cultures of avian retinal cells: Involvement of the cyclic AMP/PKA pathway. J. Neurochem 2010, 113, 661–673. [Google Scholar]
- Brito, R.; Pereira, M.R.; Paes-de-Carvalho, R.; da Calaza, K.C. Expression of A1 adenosine receptors in the developing avian retina: In vivo modulation by A(2A) receptors and endogenous adenosine. J. Neurochem 2012, 123, 239–249. [Google Scholar]
- Castillo, C.A.; Leon, D.; Ruiz, M.A.; Albasanz, J.L.; Martin, M. Modulation of adenosine A1 and A2A receptors in C6 glioma cells during hypoxia: Involvement of endogenous adenosine. J. Neurochem 2008, 105, 2315–2329. [Google Scholar]
- Palmer, T.M.; Gettys, T.W.; Jacobson, K.A.; Stiles, G.L. Desensitization of the canine A2a adenosine receptor: delineation of multiple processes. Mol. Pharmacol 1994, 45, 1082–1094. [Google Scholar]
- Palmer, T.M.; Stiles, G.L. Identification of an A2a adenosine receptor domain specifically responsible for mediating short-term desensitization. Biochemistry 1997, 36, 832–838. [Google Scholar]
- Mundell, S.J.; Luty, J.S.; Willets, J.; Benovic, J.L.; Kelly, E. Enhanced expression of G protein-coupled receptor kinase 2 selectively increases the sensitivity of A2A adenosine receptors to agonist-induced desensitization. Br. J. Pharmacol 1998, 125, 347–356. [Google Scholar]
- Khoa, N.D.; Postow, M.; Danielsson, J.; Cronstein, B.N. Tumor necrosis factor-alpha prevents desensitization of Galphas-coupled receptors by regulating GRK2 association with the plasma membrane. Mol. Pharmacol 2006, 69, 1311–1319. [Google Scholar]
- Mundell, S.J.; Kelly, E. The effect of inhibitors of receptor internalization on the desensitization and resensitization of three Gs-coupled receptor responses. Br. J. Pharmacol 1998, 125, 1594–1600. [Google Scholar]
- Matharu, A.L.; Mundell, S.J.; Benovic, J.L.; Kelly, E. Rapid agonist-induced desensitization and internalization of the A(2B) adenosine receptor is mediated by a serine residue close to the COOH terminus. J. Biol. Chem 2001, 276, 30199–30207. [Google Scholar]
- Mundell, S.J.; Benovic, J.L.; Kelly, E. A dominant negative mutant of the G protein-coupled receptor kinase 2 selectively attenuates adenosine A2 receptor desensitization. Mol. Pharmacol 1997, 51, 991–998. [Google Scholar]
- Mundell, S.J.; Loudon, R.P.; Benovic, J.L. Characterization of G protein-coupled receptor regulation in antisense mRNA-expressing cells with reduced arrestin levels. Biochemistry 1999, 38, 8723–8732. [Google Scholar]
- Mundell, S.J.; Matharu, A.L.; Kelly, E.; Benovic, J.L. Arrestin isoforms dictate differential kinetics of A2B adenosine receptor trafficking. Biochemistry 2000, 39, 12828–12836. [Google Scholar]
- Claing, A.; Perry, S.J.; Achiriloaie, M.; Walker, J.K.; Albanesi, J.P.; Lefkowitz, R.J.; Premont, R.T. Multiple endocytic pathways of G protein-coupled receptors delineated by GIT1 sensitivity. Proc. Natl. Acad. Sci. USA 2000, 97, 1119–1124. [Google Scholar]
- Peters, D.M.; Gies, E.K.; Gelb, C.R.; Peterfreund, R.A. Agonist-induced desensitization of A2B adenosine receptors. Biochem. Pharmacol 1998, 55, 873–882. [Google Scholar]
- Trincavelli, M.L.; Marroni, M.; Tuscano, D.; Ceruti, S.; Mazzola, A.; Mitro, N.; Abbracchio, M.P.; Martini, C. Regulation of A2B adenosine receptor functioning by tumour necrosis factor a in human astroglial cells. J. Neurochem 2004, 91, 1180–1190. [Google Scholar]
- Moriyama, K.; Sitkovsky, M.V. Adenosine A2A receptor is involved in cell surface expression of A2B receptor. J. Biol. Chem 2010, 285, 39271–39288. [Google Scholar]
- Ferguson, G.; Watterson, K.R.; Palmer, T.M. Subtype-specific kinetics of inhibitory adenosine receptor internalization are determined by sensitivity to phosphorylation by G protein-coupled receptor kinases. Mol. Pharmacol 2000, 57, 546–552. [Google Scholar]
- Palmer, T.M.; Benovic, J.L.; Stiles, G.L. Agonist-dependent phosphorylation and desensitization of the rat A3 adenosine receptor. Evidence for a G-protein-coupled receptor kinase-mediated mechanism. J. Biol. Chem 1995, 270, 29607–29613. [Google Scholar]
- Palmer, T.M.; Stiles, G.L. Identification of threonine residues controlling the agonist-dependent phosphorylation and desensitization of the rat A(3) adenosine receptor. Mol. Pharmacol 2000, 57, 539–545. [Google Scholar]
- Trincavelli, M.L.; Tuscano, D.; Marroni, M.; Falleni, A.; Gremigni, V.; Ceruti, S.; Abbracchio, M.P.; Jacobson, K.A.; Cattabeni, F.; Martini, C. A3 adenosine receptors in human astrocytoma cells: Agonist-mediated desensitization, internalization, and down-regulation. Mol. Pharmacol 2002, 62, 1373–1384. [Google Scholar]
- Trincavelli, M.L.; Tuscano, D.; Marroni, M.; Klotz, K.N.; Lucacchini, A.; Martini, C. Involvement of mitogen protein kinase cascade in agonist-mediated human A(3) adenosine receptor regulation. Biochim. Biophys. Acta 2002, 1591, 55–62. [Google Scholar]
- Ford, M.S.; Maggirwar, S.B.; Rybak, L.P.; Whitworth, C.; Ramkumar, V. Expression and function of adenosine receptors in the chinchilla cochlea. Hear. Res 1997, 105, 130–140. [Google Scholar]
- Nie, Z.; Mei, Y.; Ford, M.; Rybak, L.; Marcuzzi, A.; Ren, H.; Stiles, G.L.; Ramkumar, V. Oxidative stress increases A1 adenosine receptor expression by activating nuclear factor kappa B. Mol. Pharmacol 1998, 53, 663–669. [Google Scholar]
- Ramkumar, V.; Whitworth, C.A.; Pingle, S.C.; Hughes, L.F.; Rybak, L.P. Noise induces A1 adenosine receptor expression in the chinchilla cochlea. Hear. Res 2004, 188, 47–56. [Google Scholar]
- Pingle, S.C.; Mishra, S.; Marcuzzi, A.; Bhat, S.G.; Sekino, Y.; Rybak, L.P.; Ramkumar, V. Osmotic diuretics induce adenosine A1 receptor expression and protect renal proximal tubular epithelial cells against cisplatin-mediated apoptosis. J. Biol. Chem 2004, 279, 43157–43167. [Google Scholar]
- Sundaram, U.; Hassanain, H.; Suntres, Z.; Yu, J.G.; Cooke, H.J.; Guzman, J.; Christofi, F.L. Rabbit chronic ileitis leads to up-regulation of adenosine A1/A3 gene products, oxidative stress, and immune modulation. Biochem. Pharmacol 2003, 65, 1529–1538. [Google Scholar]
- Lai, D.M.; Tu, Y.K.; Liu, I.M.; Cheng, J.T. Increase of adenosine A1 receptor gene expression in cerebral ischemia of Wistar rats. Neurosci. Lett 2005, 387, 59–61. [Google Scholar]
- Lee, N.H.; Weinstock, K.G.; Kirkness, E.F.; Earle-Hughes, J.A.; Fuldner, R.A.; Marmaros, S.; Glodek, A.; Gocayne, J.D.; Adams, M.D.; Kerlavage, A.R.; et al. Comparative expressed-sequence-tag analysis of differential gene expression profiles in PC-12 cells before and after nerve growth factor treatment. Proc. Natl. Acad. Sci. USA 1995, 92, 8303–8307. [Google Scholar]
- Nie, Z.; Mei, Y.; Malek, R.L.; Marcuzzi, A.; Lee, N.H.; Ramkumar, V. A role of p75 in NGF-mediated down-regulation of the A(2A) adenosine receptors in PC12 cells. Mol. Pharmacol 1999, 56, 947–954. [Google Scholar]
- Chu, Y.Y.; Tu, K.H.; Lee, Y.C.; Kuo, Z.J.; Lai, H.L.; Chern, Y. Characterization of the rat A2a adenosine receptor gene. DNA Cell Biol 1996, 15, 329–337. [Google Scholar]
- Malek, R.L.; Nie, Z.; Ramkumar, V.; Lee, N.H. Adenosine A(2A) receptor mRNA regulation by nerve growth factor is TrkA-, Src-, and Ras-dependent via extracellular regulated kinase and stress-activated protein kinase/c-Jun NH(2)-terminal kinase. J. Biol. Chem 1999, 274, 35499–35504. [Google Scholar]
- Sha, W.C.; Liou, H.C.; Tuomanen, E.I.; Baltimore, D. Targeted disruption of the p50 subunit of NF-kappa B leads to multifocal defects in immune responses. Cell 1995, 80, 321–330. [Google Scholar]
- Jhaveri, K.A.; Reichensperger, J.; Toth, L.A.; Sekino, Y.; Ramkumar, V. Reduced basal and lipopolysaccharide-stimulated adenosine A1 receptor expression in the brain of nuclear factor-kappaB p50−/− mice. Neuroscience 2007, 146, 415–426. [Google Scholar]
- Xie, X.; Jhaveri, K.A.; Ding, M.; Hughes, L.F.; Toth, L.A.; Ramkumar, V. Expression of striatal adenosine and dopamine receptors in mice deficient in the p50 subunit of NF-kappaB. Life Sci 2007, 81, 1031–1041. [Google Scholar]
- Xie, X.; Mhaskar, Y.; Arbogast, L.A.; Trammell, R.A.; Hughes, L.F.; Toth, L.A. Adenosine receptor antagonists and behavioral activation in NF-kappaB p50 subunit knockout mice. Life Sci 2009, 85, 226–234. [Google Scholar]
- Ferre, S.; Baler, R.; Bouvier, M.; Caron, M.G.; Devi, L.A.; Durroux, T.; Fuxe, K.; George, S.R.; Javitch, J.A.; Lohse, M.J.; et al. Building a new conceptual framework for receptor heteromers. Nat. Chem. Biol 2009, 5, 131–134. [Google Scholar]
- Ciruela, F.; Casado, V.; Mallol, J.; Canela, E.I.; Lluis, C.; Franco, R. Immunological identification of A1 adenosine receptors in brain cortex. J. Neurosci. Res 1995, 42, 818–828. [Google Scholar]
- Namba, K.; Suzuki, T.; Nakata, H. Immunogold electron microscopic evidence of in situ formation of homo- and heteromeric purinergic adenosine A1 and P2Y2 receptors in rat brain. BMC Res. Notes 2010, 3, 323. [Google Scholar]
- Gracia, E.; Moreno, E.; Cortes, A.; Lluis, C.; Mallol, J.; McCormick, P.J.; Canela, E.I.; Casado, V. Homodimerization of adenosine A(1) receptors in brain cortex explains the biphasic effects of caffeine. Neuropharmacology 2013, 71, 56–69. [Google Scholar]
- Canals, M.; Burgueno, J.; Marcellino, D.; Cabello, N.; Canela, E.I.; Mallol, J.; Agnati, L.; Ferre, S.; Bouvier, M.; Fuxe, K.; et al. Homodimerization of adenosine A2A receptors: Qualitative and quantitative assessment by fluorescence and bioluminescence energy transfer. J. Neurochem 2004, 88, 726–734. [Google Scholar]
- Ciruela, F.; Casado, V.; Rodrigues, R.J.; Lujan, R.; Burgueno, J.; Canals, M.; Borycz, J.; Rebola, N.; Goldberg, S.R.; Mallol, J.; et al. Presynaptic control of striatal glutamatergic neurotransmission by adenosine A1-A2A receptor heteromers. J. Neurosci 2006, 26, 2080–2087. [Google Scholar]
- Cristovao-Ferreira, S.; Navarro, G.; Brugarolas, M.; Perez-Capote, K.; Vaz, S.H.; Fattorini, G.; Conti, F.; Lluis, C.; Ribeiro, J.A.; McCormick, P.J.; et al. A1R-A2AR heteromers coupled to Gs and G i/0 proteins modulate GABA transport into astrocytes. Purinergic Signal 2013, 9, 433–449. [Google Scholar]
- Dobson, J.G., Jr. Mechanism of adenosine inhibition of catecholamine-induced responses in heart. Circ. Res 1983, 52, 151–160. [Google Scholar]
- Komatsu, S.; Dobson, J.G., Jr.; Ikebe, M.; Shea, L.G.; Fenton, R.A. Crosstalk between adenosine A1 and beta1-adrenergic receptors regulates translocation of PKCepsilon in isolated rat cardiomyocytes. J. Cell. Physiol 2012, 227, 3201–3207. [Google Scholar]
- Chandrasekera, P.C.; Wan, T.C.; Gizewski, E.T.; Auchampach, J.A.; Lasley, R.D. Adenosine A1 receptors heterodimerize with beta1- and beta2-adrenergic receptors creating novel receptor complexes with altered G protein coupling and signaling. Cell. Signal 2013, 25, 736–742. [Google Scholar]
- Yoshioka, K.; Saitoh, O.; Nakata, H. Heteromeric association creates a P2Y-like adenosine receptor. Proc. Natl. Acad. Sci. USA 2001, 98, 7617–7622. [Google Scholar]
- Yoshioka, K.; Saitoh, O.; Nakata, H. Agonist-promoted heteromeric oligomerization between adenosine A(1) and P2Y(1) receptors in living cells. FEBS Lett 2002, 523, 147–151. [Google Scholar]
- Yoshioka, K.; Hosoda, R.; Kuroda, Y.; Nakata, H. Hetero-oligomerization of adenosine A1 receptors with P2Y1 receptors in rat brains. FEBS Lett 2002, 531, 299–303. [Google Scholar]
- Fernandez-Ruiz, J. The endocannabinoid system as a target for the treatment of motor dysfunction. Br. J. Pharmacol 2009, 156, 1029–1040. [Google Scholar]
- Carriba, P.; Ortiz, O.; Patkar, K.; Justinova, Z.; Stroik, J.; Themann, A.; Muller, C.; Woods, A.S.; Hope, B.T.; Ciruela, F.; et al. Striatal adenosine A2A and cannabinoid CB1 receptors form functional heteromeric complexes that mediate the motor effects of cannabinoids. Neuropsychopharmacology 2007, 32, 2249–2259. [Google Scholar]
- Navarro, G.; Carriba, P.; Gandia, J.; Ciruela, F.; Casado, V.; Cortes, A.; Mallol, J.; Canela, E.I.; Lluis, C.; Franco, R. Detection of heteromers formed by cannabinoid CB1, dopamine D2, and adenosine A2A G-protein-coupled receptors by combining bimolecular fluorescence complementation and bioluminescence energy transfer. Sci. World J 2008, 8, 1088–1097. [Google Scholar]
- Martire, A.; Tebano, M.T.; Chiodi, V.; Ferreira, S.G.; Cunha, R.A.; Kofalvi, A.; Popoli, P. Pre-synaptic adenosine A2A receptors control cannabinoid CB1 receptor-mediated inhibition of striatal glutamatergic neurotransmission. J. Neurochem 2011, 116, 273–280. [Google Scholar]
- Tebano, M.T.; Martire, A.; Chiodi, V.; Pepponi, R.; Ferrante, A.; Domenici, M.R.; Frank, C.; Chen, J.F.; Ledent, C.; Popoli, P. Adenosine A2A receptors enable the synaptic effects of cannabinoid CB1 receptors in the rodent striatum. J. Neurochem 2009, 110, 1921–1930. [Google Scholar]
- Hillion, J.; Canals, M.; Torvinen, M.; Casado, V.; Scott, R.; Terasmaa, A.; Hansson, A.; Watson, S.; Olah, M.E.; Mallol, J.; et al. Coaggregation, cointernalization, and codesensitization of adenosine A2A receptors and dopamine D2 receptors. J. Biol. Chem 2002, 277, 18091–18097. [Google Scholar]
- Borroto-Escuela, D.O.; Romero-Fernandez, W.; Tarakanov, A.O.; Ciruela, F.; Agnati, L.F.; Fuxe, K. On the existence of a possible A2A-D2-beta-Arrestin2 complex: A2A agonist modulation of D2 agonist-induced beta-arrestin2 recruitment. J. Mol. Biol 2011, 406, 687–699. [Google Scholar]
- Canals, M.; Marcellino, D.; Fanelli, F.; Ciruela, F.; de Benedetti, P.; Goldberg, S.R.; Neve, K.; Fuxe, K.; Agnati, L.F.; Woods, A.S.; et al. Adenosine A2A-dopamine D2 receptor-receptor heteromerization: Qualitative and quantitative assessment by fluorescence and bioluminescence energy transfer. J. Biol. Chem 2003, 278, 46741–46749. [Google Scholar]
- Kamiya, T.; Saitoh, O.; Yoshioka, K.; Nakata, H. Oligomerization of adenosine A2A and dopamine D2 receptors in living cells. Biochem. Biophys. Res. Commun. 2003, 306, 544–549. [Google Scholar]
- Cabello, N.; Gandia, J.; Bertarelli, D.C.; Watanabe, M.; Lluis, C.; Franco, R.; Ferre, S.; Lujan, R.; Ciruela, F. Metabotropic glutamate type 5, dopamine D2 and adenosine A2a receptors form higher-order oligomers in living cells. J. Neurochem 2009, 109, 1497–1507. [Google Scholar]
- Gines, S.; Hillion, J.; Torvinen, M.; Le Crom, S.; Casado, V.; Canela, E.I.; Rondin, S.; Lew, J.Y.; Watson, S.; Zoli, M.; et al. Dopamine D1 and adenosine A1 receptors form functionally interacting heteromeric complexes. Proc. Natl. Acad. Sci. USA 2000, 97, 8606–8611. [Google Scholar]
- Toda, S.; Alguacil, L.F.; Kalivas, P.W. Repeated cocaine administration changes the function and subcellular distribution of adenosine A1 receptor in the rat nucleus accumbens. J. Neurochem 2003, 87, 1478–84. [Google Scholar]
- Ciruela, F.; Ferre, S.; Casado, V.; Cortes, A.; Cunha, R.A.; Lluis, C.; Franco, R. Heterodimeric adenosine receptors: A device to regulate neurotransmitter release. Cell. Mol. Life Sci 2006, 63, 2427–2431. [Google Scholar]
- Ferre, S.; Quiroz, C.; Orru, M.; Guitart, X.; Navarro, G.; Cortes, A.; Casado, V.; Canela, E.I.; Lluis, C.; Franco, R. Adenosine A(2A) Receptors and A(2A) Receptor Heteromers as Key Players in Striatal Function. Front Neuroanat 2011, 5, 36. [Google Scholar]
- Franco, R.; Lluis, C.; Canela, E.I.; Mallol, J.; Agnati, L.; Casado, V.; Ciruela, F.; Ferre, S.; Fuxe, K. Receptor-receptor interactions involving adenosine A1 or dopamine D1 receptors and accessory proteins. J. Neural. Transm 2007, 114, 93–104. [Google Scholar]
- Porkka-Heiskanen, T.; Strecker, R.E.; Thakkar, M.; Bjorkum, A.A.; Greene, R.W.; McCarley, R.W. Adenosine: A mediator of the sleep-inducing effects of prolonged wakefulness. Science 1997, 276, 1265–1268. [Google Scholar]
- Retey, J.V.; Adam, M.; Honegger, E.; Khatami, R.; Luhmann, U.F.; Jung, H.H.; Berger, W.; Landolt, H.P. A functional genetic variation of adenosine deaminase affects the duration and intensity of deep sleep in humans. Proc. Natl. Acad. Sci. USA 2005, 102, 15676–15681. [Google Scholar]
- Basheer, R.; Porkka-Heiskanen, T.; Stenberg, D.; McCarley, R.W. Adenosine and behavioral state control: Adenosine increases c-Fos protein and AP1 binding in basal forebrain of rats. Brain Res. Mol. Brain Res 1999, 73, 1–10. [Google Scholar]
- Portas, C.M.; Thakkar, M.; Rainnie, D.G.; Greene, R.W.; McCarley, R.W. Role of adenosine in behavioral state modulation: A microdialysis study in the freely moving cat. Neuroscience 1997, 79, 225–235. [Google Scholar]
- Porkka-Heiskanen, T.; Strecker, R.E.; McCarley, R.W. Brain site-specificity of extracellular adenosine concentration changes during sleep deprivation and spontaneous sleep: An in vivo microdialysis study. Neuroscience 2000, 99, 507–517. [Google Scholar]
- Benington, J.H.; Kodali, S.K.; Heller, H.C. Stimulation of A1 adenosine receptors mimics the electroencephalographic effects of sleep deprivation. Brain Res 1995, 692, 79–85. [Google Scholar]
- Schwierin, B.; Borbely, A.A.; Tobler, I. Effects of N6-cyclopentyladenosine and caffeine on sleep regulation in the rat. Eur. J. Pharmacol 1996, 300, 163–171. [Google Scholar]
- Halassa, M.M.; Florian, C.; Fellin, T.; Munoz, J.R.; Lee, S.Y.; Abel, T.; Haydon, P.G.; Frank, M.G. Astrocytic modulation of sleep homeostasis and cognitive consequences of sleep loss. Neuron 2009, 61, 213–219. [Google Scholar]
- Huang, Z.L.; Qu, W.M.; Eguchi, N.; Chen, J.F.; Schwarzschild, M.A.; Fredholm, B.B.; Urade, Y.; Hayaishi, O. Adenosine A2A, but not A1, receptors mediate the arousal effect of caffeine. Nat. Neurosci 2005, 8, 858–859. [Google Scholar]
- Oishi, Y.; Huang, Z.L.; Fredholm, B.B.; Urade, Y.; Hayaishi, O. Adenosine in the tuberomammillary nucleus inhibits the histaminergic system via A1 receptors and promotes non-rapid eye movement sleep. Proc. Natl. Acad. Sci. USA 2008, 105, 19992–19997. [Google Scholar]
- Jhaveri, K.A.; Ramkumar, V.; Trammell, R.A.; Toth, L.A. Spontaneous, homeostatic, and inflammation-induced sleep in NF-kappaB p50 knockout mice. Am. J. Physiol. Regul. Integr. Comp. Physiol 2006, 291, R1516–R1526. [Google Scholar]
- Bryant, G.M.; Barron, S.E.; Norris, C.H.; Guth, P.S. Adenosine is a modulator of hair cell-afferent neurotransmission. Hear. Res 1987, 30, 231–237. [Google Scholar]
- Dulon, D.; Mollard, P.; Aran, J.M. Extracellular ATP elevates cytosolic Ca2+ in cochlear inner hair cells. Neuroreport 1991, 2, 69–72. [Google Scholar]
- Ramkumar, V.; Ravi, R.; Wilson, M.C.; Gettys, T.W.; Whitworth, C.; Rybak, L.P. Identification of A1 adenosine receptors in rat cochlea coupled to inhibition of adenylyl cyclase. Am. J. Physiol 1994, 267, C731–C737. [Google Scholar]
- Ford, M.S.; Nie, Z.; Whitworth, C.; Rybak, L.P.; Ramkumar, V. Up-regulation of adenosine receptors in the cochlea by cisplatin. Hear. Res 1997, 111, 143–152. [Google Scholar]
- Whitworth, C.A.; Ramkumar, V.; Jones, B.; Tsukasaki, N.; Rybak, L.P. Protection against cisplatin ototoxicity by adenosine agonists. Biochem. Pharmacol 2004, 67, 1801–1807. [Google Scholar]
- Wong, A.C.; Guo, C.X.; Gupta, R.; Housley, G.D.; Thorne, P.R.; Vlajkovic, S.M. Post exposure administration of A(1) adenosine receptor agonists attenuates noise-induced hearing loss. Hear. Res 2010, 260, 81–88. [Google Scholar]
- Mukherjea, D.; Whitworth, C.A.; Nandish, S.; Dunaway, G.A.; Rybak, L.P.; Ramkumar, V. Expression of the kidney injury molecule 1 in the rat cochlea and induction by cisplatin. Neuroscience 2006, 139, 733–740. [Google Scholar]
- Mukherjea, D.; Jajoo, S.; Kaur, T.; Sheehan, K.E.; Ramkumar, V.; Rybak, L.P. Transtympanic administration of short interfering (si)RNA for the NOX3 isoform of NADPH oxidase protects against cisplatin-induced hearing loss in the rat. Antioxid. Redox Signal 2010, 13, 589–598. [Google Scholar]
- Vlajkovic, S.M.; Housley, G.D.; Thorne, P.R. Adenosine and the auditory system. Curr. Neuropharmacol 2009, 7, 246–256. [Google Scholar]
- Ohinata, Y.; Miller, J.M.; Schacht, J. Protection from noise-induced lipid peroxidation and hair cell loss in the cochlea. Brain Res 2003, 966, 265–273. [Google Scholar]
- Rybak, L.P.; Whitworth, C.A.; Mukherjea, D.; Ramkumar, V. Mechanisms of cisplatin-induced ototoxicity and prevention. Hear. Res 2007, 226, 157–167. [Google Scholar]
- Barnes, P.J.; Karin, M. Nuclear factor-kappaB: A pivotal transcription factor in chronic inflammatory diseases. N. Engl. J. Med 1997, 336, 1066–1071. [Google Scholar]
- Kaur, T.; Mukherjea, D.; Sheehan, K.; Jajoo, S.; Rybak, L.P.; Ramkumar, V. Short interfering RNA against STAT1 attenuates cisplatin-induced ototoxicity in the rat by suppressing inflammation. Cell Death Dis 2011, 2, e180. [Google Scholar]
- Mukherjea, D.; Jajoo, S.; Sheehan, K.; Kaur, T.; Sheth, S.; Bunch, J.; Perro, C.; Rybak, L.P.; Ramkumar, V. NOX3 NADPH oxidase couples transient receptor potential vanilloid 1 to signal transducer and activator of transcription 1-mediated inflammation and hearing loss. Antioxid. Redox Signal 2011, 14, 999–1010. [Google Scholar]
- Vlajkovic, S.M.; Abi, S.; Wang, C.J.; Housley, G.D.; Thorne, P.R. Differential distribution of adenosine receptors in rat cochlea. Cell Tissue Res 2007, 328, 461–471. [Google Scholar]
- Panjehpour, M.; Hemati, S.; Forghani, M.A. Expression of A1 and A3 adenosine receptors in human breast tumors. Tumori 2012, 98, 137–141. [Google Scholar]
- Mirza, A.; Basso, A.; Black, S.; Malkowski, M.; Kwee, L.; Pachter, J.A.; Lachowicz, J.E.; Wang, Y.; Liu, S. RNA interference targeting of A1 receptor-overexpressing breast carcinoma cells leads to diminished rates of cell proliferation and induction of apoptosis. Cancer Biol. Ther 2005, 4, 1355–1360. [Google Scholar]
- Khoo, H.E.; Ho, C.L.; Chhatwal, V.J.; Chan, S.T.; Ngoi, S.S.; Moochhala, S.M. Differential expression of adenosine A1 receptors in colorectal cancer and related mucosa. Cancer Lett 1996, 106, 17–21. [Google Scholar]
- Gessi, S.; Varani, K.; Merighi, S.; Morelli, A.; Ferrari, D.; Leung, E.; Baraldi, P.G.; Spalluto, G.; Borea, P.A. Pharmacological and biochemical characterization of A3 adenosine receptors in Jurkat T cells. Br. J. Pharmacol 2001, 134, 116–126. [Google Scholar]
- Merighi, S.; Varani, K.; Gessi, S.; Cattabriga, E.; Iannotta, V.; Ulouglu, C.; Leung, E.; Borea, P.A. Pharmacological and biochemical characterization of adenosine receptors in the human malignant melanoma A375 cell line. Br. J. Pharmacol 2001, 134, 1215–1226. [Google Scholar]
- Synowitz, M.; Glass, R.; Farber, K.; Markovic, D.; Kronenberg, G.; Herrmann, K.; Schnermann, J.; Nolte, C.; van Rooijen, N.; Kiwit, J.; et al. A1 adenosine receptors in microglia control glioblastoma-host interaction. Cancer Res 2006, 66, 8550–8557. [Google Scholar]
- Saito, M.; Yaguchi, T.; Yasuda, Y.; Nakano, T.; Nishizaki, T. Adenosine suppresses CW2 human colonic cancer growth by inducing apoptosis via A(1) adenosine receptors. Cancer Lett 2010, 290, 211–215. [Google Scholar]
- D’Ancona, S.; Ragazzi, E.; Fassina, G.; Mazzo, M.; Gusella, M.; Berti, T. Effect of dipyridamole, 5′-(N-ethyl)-carboxamidoadenosine and 1,3-dipropyl-8-(2-amino-4-chlorophenyl)- xanthine on LOVO cell growth and morphology. Anticancer Res 1994, 14, 93–97. [Google Scholar]
- Shaban, M.; Smith, R.A.; Stone, T.W. Purine suppression of proliferation of Sertoli-like TM4 cells in culture. Cell Prolif 1995, 28, 673–682. [Google Scholar]
- Etique, N.; Grillier-Vuissoz, I.; Lecomte, J.; Flament, S. Crosstalk between adenosine receptor (A2A isoform) and ERalpha mediates ethanol action in MCF-7 breast cancer cells. Oncol. Rep 2009, 21, 977–981. [Google Scholar]
- Gessi, S.; Sacchetto, V.; Fogli, E.; Merighi, S.; Varani, K.; Baraldi, P.G.; Tabrizi, M.A.; Leung, E.; Maclennan, S.; Borea, P.A. Modulation of metalloproteinase-9 in U87MG glioblastoma cells by A3 adenosine receptors. Biochem. Pharmacol 2010, 79, 1483–1495. [Google Scholar]
- Gessi, S.; Merighi, S.; Varani, K.; Cattabriga, E.; Benini, A.; Mirandola, P.; Leung, E.; MacLennan, S.; Feo, C.; Baraldi, S.; et al. Adenosine receptors in colon carcinoma tissues and colon tumoral cell lines: Focus on the A(3) adenosine subtype. J. Cell Physiol 2007, 211, 826–836. [Google Scholar]
- Peterfreund, R.A.; Gies, E.K.; Fink, J.S. Protein kinase C regulates adenosine A2a receptor mRNA expression in SH-SY5Y cells. Eur. J. Pharmacol 1997, 336, 71–80. [Google Scholar]
- Mediavilla-Varela, M.; Luddy, K.; Noyes, D.; Khalil, F.K.; Neuger, A.M.; Soliman, H.; Antonia, S. Antagonism of adenosine A2A receptor expressed by lung adenocarcinoma tumor cells and cancer associated fibroblasts inhibits their growth. Cancer Biol. Ther 2013, 14, 860–868. [Google Scholar]
- Ahmad, A.; Ahmad, S.; Glover, L.; Miller, S.M.; Shannon, J.M.; Guo, X.; Franklin, W.A.; Bridges, J.P.; Schaack, J.B.; Colgan, S.P.; et al. Adenosine A2A receptor is a unique angiogenic target of HIF-2alpha in pulmonary endothelial cells. Proc. Natl. Acad. Sci. USA 2009, 106, 10684–10689. [Google Scholar]
- Montesinos, M.C.; Desai, A.; Chen, J.F.; Yee, H.; Schwarzschild, M.A.; Fink, J.S.; Cronstein, B.N. Adenosine promotes wound healing and mediates angiogenesis in response to tissue injury via occupancy of A(2A) receptors. Am. J. Pathol 2002, 160, 2009–2018. [Google Scholar]
- Ohta, A.; Gorelik, E.; Prasad, S.J.; Ronchese, F.; Lukashev, D.; Wong, M.K.; Huang, X.; Caldwell, S.; Liu, K.; Smith, P.; et al. A2A adenosine receptor protects tumors from antitumor T cells. Proc. Natl. Acad. Sci. USA 2006, 103, 13132–13137. [Google Scholar]
- Merighi, S.; Mirandola, P.; Milani, D.; Varani, K.; Gessi, S.; Klotz, K.N.; Leung, E.; Baraldi, P.G.; Borea, P.A. Adenosine receptors as mediators of both cell proliferation and cell death of cultured human melanoma cells. J. Invest. Dermatol 2002, 119, 923–933. [Google Scholar]
- Yasuda, Y.; Saito, M.; Yamamura, T.; Yaguchi, T.; Nishizaki, T. Extracellular adenosine induces apoptosis in Caco-2 human colonic cancer cells by activating caspase-9/-3 via A(2a) adenosine receptors. J. Gastroenterol 2009, 44, 56–65. [Google Scholar]
- Grant, M.B.; Tarnuzzer, R.W.; Caballero, S.; Ozeck, M.J.; Davis, M.I.; Spoerri, P.E.; Feoktistov, I.; Biaggioni, I.; Shryock, J.C.; Belardinelli, L. Adenosine receptor activation induces vascular endothelial growth factor in human retinal endothelial cells. Circ. Res 1999, 85, 699–706. [Google Scholar]
- Ryzhov, S.; Novitskiy, S.V.; Zaynagetdinov, R.; Goldstein, A.E.; Carbone, D.P.; Biaggioni, I.; Dikov, M.M.; Feoktistov, I. Host A(2B) adenosine receptors promote carcinoma growth. Neoplasia 2008, 10, 987–995. [Google Scholar]
- Merighi, S.; Simioni, C.; Gessi, S.; Varani, K.; Mirandola, P.; Tabrizi, M.A.; Baraldi, P.G.; Borea, P.A. A(2B) and A(3) adenosine receptors modulate vascular endothelial growth factor and interleukin-8 expression in human melanoma cells treated with etoposide and doxorubicin. Neoplasia 2009, 11, 1064–1073. [Google Scholar]
- Kong, T.; Westerman, K.A.; Faigle, M.; Eltzschig, H.K.; Colgan, S.P. HIF-dependent induction of adenosine A2B receptor in hypoxia. FASEB J 2006, 20, 2242–2250. [Google Scholar]
- Ma, D.F.; Kondo, T.; Nakazawa, T.; Niu, D.F.; Mochizuki, K.; Kawasaki, T.; Yamane, T.; Katoh, R. Hypoxia-inducible adenosine A2B receptor modulates proliferation of colon carcinoma cells. Hum. Pathol 2010, 41, 1550–1557. [Google Scholar]
- Jajoo, S.; Mukherjea, D.; Watabe, K.; Ramkumar, V. Adenosine A(3) receptor suppresses prostate cancer metastasis by inhibiting NADPH oxidase activity. Neoplasia 2009, 11, 1132–1145. [Google Scholar]
- Madi, L.; Ochaion, A.; Rath-Wolfson, L.; Bar-Yehuda, S.; Erlanger, A.; Ohana, G.; Harish, A.; Merimski, O.; Barer, F.; Fishman, P. The A3 adenosine receptor is highly expressed in tumor versus normal cells: Potential target for tumor growth inhibition. Clin. Cancer Res 2004, 10, 4472–4479. [Google Scholar]
- Merighi, S.; Benini, A.; Mirandola, P.; Gessi, S.; Varani, K.; Leung, E.; Maclennan, S.; Borea, P.A. Adenosine modulates vascular endothelial growth factor expression via hypoxia-inducible factor-1 in human glioblastoma cells. Biochem. Pharmacol 2006, 72, 19–31. [Google Scholar]
- Kohno, Y.; Sei, Y.; Koshiba, M.; Kim, H.O.; Jacobson, K.A. Induction of apoptosis in HL-60 human promyelocytic leukemia cells by adenosine A(3) receptor agonists. Biochem. Biophys. Res. Commun 1996, 219, 904–910. [Google Scholar]
- Bar-Yehuda, S.; Stemmer, S.M.; Madi, L.; Castel, D.; Ochaion, A.; Cohen, S.; Barer, F.; Zabutti, A.; Perez-Liz, G.; Del Valle, L.; et al. The A3 adenosine receptor agonist CF102 induces apoptosis of hepatocellular carcinoma via de-regulation of the Wnt and NF-kappaB signal transduction pathways. Int. J. Oncol 2008, 33, 287–295. [Google Scholar]
- Madi, L.; Bar-Yehuda, S.; Barer, F.; Ardon, E.; Ochaion, A.; Fishman, P. A3 adenosine receptor activation in melanoma cells: Association between receptor fate and tumor growth inhibition. J. Biol. Chem 2003, 278, 42121–42130. [Google Scholar]
- Ohana, G.; Bar-Yehuda, S.; Arich, A.; Madi, L.; Dreznick, Z.; Rath-Wolfson, L.; Silberman, D.; Slosman, G.; Fishman, P. Inhibition of primary colon carcinoma growth and liver metastasis by the A3 adenosine receptor agonist CF101. Br. J. Cancer 2003, 89, 1552–1558. [Google Scholar]
- Fishman, P.; Bar-Yehuda, S.; Ardon, E.; Rath-Wolfson, L.; Barrer, F.; Ochaion, A.; Madi, L. Targeting the A3 adenosine receptor for cancer therapy: Inhibition of prostate carcinoma cell growth by A3AR agonist. Anticancer Res 2003, 23, 2077–2083. [Google Scholar]
- Harish, A.; Hohana, G.; Fishman, P.; Arnon, O.; Bar-Yehuda, S. A3 adenosine receptor agonist potentiates natural killer cell activity. Int. J. Oncol 2003, 23, 1245–1249. [Google Scholar]


© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sheth, S.; Brito, R.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. Adenosine Receptors: Expression, Function and Regulation. Int. J. Mol. Sci. 2014, 15, 2024-2052. https://doi.org/10.3390/ijms15022024
Sheth S, Brito R, Mukherjea D, Rybak LP, Ramkumar V. Adenosine Receptors: Expression, Function and Regulation. International Journal of Molecular Sciences. 2014; 15(2):2024-2052. https://doi.org/10.3390/ijms15022024
Chicago/Turabian StyleSheth, Sandeep, Rafael Brito, Debashree Mukherjea, Leonard P. Rybak, and Vickram Ramkumar. 2014. "Adenosine Receptors: Expression, Function and Regulation" International Journal of Molecular Sciences 15, no. 2: 2024-2052. https://doi.org/10.3390/ijms15022024
APA StyleSheth, S., Brito, R., Mukherjea, D., Rybak, L. P., & Ramkumar, V. (2014). Adenosine Receptors: Expression, Function and Regulation. International Journal of Molecular Sciences, 15(2), 2024-2052. https://doi.org/10.3390/ijms15022024




