A Novel Peroxidase CanPOD Gene of Pepper Is Involved in Defense Responses to Phytophtora capsici Infection as well as Abiotic Stress Tolerance
Abstract
:1. Introduction
2. Results and Discussion
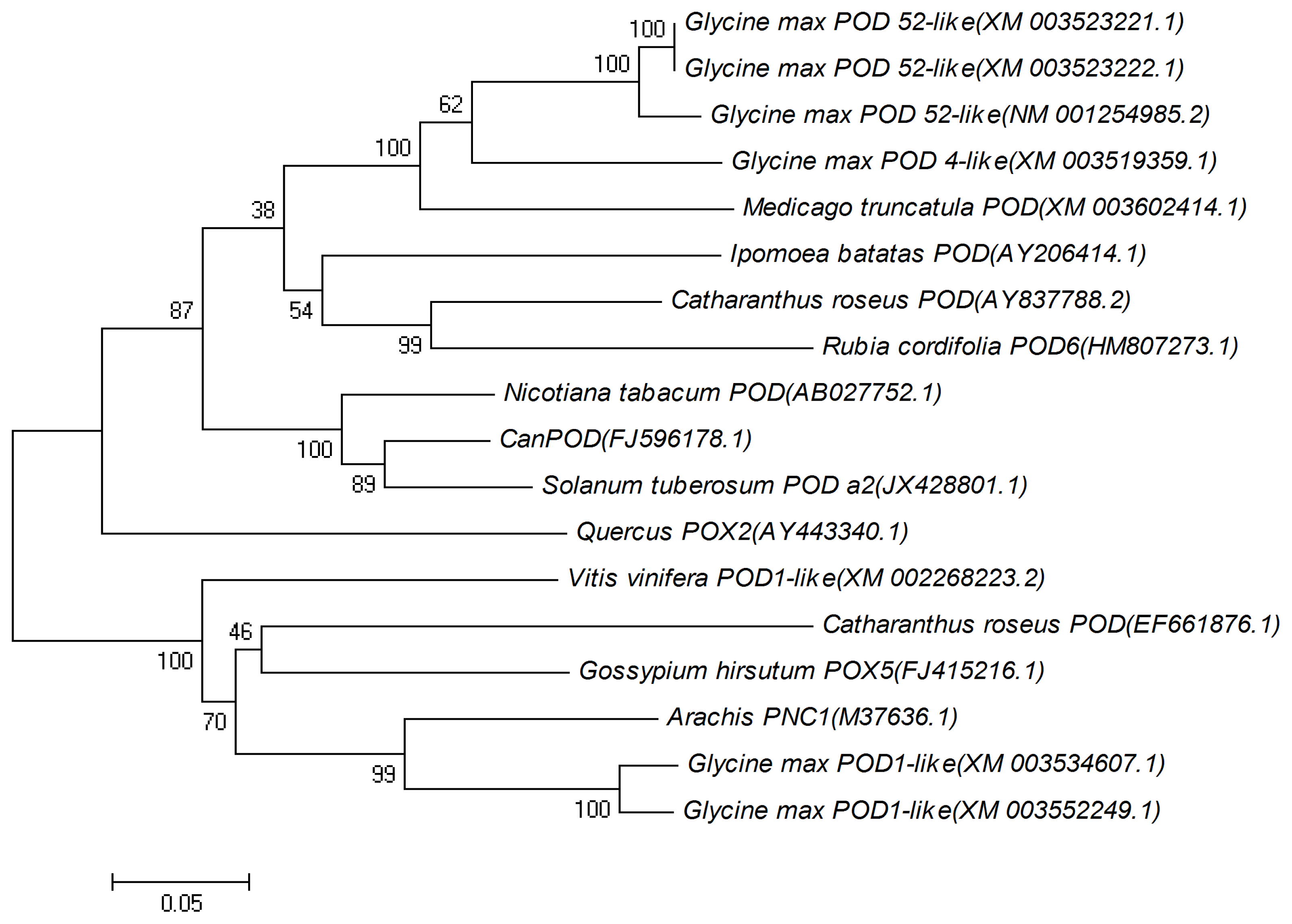
2.1. Cloning and Sequence Analysis of CanPOD Gene
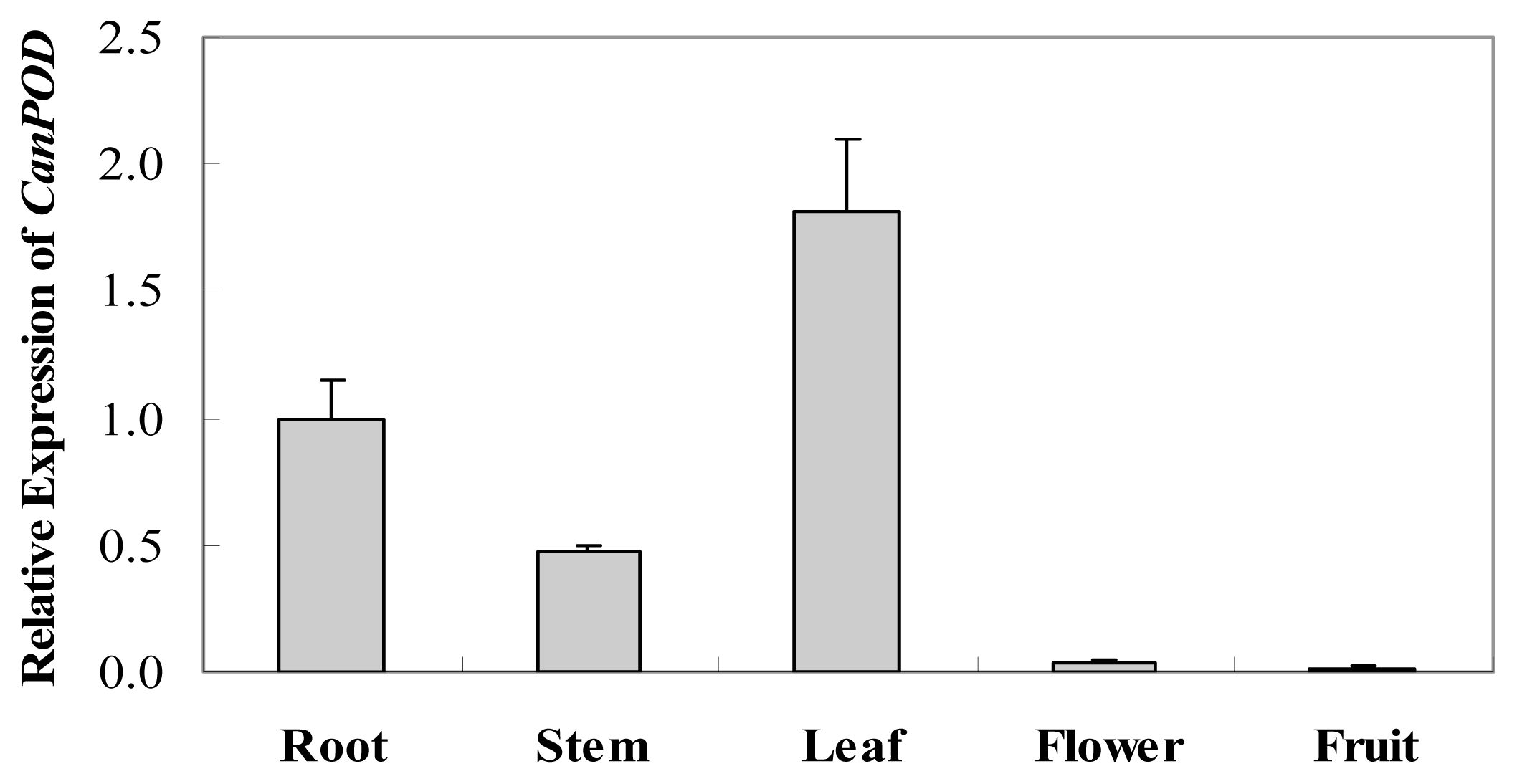
2.2. Tissue-Specific Expression of CanPOD Gene in Different Tissues of Pepper Plants
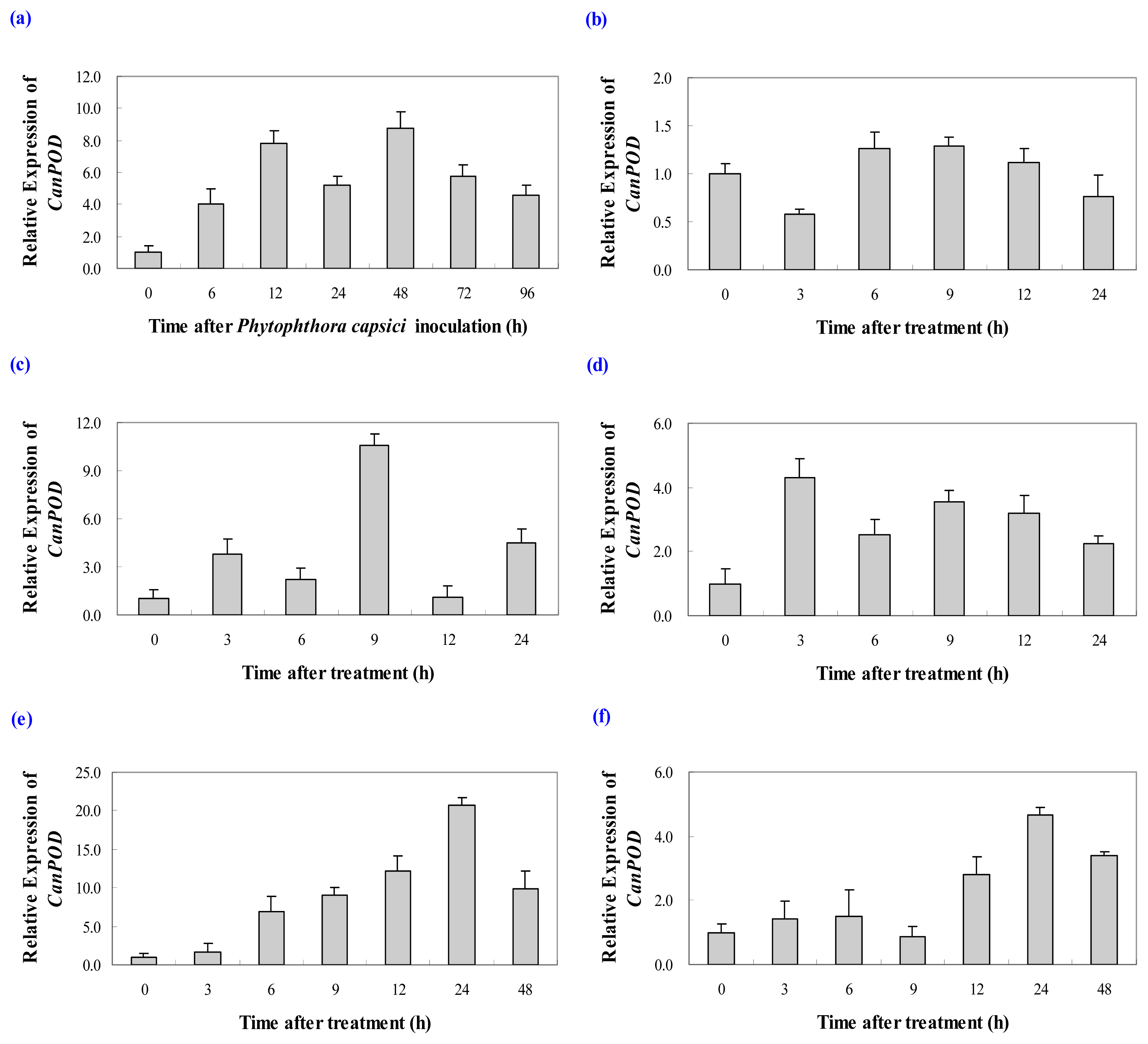
2.3. Expression Profiles of CanPOD Gene in Response to Biotic and Different Aboitic Stress Treatments
2.3.1. Expression Analysis of CanPOD Gene in Response to Phytophthora capsici Infection
2.3.2. Expression Analysis of CanPOD Gene in Response to Cold, Salt and Drought Stress Treatments
2.3.3. Expression Analysis of CanPOD Gene in Response to SA and MeJA Treatments
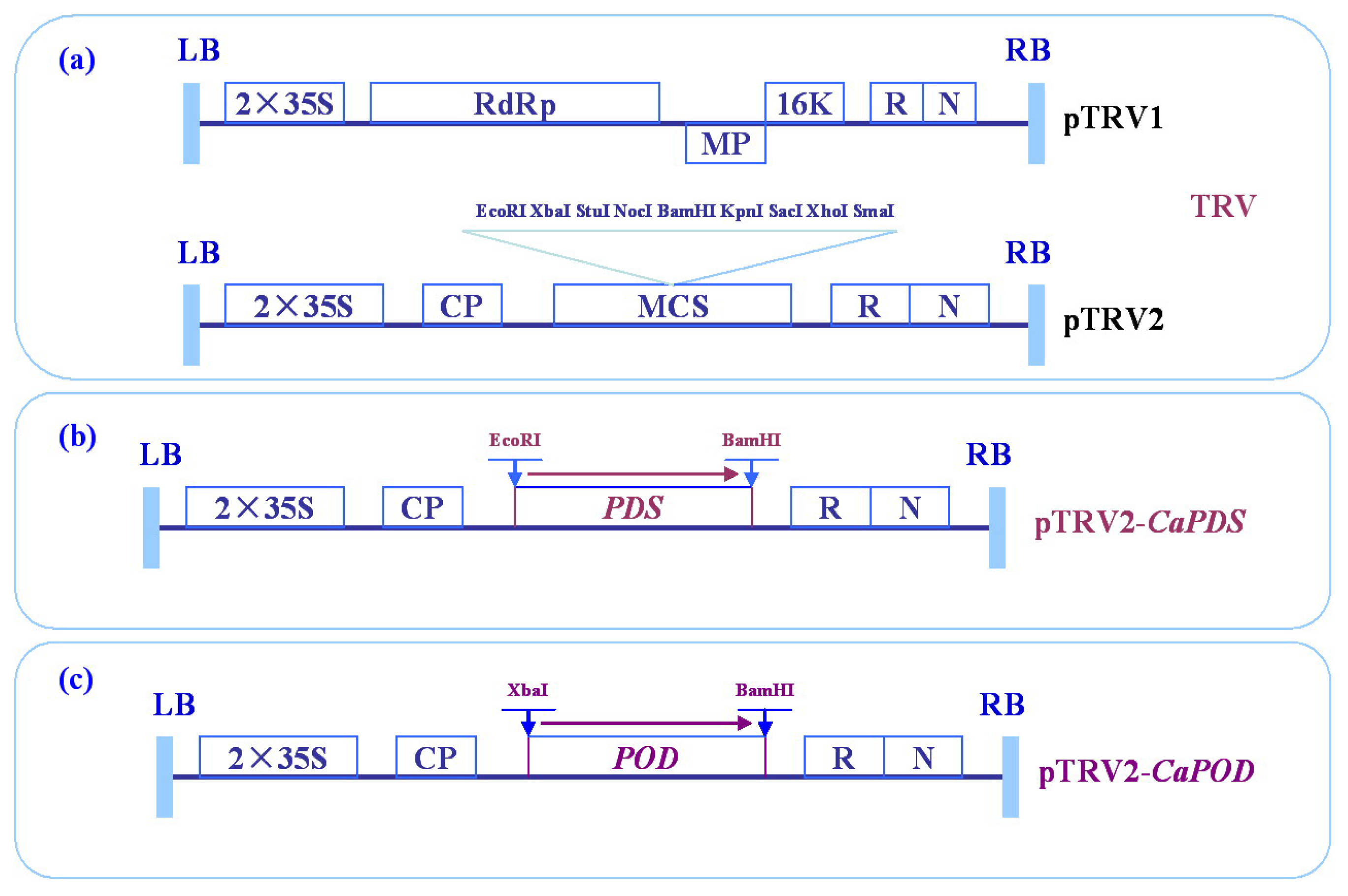
2.4. Virus-Induced Gene Silencing (VIGS) of CanPOD Gene in Pepper Plants
3. Experimental Section
3.1. Plant Materials and Growth Conditions
3.2. Pathogen Preparation and Inoculation Procedures
3.3. Cloning and Sequence Analysis of CanPOD Gene
3.4. CanPOD Gene Expression Patterns Analysis
3.4.1. Tissue-Specific Expression of CanPOD Gene
3.4.2. Stress Treatments
3.5. RNA Isolation and Real-Time RT-PCR Analysis
3.6. Virus-Induced Gene Silencing (VIGS) Analysis of CanPOD Gene in Pepper Plants
3.7. Peroxidase (EC 1.11.1.7) Activity Assay
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Lamour, K.H.; Stam, R.; Jupe, J.; Huitema, E. The oomycete broad-host-range pathogen Phytophthora capsici. Mol. Plant Pathol 2012, 13, 329–337. [Google Scholar]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar]
- Sarowar, S.; Kim, E.N.; Kim, Y.J.; Ok, K.D.; Kim, K.D.; Hwang, B.K.; Shin, J.S. Overexpression of a pepper ascorbate peroxidase-like 1 gene in tobacco plants enhances tolerance to oxidative stress and pathogens. Plant Sci 2005, 1, 55–63. [Google Scholar]
- Dang, F.F.; Wang, Y.N.; Yu, L.; Eulgem, T.; Lai, Y.; Liu, Z.Q.; Wang, X.; Qiu, A.L.; Zhang, T.X.; Lin, J.; et al. CaWRKY40, a WRKY protein of pepper, plays an important role in the regulation of tolerance to heat stress and resistance to Ralstonia solanacearum infection. Plant Cell Environ. 2012. [Google Scholar] [CrossRef]
- Passardi, F.; Penel, C.; Dunand, C. Performing the paradoxical: How plant peroxidases modify the cell wall. Trends Plant Sci 2004, 11, 534–540. [Google Scholar]
- Tognolli, M.; Penel, C.; Greppin, H.; Simon, P. Analysis and expression of the class III peroxidase large gene family in Arabidopsis thaliana. Gene 2002, 288, 129–138. [Google Scholar]
- Welinder, K.G.; Justesen, A.F.; Inger, V.H.; Kjærsgård, I.V.H.; Jensen, R.B.; Rasmussen, S.K.; Jespersen, H.M.; Duroux, L. Structural diversity and transcription of class III peroxidases from Arabidopsis thaliana. Eur. J. Biochem 2002, 269, 6063–6381. [Google Scholar]
- Hiraga, S.; Sasaki, K.; Ito, H.; Ohashi, Y.; Matsui, H. A large family of class III plant peroxidases. Plant Cell Physiol 2001, 42, 462–468. [Google Scholar]
- Valerio, L.; Meyer, M.D.; Penel, C.; Dunand, C. Expression analysis of the Arabidopsis peroxidase multigenic family. Phytochemistry 2004, 65, 1331–1342. [Google Scholar]
- Kim, S.S.; Choi, S.Y.; Park, J.H.; Lee, D.J. Regulation of the activity of Korean radish cationic peroxidase promoter during dedifferentiation and differentiation. Plant Physiol. Biochem 2004, 42, 763–772. [Google Scholar]
- Bae, H.; Roberts, D.P.; Lim, H.S.; Strem, M.D.; Park, S.C.; Ryu, C.M.; Melnick, R.L.; Bailey, B.A. Endophytic trichoderma isolates from tropical environments delay disease onset and induce resistance against Phytophthora capsici in hot pepper using multiple mechanisms. Mol. Plant-Microbe Interact 2011, 24, 336–351. [Google Scholar]
- Passardi, F.; Cosio, C.; Penel, C.; Dunand, C. Peroxidases have more functions than a Swiss army knife. Plant Cell Rep 2005, 24, 255–265. [Google Scholar]
- Veloso, J.; Díaz, J. Fusarium oxysporum Fo47 confers protection to pepper plants against Verticillium dahliae and Phytophthora capsici, and induces the expression of defence genes. Plant Pathol 2012, 61, 281–288. [Google Scholar]
- Choi, H.W.; Kim, Y.J.; Lee, S.C.; Hong, J.K.; Hwang, B.K. Hydrogen peroxide generation by the pepper extracellular peroxidase CaPO2 activates local and systemic cell death and defense response to bacterial pathogens. Plant Physiol 2007, 145, 890–904. [Google Scholar]
- Bae, E.K.; Lee, H.; Lee, J.S.; Noh, E.W.; Jo, J. Molecular cloning of a peroxidase gene from poplar and its expression in response to stress. Tree Physiol 2006, 26, 1405–1412. [Google Scholar]
- Hiraga, S.; Yamamoto, K. Diverse expression profiles of 21 rice peroxidase genes. FEBS Lett 2000, 471, 245–250. [Google Scholar]
- Ito, H.; Hiraga, S.; Tsugawa, H.; Matsui, H.; Honma, M.; Otsuki, Y.; Murakami, T.; Ohashi, Y. Xylem-specific expression of wound-inducible rice peroxidase genes in transgenic plants. Plant Sci 2000, 155, 85–100. [Google Scholar]
- Kim, K.Y.; Kwon, H.K.; Kwon, S.Y.; Lee, H.S.; Hur, Y.; Bang, J.W.; Choi, K.S.; Kwak, S.S. Differential expression of four sweet potato peroxidase genes in response to abscisic acid and ethephon. Phytochemistry 2000, 54, 19–22. [Google Scholar]
- Yoshida, K.; Kaothien, P.; Matsui, T.; Kawaoka, A.; Shinmyo, A. Molecular biology and application of plant Peroxidase genes. Appl. Microbiol. Biot 2003, 60, 665–670. [Google Scholar]
- Park, S.Y.; Ryu, S.H.; Kwon, S.Y.; Lee, H.S.; Kim, J.G.; Kwak, S.S. Differential expression of six novel peroxidase cDNAs from cell cultures of sweetpotato in response to stress. Mol. Genet. Genomics 2003, 269, 542–552. [Google Scholar]
- Matin, M.N.; Pandeya, D.; Baek, K.H.; Lee, D.S.; Lee, J.H.; Kang, H.; Kang, S.G. Phenotypic and genotypic analysis of rice lesion mimic mutants. Plant Pathology J 2010, 26, 159–169. [Google Scholar]
- Sang, M.K.; Kim, J.G.; Kim, K.D. Biocontrol activity and induction of systemic resistance in pepper by compost water extracts against Phytophthora capsici. Phytopathology 2010, 100, 774–783. [Google Scholar]
- Sang, M.K.; Kim, K.D. Biocontrol activity and primed systemic resistance by compost water extracts against anthracnoses of pepper and cucumber. Phytopathology 2011, 101, 732–740. [Google Scholar]
- Way, H.M.; Kazan, K.; Goulter, K.C.; Birch, R.G.; Manners, J.M. Expression of the Shpx2 peroxidase gene of Stylosanthes humilis in transgenic tobacco leads to enhanced resistance to Phytophthora parasitica pv. nicotianae and Cercospora nicotianae. Mol. Plant Pathol 2000, 1, 223–232. [Google Scholar]
- Liu, G.; Sheng, X.; Greenshields, D.L.; Ogieglo, A.; Kaminskyj, S.; Selvaraj, G.; Wei, Y. Profiling of wheat class III peroxidase genes derived from powdery mildew-attacked epidermis reveals distinct sequence-associated expression patterns. Mol. Plant-Microbe Interact 2005, 18, 730–741. [Google Scholar]
- Kim, Y.H.; Kim, C.Y.; Song, W.K.; Park, D.S.; Kwon, S.Y.; Lee, H.S.; Bang, J.B.; Kwak, S.S. Overexpression of sweetpotato swpa4 peroxidase results in increased hydrogen peroxide production and enhances stress tolerance in tobacco. Planta 2008, 227, 867–881. [Google Scholar]
- Choi, H.; Hwang, B. The pepper extracellular peroxidase CaPO2 is required for salt, drought and oxidative stress tolerance as well as resistance to fungal pathogens. Planta 2012, 235, 1369–1382. [Google Scholar]
- Do, H.M.; Hong, J.K.; Jung, H.W.; Kim, S.H.; Ham, J.H.; Hwang, B.K. Expression of peroxidase-like genes, H2O2 production, and peroxidase activity during the hypersensitive response to Xanthomonas campestris pv. vesicatoria in Capsicum annuum. Mol. Plant-Microbe Interact 2003, 16, 196–205. [Google Scholar]
- Chmielowska, J.; Veloso, J.; Gutierrez, J.; Silvar, C.; Diaz, J. Cross-protection of pepper plants stressed by copper against a vascular pathogen is accompanied by the induction of a defence response. Plant Sci 2010, 178, 176–182. [Google Scholar]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot-London 2003, 91, 179–194. [Google Scholar]
- Baltruschat, H.; Fodor, J.; Harrach, B.D.; Niemczyk, E.; Barna, B.; Gullner, G.; Janeczko, A.; Kogel, K.H.; Schäfer, P.; Schwarczinger, I.; et al. Salt tolerance of barley induced by the root endophyte Piriformospora indica is associated with a strong increase in antioxidants. New Phytol 2008, 180, 501–510. [Google Scholar]
- Murata, N.; Ishizaki, N.O.; Higashi, S.; Hayashi, H.; Tasaka, Y.; Nishida, I. Genetically engineered alteration in the chilling sensitivity of plants. Nature 1992, 356, 710–713. [Google Scholar]
- Sreenivasulu, N.; Grimm, B.; Wobus, U.; Weschke, W. Differential response of antioxidant compounds to salinity stress in salt tolerant and salt sensitive seedlings of foxtail millet (Setaria italica). Physiol Plantarum 2000, 109, 435–442. [Google Scholar]
- Menezes-Benavente, L.; Teixeira, F.K.; Kamei, C.L.A.; Pinheiro, M.M. Salt stress induces altered expression of genes encoding antioxidant enzymes in seedlings of a Brazilian indica rice (Oryza sativa L.). Plant Sci 2004, 166, 323–331. [Google Scholar]
- Koussevitzky, S.; Suzuki, N.; Huntington, S.; Armijo, L.; Sha, W.; Cortes, D.; Shulaev, V.; Mittler, R. Ascorbate peroxidase 1 plays a key role in the response of Arabidopsis thaliana to stress combination. J. Biol. Chem 2008, 283, 34197–34203. [Google Scholar]
- Donahue, J.L.; Okpodu, C.M.; Cramer, C.L.; Grabau, E.A.; Alscher, R.G. Responses of antioxidants to paraquat in pea leaves. Plant Physiol 1997, 113, 249–257. [Google Scholar]
- Cazale, A.C.; Rouet-Mayer, M.A.; Barbier-Brygoo, H.; Mathieu, Y.; Lauriere, C. Oxidative burst and hypoosmotic stress in tobacco cell suspension. Plant Physiol 1998, 116, 659–669. [Google Scholar]
- An, S.H.; Sohn, K.H.; Choi, H.W.; Hwang, I.S.; Lee, S.C.; Hwang, B.K. Pepper pectin methylesterase inhibitor protein CaPMEI1 is required for antifungal activity, basal disease resistance and abiotic stress tolerance. Planta 2008, 228, 61–78. [Google Scholar]
- Lee, S.C.; Hwang, B.K. Functional roles of the pepper antimi-crobial protein gene, CaAMP1, in abscisic acid signaling and salt and drought tolerance in Arabidopsis. Planta 2009, 229, 383–391. [Google Scholar]
- Choi, D.S.; Hwang, B.K. Proteomics and functional analyses of pepper abscisic acid-responsive 1 (ABR1), which is involved in cell death and defense signaling. Plant Cell 2011, 23, 823–842. [Google Scholar]
- Glazebrook, J. Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu. Rev. Phytopathol 2005, 43, 205–227. [Google Scholar]
- Mur, L.A.J.; Kenton, P.; Atzorn, R.; Miersch, O.; Wasternack, C. The outcomes of concentration-specific interactions between salicylate and jasmonate signaling include synergy, antagonism, and oxidative stress leading to cell death. Plant Physiol 2006, 140, 249–262. [Google Scholar]
- Liu, Y.; Schiff, M.; Dinesh-Kumar, S.P. Virus-induced gene silencing in tomato. Plant J 2002, 31, 777–786. [Google Scholar]
- Chung, E.; Seong, E.; Kim, Y.C.; Chung, E.J.; Oh, S.K.; Lee, S.; Park, J.M.; Joung, Y.H.; Choi, D. A method of high frequency virus-induced gene silencing in chili pepper (Capsicum annuum L.cv.Bukang). Mol. Cells 2004, 17, 377–380. [Google Scholar]
- An, S.H.; Choi, H.W.; Hwang, I.S.; Hong, J.K.; Hwang, B.K. A novel pepper membrane-located receptor-like protein gene CaMRP1 is required for disease susceptibility, methyl jasmonate insensitivity and salt tolerance. Plant Mol. Biol 2008, 67, 519–533. [Google Scholar]
- Hwang, I.S.; Hwang, B.K. The pepper 9-lipoxygenase gene CaLOX1 functions in defense and cell death responses to microbial pathogens. Plant Physiol 2010, 152, 948–967. [Google Scholar]
- Kim, H.J.; Nahm, S.H.; Lee, H.R.; Yoon, G.B.; Kim, K.T.; Kang, B.C.; Choi, D.; Kweon, O.Y.; Cho, M.C.; Kwon, J.K. BAC-derived markers converted from RFLP linked to Phytophthora capsici resistance in pepper (Capsicum annuum L.). Theor. Appl. Genet 2008, 118, 15–27. [Google Scholar]
- Bioinformatics Tool. Available online: http:/blast.ncbi.nlm.nih.gov (accessed on 15 December 2012.
- Bioinformatics Tool. Available online: http://www.ncbi.nlm.nih.gov/stucture/cdd.shtml accessed on 15 December 2012.
- Wan, H.J.; Yuan, W.; Ruan, M.; Ye, Q.; Wang, R.; Li, Z.; Zhou, G.; Yao, Z.; Zhao, J.; Liu, S.; Yang, Y. Identification of reference genes for reverse transcription quantitative real-time PCR normalization in pepper (Capsicum annuum L.). Biochem. Biophy. Res. Co 2011, 416, 24–30. [Google Scholar]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C (T)) Method. Methods 2001, 25, 402–408. [Google Scholar]
- Liu, Y.; Schiff, M.; Marathe, R.; Dinesh-Kumar, S.P. Tobacco Rar1, EDS1 and NPR1/NIM1 like genes are required for N-mediated resistance to tobacco mosaic virus. Plant J 2002, 30, 415–429. [Google Scholar]
- Cunningham, F.X.; Gantt, E. Genes and enzymes of cartotenoid biosynthesis in plants. Plant Mol. Biol 1998, 49, 557–583. [Google Scholar]
- Beffa, R.; Martin, H.V.; Pilet, P.E. In vitro oxidation of indoleacetic acid by soluble auxin-oxidasesand peroxidases from maize roots. Plant Physiol 1990, 94, 485–491. [Google Scholar]




| Gene | Primer | Sequence (5′-3′) |
|---|---|---|
| CaUbi3 | QCaUbi3-F | TGTCCATCTGCTCTCTGTTG |
| QCaUbi3-R | CACCCCAAGCACAATAAGAC | |
| CanPOD | QCanPOD-F | CAAGGTTCAATGTGTGTTACC |
| QCanPOD-R | ATGATGATACAAATACAGTGCC |
© 2013 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, J.-E.; Liu, K.-K.; Li, D.-W.; Zhang, Y.-L.; Zhao, Q.; He, Y.-M.; Gong, Z.-H. A Novel Peroxidase CanPOD Gene of Pepper Is Involved in Defense Responses to Phytophtora capsici Infection as well as Abiotic Stress Tolerance. Int. J. Mol. Sci. 2013, 14, 3158-3177. https://doi.org/10.3390/ijms14023158
Wang J-E, Liu K-K, Li D-W, Zhang Y-L, Zhao Q, He Y-M, Gong Z-H. A Novel Peroxidase CanPOD Gene of Pepper Is Involved in Defense Responses to Phytophtora capsici Infection as well as Abiotic Stress Tolerance. International Journal of Molecular Sciences. 2013; 14(2):3158-3177. https://doi.org/10.3390/ijms14023158
Chicago/Turabian StyleWang, Jun-E, Ke-Ke Liu, Da-Wei Li, Ying-Li Zhang, Qian Zhao, Yu-Mei He, and Zhen-Hui Gong. 2013. "A Novel Peroxidase CanPOD Gene of Pepper Is Involved in Defense Responses to Phytophtora capsici Infection as well as Abiotic Stress Tolerance" International Journal of Molecular Sciences 14, no. 2: 3158-3177. https://doi.org/10.3390/ijms14023158




