A Case of Stage III c Ovarian Clear Cell Carcinoma: The Role for Predictive Biomarkers and Targeted Therapies
Abstract
:1. Introduction
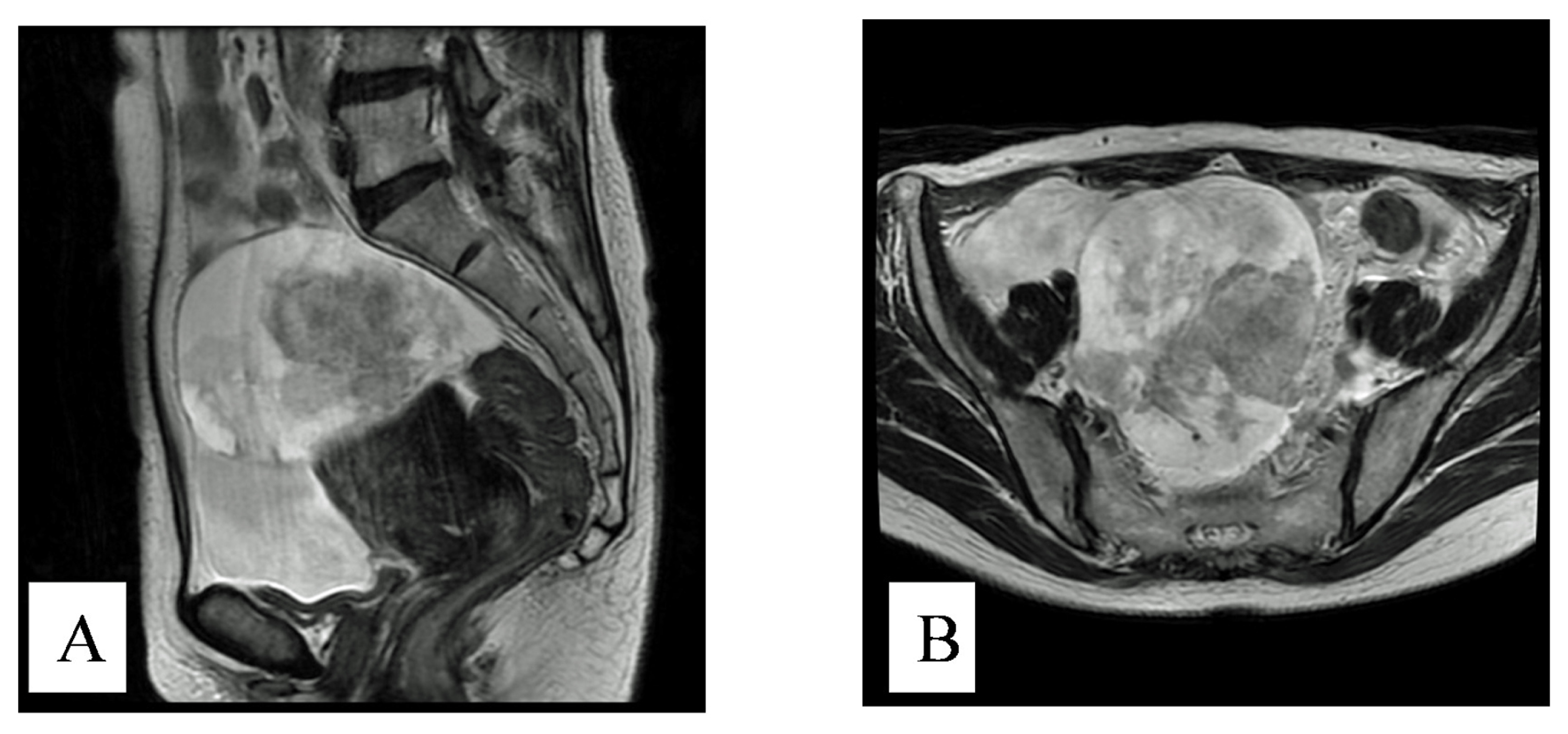
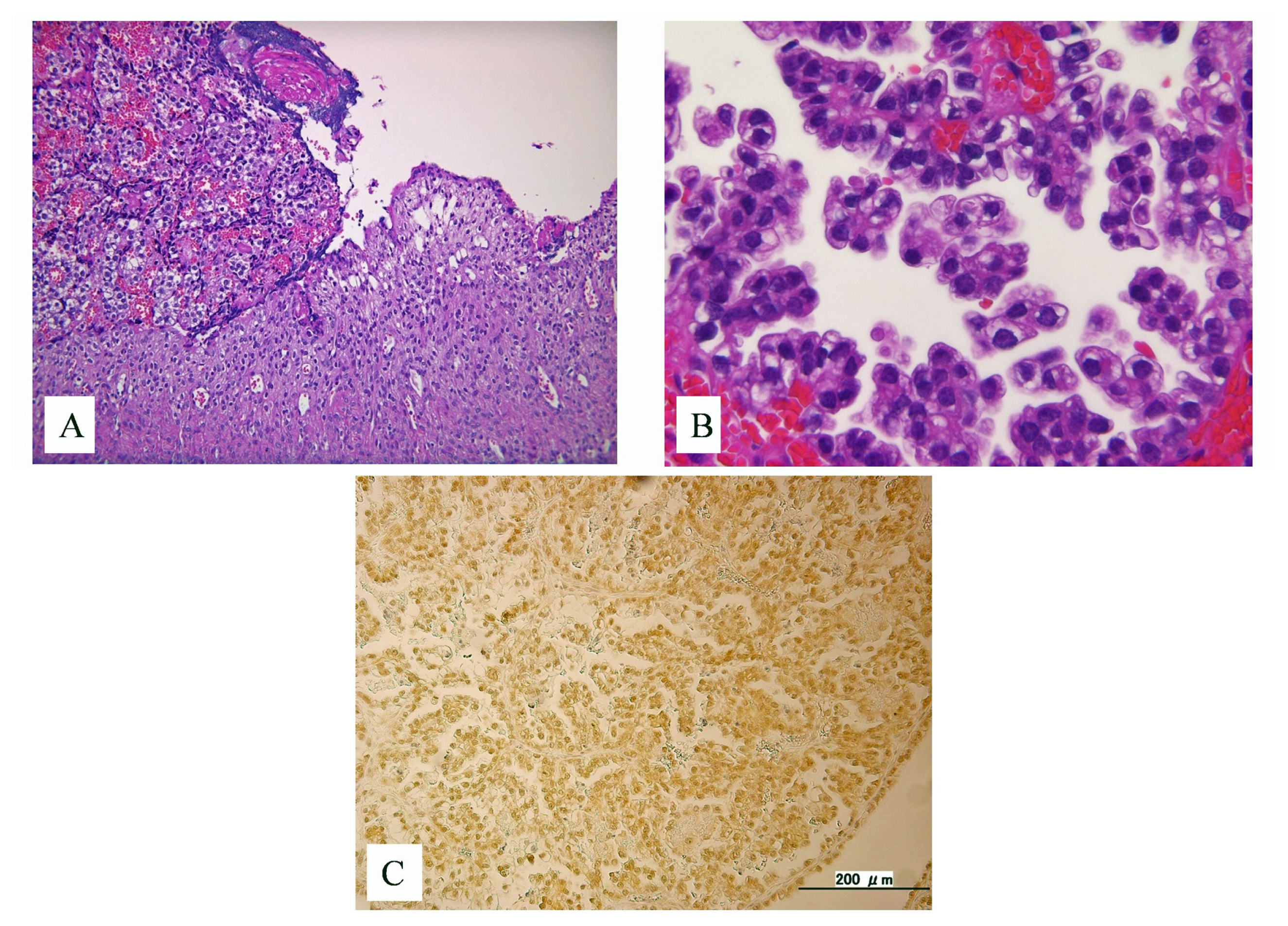
2. Case History
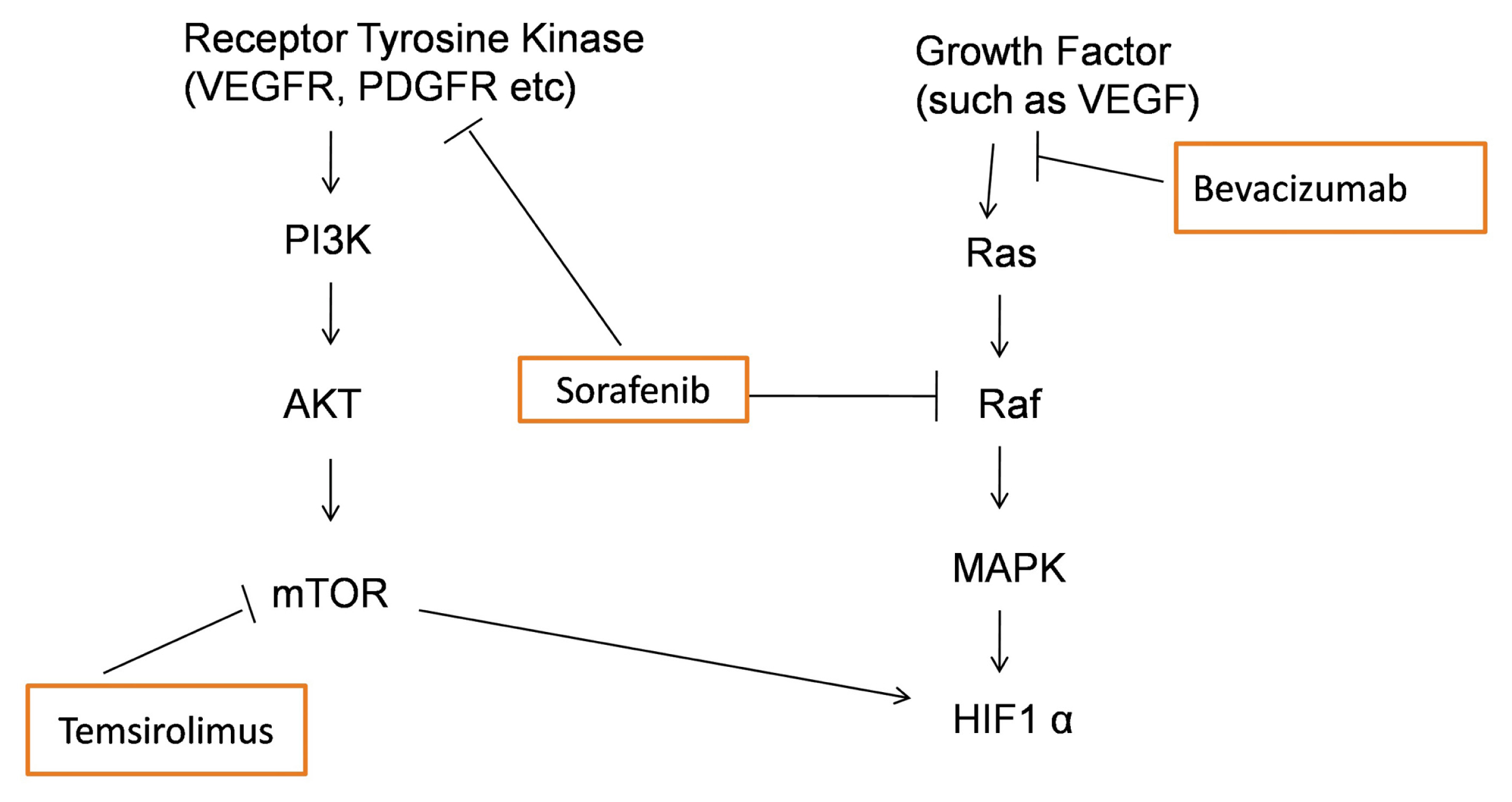
3. Discussion
4. Conclusions
Conflict of Interest
References
- Itamochi, H.; Kigawa, J.; Terakawa, N. Mechanisms of chemoresistance and poor prognosis in ovarian clear cell carcinoma. Cancer Sci 2008, 99, 653–658. [Google Scholar]
- Sugiyama, T.; Kamura, T.; Kigawa, J.; Terakawa, N.; Kikuchi, Y.; Kita, T.; Suzuki, M.; Sato, I.; Taguchi, K. Clinical characteristics of clear cell carcinoma of the ovary: A distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer 2000, 88, 2584–2589. [Google Scholar]
- Jones, S.; Wang, T.L.; Shih, I.-M.; Mao, T.L.; Nakayama, K.; Roden, R.; Glas, R.; Slamon, D.; Diaz, L.A., Jr; Vogelstein, B.; et al. Frequent mutations of chromatin remodeling gene ARID1A in ovarian clear cell carcinoma. Science 2010, 330, 228–231. [Google Scholar]
- Rahman, M.; Nakayama, K.; Rahman, M.T.; Nakayama, N.; Ishikawa, M.; Katagiri, A.; Iida, K.; Nakayama, S.; Otsuki, Y.; Shih, I.-M.; et al. Clinicopathologic and biological analysis of PIK3CA mutation in ovarian clear cell carcinoma. Hum. Pathol 2012, 43, 2197–2206. [Google Scholar]
- Uno, K.; Homma, S.; Satoh, T.; Nakanishi, K.; Abe, D.; Matsumoto, K.; Oki, A.; Tsunoda, H.; Yamaguchi, I.; Nagasawa, T.; et al. Tissue factor expression as a possible determinant of thromboembolism in ovarian cancer. Br. J. Cancer 2007, 96, 290–295. [Google Scholar]
- Kurman, R.J.; Shih, I.-M. Molecular pathogenesis and extraovarian origin of epithelial ovarian cancer—Shifting the paradigm. Hum. Pathol 2011, 42, 918–931. [Google Scholar]
- Lalwani, N.; Prasad, S.R.; Vikram, R.; Shanbhogue, A.K.; Huettner, P.C.; Fasih, N. Histologic, molecular, and cytogenetic features of ovarian cancers: Implications for diagnosis and treatment. Radiographics 2011, 31, 625–646. [Google Scholar]
- Han, E.S.; Lin, P.; Wakabayashi, M. Current status on biologic therapies in the treatment of epithelial ovarian cancer. Curr. Treat. Options Oncol 2009, 10, 54–66. [Google Scholar]
- Markman, M. Antiangiogenic drugs in ovarian cancer. Expert Opin. Pharmacother 2009, 10, 2269–2277. [Google Scholar]
- Behbakht, K.; Sill, M.W.; Darcy, K.M.; Rubin, S.C.; Mannel, R.S.; Waggoner, S.; Schilder, R.J.; Cai, K.Q.; Godwin, A.K.; Alpaugh, R.K. Phase II trial of the mTOR inhibitor, temsirolimus and evaluation of circulating tumor cells and tumor biomarkers in persistent and recurrent epithelial ovarian and primary peritoneal malignancies: A gynecologic oncology group study. Gynecol. Oncol 2011, 123, 19–26. [Google Scholar]
- Matsumura, N.; Mandai, M.; Okamoto, T.; Yamaguchi, K.; Yamamura, S.; Oura, T.; Baba, T.; Hamanishi, J.; Kang, H.S.; Matsui, S.; et al. Sorafenib efficacy in ovarian clear cell carcinoma revealed by transcriptome profiling. Cancer Sci 2010, 101, 2658–2663. [Google Scholar]
- Janku, F.; Wheler, J.J.; Westin, S.N.; Moulder, S.L.; Naing, A.; Tsimberidou, A.M.; Fu, S.; Falchook, G.S.; Hong, D.S.; Garrido-Laguna, I.; et al. PI3K/AKT/mTOR inhibitors in patients with breast and gynecologic malignancies harboring PIK3CA mutations. J. Clin. Oncol 2012, 30, 777–782. [Google Scholar]
- Takano, M.; Kikuchi, Y.; Kudoh, K.; Goto, T.; Furuya, K.; Kikuchi, K.; Kita, T.; Fujiwara, K.; Shiozawa, T.; Aoki, D. Weekly administration of temsirolimus for heavily pretreated patients with clear cell carcinoma of the ovary: A report of six cases. Int. J. Clin. Oncol 2011, 16, 605–609. [Google Scholar]
- Shoji, K.; Oda, K.; Kashiyama, T.; Ikeda, Y.; Nakagawa, S.; Sone, K.; Miyamoto, Y.; Hiraike, H.; Tanikawa, M.; Miyasaka, A.; et al. Genotype-dependent efficacy of a dual PI3K/mTOR inhibitor, NVP-BEZ235, and an mTOR inhibitor, RAD001,in endometrial carcinomas. PLoS One 2012, 7, e37431. [Google Scholar]
- Katagiri, A.; Nakayama, K.; Rahman, M.T.; Rahman, M.; Katagiri, H.; Nakayama, N.; Ishikawa, M.; Ishibashi, T.; Iida, K.; Kobayashi, H.; et al. Loss of ARID1A expression is related to shorter progression-free survival and chemoresistance in ovarian clear cell carcinoma. Mod. Pathol 2012, 25, 282–288. [Google Scholar]
- Yamaguchi, K.; Mandai, M.; Oura, T.; Matsumura, N.; Hamanishi, J.; Baba, T.; Matsui, S.; Murphy, S.K.; Konishi, I. Identification of an ovarian clear cell carcinoma gene signature that reflects inherent disease biology and the carcinogenic processes. Oncogene 2010, 29, 1741–1752. [Google Scholar]

| ARID1A | Mutation |
|---|---|
| PPP2R1A | Wildtype |
| PIK3CA | Mutation |
| KRAS | Wildtype |
| HIF1α | High expression |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rahman, M.; Nakayama, K.; Ishibashi, T.; Ishikawa, M.; Rahman, M.T.; Katagiri, H.; Katagiri, A.; Iida, K.; Kikuchi, Y.; Miyazaki, K. A Case of Stage III c Ovarian Clear Cell Carcinoma: The Role for Predictive Biomarkers and Targeted Therapies. Int. J. Mol. Sci. 2013, 14, 6067-6073. https://doi.org/10.3390/ijms14036067
Rahman M, Nakayama K, Ishibashi T, Ishikawa M, Rahman MT, Katagiri H, Katagiri A, Iida K, Kikuchi Y, Miyazaki K. A Case of Stage III c Ovarian Clear Cell Carcinoma: The Role for Predictive Biomarkers and Targeted Therapies. International Journal of Molecular Sciences. 2013; 14(3):6067-6073. https://doi.org/10.3390/ijms14036067
Chicago/Turabian StyleRahman, Munmun, Kentaro Nakayama, Tomoka Ishibashi, Masako Ishikawa, Mohammed Tanjimur Rahman, Hiroshi Katagiri, Atsuko Katagiri, Kouji Iida, Yoshihiro Kikuchi, and Kohji Miyazaki. 2013. "A Case of Stage III c Ovarian Clear Cell Carcinoma: The Role for Predictive Biomarkers and Targeted Therapies" International Journal of Molecular Sciences 14, no. 3: 6067-6073. https://doi.org/10.3390/ijms14036067




