Identification of Amplified Fragment Length Polymorphism (AFLP) Markers Tightly Associated with Drought Stress Gene in Male Sterile and Fertile Salvia miltiorrhiza Bunge
Abstract
:1. Introduction
2. Results and Discussion
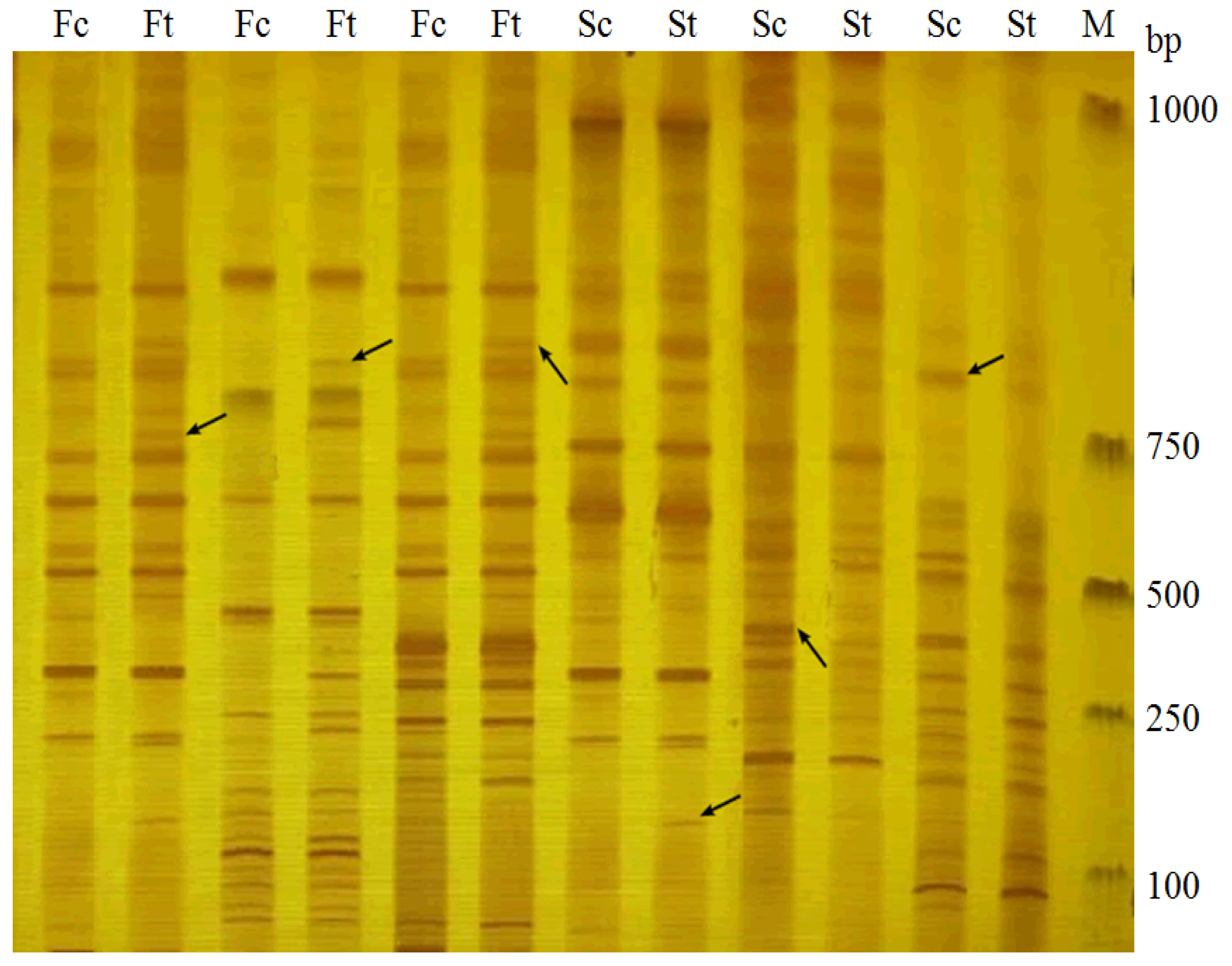
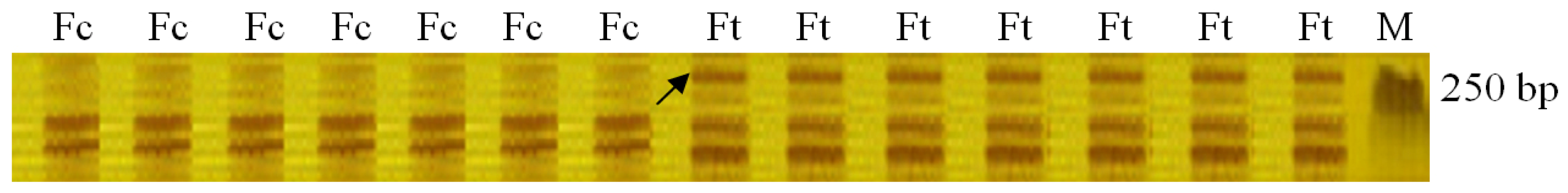
2.1. Identification of AFLP Marker Linked to Drought Stress Gene in Fertile S. miltiorrhiza
2.2. Identification of AFLP Marker Linked to Drought Stress Gene in Sterile S. miltiorrhiza
2.3. Sequence Features of Drought Stress Gene Amplified by E9/M3 and E2/M5 Primer Combinations
2.4. Discussion
3. Experimental Section
3.1. Plant Material
3.2. Drought stress Treatments
3.3. AFLP Fragment Sequencing and Linkage Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Chaves, M.M.; Flexas, J.; Pinheiro, C. Photosynthesis under drought and salt stress: Regulation mechanisms from whole plant to cell. Ann. Bot 2009, 103, 551–560. [Google Scholar]
- Manuel, J. Drought in the southeast: Lessens for water management. Environ. Health Persp 2008, 116, A168–A171. [Google Scholar]
- Cai, X.L.; Wu, S.L.; He, H.; Jiang, C.Y.; Sun, X.; Gao, H.Y.; Zheng, G.F. Change of extreme climate events in Shaanxi Province under global warming background. J. Desert Res 2012, 32, 1095–1101. [Google Scholar]
- National Committee of China Pharmacopeia, China Pharmacopeia; Chemical Industry Press: Beijing, China, 2010; p. 52.
- Liu, H.; Wang, X.; Wang, D.; Zou, Z.; Liang, Z. Effect of drought stress on growth and accumulation of active constituents in Salvia miltiorrhiza Bunge. Ind. Crops Prod 2011, 33, 84–88. [Google Scholar]
- Parish, R.W.; Phan, H.A.; Lacuone, S.; Li, S.F. Tapetal development and abiotic stress: A centre of vulnerability. Funct. Plant Biol 2012, 39, 553–559. [Google Scholar]
- Webster, H.; Keeble, G.; Dell, B.; Fosu-Nyarko, J.; Mukai, Y.; Moolhuijzen, P.; Bellgard, M.; Jia, J.; Kong, X.; Feuillet, C.; et al. Genome-level identification of cell wall invertase genes in wheat for the study of drought tolerance. Funct. Plant Biol 2012, 39, 569–579. [Google Scholar]
- Shu, Z.; Liang, Z.; Sun, Q.; Zhang, X.; Fu, L. Identification and pollen development anatomy of the male sterility line Sh-B in Salvia miltiorrhiza. Acta Bot. Boreal Occident Sin 2006, 26, 2202–2207. [Google Scholar]
- Shu, Z.; Liang, Z.; Sun, Q.; Fu, L.; Zhang, X.; Ye, Z.; Wei, X. Biological characteristics of Salvia miltiorrhiza male sterile line Sh-B. J. Northwest A F Univ 2007, 35, 175–179. [Google Scholar]
- Shu, Z.; Wang, Z.; Mu, X.; Liang, Z.; Guo, H. A dominant gene for male sterility in Salvia miltiorrhiza Bunge. PLoS One 2012, 7, e50903. [Google Scholar]
- Liu, Z.W.; Fu, T.D.; Tu, J.X.; Chen, B.Y. Inheritance of seed colour and identification and AFLP markers linked to the seed in rapeseed (Brassica napus L.). Theor. Appl. Genet 2005, 110, 303–310. [Google Scholar]
- Siosemardeh, A.; Osmani, Z.; Bahramnejad, B.; Vahabi, K.; Roohi, E. Identification of AFLP markers associated with stress tolerance index in Sardari wheat ecotypes. J. Agric. Sci. Technol 2012, 14, 629–643. [Google Scholar]
- Michelmore, R.W.; Paran, I.; Lesseli, R.V. Identification of markers linked to disease-resistance genes by bulked segregant analysis: a rapid method to detect markers in specific genomic regions by using segregating populations. Proc. Natl. Acad. Sci. USA 1991, 88, 9828–9832. [Google Scholar]
- Negi, M.S.; Devic, M.; Delseny, M.; Lakshmikumaran, M. Identification of AFLP fragments linked to seed coat colour in Brassica junca and conversion to a SCAR marker for rapid selection. Theor. Appl. Genet 2009, 101, 146–152. [Google Scholar]
- Clément, C.; Burrus, M.; Audran, J.C. Floral organ growth and carbohydrate content during pollen development in Lilium. Am. J. Bot 1996, 83, 459–469. [Google Scholar]
- Nguyen, G.N.; Hailstones, D.L.; Wilkes, M.; Sutton, B.G. Role of carbohydrate metabolism in drought-induced male sterility in rice anthers. J. Agron. Crop Sci 2010, 196, 346–357. [Google Scholar]
- Hermo, L.; Pelletier, R.M.; Cyr, D.G.; Smith, C.E. Surfing the wave, cycle, life history, and genes/proteins expressed by testicular germ cells. Part 4: Intercellular bridges, mitochondria, nuclear envelope, apoptosis, ubiquitination, membrane/voltage-gated channels, methylation/acetylation, and transcription factors. Microsc. Res. Techniq 2010, 73, 364–408. [Google Scholar]
- Baroux, C.; Spillane, C.; Crossniklaus, U. Genomic imprinting during seed development. Adv. Genet 2002, 46, 165–214. [Google Scholar]
- Saze, H.; Scheidm, O.M.; Pazskowski, J. Maintenance of CpG methylation is essential for epigenetic inheritance during plant gametogenesis. Nat. Genet 2003, 34, 65–69. [Google Scholar]
- Huang, W.; Yu, M.; Jiao, Y.; Ma, J.; Ma, M.; Wang, Z.; Wu, H.; Tan, D. Mitochondrial transcription termination factor 2 binds to entire mitochondrial DNA and negatively regulates mitochondrial gene express. Acta Biochim. Biophys. Sin 2011, 43, 472–479. [Google Scholar]
- Roberti, M.; Polosa, P.L.; Bruni, F.; Manzari, C.; Deceglie, S.; Gadaleta, M.N.; Cantatore, P. The MTERF family proteins: mitochondrial transcription regulations and beyond. Biochim. Biophys. Acta 2009, 1787, 303–311. [Google Scholar]
- Warmke, H.E.; Lee, S.L.J. Pollen abortion in T cytoplasmic male-sterile corn (Zea mays): A suggested mechanism. Science 1978, 200, 561–563. [Google Scholar]
- Saini, H.S.; Sedgley, M.; Aspinall, D. Developmental anatomy in wheat of male sterility causes induced by heat stress, water deficit or abscisic acid. Aust. J. Plant Physiol 1984, 11, 243–253. [Google Scholar]
- Sheoran, I.S.; Saini, H.S. Drought-induced male sterile in rice: Changes in carbohydrate levels and enzyme activities associated with the inhibition of starch accumulation in pollen. Sex Plant Reprod 1996, 9, 161–169. [Google Scholar]
- Nguyen, G.N.; Hailstones, D.L.; Wilkes, M.; Sutton, B.G. Drought-induced oxidative conditions in rice anthers leading to a programmed cell death and pollen abortion. J. Agron. Crop Sci 2009, 195, 157–164. [Google Scholar]
- Ji, X.; Shiran, B.; Wan, J.; Lewis, D.C.; Jenkins, C.L.D.; Condon, A.G.; Richard, R.A.; Dolferus, R. Importance of pre-anthesis anther sink strength for maintenance of grain number during reproductive stage water stress in wheat. Plant Cell Environ 2010, 33, 926–942. [Google Scholar]
- Shinozaki, K.; Yamaguchi-Shinozaki, K.; Seki, M. Regulatory network of gene expression in the drought and cold stress responses. Curr. Opin. Plant Biol 2003, 6, 410–417. [Google Scholar]
- Arroyo, A.; Bossi, F.; Finkelstein, R.R.; León, P. Three genes that affect sugar sensing (Abscisic Acid insensitive 4, Abscisic Acid Insensitive 5, and Constitutive Triple Response 1) are differentially regulated by glucose in Arabidopsis. Plant Physiol 2003, 133, 231–242. [Google Scholar]
- Ji, X.; Dong, B.; Shiran, B.; Talbot, M.J.; Edlington, J.E.; Hughes, T.; White, R.G.; Gubler, F.; Dolferus, R. Control of ABA catabolism and ABA homestasis is important for reproductive stage stress tolerance in cereals. Plant Physiol 2011, 156, 647–662. [Google Scholar]
- Frey, A.; Audran, C.; Marin, E.; Sotta, B.; Marion-Poll, A. Engineering seed dormancy by the modification of zeaxanthin epoxidase gene expression. Plant Mol. Biol 1999, 39, 1267–1274. [Google Scholar]
- Verrier, P.J.; Bird, D.; Burla, B.; Dassa, E.; Forestier, C.; Geisler, M.; Klein, M.; Kolukisaoglu, U.; Lee, Y.; Martinoia, E.; et al. Plant ABC proteins—A unified nomenclature and updated inventory. Trends Plant Sci 2008, 13, 151–159. [Google Scholar]
- Yazaki, K. ABC transporters involved in the transport of plant secondary metabolites. FEBS Lett 2006, 580, 1183–1191. [Google Scholar]
- Cheng, Y.; Dai, X.; Zhao, Y. Auxin biosynthesis by the YUCCA flavin monooxygenase controls the formation of floral organs and vascular tissues in Arabidopsis. Genes Dev 2006, 20, 1790–1799. [Google Scholar]
- Klee, H.J.; Horsch, R.B.; Hinchee, M.A.; Hein, M.B.; Hoffmann, N.L. The effects of overproduction of two Agrobacterium tumefaciens T-DNA auxin biosynthetic gene products in transgenic petunia plants. Genes Dev 1987, 1, 86–96. [Google Scholar]
- Woo, Y.M.; Park, H.J.; Suudi, M.; Yang, J.I.; Park, J.J.; Back, K.; Park, Y.M.; An, G. Constitutively wilted I, a member of the rice YUCCA gene family, is required for maintaining water homestasis and an appropriate root to shoot ratio. Plant Mol. Biol 2007, 65, 125–136. [Google Scholar]
- Dolferus, R.; Ji, X.; Richards, R.A. Abiotic stress and control of grain number in cereals. Plant Sci 2011, 181, 331–341. [Google Scholar]
- NCBI (National Center for Biotechnology Information)\BLAST Homepage. Available online: http://ncbi.nlm.nih.gov/BLAST accessed on 27 February 2013.
- Lander, E.S.; Green, P.; Abrahamson, J.; Barlow, A.; Daly, M.J.; Lincoln, S.E.; Newberg, L.A.; Newburg, L. MAPMAKER: An interactive computer package for constructing primary genetic linkage maps of experimental and natural populations. Genomics 1987, 1, 174–181. [Google Scholar]
- Lincoln, S.; Daly, M.; Lander, E. Constructing Genetic Maps with MAPMAKER/EXP 3.0; Whitehead Technical Institute: Cambridge, MA, USA, 1992. [Google Scholar]
- Kosambi, D.D. The estimation of map distance from recombination values. Ann. Eugen 1944, 12, 172–175. [Google Scholar]

| GenBank Acc. No. | Blast hits | Organism | e-value | Query cover (%) | Identity (%) |
|---|---|---|---|---|---|
| E9/M3246 | |||||
| NC003071.7 | Hypothetical protein | Arabidopsis thaliana, chromosome 2 | 0.15 | 32 | 100 |
| NC003074.8 | Hypothetical protein | Arabidopsis thaliana, chromosome 3 | 0.52 | 23 | 100 |
| NC003070.9 | 1536 bp at 5′ side: hypothetical protein; 386 bp at 3′ side: mitochondrial transcription termination factor family protein | Arabidopsis thaliana, chromosome 1 | 0.52 | 39 | 100 |
| NC003076.8 | Putative leucine-rich repeat receptor-like protein kinase | Arabidopsis thaliana, chromosome 5 | 6.3 | 17 | 95 |
| NC008396.2 | Hypothetical protein | Oryza sativa Japonica Group DNA Chromosome 3 | 0.47 | 10 | 93 |
| NC008398.2 | 38938 bp at 5′ side: hypothetical protein; 11051 bp at 3′ side: hypothetical protein | Oryza sativa Japonica Group DNA Chromosome 5 | 1.7 | 19 | 92 |
| NC008399.2 | 23149 bp at 5′ side: hypothetical protein; 2957 bp at 3′ side: hypothetical protein | Oryza sativa Japonica Group DNA Chromosome 6 | 5.8 | 10 | 92 |
| E2/M5357 | |||||
| NC003074.8 | ABC transporter B family member 16 | Arabidopsis thaliana, chromosome 3 | 0.22 | 26 | 100 |
| NC003076.8 | DNA-3-methyladenine glycosylase I | Arabidopsis thaliana, chromosome 5 | 2.7 | 12 | 91 |
| NC003075.7 | Hypothetical protein | Arabidopsis thaliana, chromosome 4 | 2.7 | 12 | 100 |
| NC003071.7 | 2397 bp at 5′ side: hypothetical protein; 1525 bp at 3′ side: laccase-4 | Arabidopsis thaliana, chromosome 2 | 2.7 | 31 | 95 |
| NC003070.9 | 18 bp at 5′ side: flavin-binding monooxygenase family protein; 462 bp at 3′ side: putative F-box/kelch-repeat protein | Arabidopsis thaliana, chromosome 1 | 2.7 | 29 | 100 |
| NC008404.2 | 17867 bp at 5′ side: hypothetical protein; 46979 bp at 3′ side: hypothetical protein | Oryza sativa Japonica Group DNA Chromosome 11 | 2.5 | 14 | 92 |
| NC008397.2 | Hypothetical protein | Oryza sativa Japonica Group DNA Chromosome 4 | 2.5 | 5 | 100 |
| NC008395.2 | Hypothetical protein | Oryza sativa Japonica Group DNA Chromosome 2 | 2.5 | 18 | 95 |
| E2/M5357 | |||||
| NC008403.2 | Hypothetical protein | Oryza sativa Japonica Group DNA Chromosome 10 | 8.7 | 5 | 100 |
| NC008402.2 | 2747 bp at 5′ side: hypothetical protein; 1409 bp at 3′ side: hypothetical protein | Oryza sativa Japonica Group DNA Chromosome 9 | 8.7 | 5 | 100 |
| E9/M3246 and E11/M4208 | – | – | 4 × 10−93 | 84 | 95 |
| E2/M5357 and E11/M4208 | – | – | 0.03 | 3 | 100 |
| E9/M3246 and E2/M5357 | – | – | 0.036 | 4 | 100 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, Y.; Guo, L.; Shu, Z.; Sun, Y.; Chen, Y.; Liang, Z.; Guo, H. Identification of Amplified Fragment Length Polymorphism (AFLP) Markers Tightly Associated with Drought Stress Gene in Male Sterile and Fertile Salvia miltiorrhiza Bunge. Int. J. Mol. Sci. 2013, 14, 6518-6528. https://doi.org/10.3390/ijms14036518
Zhang Y, Guo L, Shu Z, Sun Y, Chen Y, Liang Z, Guo H. Identification of Amplified Fragment Length Polymorphism (AFLP) Markers Tightly Associated with Drought Stress Gene in Male Sterile and Fertile Salvia miltiorrhiza Bunge. International Journal of Molecular Sciences. 2013; 14(3):6518-6528. https://doi.org/10.3390/ijms14036518
Chicago/Turabian StyleZhang, Yuejin, Lijun Guo, Zhiming Shu, Yiyue Sun, Yuanyuan Chen, Zongsuo Liang, and Hongbo Guo. 2013. "Identification of Amplified Fragment Length Polymorphism (AFLP) Markers Tightly Associated with Drought Stress Gene in Male Sterile and Fertile Salvia miltiorrhiza Bunge" International Journal of Molecular Sciences 14, no. 3: 6518-6528. https://doi.org/10.3390/ijms14036518




